HPLC IDENTIFICATION OF CAFFEOYLQUINIC ACID DERIVATIVES OF CEPHALARIA GIGANTEA
HTML Full TextHPLC IDENTIFICATION OF CAFFEOYLQUINIC ACID DERIVATIVES OF CEPHALARIA GIGANTEA
N. Tabatadze * 1, I. Tsomaia 2, A. Pichette 3 and V. Mshvildadze 3, 4
Faculty of Natural Sciences 1, Mathematics, Technology and Pharmacy, Ivane Javakhishvili Tbilisi State University, 26A. Politkovskayast, 0186 Tbilisi, Georgia.
Faculty of Chemical Technology and Metallurgy 2, Department of Pharmacy, Georgian Technical University, 75M. Kostavast, 0160 Tbilisi, Georgia.
Department of Fundamental Sciences 3, University of Quebec, 555 Bd. Université, Chicoutimi, G7H2B1, Canada.
Iovel Kutateladze Institute of Pharmacochemistry 4, Tbilisi State Medical University, 36 P. Sarajishvilist, 0159 Tbilisi, Georgia.
ABSTRACT: Six phenolic acids -chlorogenic acid (1), caffeic acid (2), 4,5 di-O-caffeoylquinic acid (3), 3,5 di-O-caffeoylquinic acid (4), 3,4 di-O-caffeoylquinic acid (5) and 3,4,5-tri-O-caffeoylquinic acid (6) were identified by HPLC-UV-MS from the roots of Cephalaria gigantea (Dipsacaceae) growing in Georgia. The aim of the present research was a qualitative identification of phenolic constituents in an anticonvulsive enriched fraction from the roots of C. gigantea, for further quantitative standardization. The HPLC profile demonstrated a good resolution of phenolic acids of the active extract within 25 min of gradient mode (ACN 5% to 40%). On the basis of retention times, using the UV and MS spectra of the corresponding standards, compounds 4 and 5 were found to be majors in the analyzed fraction and were reported for the first time from C. gigantea roots. These experimental data could be a solid base for the future quantitative standardization of the anticonvulsive fraction for the prevention and treatment of psychosomatic forms of epilepsy.
| Keywords: |
Triterpene saponins, HPLC, Phenolic acids, Caffeoylquinic acid, Anticonvulsive fraction
INTRODUCTION: Out of 60 species of the genus Cephalaria, 12 are found in Georgia, among these Cephalaria gigantea (Ledeb.) Bobr. Fam. Dipsacaceae) is a Caucasian endemic plant and is widespread throughout the Caucasus 1. The presence of alkaloids, flavonoids, phenolcarboxylic acids and triterpene compounds was demonstrated in different members of the genus Cephalaria 2.
Preliminary phytochemical analysis showed the presence of 0.2% alkaloids (including gentianine, gentianidine, gentianaine) in the plant’s roots 3.
The main constituents of the roots are triterpene saponins, represented by 15 giganteosides: A-J, J', K-N. The chemical structures of isolated compounds were established on the basis of chemical methods (hydrolyses), by 1D and 2D NMR experiments (1H, 13C, GS-COSY, GS-HMBC, GS-HMQC and GS-HSQC-TOCSY) and mass Spectrometry (MALDI-TOF, ESI-HR-MS 4-8. The presence of chlorogenic acid, caffeic acid and flavonoid glycoside quercitrin was demon-strated in the roots 9.
The fungicidal and anti-protozoal activities of individual substances and enriched fractions from the roots of Cephalaria were studied by French researchers; the alkaloid containing fractions of the roots of C. gigantea were active against Plazmodium falciparum, a parasite that causes malaria 10. The giganteoside E exhibited a significant inhibitory effect on HL-60 leukemia cells 11.
The roots of Cephalaria gigantea was used in Georgian traditional (folk) medicine as anti-epileptic remedy 12. The anticonvulsive effects of the aqueous extract of C. Gigantean was studied on pentylenetetrazol and audiogenic seizure models (The Krushinsky-Molodkina and Wistar rat strains) 13. The aqueous extract was effective in both types (single and repeated) of oral administration and injection. At the same time, a significant increase in the duration of a barbiturate induced sleep in mice shows, that the extract possesses sedative properties. An optimal method was developed for obtaining of anti convulsive, enriched fraction from C. Gigantean roots with a very low toxicity was developed. The obtained phytochemical, biological and toxicologic data gives an opportunity to develop a new bioactive product of plant origin for prevention and treatment of psychosomatic forms of epilepsy 14, 15.
MATERIALS AND METHODS: The subject of the proposed work was a qualitative identification of phenolic constituents in the anticonvulsive enriched fraction from the roots of C. gigantea, for further, quantitative standardisation of identified major markers.
Standards and Chemicals: The individual high purity standards of phenolic acids were purchased from Sigma-Aldrich. Reagent and analytical grade solvents (EtOAc, MeOH, ACN) were purchased from VWR International (Ville Mont-Royal, Québec, Canada) and used without further purification for the extraction and analyses. Deionized water was produced provided by a Milli-Q water system (Millipore, Billerica, MA, USA). The HPLC solvents were degassed under vacuum and filtered through a 0.22-micron nylon membrane.
Plant Sample Collection: The roots of Cephalaria gigantean were collected in the region of Imereti (Georgia, October 2018) and dried in the shade. A voucher specimen (No 7867) was placed at the herbarium of the department of Pharmacobotany, at the TSMU Iovel Kutateladze Institute of Pharmacochemistry (Herbarium TBPH, Tbilisi, Georgia).
Preparation of Extracts and Standards: Dried and powdered roots of C. Gigantean (350 g) were extracted with water by heating during 30 min (2 l). The aqueous extract was concentrated under vacuum and lyophilised yielded 96 g. In order, to obtain an enriched fraction of phenolic compounds from the active extract of C. gignatea, multiple treatment of the aqueous extract with ethyl acetate was realized. Namely, 10 g of the dry extract was dissolved in 80 mL of distilled water (ultrasonic bath solubilisation). The aqueous solution was placed in a 200 mL separatory funnel and extracted with 200 mL of ethyl acetate 5 times with careful stirring. The ethyl acetate layers, enriched with phenolic substances, were jointed and the organic solvent was removed by a vacuum rotating evaporator. While the final stage of drying was performed in a vacuum dryer at 50-60 °C with the presence of calcium chloride to give 2 g of enriched fraction.
The standard stock solutions were prepared by dissolving 1 mg of each standard in MeOH (1 mL), the concentration of the EtOAcfraction sample was 10 mg/mL in MeOH.
The qualitative analyses were performed on an Agilent 1200 HPLC system (Agilent Technologies, Palo Alto, CA) consisting of a degasser, a quaternary pump, an automatic injector, a temperature-controlled column compartment, a diode array detector (UV detection was performed at 365 nm) and a mass selective detector Agilent G1946 VL model equipped with an ES-APCI sourcein negative mode on a C18 reversed phase column Kinetex XB-Rp18, 250 × 4.6mm (Phenomenex), the mobile phase A (H2O+HCOOH 0.1%) and phase B (Acetonitrile + HCOOH 0.1%) were used in gradient condition (acetonitrile 5%→40%). Flow rate was 1.0 ml/min, injection volume - 10 µL and run time - 25 mn.
RESULTS AND DISCUSSION: The EtOAc phase enriched with phenolic acids, was obtained from the antiepileptic aqueous extract of the roots of Cephalaria gigantea by liquid-liquid extraction. A high performance liquid chromatography method was developed for the qualitative characterization of phenolic constituents of the EtOAc fraction.
The HPLC profile demonstrated a good resolution of phenolic acids of the enriched active fraction within 25 min of gradient mode (ACN 5% to 40%).
FIG. 1: HPLC-MS CHROMATOGRAM PROFILE OF THE EtOAc FRACTION OF CEPHALARIA GIGANTEA
Retention Times: Compound 1 - 10.9 min, compound 2 - 12.4 min., compound 3- 17.7 min, compound 4 - 18.4 min, compound 5 - 19.2 min. and compound 6 – 23.7 min.
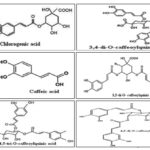
On the basis of the retention times, using the UV and MS spectra of corresponding standards, the following substances have been identified: chlorogenic acid (1), caffeic acid (2), 4,5 di-O-caffeoylquinic acid (3), 3,5 di-O-caffeoylquinic acid (4), 3,4 di-O-caffeoylquinic acid (5) and 3,4,5-tri-O-caffeoylquinic acid (6). Thus, the compounds 4 and 5 were majors in analyzed fraction. These phenolic acids were reported for the first time in C. gigantea’s roots.
FIG. 2: CHEMICAL STRUCTURES OF PHENOLIC ACIDS FROM CEPHALARIA GIGANTEA
CONCLUSION: An RP-HPLC-DAD-MS method was developed for the identification of 6 phenolic acids in the anticonvulsive enriched fraction from Cephalaria gigantea roots. The proposed HPLC assay demonstrated an optimal resolution of the components. The results showed that 3,5 di-O-caffeoylquinic acid and 3,4 di-O-caffeoylquinic acid are the dominants in the roots of C. gigantea. Although the minor phenolic acids could contribute to the total anticonvulsive activity of the Cephalaria’s extract.
These results could be a solid base for the future quantitative standardization of anticonvulsive fraction and gives an opportunity to develop a new product of plant origin for prevention and treatment of psychosomatic forms of epilepsy.
ACKNOWLEDGEMENT: The authors acknowledge Romain Jarossay for his technical assistance.
CONFLICTS OF INTEREST: The authors declare no conflict of interest.
REFERENCES:
- KetskhoveliN, Kharadze A and Gagnidze R: Flora of Georgia, Caprifoliaceae - Tbilisi, Metsniereba, Edition 2, Vol. 13, 2001.
- Tabatadze N, Mshvildadze V, Elias R, Ollivier E, Faure R, Dekanosidze G, Balansard G and Kemertelidze E: A new triterpenoid glycosides from the roots of Cephalaria gigantea Georgia Chemical Journal 2003; 3(2): 158-60.
- Aliev AM, Movsumov IS and Bagirov EX: Alkaloids from some species of Cephalaria. Chemistry of Nat. Compounds 1975; 5: 667.
- Zviadadze LD, Dekanosidze GE, Djikia OD, Kemertelidze EP and Shashkov AS: Triterpene glycosides of Cephaparia gigantea, structures of giganteosides D and G Bioorganic chemistry 1981; 7(5): 736-40.
- Zviadadze LD, Dekanosidze GE, Djikia OD and Kemertelidze EP: Triterpene glycosides of Cephaparia gigantea, structures of giganteosides E and H. Chemistry of Nat Compounds 1983; 1: 46-49.
- Tabatadze N, Mshvildadze V, Dekanosidze G, Zviadadze L, Elias R, Ollivier E, Faure R and Balansard G: Triterpenoid glycosides from the roots of Cephalaria gigantea Bulletin of the Georgian Academy of Sciences 2005; 171(2): 296-99.
- Tabatadze NA, Zviadadze LD, Dekanosidze GE and Kemertelidze EP: Triterpene glycosides of Cephaparia gigantea, structures of giganteosides I and K. Georgia Chemical Journal 2003; 3(2): 156-57.
- Tabatadze N, Elias R, Faure R, Gerkens P, De Paul-Gillet MC, Kemertelidze E, Chea A and Ollivier E: Cytotoxic triterpenoid saponins from the roots of Cephalaria gigantea. Chem Pharm Bull 2007; 55(1): 102-05.
- Tabatadze N, Tabidze B, Getia M, Mshvildadze V, Pichette A, Dekanosidze G and Kemertelidze E: HPLC analysis of an anticonvulsant fraction from the roots of Cephalaria gigantea. Bulletin of the. Georgian National Academy of Sciences 2017; 11(2): 118-22.
- Tabatadze N, Zviadadze L, Favel A, Delmas F, Di Giorgio C, Balansard G and Kemertelidze E: Biologically active triterpene saponins from Cephalaria gigantea. Bulletin of the Georgian academy of Sciences 2002; 165(2): 318-20.
- Gerkens P, Dobson R, Tabatadze N, Mshvildadze V, Elias R, Peulen OJ, Jolois OM and De Paul-Gillet MC: Apoptosis and cytolysis induced by giganteosides and hederacolchisides in HL-60 cells. Anticancer Research 2007; 27: 2529-34.
- Zviadadze LD, Dekanosidze GE and Kemertelidze EP: Photochemical investigation of saponins from Cephalaria gigantea. Biological active compounds from. Georgian Flora 1976; 13: 143-46.
- Gogitidze NM, Mushkiashvili NI, Gedevanishvili MD, Tabatadze NA and Dekanosidze GE: Antiseizure activity of Cephalaria gigantea root extract. Georgian Medic News 2017; 264(3): 127-31.
- Sixarulidze I, Gogitidze N, Mushkiashvili N, Gabelaia M, Vachnadze N and Dekanosidze G: Anticonvulsant extract from the roots of Cephalaria gigantea. Allergology and Immunology 2010; 11(2): 170-72.
- Tabatadze N, Vachnadze N, Tabidze B, Getia M, Gogitidze N, Mshvildadze V and Dekanosidze G: Chemical content and pharmacological active compounds from the roots of Cephalaria gigantea growing in Georgia. Experimental and Clinical Medicine 2014; 4: 93-97.
How to cite this article:
Tabatadze N, Tsomaia I, Pichette A and Mshvildadze V: HPLC identification of caffeoylquinic acid derivatives of Cephalaria gigantea. Int J Pharmacognosy 2021; 8(4): 155-58. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(4).155-58.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
155-158
695
992
English
IJP
N. Tabatadze *, I. Tsomaia, A. Pichette and V. Mshvildadze
Faculty of Natural Sciences, Mathematics, Technology and Pharmacy, Ivane Javakhishvili Tbilisi State University, 26A. Politkovskayast, 0186 Tbilisi, Georgia.
nino_tabatadze@yahoo.com
27 November 2020
23 April 2021
29 April 2021
10.13040/IJPSR.0975-8232.IJP.8(4).155-58
30 April 2021