FORMULATION AND ASSESSMENT OF HERBAL CAPSULE USING MEDICINAL PLANT EXTRACTS
HTML Full TextFORMULATION AND ASSESSMENT OF HERBAL CAPSULE USING MEDICINAL PLANT EXTRACTS
Deep Kant *, M. Himaja Trivedi and Satendra Sharma
Shri Ramnath Singh Institute of Pharmaceutical Science and Technology, Sitholi, Gwalior, Madhya Pradesh, India.
ABSTRACT: The present study aimed to formulate and evaluate a polyherbal capsule using aqueous extracts of Vinca rosea, Curcuma longa, and Dalbergia sissoo. Plant materials were collected, authenticated, dried, and extracted using a traditional aqueous extraction method. Pre-formulation studies were conducted to assess powder flow properties, including bulk density, tapped density, Carr’s index, Hausner’s ratio, and angle of repose. Six formulations (PC1–PC6) were prepared by wet granulation and filled into size “0” hard gelatin capsules using a manual plate method. The capsules were evaluated for organoleptic characteristics, weight variation, disintegration time, moisture content, drug content, in-vitro dissolution, and stability under varying temperature and humidity conditions. Among all formulations, PC1 exhibited superior flow properties, uniform weight, rapid disintegration, and maximum drug release (94.77% at 30 minutes). Stability studies revealed good stability under ambient and moderately elevated temperatures, while degradation occurred at higher temperature and humidity. Overall, the results demonstrate that the developed polyherbal capsule is pharmaceutically acceptable, stable under recommended storage conditions, and holds promise for effective oral herbal therapy.
Keywords: Vinca rosea, Curcuma longa, and Dalbergia sissoo, Capsule
INTRODUCTION: Herbal medicines play a vital role in both traditional and modern healthcare systems due to their therapeutic efficacy, natural origin, and relatively low risk of adverse effects 1. Medicinal plants are rich sources of bioactive phytochemicals such as alkaloids, flavonoids, phenolics, and terpenoids, which contribute to diverse pharmacological activities 2, 3.
Vinca rosea, Curcuma longa, and Dalbergia sissoo are well-known medicinal plants widely used in traditional medicine for their anticancer, anti-inflammatory, antioxidant, antimicrobial, and immunomodulatory properties 4-8. The growing demand for standardized and scientifically validated herbal formulations has emphasized the need to develop suitable dosage forms that ensure safety, efficacy, and patient compliance 9.
Capsules are one of the most preferred oral dosage forms for herbal preparations as they provide accurate dosing, ease of administration, masking of unpleasant taste, and protection of active constituents from environmental factors 10. Hard gelatin capsules are particularly suitable for powdered herbal extracts due to their simplicity of formulation and manufacturing. However, the successful formulation of herbal capsules requires careful consideration of powder flow properties, excipient compatibility, capsule size, and filling techniques to achieve uniformity and stability of the final product 11.
The present study focuses on the formulation and assessment of a polyherbal capsule using extracts of Vinca rosea, Curcuma longa, and Dalbergia sissoo. The work involves the preparation of powdered extracts, selection of appropriate excipients, and evaluation of physicochemical parameters such as bulk density, tapped density, flowability, and capsule quality. This study aims to develop a standardized herbal capsule with acceptable pharmaceutical characteristics and potential therapeutic benefits.
MATERIALS AND METHODS:
Collection of Plant Materials: The aerial parts (leaves and stems) of Vinca rosea were collected from the local region of Lucknow, and a voucher specimen was deposited in the herbarium of the Botany Department. As the fresh material could not be stored for long, the leaves and stems were washed with water and dried in a hot-air oven at 60 °C for approximately three days until a constant weight was obtained. Drying concentrates non-volatile bioactive compounds by removing water, making dried material more suitable for extraction and storage than fresh plant parts. Rhizomes of Curcuma longa were similarly collected from Lucknow and authenticated. Prior to drying, the rhizomes were cured by boiling or steaming for 30–45 minutes to enhance color and reduce drying time, followed by drying in sunlight or in a hot-air oven at 50–60 °C until hard and brittle. Leaves of Dalbergia sissoo were collected and authenticated, then washed and oven-dried using the same procedure as for Vinca rosea 12.
Extraction: To prepare the aqueous extracts, 10 g of the aerial parts of Vinca rosea, 10 g of rhizomes of Curcuma longa, and 10 g of leaves of Dalbergia sissoo were separately weighed and added to 500 ml of boiling water. Each plant material was boiled gently for 10 minutes and then allowed to cool overnight, following traditional extraction procedures. The mixtures were subsequently filtered through a Buchner funnel using Whatman No. 1 filter paper. The filtrates were frozen in a dry ice–acetone bath. Once completely dried, the extracts were collected, transferred into moisture-free containers, sealed, and stored in a desiccator until further use for formulation or analysis 13.
Qualitative Analysis: Biochemical tests for the screening and identification of active components have been done using standard protocols. For each test, 100 μl/ml concentration of each extract were used for the analysis of biochemical compounds. Each extract was subjected to biochemical tests as per the methods described below for the identification of different compounds present in the plant extract 14.
Pre-formulation Study:
Determination of the Density of the Granules: Powder flow properties were evaluated by comparing bulk (poured) density and tapped density using Carr’s compressibility index, an empirical parameter for assessing flowability 15. Carr’s index was calculated using the equation:
Carr’s index (%) = (Tapped density – Bulk density) / Tapped density
Bulk density was determined by pouring a known mass of powder, previously passed through a 180-mesh sieve to break agglomerates, into a graduated cylinder and recording the unsettled volume (V₀). Tapped density was measured by mechanically tapping the cylinder containing the powder until minimal volume change was observed, and the final tapped volume (V₁₂₅₀) was recorded. Bulk density was calculated as m/V₀ and tapped density as m/V₁₂₅₀ (g/ml). Flowability was further assessed by determining the angle of repose (θ). Ten grams of powder were allowed to flow from a glass cylinder onto a flat plate to form a conical mound. The height (h) and radius (r) of the mound were measured, and the angle of repose was calculated using tan θ = h/r 16, 17.
Formulation Development:
Selection of Capsule Size: The capsule size was selected based on the volume of material to be filled. Hard gelatin capsules of sizes “0” to “4” are commonly available, and their corresponding body volumes. For pharmaceutical preparations, capsule sizes larger than “0” are rarely used due to swallowing difficulties, while size “5” is seldom employed because of limitations in automatic filling processes. Hence, size “0” capsules were selected for this study 18.
Selection of Filling Method: The choice of filling method depended on the available equipment. Capsule filling can be performed using manual or automatic machines. In the present study, a manual plate method was employed using a Feton hand-operated capsule filler. This method, widely used in pharmacies and development laboratories, involved plastic plates designed to hold 50 size “0” capsules. Capsule bodies were filled with powder by spreading and gentle shaking to ensure uniform filling and eliminate air pockets. Caps were then replaced and locked by gentle pressure. The final fill weight was achieved through a trial-and-error approach 19.
Selection of Excipients: Hard gelatin capsules are commonly filled with powder blends of active ingredients and excipients. Based on pre-formulation studies, sparingly soluble and hygroscopic plant extracts required suitable diluents such as mannitol or anhydrous lactose. Glidants (e.g., colloidal silica) were used to enhance flow, while lubricants like magnesium stearate were included as needed to reduce adhesion during filling 20.
TABLE 1: FORMULATION OF POLYHERBAL GRANULES BY WET GRANULATION METHOD
| Ingredients | PC 1 (mg) | PC 2 (mg) | PC 3 (mg) | PC 4 (mg) | PC 5 (mg) | PC 6 (mg) |
| Vinca rosea | 50 | 50 | 50 | 60 | 60 | 60 |
| Curcuma longa | 60 | 60 | 60 | 50 | 50 | 50 |
| Dalbergia sissoo | 20 | 25 | 30 | 20 | 25 | 30 |
| Lactose / Mannitol | 235 | 225 | 215 | 205 | 195 | 185 |
| Pregelatinised Starch | 0 | 10 | 20 | 30 | 40 | 50 |
| Talc | 20 | 20 | 20 | 20 | 20 | 20 |
| Sodium Benzoate | 5 | 5 | 5 | 5 | 5 | 5 |
Capsule Evaluation Parameters: The parameters like organoleptic characters, average weight, weight variation, disintegration time, moisture content and dissolution rate were determined in order to evaluate the capsules of polyherbal granules 21, 22.
Stability Study: To determine the durability of the pharmaceutical formulations, they must be exposed to augmented temperature, humidity and light intensities. Researchers examined the effects of extrinsic factors on the capsule's physical, chemical, and medicinal properties 23.
RESULTS AND DISCUSSION:
Extraction: The aqueous extracts of the selected medicinal plants Vinca rosea (leaves and stem), Curcuma longa (rhizomes), and Dalbergia sissoo (leaves) were successfully prepared, and their yields. The highest yield was obtained from Vinca rosea, with an average extract amount of 743.67 ± 10.01 mg, followed by Curcuma longa at 407.54 ± 13.43 mg, and Dalbergia sissoo yielding 312.67 ± 7.64 mg.
Pre-formulation Study: The pre-formulation parameters of the polyherbal capsules (PC 1–PC 6) were evaluated to assess the flow properties and compressibility of the powder blends before capsule filling. Bulk density ranged from 0.44 ± 0.001 g/ml (PC 1) to 0.68 ± 0.003 g/ml (PC 6), while tapped density values varied from 0.33 ± 0.003 g/ml (PC 5) to 0.67 ± 0.002 g/ml (PC 1 and PC 2). The Carr’s index (%), an indicator of powder compressibility, ranged from 10.56 ± 1.12% (PC 1) to 24.12 ± 2.42% (PC 6). According to general guidelines, Carr’s index values below 15% indicate good flow, 15–25% suggest fair to passable flow, and values above 25% indicate poor flow. Most formulations showed fair flow properties, with PC 1 and PC 2 exhibiting the best flow characteristics. The Hausner’s ratio, another measure of flowability, ranged from 1.16 ± 0.002 (PC 2) to 1.41 ± 0.001 (PC 6). A Hausner’s ratio below 1.25 indicates good flow, whereas values above 1.25 suggest poor flow. Based on these results, PC 1 and PC 2 demonstrated optimal flow, while PC 4, PC 5, and PC 6 exhibited relatively lower flow properties, likely due to increased cohesiveness from higher pregelatinised starch content. The angle of repose varied from 21.65 ± 2.065° (PC 1) to 37.62 ± 3.76° (PC 6). Angles of repose below 25° indicate excellent flow, 25–30° good flow, 30–40° fair to passable flow, and above 40° poor flow. PC 1 and PC 2 again showed excellent to good flow, whereas PC 4–PC 6 showed fair flow, consistent with Carr’s index and Hausner’s ratio findings.
TABLE 2: PRE-FORMULATION
| Parameters | PC 1 | PC 2 | PC 3 | PC 4 | PC 5 | PC 6 |
| Bulk density (g/ml) | 0.44±0.001 | 0.45±0.002 | 0.57±0.001 | 0.52±0.002 | 0.59±0.003 | 0.68±0.003 |
| Tapped density (g/ml) | 0.67±0.002 | 0.67±0.002 | 0.54±0.003 | 0.51±0.001 | 0.33±0.003 | 0.61±0.001 |
| Carr’s index (%) | 10.56±1.12 | 13.65±0.98 | 19.54±2.23 | 23.97±1.80 | 21.11±1.95 | 24.12±2.42 |
| Hausner’s ratio | 1.25±0.001 | 1.16±0.002 | 1.29±0.003 | 1.38±0.002 | 1.32±0.002 | 1.41±0.001 |
| Angle of repose (°) | 21.65±2.065 | 26.94±3.54 | 30.21±2.54 | 33.11±3.76 | 35.53±5.32 | 37.62±3.76 |
Overall, the pre-formulation studies suggest that formulations with lower pregelatinised starch content (PC 1 and PC 2) exhibit superior flow and compressibility, which are critical for uniform capsule filling. Formulations with higher starch content (PC 5 and PC 6) showed relatively reduced flow properties but remain within acceptable limits for capsule manufacturing. These results provide a strong foundation for the development of uniform, stable, and reproducible polyherbal capsules.
Evaluation Polyherbal Formulation: Organoleptic characteristics revealed that the capsules had a black-colored cap and a white body filled with brown-colored powder. The capsules were bitter in taste and exhibited a characteristic herbal odour, consistent with the presence of bitter bioactive constituents in the herbal extracts. The capsules were of 0 size, with an average weight of 423 g and a weight variation range of 415–432 g, which is within acceptable limits as per the Indian Pharmacopoeia (IP). This indicates uniformity in capsule filling and content, essential for dose accuracy and therapeutic efficacy.
TABLE 3: EVALUATION PARAMETERS OF POLYHERBAL CAPSULES
| Parameters | Observations |
| Organoleptic characters | Capsules with black cap and white body filled with brown powder |
| Size | 0 size |
| Taste | Bitter |
| Odour | Characteristic |
| Average weight (g) | 423 |
| Weight variation (g) | 415–432 |
| Disintegration time (min) | 9.65 |
| Moisture content (%) | 4.87 |
| Drug content | Within IP limits |
The disintegration time was 9.65 minutes, suggesting that the capsules disintegrate efficiently in aqueous media, facilitating the release and absorption of active constituents. Moisture content was 4.87%, indicating adequate protection from microbial growth and maintaining the stability of the herbal powder. Finally, the drug content was found to be within IP limits, confirming consistent incorporation of active herbal ingredients in the formulation.
Overall, the results demonstrate that the polyherbal capsules possess satisfactory organoleptic, physical, and physicochemical properties. The uniform weight, appropriate disintegration time, acceptable moisture content, and consistent drug content indicate that the formulation is suitable for oral administration and likely to deliver reproducible therapeutic effects.
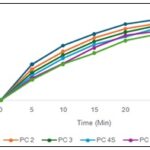
In-vitro Dissolution Study of Polyherbal Capsules: The in-vitro dissolution profiles of six polyherbal capsule formulations (PC 1–PC 6) were evaluated over a 30-minute period. The dissolution percentage increased progressively with time for all formulations, indicating a gradual release of the active constituents from the capsules.
At 5 minutes, PC 1 showed the highest release (39.645%), followed by PC 2 (34.490%) and PC 6 (22.418%), suggesting faster initial drug release from formulations with lower excipient content or higher solubility of the herbal extracts. By 15 minutes, PC 1 and PC 2 continued to show higher cumulative release (73.156% and 69.001%, respectively) compared to PC 5 and PC 6 (58.153% and 52.112%), indicating that formulations with higher proportions of pregelatinised starch or fillers had a slower release rate. At the end of 30 minutes, the cumulative drug release was highest for PC 1 (94.772%), followed by PC 2 (91.617%) and PC 3 (88.613%), whereas PC 6 showed the lowest release (76.083%). This trend indicates that the formulation composition, particularly the ratio of herbal extract to excipients like lactose, mannitol, and pregelatinised starch, significantly influences the dissolution rate. Formulations with lower filler content and optimized excipient ratios exhibited more efficient disintegration and release of active constituents.
FIG. 1: DISSOLUTION STUDY OF POLYHERBAL CAPSULES
Overall, the dissolution study demonstrated that all polyherbal capsule formulations released their contents progressively, with PC 1 showing the most rapid and complete drug release. These results suggest that PC1 is likely to provide better bioavailability and therapeutic efficacy. Formulation optimization focusing on excipient ratios can further improve dissolution characteristics for the remaining formulations.
Stability of Polyherbal Capsules at Different Temperatures: The polyherbal capsules were subjected to different storage temperatures to assess their thermal stability. At ambient temperature (30 °C) and warm conditions (35 °C), the capsules showed no signs of degradation even after 6 hours, indicating good stability under normal storage conditions.
Similarly, at accelerated testing at 50 °C, the capsules remained stable for the entire 6-hour duration. However, at higher temperatures, degradation was observed: at 55 °C, capsules began to degrade after 4 hours, and at 60 °C, degradation occurred as early as 2 hours. This indicates that the polyherbal capsule formulations are sensitive to high-temperature conditions, and their shelf-life may be compromised if exposed to excessive heat.
TABLE 4: STABILITY TEST OF POLYHERBAL CAPSULE AT DIFFERENT TEMPERATURE
| Storage Condition | Testing Temperature (°C) | Time Duration (h) | Results |
| Ambient | 30 | 0.5, 1, 3, 6 | No degradation in 6 h |
| Warm | 35 | 0.5, 1, 3, 6 | No degradation in 6 h |
| Accelerated | 50 | 0.5, 1, 3, 6 | No degradation in 6 h |
| Accelerated | 55 | 0.5, 1, 3, 6 | Degraded after 4 h |
| Accelerated | 60 | 0.5, 1, 3, 6 | Degraded after 2 h |
Overall, these results demonstrate that the capsules are thermally stable under normal and moderately elevated temperatures, but care should be taken to avoid exposure to temperatures above 50 °C. This information is critical for recommending appropriate storage conditions to maintain efficacy and shelf-life.
Effect of Temperature and Humidity on Polyherbal Capsule Stability: The polyherbal capsules were evaluated for stability under varying combinations of temperature (30–65 °C) and relative humidity (30–90%). At 30 °C, no degradation was observed at any humidity level (30–90%), indicating good stability under standard ambient conditions. At 35 °C, slight degradation (+) was observed at higher humidity levels (70% and 90%), while lower humidity (30–50%) showed no change. This indicates that increased moisture at moderately elevated temperatures begins to affect the integrity of the capsules. At 55 °C, degradation was more pronounced at high humidity: slight degradation (+) at 70% and moderate degradation (++) at 90%. Similarly, at 65 °C, the capsules exhibited moderate (++) degradation at 70% humidity and severe (+++) degradation at 90% humidity. These results demonstrate that both high temperature and high humidity accelerate degradation of the capsules, likely due to enhanced moisture uptake and chemical instability of the herbal constituents. Overall, the study highlights that polyherbal capsules are stable under ambient conditions, but elevated temperature and humidity adversely affect stability. For optimal shelf-life and efficacy, storage in cool, dry conditions is recommended.
TABLE 5: STABILITY STUDY AT DIFFERENT HUMIDITY WITH RESPECT TO DIFFERENT TEMPERATURE
| Temperature (°C) | 30% Humidity | 50% Humidity | 70% Humidity | 90% Humidity |
| 30 | – | – | – | – |
| 35 | – | – | + | + |
| 55 | – | – | + | ++ |
| 65 | – | – | ++ | +++ |
Note: (+) Degradation and (-) No change.
CONCLUSION: The aqueous extracts of Vinca rosea, Curcuma longa, and Dalbergia sissoo contain diverse bioactive phytochemicals, particularly alkaloids, flavonoids, terpenoids, and steroids, which contribute to their significant anticancer activity. The developed polyherbal capsules exhibited satisfactory preformulation, physicochemical, and organoleptic properties, with efficient disintegration and progressive drug release. Stability studies confirmed that the capsules are photostable and thermally stable under normal storage conditions but sensitive to extreme heat and high humidity. Overall, this study validates the scientific rationale for combining these three medicinal plants into a standardized polyherbal capsule, demonstrating potential for complementary and synergistic anticancer effects. Formulation PC 1, with optimal excipient ratios, emerged as the most promising candidate for further development and potential therapeutic application.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Backes E, Pereira C, Barros L, Prieto MA, Genena AK and Barreiro MF: Recovery of bioactive anthocyanin pigments from Ficus carica L. peel by heat, microwave, and ultrasound based extraction techniques. Food Res Int Ott Ont 2018; 113: 197–209. 10.1016/j.foodres.2018.07.016
- Barnes M, Sulé-Suso J, Millett J and Roach P: Fourier transform infrared spectroscopy as a non-destructive method for analysing herbarium specimens. Biol Lett 2023; 19: 20220546. 10.1098/rsbl.2022.0546
- Aruna MS, Prabha MS, Priya NS and Nadendla R: Catharanthus roseus: ornamnetal plant now medicinal Boutique. Journal of Drug Delivery and Therapeutics 2015; (3): 1-4.
- Allamsettty J, Pedada SP, Pedada N and Dhanunjayarao K: A basic review on vinca rosea. International Journal of Pharmacognosy and Chemistry 2020; (1): 30-36.
- Chen R, Peng X, Du W, Wu Y, Huang B and Xue L: Curcumin Attenuates Cardiomyocyte Hypertrophy Induced by High Glucose and Insulin via the PPARγ/Akt/NO Signaling Pathway. Diabetes Res Clin Pract 2015; 108(2): 235–242. 10.1016/j.diabres.2015.02.012
- Cheng AL, Hsu CH, Lin JK, Hsu MM, Ho YF and Shen TS: Phase I Clinical Trial of Curcumin, a Chemopreventive Agent, in Patients with High-Risk or Pre-malignant Lesions. Anticancer Res 2001; 21(4B): 2895–2900.
- Choi YH, Yan GH, Chai OH and Song CH: Inhibitory Effects of Curcumin on Passive Cutaneous Anaphylactoid Response and Compound 48/80-induced Mast Cell Activation. Anat. Cel Biol 2010; 43(1): 36–43. 10.5115/acb.2010.43.1.36
- Bijauliya RK, Jain SK and Alok S: Antilithiatic activity of ethanolic leaves extract of Dalbergia sissoo Linn. ethylene glycol induced lithiasis in rats. Int J Pharm Sci & Res 2018; 9(11): 4923-29. doi: 10.13040/IJPSR.0975-8232.9(11).4923-29.
- Bijauliya RK, Jain SK, Alok S, Dixit VK, Singh D and Singh M: Dalbergia sissoo Linn. An overview morphology, phytochemistry and pharmacology. Int J Pharm Sci Res 2017; 8(4): 1522-33.doi: 10.13040/IJPSR.0975-8232.8(4).1522-33.
- Woo SW, Hwang SJ and Cho CW: Liquid-filled hard capsule formulation of choline alfoscerate: preparation and in-vitro/in-vivo evaluation. Journal of Pharmaceutical Investigation 2023; 53(4): 517-26.
- da Fonseca LS, Silveira RP, Deboni AM, Benvenutti EV, Costa TM, Guterres SS and Pohlmann AR: Nanocapsule@ xerogel microparticles containing sodium diclofenac: a new strategy to control the release of drugs. International Journal of Pharmaceutics 2008; 358(1-2): 292-5.
- Jang ES, Nam Y, Lee YJ, Jang C and Lee SR: Phytochemicals with anticancer activity from poisonous plants: potential leads for cancer therapy. Phytochemistry Reviews 2025; 24(4): 3265-87.
- Rathnayake PC, Senavirathna DC, Milhan MM, De Vass Gunawaradane S, Seneviratne NN, Perera DN, Rajagopalan U, Senathilake KS and Samarakoon SR: In-vitro anti-cancer properties of a commercially available polyherbal nutraceutical (Vernolac) capsule on cancer stem cell-like (NTERA-2 cl. D1) cells. bioRxiv 2025; 2025-07.
- Karunamoorthy K and Vasudevan J: Anticancer potential of iron-based polyherbal formulation against lung and colon cancer cell lines. Journal of Ethnopharmacology 2025; 119999.
- Onengiyeofori Ibama, Aleruchi-Didia Tarila Ngowari, Nyebuchi Jonathan and Onengiye Davies-Nwalele: “Revive Herbal Capsule: A Review on the Mechanism of Action, Constituent Herbs and Phytochemicals”. Journal of Complementary and Alternative Medical Research 2021; 16(1): 17–33. https://doi.org/10.9734/jocamr/2021/v16i130278.
- Širola I, Lukšić J, Simunić B and Kujundžić N: Effect of Crystal Size and Shape on Bulk Density of Pharmaceutical Powders. Journal of Crystal Growth 1997; 181(4): 403–409. DOI: 10.1016/S0022-0248(97)00299-6.
- Abuzinadah MF and Ahmad A: Pharmacological studies on the efficacy of a thymoquinone‐containing novel polyherbal formulation against cisplatin‐induced hepatorenal toxicity in rats. Journal of Food Biochemistry 2020; 44(2): 13131.
- Selvakumar S and Sarkar B: In-vitro cytotoxicity analysis of chloroform extract of novel poly herbal formulation. Int J Pharmacogn Phytochem Res 2017; 9(2): 193-6.
- Al-Rimawi F, Rishmawi S, Ariqat SH, Khalid MF, Warad I and Salah Z: Anticancer activity, antioxidant activity, and phenolic and flavonoids content of wild Tragopogon porrifolius plant extracts. Evidence‐based Complementary and Alternative Medicine 2016; 2016(1): 9612490.
- Saravanan VS, Selvam P, Parthiban P, Bose PS and Raj SP: Anti cancer activity of polyherbal formulation. Int J Pharm Pharm Anal 2015; 2.
- Solowey E, Lichtenstein M, Sallon S, Paavilainen H, Solowey E and Lorberboum-Galski H: Evaluating medicinal plants for anticancer activity. The Scientific World Journal 2014; 2014(1): 721402.
- Yasmeen S and Gupta P: Cosmeceutical and anticancer potential of aqueous extracts of Dalbergia sissoo Roxb. aerial parts. Journal of Herbal Medicine 2021; 29: 100456.
- Widyananda MH, Puspitarini S, Rohim A, Khairunnisa FA, Jatmiko YD, Masruri M and Widodo N: Anticancer potential of turmeric (Curcuma longa) ethanol extract and prediction of its mechanism through the Akt1 pathway. F1000 Research 2022; 11: 1000.
How to cite this article:
Kant D, Trivedi MH and Sharma S: Formulation and assessment of herbal capsule using medicinal plant extracts. Int J Pharmacognosy 2026; 13(2): 119-25. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(2).119-25.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
119-125
520 KB
181
English
IJP
Deep Kant *, M. Himaja Trivedi and Satendra Sharma
Shri Ramnath Singh Institute of Pharmaceutical Science and Technology, Sitholi, Gwalior, Madhya Pradesh, India.
rkpharma3791@gmail.com
28 January 2026
25 February 2026
26 February 2026
10.13040/IJPSR.0975-8232.IJP.13(2).119-25
28 February 2026