EXTRACTION, PURIFICATION AND DETERMINATION OF PHYTO-COMPONENT OF FATTY ACIDS FROM IRVINGIA GABONENSIS SEEDS
HTML Full TextEXTRACTION, PURIFICATION, AND DETERMINATION OF PHYTO-COMPONENT OF FATTY ACIDS FROM IRVINGIA GABONENSIS SEEDS
A. O. Shittu * 1 and N. S. Njinga 2
Department of Pharmaceutics and Industrial Pharmacy 1, Department of Pharmaceutical and Medicinal Chemistry 2, Faculty of Pharmaceutical Sciences, University of Ilorin, Nigeria.
ABSTRACT: Objective: This study was designed to extract, purify and determine the phyto-components in the petroleum ether extract of Irvingia gabonensis seeds (IGS). A multiple batch extraction procedure was employed using petroleum ether as a solvent. A specified quantity of the IGS was size reduced, dispersed in freshly distilled water by heating to a temperature of 80 ºC. The dispersion was then filtered through a clean muslin cloth. The fats content was separated from the Biopolymer by first dispersing in 90% alcohol, filtered and dried at 40 ºC. The fat component of the residue was finally extracted with petroleum ether. The GC-MS analysis of the petroleum ether extract of IGS was performed using an Agilent G1701 GC/MSD system. This investigation was done to determine the possible chemical components from IGS by GC-MS. This analysis revealed that the petroleum ether extract of IGS contained mainly fatty acid compounds–lauric acid (36.82%), myristic acid (31.52%), benzyl dodecanoate (9.06%) and palmitic acid (4.03%), n-Decanoic acid, 1-(hydroxyl-methyl)- 1, 2- ethanediyl ester, tetradecanoic acid, 2- hydroxy-1, 3-propanediyl ester. The results from the present study show that IGS contains various bioactive compounds and is recommended as a plant of phytopharmaceutical importance.
| Keywords: |
Extraction, Purification, GC-MS, Phytochemistry, Fatty acids, Irvingia gabonensis seeds
INTRODUCTION: The Irvingia gabonensis seeds has been studied and said to have some industrial potential. Although the two species’ seeds do differ in their composition, the differences are not always found to be significant, and some studies fail to identify which species is being analyzed. This is particularly so for those that were undertaken before I. wombolu was recognized as a separate species, therefore in some cases figures given for I. gabonensis are actually for I. gabonensis var. excelsa (I. wombolu).
Onyeike and co-worker 1 report that the crude fat content of I. gabonensis seeds is 62.25% ± 0.55, proving them to be ‘very good oilseeds’. Amubode and Fetuga 2 analyzed the amino acids in I. gabonensis and came up with the following:
Crude Protein: Tryptophan, lysine, histidine, arginine, aspartic acid, threonine, serine, glutamic acid, proline, glycine, alanine, valine, methionine, cystine, isoleucine, tyrosine, phenylalanine. omogbai’s 3 study of the lipid and fatty acid composition of Nigerian tropical seeds included I. gabonensis.
Fruits: The juicy fruit pulp of I. gabonensisis rich in vitamin C and is widely reported to be consumed as a dessert fruit or snack throughout Western and Central Africa 3, 4.
Seeds: The kernels of I. gabonensis and I. wombolu are classed as oilseeds. They are ground with a pestle and mortar or on a stone into a paste or cake called ‘dika bread,’ which is used as a soup, stew or sauce additive, for flavoring and thickening 5, 6. The kernels are highly valued for the slimy consistency they produce. Okafor 7 notes that whilst kernels from both Irvingia spp. are used in soup making, I. gabonensis kernels can only be used when fresh since they become too slimy over time. Irvingia kernels form an important part of the West and Central African diet, providing carbohydrate and protein 1.
Fat extracted from the kernels can be used for food applications, such as in cooking oil or margarine, and is also suitable for pharmaceuticals, cosmetics, and soap 8. The potential industrial applications of bush mango kernel fat listed by Joseph 9, include cooking oil, margarine, perfume, soap, and pharmaceuticals. Aside from its role as a thickener, the residual kernel cake could also be used as a binder in food or pharmaceutical products 9. Ndjouenkeu and co-worker 10 extracted the polysaccharides from Irvingia kernels, and an analysis of their properties concluded that they have potential as an industrial gum.
MATERIALS AND METHODS:
Extractions of Irvingia gabonensis (Ogbono) Gum: One kilogram of Irvingia gabonensis seed was size reduced using an electric blender. The granules were then transferred into a dried clean beaker (1000 ml capacity). Water was added up to 1000 ml mark, and with the aid of the hot plate, it was heated until the temperature of 80 ºC was reached. The solution was constantly stirred and left to stand for 24 h for proper dispersion in distilled water. After 24 h, the dispersion was filtered through a clean muslin cloth, the filtrate obtained contains: biopolymer, fat, and water
About ten (10) liters of 95% ethanol was added to the filtrate until the mixture of biopolymer and fat crumbled in the ethanol indicating complete precipitation and total separation from water. The residue was collected, while the filtrate was discarded. The residue extracted was properly spread on a clean brown paper and air dried for 24 h for complete drying, it was dried in an oven at a temperature of 40 ºC for 20 min.
Defatting of the Gum: The extracted biopolymer contains fat. It was defatted by the use of petroleum ether. This was done by adding 100 ml of petroleum ether onto the 100 g of extracted biopolymer with stirring, after which granular sediments (gum) were filtered from the petroleum ether. Six batches of the defatting were done using 100 ml each until the biopolymer was free of fat (600 ml of petroleum ether was used). The biopolymer obtained was air dried for 24 h, after which it was dried in the oven at a temperature of 40 ºC for 30 min. The dried, purified biopolymer was then size reduced using a dry porcelain mortar and pestle. The weight of the dried granules obtained was 30 g, and the percentage yield was determined.
Recovering the Fat from Petroleum Ether: The several batches of the petroleum ether used for the purification of the biopolymer were collected together in a 1000 ml capacity beaker and left in the open air for 72 h, after which pet ether had evaporated leaving behind a semisolid mass which is fat.
Percentage yield (%) = [final weight / initial weight] × 100.
GC-MS Analysis: GC-MS analysis of the petroleum ether extract of IGS was performed using a Agilent G1701 GC/MSD system comprising an AOC-20i auto-sampler and a Gas Chromatograph interfaced to a Mass Spectrometer (GC-MS) equipped with a Elite-5MS (5% diphenyl / 95% dimethyl polysiloxane) fused a capillary column (30 × 0.25 μm ID × 0.25 μmdf). For GC-MS detection, an electron ionization system was operated in electron impact mode with ionization energy of 70 eV. Helium gas (99.999%) was used as a carrier gas at a constant flow rate of 1 ml/min, and an injection volume of 2 μl was employed (a split ratio of 10:1).
The injector temperature was maintained at 250 °C, the ion-source temperature was 200 °C, the oven temperature was programmed from 100 °C (isothermal for 2 min), with an increase of 4 °C / min to 300 °C, ending with a 10 min isothermal at 300 °C. Mass spectra were taken at 70 eV; a scan interval of 0.5 s and fragments from 45 to 450 Da. The solvent delay was 0 to 2 min, and the total GC/MS running time was 60 min.
The relative percentage amount of each component was calculated by comparing its average peak area to the total areas. The mass-detector used in this analysis was Turbo-Mass Gold-Perkin-Elmer, and the software adapted to handle mass spectra and chromatograms was Agilent G1701 GC/MSD ChemStation, and the library used was Agilent IO Libraries version 15.5.
Identification of Phytocomponents: Interpretation on mass-spectrum GC-MS was conducted using the database of Agilent IO Libraries version 15.5 National Institute Standard and Technology (NIST) having more than 62,000 patterns. The spectrum of the unknown components was compared with the spectrum of known components stored in the NIST library. The name, molecular weight, and structure of the components of the test materials were ascertained.
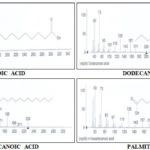
RESULTS AND DISCUSSION: GC chromatogram analysis of the petroleum ether extract of IGS Fig. 1 showed 12 peaks which indicates the presence of 12 phytochemical constituents Table 1. On comparison of the mass spectra of the constituents with the Agilent IO library, eight (8) phytocompounds were characterized and identified Fig. 2. The various phytochemicals which contribute to the medicinal activities of the plant were shown in Fig. 2. The mass spectra of all the phytochemicals identified in the petroleum ether extract of IGS were presented in Fig. 2. Of the 8 compounds identified, the most prevailing compounds were lauric acid (36.82%), myristic acid (31.52%), benzyl dodecanoate (9.06%) and palmitic acid (4.03%). Among these compounds, three compounds ‘lauric acid, myristic acid and palmitic acid’ were reported to have antimicrobial activity.
FIG. 1: MASS SPECTRUM OF PHYTOCOMPONENTS IDENTIFIED BY GC-MS IN THE PETROLEUM ETHER EXTRACTS OF IGS
TABLE 1: PHYTOCOMPONENTS AS IDENTIFIED IN THE IGS PETROLEUM ETHER EXTRACT BY GC-MS
| S. no. | RT | Name of compound | Molecular formula | MW | Peak area % |
| 1 | 5.76 | n-Decanoic acid | C10H20O2 | 172 | 2.09 |
| 2 | 11.19 | n-Dodecanoic acid (lauric acid) | C12H24O2 | 200 | 36.82 |
| 3 | 15.97 | n-Tetradecanoic acid (myristic acid) | C14H28O2 | 228 | 31.52 |
| 4 | 19.87 | n-hexadecanoic acid (palmitic acid) | C16H32O2 | 256 | 4.03 |
| 5 | 23.67 | benzyl dodecanoate | C19H30O2 | 290 | 9.06 |
| 6 | 27.95 | benzyl dodecanoate | C19H30O2 | 227 | 2.11 |
| 7 | 41.60 | Dodecanoic acid, 1-(hydroxymethyl)-1,2-ethanediyl ester | C27H52O5 | 456 | 1.75 |
| 8 | 41.94 | Dodecanoic acid, 1-(hydroxymethyl)-1,2-ethanediyl ester | C27H52O5 | 456 | 2.01 |
| 9 | 44.58 | Dodecanoic acid, 1-(hydroxymethyl)-1,2-ethanediyl ester | C27H52O5 | 456 | 2.24 |
| 10 | 44.93 | Tetradecanoic acid, 2-hydroxy-1,3-propanediyl ester | C31H60O5 | 512 | 5.28 |
| 11 | 47.66 | Tetradecanoic acid, 2-hydroxy-1,3-propanediyl ester | C31H60O5 | 512 | 1.96 |
| 12 | 52.26 | Dodecanoic acid, 1,2,3-propanetriyl ester | C39H74O5 | 638 | 1.13 |
The GC of the petroleum ether extract of IGS showed the presence of 12 compounds as shown in Fig. 1. Most of the phytocomponents found were fatty acid which include n-Decanoic acid, n-Dodecanoic acid (lauric acid), n-Tetradecanoic acid (myristic acid), n-hexadecanoic acid (palmitic acid), benzyl dodecanoate, Dodecanoic acid, 1-(hydroxymethyl)- 1, 2- ethanediyl ester, Tetra-decanoic acid, 2- hydroxy-1,3-propanediyl ester Fig. 2. Some of the fatty acids could not be resolved, but their mass spectrum indicated the presence of a benzyl dodecanoate, dodecanoic acid, 1-(hydroxyl-methyl)-1, 2-ethanediyl ester and tetradecanoic acid, 2-hydroxy-1, 3-propanediyl ester as shown in Fig. 3. The most abundant of the compounds include lauric acid followed by myristic acid Table 1. Phytocomponents identified in the petroleum ether extracts of IGS by GC-MS is shown in Fig. 2.
FIG. 2: MASS SPECTRUM AND STRUCTURE OF PHYTOCOMPONENTS IDENTIFIED BY GC-MS IN THE PETROLEUM ETHER EXTRACTS OF IGS
Lauric, palmitic and myristic acids are known to have potential antibacterial and antifungal properties 11, 12 thus suggesting the possible antibacterial and antifungal activity of this extract. Myristic acids have also been reported to have larvicidal and repellent activity 13 and palmitic acid reported to have anti-inflammatory, 14 antioxidant, hypocholesterolemic nematicide, pesticide, anti-androgenic flavor, hemolytic, 5-alpha-reductase inhibitor, 15 potent mosquito larvicide activities 16. Decanoic acid is a fatty acid can form salt or ester with a drug which will increase its affinity for fatty tissue and its lipophilicity. This thus makes this extract well information of prodrug and depot injection by using its decanoate form. The activities of these compounds are a function of the lipophilic properties of their constituent, the properties of their functional groups, and their aqueous solubility 17.
FIG. 3: MASS SPECTRUM AND STRUCTURE OF PHYTOCOMPONENTS IDENTIFIED BY GC-MS IN THE PETROLEUM ETHER EXTRACTS OF IGS
CONCLUSION: The various bioactive compounds presence in the petroleum ether extract of IGG justifies its use for various ailments by traditional practitioners. That notwithstanding, isolation of individual phytochemical constituents and subjecting it to the biological activity will definitely give fruitful results. From the results, it could be concluded that Irvingia gabonensis fats contain various bioactive compounds.
Therefore, it is recommended as a plant of phytopharmaceutical and medicinal importance. Future work shall be directed on isolation and designing a stable formulation for the bioactive compounds.
ACKNOWLEDGEMENT: The authors thank all the subjects involved in this study. Special thanks to the staff of Physical Chemistry, of the University of Ilorin for their support.
CONFLICT OF INTEREST: There is no conflict of interest. The entire article is the original copy of the outcome of the research carried out by my group.
REFERENCES:
- Onyeike EN, Olungwe T and Uwakwe AA: Effect of heat-treatment and defatting on the proximate composition of some Nigerian local soup thickeners. Food Chemistry 1995; 53(2): 173-175.
- Amubode FO and Fetuga BL: Amino acid composition of seeds of some lesser known tree crops. Food Chemistry 1984; 13(4): 299-307.
- Omogbai FE: Lipid composition of tropical seeds used in the Nigerian diet. Journal of the Science of Food and Agriculture 1990; 50(2): 253-255.
- Ejiofor MAN: Nutritional values of Ogbono (Irvingia gabonensis excelsa). ICRAF-IITA Conference on Irvingia gabonensis; Ibadan, Nigeria 1994.
- Vivien J and Faure JJ: Fruitiers Sauvagesd. Afrique 1996.
- Agbor LON: Marketing trends and potentials for Irvingia gabonensis products in Nigeria. ICRAF-IITA Conference on Irvingia gabonensis; Ibadan, Nigeria; 1994.
- Okafor JC: Varietal delimitation in Irvingia gabonensis (Irvingiaceae). Bulletin du Jardin Botanique National de Belgique 1975; 45(1-2): 211-221.
- Ejiofor MAN, Onwubuke SN and Okafor JC: Developing improved methods of processing and utilization of kernels of Irvingia gabonensis (var. gabonensis and var. excelsa). The International Tree Crops Journal 1987; 4: 283-290.
- Joseph JK: Physico-chemical attributes of wild mango (Irvingia gabonensis) seeds. Bioresource Technology 1995; 53(2): 179-181.
- Ndjouenkeu R, Goycoolea FM, Morris ER and Akingbala JO: Rheology of okra (Hibiscus esculentus) and dika nut (Irvingia gabonensis) polysaccharides. Carbohydrates Polymers 1996; 29: 263-269
- McGaw LJ, Jäger AK and Van Staden J: Isolation of antibacterial fatty acids from Schotia brachypetala. Fitoter 2002; 73: 431-433.
- Seidel V and Taylor PW: In-vitro activity of extracts and constituents of gonium against rapidly growing mycobacteria. Int J Antimicrob Agen 2004; 23: 613-619.
- Sivakumar R, Jebanesan A, Govindarajan M and Rajasekar P: Larvicidal and repellent activity of tetradecanoic acid against Aedesa egypti (Linn.) and Culex quinquefasciatus (Say.) (Diptera: Culicidae). Asian Pac J Trop Med 2011; 4: 706-710.
- Aparna V, Dileep KV, Mandal PK, Karthe P, Sadasivan C and Haridas M: Anti-inflammatory property of n-hexadecanoic acid: Structural evidence and kinetic assessment. Chem Biol Drug Des 2012; 80: 4340439.
- Kumar PP, Kumaravel S and Lalitha C: Screening of antioxidant activity, total phenolics and GC-MS study of Vitex negundo. Afr J Biochem Res 2010; 4: 191-195.
- Rahuman AA, Gopalakrishnan G, Ghouse BS, Arumugam, S and Himalayan B: Effect of Feronia limonia on mosquito larvae. Fitoterapia 2000; 71: 553-555.
- Mahato SB and Sen S: Advances in triterpenoid research, 1990-1994. Phytochemistry 1997; 44: 115-23.
How to cite this article:
Shittu AO and Njinga NS: Extraction, purification and determination of phyto-component of fatty acids from Irvingia gabonensis seeds. Int J Pharmacognosy 2018; 5(9): 590-95. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(9).590-95.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
590-595
660
1580
English
IJP
A. O. Shittu * and N. S. Njinga
Department of Pharmaceutics and Industrial Pharmacy, Faculty of Pharmaceutical Sciences, University of Ilorin, Nigeria.
neobiogate@yahoo.com
12 June 2018
04 July 2018
09 July 2018
10.13040/IJPSR.0975-8232.IJP.5(9).590-95
01 September 2018