EXTRACTION, NANOFORMULATION AND EVALUATION OF CURCUMIN
HTML Full TextEXTRACTION, NANOFORMULATION AND EVALUATION OF CURCUMIN
Sri Harsha Mainum * and P. Shashikala
University College of Technology, Osmania University, Hyderabad - 500007, Andhra Pradesh, India.
ABSTRACT: Nanosuspensions have the versatile potential for efficient exploitation of different drug delivery formulations and routes because of the properties provided by their small size. In the present study, curcumin extraction was carried out from Curcuma longa (crude curcuminoids) using soxhlet extraction method with ethanol as a solvent which gives a high yield. Formulations of nanosuspensions with the nanoprecipitation technique are made using polycaprolactone (PCL) as a polymer, with extracted natural curcumin, and with synthetic curcumin. A comparison is made for parameters such as product yield, drug content, drug entrapment efficiency and in-vitro studies between the formulation of nanosuspensions with naturally extracted curcumin and synthetic curcumin. Average particle size obtained ranging from 297.4 nm to 973.1 nm for different formulations with natural curcumin and 253.8 nm to 850.1 nm for different formulations with synthetic curcumin. Entrapment efficiency of nanosuspension ranged between 66.7% to 80.78% for natural curcumin and 68.78% to 80.86% for synthetic curcumin. Drug content ranged from 67.41% to 85.70% for formulation with natural drug and 68.27% to 83.27% for formulation with the synthetic drug. The zeta potential values show good stability and negative surface charge in the range of -72.95mv to -8.59mv. The prepared nanosuspension showed enhanced dissolution which may lead to enhanced oral bioavailability.
| Keywords: |
Antinociceptive, Spirulina platensis, Writhing test, Paw licking test
INTRODUCTION: Extraction, as the term used pharmaceutically, involves the separation of medicinally active portions of plant or animal tissues from the inactive or inert components. The techniques to extraction process are maceration, percolation, Soxhlet extraction, decoction, continuous countercurrent extraction, etc. Curcumin from rhizome Curcuma longa (raw turmeric) can be extracted using maceration, percolation, and Soxhlet extraction techniques 1.
Curcumin is an orange yellow crystalline powder with three colouring components in various portions which are all dicinnamoyimethane derivatives. (i) 1, 7- Bis-(4-hydroxy- 3-methoxy phenyl)-hepta-1, 6-diene- 3, 5 dione = diferuloyl methane (Chemical formula: C21H20O6, Formula weight: 368). (ii) 1- (4-hydroxyphenyl) -7 -(4-hydroxy 3- methoxyphenyl)-hepta-1, 6-diene-3, 5 dione = phydroxycinnamoyl ferulo methane (Chemical formula: C20H18O5, Formula weight: 338). (iii) 1, 7-Bis-(4-hydroxyphenyl) - hepta-1, 6-diene-3, 5dione=p, pdihydroxy dicinnamoyl methane (Chemical formula: C19H16O4, Formula weight: 308).
Nanopharmaceuticals are the pharmaceuticals which are designed by using nanotechnology techniques. The applications of nanopharmaceuticals range from smart material for tissue engineering tools to drug delivery systems, to the production of nanomaterials. Nanopharmaceuticals are the future of healthcare and have great promise. Nano-suspensions are one of the preferred dosage forms of oral bioavailability for poor water-soluble drugs and to increase the therapeutic performance of these drugs in any route of administration 2. Nanosuspension 3 is fine dispersion of uniform-sized solid particles in an aqueous vehicle.
A nanosuspension not only solves the problems of poor solubility and bioavailability but also alters the pharmacokinetics of drug and thus improves drug safety and efficacy. The techniques to prepare nanosuspension 4 are supercritical fluid technique, melt emulsification technique, solvent evaporation technique, etc. These can be used to enhance the solubility of drugs that are poorly soluble in water as well as lipid media.
As a result of increased solubility, the rate of flooding of the active compound increases and the maximum plasma level is reached faster. This approach is useful for molecules with poor solubility, poor permeability, or both, which poses a significant challenge for the formulators. The reduced particle size renders the possibility of intravenous administration of poorly soluble drugs without any blockade of the blood capillaries. Advantages of nanosuspension are suitable for hydrophilic drugs, higher drug loading can be achieved, and dose reduction is possible. Nanosuspension increases dissolution rate and absorption of the drug due to smaller particle size and larger surface area. For large-scale production of nanosuspension, media milling and high-pressure homogenization technology have been successfully used.
MATERIALS AND METHODS:
Materials: Model drug synthetic curcumin from Sigma Labourites, Hyderabad. Polymer poly-caprolactone and polyvinyl alcohol from Sigma Aldrich, Bangalore. Sodium tripolyphosphate from finer chemicals, Hyderabad. Ethanol from srinivasa scientific, Hyderabad. Hexane, acetone, dichloro-methane from qualigans fine chemicals, Mumbai.
Methods:
Extraction of Curcumin: 5, 6 The Soxhlet extractor is filled with 150 gm of turmeric powder, and the round bottom flask is filled with 175 ml of ethyl alcohol. The heating mantel and temperature start the extraction is maintained at the set point of 60°C. The extraction is continued for 3 to 4 h until the solvent which fills siphon cycle unit is filled with almost colorless solvent.
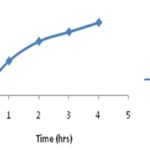
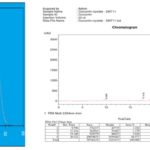
Then, remove the round bottom flask from Soxhlet extractor and transfer it to a beaker. Concentrated the extract with continuous stirring at room temperature, until ethyl alcohol is evaporated off from the extract. Then, 50 ml of hexane is added to the concentrated extract and stir the solution using a magnetic stirrer at 500 rpm. Water is added slowly to the solution until curcumin precipitate is observed. Filter the precipitate of curcumin using filtration and recrystallize from ethanol. The obtained precipitate is dried, and characterization is done by using UV-spectrophotometer and HPLC 7, 8. The extraction results are shown in Table 1.
The increasing range of extraction of curcumin and characterization results of UV-spectrophotometer and HPLC are showed in Fig. 1 and 2.
TABLE 1: OBSERVED RESULTS FOR VALIDATING THE EXTRACTION PROCEDURE
| S.
no. |
Time for extraction | Raw Turmeric
powder (gm) |
Solvent vol.(ml) | Product
yield (gm) |
%
Yield |
%
Extracted |
| 1 | 1 h | 50 | 150 | 1.2 | 2.4% | 41.49% |
| 2 | 2 h | 50 | 150 | 1.9 | 3.8% | 64.83% |
| 3 | 3 h | 50 | 150 | 3.25 | 6.5% | 76.11% |
| 4 | 4 h | 50 | 150 | 4.13 | 8.6% | 87.3% |
FIG. 1: RESULTS OF EXTRACTION OF CURCUMIN
FIG. 2: UV-VISIBLE WAVELENGTH AND HPLC RESULTS OF CURCUMIN AT MAXIMUM (λ max) ABSORBANCE
Preparation of Nanosuspension Using Nano-precipitation Technique: Nanosuspension of curcumin was prepared by using curcumin drug and different polymer concentration (0.8%, 1.6%, 3.2% & 4.8%) by nanoprecipitation technique as shown in. Required quantities of drug, polymer, cross Table 2 linking agent and stabilizer are added under continuous stirring for 8h and at last, water is added as anti-solvent agent. The prepared nano-suspension is placed in an airtight container and stored in a cool place.
TABLE 2: FORMULATIONS WITH CURCUMIN DRUG AND ITS PRODUCT YIELD
| S.
no. |
Polymer
conc. |
Synthetic curcumin conc. | PVA
conc. |
STPP
conc. |
Stirring
Speed |
Time of
Stirring |
Product yield on a
natural drug |
Product yield of synthetic drug |
| 1 | 0.8% w/v | 100mg | 4% w/v | 1% w/v | 1200 rpm | 8 h | 69.5% | 72.9% |
| 2 | 1.6% w/v | 100mg | 4% w/v | 1% w/v | 1200 rpm | 8 h | 76.3% | 82.2% |
| 3 | 3.2% w/v | 100mg | 4% w/v | 1% w/v | 1200 rpm | 8 h | 78.4% | 83.3% |
| 4 | 4.8% w/v | 100mg | 4% w/v | 1% w/v | 1200 rpm | 8 h | 83.3% | 86.1% |
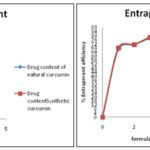
Estimation of Drug Content and Entrapment Efficiency: 100mg of Drug-loaded nanosuspension is taken and dissolved in 100 ml of suitable buffer solution of pH 7.4. Now, these samples are analyzed against a blank spectrophotometrically at 421 nm using Shimadzu UV spectrophotometer. Dilute the stock solution until the minimum absorbance reaches to 0.899Abs or below. Then the last addition of solvent is taken as a dilution factor. This procedure is repeated for the evaluation of entrapment efficiency with the stock solution samples are centrifuged in a centrifuge tube for 1 h at room temperature and 5000 rpm. The calculations of drug content and entrapment efficiency values are shown in Table 3.
In-vitro Release Studies: 9 10 In-vitro drug release was carried out using dissolution test apparatus USP XXII using a dialysis membrane to hold nanosuspension. The dissolution consisted of phosphate buffer (pH 7.4) for 9 h. An amount of 900 ml of the dissolution fluids was used at 37 ± 1 °C with a stirring speed 70 ± 2 rpm. Aliquots of 5 ml were withdrawn at predetermined time intervals and an equivalent amount of free dissolution media maintained at the same temperature was replaced.
TABLE 3: DRUG CONTENT AND ENTRAPMENT EFFICIENCY FOR FORMULATIONS OF NANOSUSPENSION WITH CURCUMIN
| Sample name | Samples | % Drug content | % Entrapment efficiency | ||
| Formulation with natural curcumin | Formulation with synthetic curcumin | Formulation with natural curcumin | Formulation with synthetic curcumin | ||
| Cur-S1 | 0.8% PCL | 67.41% | 68.27% | 66.75% | 67.75% |
| Cur-S2 | 1.6% PCL | 70.34% | 71.72% | 71.25% | 70.89% |
| Cur-S3 | 3.2% PCL | 80.10% | 81.10% | 77.57% | 78.06% |
| Cur-S4 | 4.8% PCL | 85.75% | 83.75% | 80.78% | 80.86% |
FIG 3: COMPARISON OF DRUG CONTENT AND ENTRAPMENT EFFICIENCY OF NANOSUSPENSION BETWEEN SYNTHETIC DRUG AND NATURAL DRUG FORMULATIONS
The samples were analyzed by measuring the absorbance at 421 nm by UV spectrophotometer. The drug release profile is shown in Fig. 4.
RESULTS AND DISCUSSION: The present investigation was undertaken to formulate and evaluate the nanosuspension with natural drug and synthetic drug for sustained release dosage form. Extraction studies indicated that 8.6% of curcumin were extracted, which normally has 6-9% in a natural Curcuma longa. The entrapment efficiency indicates good compatibility between drug, polymer, and stabilizer. The results of drug content, entrapment efficiency are shown in Table 3.
The results drug content was between 67 ± 0.5% to 83 ± 0.5%, and the results of entrapment efficiency were between 66 ± 0.5% to 80 ± 0.5% which indicates the good to excellent drug content and entrapment between polymer and drug. The selected formulations based on near entrapment efficiency between natural curcumin and synthetic curcumin were subjected to in-vitro studies and subjected various evaluation parameters. The results obtained for in-vitro studies were shown in Table 4, and kinetic models evaluation parameters are shown in Table 5.
Upon model fitting analysis of nanosuspension, Korsmeyer Peppa's model is with ‘n’ values 0.831 to 0.880 and average correlation coefficient ‘R2’ 0.970 to 0.980. The obtained in-vitro results of selected formulations are evaluated with various kinetics models like zero-order kinetics, first-order kinetics, Higuchi’s model and Korsmeyer Peppa's model Fig. 5 to 6 11, 12.
TABLE 4: DRUG RELEASE PROFILE OF FORMULATED NANOSUSPENSION
| Time (h) | % Drug Release of Cur-N2 | % Drug Release of
Cur-S2 |
% Drug Release of
Cur-N3 |
%Drug Release of
Cur-S3 |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 5.1 | 5.7 | 5.2 | 5.7 |
| 2 | 7.4 | 9.6 | 11.7 | 11.3 |
| 3 | 12.23 | 16.41 | 22.3 | 18.4 |
| 4 | 21.59 | 23.6 | 27.9 | 26.5 |
| 5 | 37.23 | 38.4 | 42.9 | 34.6 |
| 6 | 57.5 | 51.2 | 59.4 | 49.04 |
| 7 | 68.79 | 68.1 | 68.8 | 64.8 |
| 8 | 78.6 | 73.3 | 77.3 | 81.3 |
| 9 | 80.4 | 81.1 | 80.7 | 83.4 |
TABLE 5: KINETIC DATA FOR IN-VITRO STUDIES
| Model and Parameter | CUR
N2 |
CUR
N3 |
Average
of R2 |
CUR
S2 |
CUR
S3 |
Average
of R2 |
Model and Parameter |
| Zero-order kinetics | R2 | 0.952 | 0.981 | 0.966 | 0.971 | 0.967 | 0.969 |
| K0 (h-1) | 10.28 | 9.956 | 9.786 | 9.983 | |||
| First-order kinetics | R2 | 0.930 | 0.958 | 0.944 | 0.937 | 0.892 | 0.914 |
| K1(h-1) | -0.096 | -0.092 | -0.090 | -0.098 | |||
| Higuchi kinetics | R2 | 0.796 | 0.861 | 0.828 | 0.824 | 0.816 | 0.82 |
| KH (h-1/2) | 30.73 | 30.47 | 29.46 | 29.97 | |||
| Korsmeyer Peppas kinetics | R2 | 0.951 | 0.993 | 0.972 | 0.977 | 0.988 | 0.982 |
| n | 0.843 | 0.831 | 0.831 | 0.879 |
FIG. 4: DRUG RELEASE PROFILE FOR DIFFERENT SAMPLES
FIG. 5: FIRST ORDER KINETICS AND HIGUCHI’S MODEL FOR SELECTED FORMULATIONS
FIG. 6: KORSMEYER PEPPAL MODEL FOR SELECTED FORMULATIONS
This indicates that the release of curcumin loaded polycaprolactone nanosuspension follow zero order kinetics with sustained release pattern.
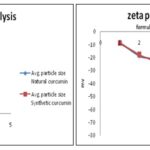
The results particle size distribution according to the Beckman colter nanosize analyzer 13 showed 297 ± 0.5 nm to 973 ± 0.5 nm which are in the range of 1 nm to 1000 nm Table 6. The charge of the particle was determined by zeta potential analyzer as shown in Table 6.
Comparison of all evaluation studies of particle size analysis and zeta potential analysis are shown in Fig. 7.
TABLE 6: AVERAGE PARTICLE SIZE FOR NANOSUSPENSIONS FORMULATIONS
| Sample
name |
Average Particle Size | Zeta Potental | ||
| Natural Drug,
nm |
Synthetic Drug,
nm |
Natural Curcumin, mv | Synthetic curcumin, mv | |
| 0.8% w/v | 297.4 | 253.8 | -8.41 | -9.19 |
| 1.6% w/v | 410.6 | 413.2 | -18.95 | -17.96 |
| 3.2% w/v | 668.5 | 667.5 | -26.42 | -28.36 |
| 4.8% w/v | 973.1 | 850.1 | -70.59 | -72.59 |
FIG. 7: RESULTS OF AVERAGE PARTICLE SIZE AND ZETA POTENTIAL ANALYSIS FOR NANO-SUSPENSIONS FORMULATIONS
CONCLUSION: Extraction, formulation, and evaluation of nanosuspension containing curcumin were found to be potential, cost-effective and satisfactory in-vitro release studies. It may enable to release the drug in a sustained pattern for a prolonged time and thereby to accompany some of the benefits like target delivery of the drug, reduction of dose-related side effects and better patient compliance.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Dean JR: Extraction techniques in analytical sciences. Extraction techniques of medicinal plants 1: 228-235
- Stanford MD and Gale BK: Nanotechnology and its applications July 2005.
- Prabhakar CH: A review in nanosuspensions in drug delivery, 2011; 2.
- Nagavarma BVN: Different techniques for preparation of polymeric nanoparticles- a review. 2007; 5(3).
- Rydberg J and Cox’s M: Solvent extraction principles and practices, Edition 2nd, 286-290.
- Rydberg J and Cox’s M: Solvent extraction principles and practices, Edition 2nd, 286-290.
- Braga MEM: Accelerated solvent extraction and Fractioned extraction to obtain the Curcuma longa volatile oil and oleoresin. 2007.
- Sharma K: Development and Validation of UV spectrophotometric method for the estimation of curcumin in bulk drug and pharmaceutical dosage forms. 2012; 4: 9344.
- Kumar KS and Gnanaprakash D: Chitosan-gold nano-particles as delivery systems for curcumin 2012; 3.
- Mohite JM: Novel approaches in development and in-vitro evaluation of mucoadhesive buccal tablet of curcumin. 2012; 4.
- Jithan A: Enhanced liver delivery and sustained release of curcumin with drug-loaded nanoparticles after intravenous administration in rats. Research article Published by, 1998.
How to cite this article:
Mainum SH and Shashikala P: Extraction, nanoformulation and evaluation of curcumin. Int J Pharmacognosy 2014; 1(8): 520-27. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(8).520-27.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
520-527
852
2373
English
IJP
S. H. Mainum * and P. Shashikala
University College of Ttechnology, Osmania University, Hyderabad, Andhra Pradesh, India.
sriharsha.mainum@gmail.com
12 March 2014
13 June 2014
28 July 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(8).520-27
01 August 2014