EXPLORING AYURVEDA AND TRADITIONAL MEDICINES: EFFICACY OF POLYHERBAL ANTIDIABETIC TABLETS AND THEIR ANTIOXIDANT PROPERTIES
HTML Full TextEXPLORING AYURVEDA AND TRADITIONAL MEDICINES: EFFICACY OF POLYHERBAL ANTIDIABETIC TABLETS AND THEIR ANTIOXIDANT PROPERTIES
Shiom Ramesh Mane *, Chinmay Shivaji Patil, Priti Rajagonda Patil, Snehal Sanjay Patil, Pranali Piraji Bhosale, Poonam Nilesh Chougule and Sachinkumar Patil
Ashokrao Mane College of Pharmacy, Peth, Vadgaon, Maharashtra, India.
ABSTRACT: Background: Diabetes mellitus, or simply diabetes, is a set of endocrine ailments characterized by persistently elevated blood sugar levels. Diabetes is caused by either the pancreas not making enough insulin or the body's cells becoming resistant to the hormone's effects. Thirst, polyuria, weight loss, and impaired eyesight are common indications of Diabetes. Combining herbal therapy seems to be beneficial in the treatment of type 2 diabetes and related issues. Aim: The goal of the current research was to formulate a polyherbal anti-diabetic formulation using Withania coagulans, Emblica officinalis, and Trigonella foenum-graecum. Method: Material like pulverized powder of Trigonella foenum-graecum Linn, Withania coagulans Dunal, and Emblica officinalis were used in the present study for phytochemical screening, tablet punching, and in-vitro study along with the flow properties of Polyherbal granules. Polyherbal tablets were used in the in-vitro antioxidant DPPH experiment and the in-vitro anti-diabetic effect was examined using alpha-amylase enzyme inhibitory activity. Result: The flow properties of the developed Polyherbal granules were remarkable. In an in-vitro investigation, polyherbal tablets were examined for anti-diabetic effectiveness. The polyherbal formulation was high in polyphenols and flavonoids. When compared to acarbose, it likewise displayed great antioxidant properties as well as significant -glucosidase and -amylase activities. The Polyherbal Granules had a peak at 278 nm, indicating the presence of flavonoids and phenols, according to the UV-Vis spectra. More research using an in-vivo model is needed, however, to examine the role of free radicals and anti-diabetic effects in the treatment of type 2 diabetes.
Keywords: Traditional medicine, Ayurveda, Polyherbal tablet, Paneer phool, Amla powder, Fenugreek seeds
INTRODUCTION: Diabetes mellitus, a complex and pervasive metabolic disorder, remains a formidable global health challenge. This chronic condition, characterized by abnormal blood glucose levels, affects millions of individuals worldwide, with its prevalence steadily rising. As of 2022, it was estimated that over 400 million people were living with diabetes, and this number is expected to increase in the coming years.
The multifaceted nature of diabetes, its associated complications, and the immense burden it places on healthcare systems make it a subject of continuous and critical research 1. Pharmacognosy has become one of the pillars of fields such as pharmacy, medicine, natural product chemistry, and many others, allowing them to identify the relevance of plants as sources of medicine.
Traditional medicine is still widely used in developing nations, while complementary and alternative medicine use is fast expanding in developed countries 2. However, the knowledge system of Ayurveda differs greatly from that of biomedicine. Ayurveda never loses sight of the whole, whereas biomedicines till focuses exclusively on individual system components 3. The study of diabetes encompasses a wide spectrum of disciplines, from clinical medicine and pharmacology to epidemiology, genetics, and public health. Researchers, healthcare professionals, and policymakers worldwide are engaged in a concerted effort to better understand the intricacies of diabetes, develop innovative treatment modalities, and implement effective prevention strategies 4. The unmatched chemical variation of natural materials, whether as standardized plant extracts or as pure chemicals, provides numerous chances for novel therapeutic discoveries. A specific ratio of various plants (polyherbal) results in a desired medicinal effect since the highly efficient phytochemical components of individual plants are inadequate to provide the desired result 5, 6. The use of natural ingredients, such as Withania coagulans (Withania coagulans), Fenugreek seeds (Trigonella foenum-graecum), and amla fruit (Emblica officinalis), have been acknowledged for their potential health benefits.
Pharmacognosy & Phytochemistry of Withania coagulans: Withania coagulans known as Paneer Phool or Indian Rennet, is a plant native to arid regions of South Asia, including India and Pakistan. It belongs to the Solanaceae family and is renowned for its various medicinal properties. In recent years, this plant has garnered significant attention for its potential role in the management of diabetes, among other health benefits 7.
Traditional medicine systems, such as Ayurveda and Unani, have long recognized the therapeutic properties of Withania coagulans. In these systems, it is often used to address a wide range of health concerns, including diabetes 8. Its traditional uses and contemporary research suggest that it may hold promise as a natural and alternative approach to diabetes management 9.
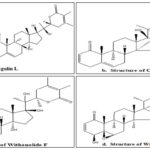
The potential antidiabetic effects of Withania coagulans are attributed to its bioactive compounds, which include Coagulin L 10, 11, Coagulin C 10, 12, Withanolide F 10, Withaferin A 13, alkaloids, flavonoids, and polyphenols. These compounds are believed to play a role in improving insulin sensitivity, regulating blood glucose levels, and reducing complications associated with diabetes. As diabetes continues to be a global health concern, there is a growing interest in exploring natural remedies like Withania coagulans as potential complementary or alternative treatments 14, 15.
FIG. 1: STRUCTURE OF PHYTOCONSTITUENTS OF WITHANIA COAGULANS
Pharmacognosy & Phytochemistry of Trigonella foenum-graecum Linn: Trigonella foenum-graecum Linn, commonly known as fenugreek, is an annual herb belonging to the Fabaceae family. It has a long history of use as both a culinary spice and a traditional medicinal herb. Fenugreek seeds are well-regarded for their distinctive aromatic flavour and are often employed in various culinary dishes in regions such as India, the Middle East, and North Africa. Beyond its culinary applications, fenugreek has gained recognition for its potential as an antidiabetic agent 16. The use of fenugreek in traditional medicine systems, including Ayurveda, has persisted for centuries. In these systems, fenugreek seeds have been recommended for their various health benefits, particularly in the management of diabetes. The growing global prevalence of diabetes and the associated demand for alternative and complementary therapies have sparked scientific interest in fenugreek's potential as a natural antidiabetic agent 17. The antidiabetic properties of fenugreek are primarily attributed to its bioactive constituents, including alkaloids, flavonoids, and a group of unique soluble dietary fibres known as galactomannans. These compounds are believed to have a positive impact on blood glucose regulation and insulin sensitivity. As diabetes remains a significant public health challenge worldwide, there is a continuous exploration of natural remedies like fenugreek as potential allies in the ongoing battle against this metabolic disorder 18.
FIG. 2: STRUCTURE OF PHYTOCONSTITUENTS OF TRIGONELLA FOENUM-GRAECUM LINN
Pharmacognosy & Phytochemistry of Emblica officinalis: Amla fruit, scientifically known as Emblica officinalis and commonly referred to as Indian gooseberry, is a highly revered and versatile fruit in traditional Indian and Ayurvedic medicine. Amla is renowned for its numerous health-promoting properties and has been used for centuries in various medicinal formulations. In recent years, it has garnered attention as a potential antidiabetic agent due to its rich phytochemical composition and multifaceted health benefits 19. Amla has a significant presence in traditional Indian medicine systems, where it is considered an essential component in various remedies and formulations. Its traditional use encompasses a wide range of health concerns, including diabetes. The therapeutic potential of Amla fruit as an antidiabetic agent has sparked scientific interest, offering a natural and holistic approach to diabetes management 20.
The multifaceted properties of Amla fruit can be attributed to its unique phytochemical profile, which includes a high concentration of ascorbic acid (vitamin C), gallic acid, ellagic acid, tannins, and various polyphenolic compounds. These constituents have demonstrated antioxidant, anti-inflammatory, and antihyperglycemic effects, making Amla a promising candidate for supporting individuals with diabetes. In the face of the global diabetes epidemic and the growing interest in natural and alternative remedies, Amla fruit stands out as a potential ally in the management of diabetes and its associated complications 21, 22.
FIG. 3: STRUCTURE OF PHYTOCONSTITUENTS OF EMBLICA OFFICINALIS
FIG. 4: GEOGRAPHICAL LOCATIONS OF POLYHERBAL DRUGS
MATERIALS AND METHODOLOGY:
Plant Material Collection and Plan of Study: Materials Like pulverized powder of Fenugreek seeds, flowers of Paneer Phool, and dried powder of Amla were used inthe present study which were purchased from Aditya Herbal (Bavdekar Ayurvedic Medical) near Kolhapur Municipal Corporation, Kolhapur. Firstly, the phytochemical screening of crude drug was done then a polyherbal mixture was formed and the flow properties of the mixture were studied.
Then formulation table was formed and executed into 5 batches of different excipients and their concentration. Later, the polyherbal tablets were punched and an evaluation study of the tablets was performed. Lastly, the tablets were sent to the Biocyte Research Laboratory for in-vitro study of Anti-diabetics and Anti-microbial Activity.
Chemicals and Reagents: The study used sodium nitroprusside, potassium tartrate, potassium ferricyanide, ascorbic acid, acetic acid, ammonium molybdate, ferric chloride (FeCl3), lactose monohydrate, methylparaben, methanol, 4-nitrophenyl β-D-glucopyranoside, propylparaben, potassium bromide, potassium ferricyanide, phosphate buffer, starch, sulfuric acid, sodium nitroprusside, sodium potassium tartrate, sodium phosphate, sodium hydroxide (NaOH), sodium carbonate (Na2CO3), and tri-chloroacetic acid were utilized in the work. All these substances were issued from the Pharmaceutics Laboratory of Ashokrao Mane College of Pharmacy.
TABLE 1: COMPOSITION OF POLYHERBAL TABLET
| Sr. no. | Name of Content | Quantity (in mg) | ||||
| F1 | F2 | F3 | F4 (Mannitol) | F5 (Gelatin) | ||
| 1. | Fenugreek | 80 | 95 | 90 | 95 | 95 |
| 2. | Panir phool | 80 | 95 | 90 | 95 | 95 |
| 3. | Amla | 80 | 70 | 75 | 70 | 70 |
| 4. | Lactose | 150 | 144.5 | 140 | 144.5 | 144.5 |
| 5. | Talc | 05 | 5.5 | 05 | 5.5 | 5.5 |
| 6. | Starch | 75 | 70.5 | 75 | Q.S. | Q.S. |
| 7. | Mannitol | 00 | 00 | 00 | 70.5 | 00 |
| 8. | Gelatin | 00 | 00 | 00 | 00 | 70.5 |
| 9. | Mg Stearate | 8 | 08 | 08 | 08 | 08 |
| 10. | Liquorice | 20 | 10 | 15 | 10 | 10 |
| 11. | Methyl Paraben | 1.5 | 1.5 | 02 | 1.5 | 1.5 |
| 12. | Peppermint Oil | Q.S. | Q.S. | Q.S. | Q.S. | Q.S. |
| 13. | Total | 500 | 500 | 500 | 500 | 500 |
Preparation of Polyherbal Granules: Prior to use, all powder fractions (Phyllanthus emblica, Withania coagulans, and Trigonella foenum-graecum Linn.) and excipients were sieved through sieve number 80. Individual material dosages were accurately weighed out in an electronic balance before being mixed with the diluent lactose to produce a dry powder that was then passed through filter number 44. The required quantities of starch and Liquorice were added to the previously mentioned combination. Methylparaben and Peppermint Oil were mixed to make a starch paste. The wet coherent mass was formed, sieved using sieve 14, and dried in a hot air oven at 80 degrees Celsius for 15 minutes before being sieved again with sieve number 22. Finally, pure talc and magnesium stearate were utilized to lubricate the grains. Five trial batches of tablets have been formulated through the various compositions of excipients proportions for excellent belongings. The mixed powder of all 5 trial batches has been subjected to the following evaluation standards.
Preformulation Study of Granules: A conventional technique was used to assess the flow properties of Polyherbal Granules 15, and the best trial batch was chosen for pre-formulation investigations and tablet punching.
Bulk Density (δB): It is calculated by dividing the weight of powder or grains by the volume of the powder or grains. Bulk density is used to calculate container sizes, production equipment, packaging materials, and empty gelatin capsules, as well as the consistency of bulk powdered products. The bulk density of a powder changes depending on particle packing and powder concentration.
δB = Mass/ Bulk Volume
Tapped Density (δT): The tapped density is an increased bulk density obtained by mechanically tapping a container containing a powder sample. Mechanically tapping a graduated measuring cylinder or vessel containing the powder sample yields the tapped density.
δT = Mass / Tapped Volume
Compressibility Index (δC): Since, all the factors might affect the measured compressibility index, the compressibility index has been proposed as an indirect measure of bulk density, size and shape, surface area, moisture content, and cohesiveness of materials.
δC = δT – δB / δT
Hausner's Ratio (δH): It also demonstrates densification of the herbal powder combination, which may be a result of feed hopper vibration, and can be computed using the formula listed below:
δH = δT / δB
UV-visible Investigation of Polyherbal Granules and Dried Seed Extracts: The UV/Vis analyses of all dried seed extracts and PHGs were carried out using a UV/Vis spectrophotometer, as previously described 23. Individual extracts and PHG solutions were made by dissolving 50 mg in 100 mL of distilled water individually.
The resulting solution was sonicated and filtered using the first Whatman filter. The samples were scanned in the UV/Vis region at 200-800 nm wavelengths against distilled water as a blank. Characteristic peaks have been recorded.
In-vitro Antidiabetic Activities:
Alpha Amylase Inhibition: The Patil SB [2018] technique was used to study in vitro amylase inhibition. In the simplest terms, 500 μL of the test sample -NS was combined with 500 μL of 0.1M phosphate buffer (pH 6.9) with 0.5% α -amylase enzyme (fungal diastage) and allowed to react. 500 μL of 1% starch soluble, extra pure (Loba Chemie) in 0.1M phosphate buffer pH6.8 was added after a 10 minute incubation at 25° C. Once more incubated for 10 minutes at 250° C. The identical procedure was used for the controls, except that instead of the enzyme, 500 μL of buffer was used. 1000 μL of DNS (Loba Chemie) was introduced to both the test and the control, post-incubation. Standard acarbose (α-amylase enzyme inhibitor) is used as a standard drug. They spent 10 minutes in a bath of boiling water before being cooled. Using a spectrophotometer, the absorbance at 540 nm was measured. The percentage of the enzyme -amylase that was inhibited was estimated 24.
Determination of Anti-oxidant Activity: Antioxidant activity in the sample compounds was estimated for their free radical scavenging activity by using DPPH (1, 1-Diphenyl-2, Picryl-Hydrazyl) free radicals (George et al., 1996). 100μL of test compounds water was taken in the microtiter plate.
100μL of 0.1% methanolic DPPH was added over the samples at different concentrations (1000μg/ml) and incubated for 30 minutes in dark conditions. The samples were then observed for discoloration; from purple to yellow and pale pink were considered as strong and weak positive respectively and read the plate on Elisa plate reader at 490nm 25.
Tablets' Physical Evaluation: The following parameters were used to evaluate tablets:
Colour and Appearance: The colour of the tablets was evaluated visually while odour and appearance were evaluated sensually.
pH Test: By using pH paper at room temperature, the produced herbal tablet's pH in distilled water was determined.
Weight Variation Test: The average weight of 20 tablets was obtained for each variation. Each tablet's weight was tested individually. The difference from the average weight was determined and given as a percentage in each case. Not more than two tablets in the sample size depart from the average weight by a bigger percentage, and none deviate by more than double that percentage.
Hardness Test: A calibrated Monsanto hardness tester was used to measure the hardness of the tablets.
Friability Test (δF): This test is used to determine the combined impact of abrasion and stock The Roche Friabilator was employed for this test. Pre-weighed tablets (δF0) were placed in the friabilator and rotated at a speed of 25 rpm for 4 minutes. At each turn, the tablets were dropped from a distance of 6 inches. The tablets were then reweighed (δF1) after the fines were removed and the percentage of weight loss was determined.
% F = δF1 - δF1 / δF1 x 100
Thickness Test: The Vernier calipers scale was used to measure the thickness of the tablets. The thicknesses of the tablets were evaluated.
Loss on Drying Test (LOD): 5g of tablets were weighed (W0) and placed in a dry Petri plate. They were then placed in the Hot air oven for one hour. It was then removed and weighed once again (W!). The pills were then placed back inside the drying chamber and weighed after 10 minutes. This process is done until a steady weight is achieved. The formula used to calculate weight loss is:
% LOD = W0 - W! / W0x 100
RESULTS:
Preliminary Phytochemical Screening of Individual Herb: As depicted in table no 13, following outcomes were found by performing the preliminary Phytochemical Screening of individual herbs.
TABLE 2: PHYTOCHEMICAL SCREENING OF AMLA, FENUGREEK, AND PANEER PHOOL
| Sr. no. | Tests | Amla (Water Extract) | Fenugreek (n-Hexane Extract) | Withania coagulans (Acetone + n-Hexane Extract) |
| 1 | Alkaloid | - | + | + |
| 2 | Glycosides | - | + | - |
| 3 | Phenols | + | + | + |
| 4 | Resins | - | + | - |
| 5 | Saponin | + | + | + |
| 6 | Steroids | + | + | + |
| 7 | Tannin | + | + | + |
| 8 | Terpenoid | + | + | - |
| 9 | Carbohydrates | + | + | + |
| 10 | Protein | - | + | + |
| 11 | Amino test | - | - | + |
| 12 | Essential oils | - | + | + |
Flow Properties of Granules: The bulk density, tapped density, Hausner's ratio, Carr's compressibility index, and angle of repose of Polyherbal Granules were found to be consistent with their physicochemical characteristics Table 3. The Carr's compressibility index and Hausner's ratio were calculated using the bulk density and tapped density measurements.
TABLE 3: EVALUATION FLOW PROPERTIES OF POLYHERBAL GRANULES OF VARIOUS BATCHES
| Sr. no. | Parameters | F1 | F2 | F3 | F4 | F5 |
| 1. | Angle of Repose (°) | 36.4 | 32.4 | 29.4 | 30.05 | 31.00 |
| 2. | Bulk Density (gm/ml) | 0.45 | 0.41 | 0.60 | 0.40 | 0.39 |
| 3. | Tapped Density (gm/ml) | 0.60 | 0.47 | 0.71 | 0.45 | 0.46 |
| 4. | Compressibility index | 25 | 17.76 | 8.28 | 17.00 | 16.95 |
| 5. | Hausner’s Ratio | 1.35 | 1.25 | 1.09 | 1.20 | 1.22 |
| 6. | Inference | Fair | Good | Excellent | Good | Good |
Physical Evaluation of Polyherbal Tablet: The Polyherbal tablets were evaluated for the following test as in Table 4.
TABLE 4: PHYSICAL EVALUATION OF POLYHERBAL TABLET
| Sr. no. | Parameter | Observation |
| 1. | Colour | Light Brown |
| 2. | Odour | Peppermint like |
| 3. | Taste | Pleasant Taste |
| 4. | Shape | Round, Biconvex |
| 5. | pH (1% aqueous solution) | 7.0 |
TABLE 5: TABLET’S EVALUATION TEST
| Batch No | Diameter
(mm) |
Thickness
(mm) |
Hardness
(kg/cm2) |
Friability
(%) |
Weight Variation | Disintegration Test (Min) |
| F1 | 10 | 4 | 4.00 | 0.00 | 1.93 | 10 |
| F2 | 10 | 4.2 | 3.50 | 0.80 | 2.71 | 05 |
| F3 | 10 | 4.1 | 4.00 | 0.00 | 2.36 | 12 |
| F4 | 10 | 4.1 | 4.00 | 0.00 | 2.50 | 15 |
| F5 | 10 | 4.3 | 3.50 | 0.84 | 1.22 | 13 |
UV-visible Investigation of Polyherbal Granules and Dried Seed Extracts: The qualitative UV-Vis spectra of the dried seed extracts of PHGs, Withania coagulans, Emblica officinalis, and Trigonella foenum-graecum Linn revealed a peak at 274 nm with absorption values of 0.525, 0.555, 0.336, and 0.410, respectively Table 6. Previous research demonstrated that the alkaloids, flavonoids, and phenolic chemicals were identified by absorption bands at a wavelength range of 234–676 nm 23. In this instance, the existence of these secondary metabolites was suggested by peaks that emerged at 278 nm in all of the samples.
TABLE 6: UV-VIS SPECTRA SHOWING THE EXISTENCE OF SECONDARY METABOLITES
| Sample | Absorbance | Wavelength | Reference Wavelength | Phytochemical Compounds |
| Withania coagulans | 0.525 | 278 | 234-676 | Flavonoids, alkaloids & Phenolic Compounds |
| Trigonella foenum-graecum Linn | 0.555 | 278 | ||
| Emblica officinalis | 0.336 | 278 | ||
| Polyherbal granules | 0.410 | 278 |
In-vitro Antidiabetics Activities Results:
Alpha Amylase Inhibition: Polyherbal Granules' ability to block alpha-amylase was evaluated by comparing it with acarbose, a common α-amylase inhibitor. Similar to acarbose, the Polyherbal Granules inhibited the alpha-amylase enzyme in a dose-dependent manner (***p < 0.001). However, at 1000 mg dosage, the maximum percentage inhibition of Polyherbal Granules (77.20%) was smaller than that of Standard acarbose (81.42%).
TABLE 7: EFFECT OF COMPOUND BY USING ALPHA-AMYLASE INHIBITION ASSAY
| Sample code | Concentration | α-amylase enzyme inhibition assay | ||
| Absorbance at 540 nm | Mean | %inhibition | ||
| Control | 0.73
0.67 0.70 |
0.70 | ||
| StandardAcarbose | 1mg | 0.12
0.13 0.15 |
0.13 | 81.42 |
| Sample–F1 | 1mg | 0.20
0.16 0.15 |
0.17 | 74.71 |
| Sample–F2 | 1mg | 0.25
0.27 0.23 |
0.25 | 64.30 |
| Sample–F3 | 1mg | 0.20
0.22 0.21 |
0.21 | 70.00 |
| Sample F4–Mannitol | 1mg | 0.17
0.15 0.16 |
0.16 | 77.20 |
| Sample F5–Gelatin | 1mg | 0.22
0.21 0.23 |
0.22 | 68.60 |
FIG. 5: IN-VITRO ANTI-DIABETICS ACTIVITY
In-vitro Antioxidant Activities: The Standard Ascorbic acid showed 93.56 % inhibition, while 86.8%, 77.44%, 81.30%, 77.18%, and 78.21% inhibitions were shown by F1, F2, F3, F4, and F5 batches respectively.
TABLE 8: EFFECT OF SAMPLES BY USING ANTIOXIDANT ACTIVITY BY DPPH (96 WELL METHOD)
| Sample code | Concentration | Antioxidant activity by DPPH (96 well method) | ||
| Absorbance | Mean | Percentage Inhibition | ||
| Control | ---- | 2.101
1.912 1.813 |
1.942 | |
| Standard
Ascorbic acid |
1000µg | 0.150
0.106 0.120 |
0.125 | 93.56 |
| Sample–F1 | 1000µg | 0.234
0.285 0.254 |
0.257 | 86.8 |
| Sample–F2 | 1000µg | 0.425
0.434 0.456 |
0.438 | 77.44 |
| Sample–F3 | 1000µg | 0.356
0.364 0.375 |
0.365 | 81.30 |
| Sample– F4 (Mannitol) | 1000µg | 0.385
0.402 0.434 |
0.443 | 77.18 |
| Sample– F5 Gelatin) | 1000µg | 0.432
0.421 0.416 |
0.423 | 78.21 |
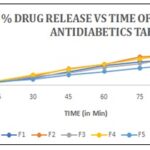
Dissolution Study of Polyherbal Tablet: The Lab India dissolution Tester DS 8000 USP equipment type II (rotating paddle) was used to determine the polyherbal tablet's dissolution profile. Using phosphate-buffered saline (PBS) (pH= 6.8) as the dissolving medium, an accurately weighed quantity of tablet was added to a 900 ml volumetric flask and spun at 100 ± 5 rpm while the temperature was set to 37 ± 0.5 °C. At predetermined intervals (0, 15, 30, 45, 60, 75, and 90 min), 1 ml aliquots of the sample were taken, diluted, and measured spectrophotometrically at 213 nm. To keep the volume constant, an equivalent volume of PBS was added in the meantime. From the previously created calibration curve, the formulation's cumulative % release was estimated.
TABLE 9: DISSOLUTION STUDY PROFILE
| Time (in Min) | Percent drug released in formulation batches | ||||
| F1 | F2 | F3 | F4 | F5 | |
| 15 | 6.83 | 5.79 | 7.27 | 9.14 | 7.92 |
| 30 | 17.37 | 22.42 | 19.72 | 25.02 | 14.97 |
| 45 | 32.45 | 41.14 | 29.77 | 39.24 | 25.56 |
| 60 | 48.74 | 51.19 | 43.45 | 52.77 | 34.3 |
| 75 | 58.75 | 70.59 | 53.89 | 65.95 | 43.27 |
| 90 | 64.44 | 75.38 | 70.38 | 79.3 | 52.77 |
FIG. 6: DISSOLUTION STUDY GRAPH
FIG. 7: POLYHERBAL MIXTURE
FIG. 8: GRANULES
FIG. 9: POLYHERBAL TABLETS
DISCUSSION: The preliminary Phytochemical Screening of several plants produced the results shown in Table 2 above. While Table 1 summarizes the different elements of the formulations for the produced polyherbal dispersible tablets. The micrometric properties of formulations containing aqueous extract powders of Paneer Phool, Fenugreek, and amla used for the preparation of polyherbal dispersible tablets revealed that formulation F-3 has passed and exhibits all excellent properties such as angle of repose (29.4°), bulk density (0.6 g/mL), tapped density (0.71 g/mL), compressibility (8.28%), Hausner ratio (1.09) was equivalent to the IP standard Table 3. Utilizing previously published standard methods, such as powder characteristics, powder micromeritics, drug content uniformity test, disintegration and dispersion time for drug excipients interaction study, the formulation containing polyherbal drugs chosen for making dispersible tablets was identified using earlier accepted procedures, like powder properties, powder micromeritics, like colour, odour, taste, shape, and pH for drug excipients interaction study Table 4 26. During the assessment and development of tablets, it was recorded that the average tablet thickness was 4.00±0.5 mm and the average diameter was 10.00±0.01 mm, while F-1, F-3, and F-4 exhibit good hardness, all of the batches pass the weight variation and friability test, and the F-2 batch has good disintegration time in under 5 minutes Table 5 and F2 Batch showed Excellent result with 75.38%drug release Table 9 27. When compared to the acarbose standard, the prepared samples demonstrated goodamylase enzyme inhibition. The F-4 Mannitol batch had the highest level of inhibition of all other batches (77.20%) in an Alpha-amylase inhibition experiment Table 7 and Fig. 6 28. In this study, samples' direct scavenging abilities were assessed using DPPH scavenging assays. Sample– Tablet of F-1 batch, at a concentration of 1000g/ml, demonstrated the best activity i.e. (86.80%) Table 8 and Fig. 5 29.
CONCLUSION: Herbal products are supposed to possess additive or synergistic benefits and mightcontaina single herboracombination of different plants. Emblica officinalis (amla) fruit, Fenugreek seeds, and Paneer Phool seeds were used to make the herbal tablet, which were tested for parameters including hardness, thickness, weight variation, LOD, and friability. Results for the angles of repose, Carr's Compressibility index, and Hausner's ratio showed that those mixtures of powder had good flow characteristics. Weight homogeneity, hardness, and friability of all batches were assessed, and they were found to meet the official criteria and the official restrictions outlined in IP 2010. The F-3 formulation surpassed other formulations in terms of in-vitro dispersion time and disintegration characteristics. The F-4 Mannitol batch showed the highest degree of inhibition, with anti-diabetic activity of 77.20% and anti-oxidant activity of 86.80%. Additional investigations on stability and additional clinical trials are recommended.
ACKNOWLEDGEMENT: The authors wish to acknowledge the management of Ashokrao Mane College of Pharmacy, Peth Vadgaon (Department of Pharmacognosy and Phytochemistry & Department of Pharmaceutics) for offering the experimental resources necessary to conduct effective studies.
CONFLICT OF INTEREST: There are no disclosed conflicts of interest for the writers. The manuscript's contents have been reviewed and approved by each co-author, and there are no competing financial interests to disclose. We confirm that the contribution is unique and is not already being considered by another publisher.
REFERENCES:
- Ong and Kanyin Liane: "Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021." The Lancet (2023).https://doi.org/10.1016/S0140-6736(23)01301-6
- Nordeng H and Havnen GC: Impact of socio-demographic factors, knowledge and attitude on the use of herbal drugs in pregnancy. Acta obstetriciaet gynecologica S candinavica 2005; 84(1): 26-33.https://doi.org/10.1080/j.0001-6349.2005.00648.x
- Hankey A: Ayurveda and the battle against chronic disease: An opportunity for Ayurveda to go mainstream? Journal of Ayurveda and Integrative Medicine. 2010; 1(1): 9.https://doi.org/10.4103%2F0975-9476.59819
- Kumar and Mahendra: "The Bidirectional Link Between Diabetes and Kidney Disease: Mechanisms and Management." Cureus 15.9 (2023). DOI: 10.7759/cureus.45615
- Garg V, Dhar VI, Sharma A and Dutt R: Facts about standardization of herbal medicine: a review. Zhongxiyijie he xuebao = Journal of Chineseintegrative medicine 2012; 10(10): 1077-83.https://doi.org/10.3736/jcim20121002
- Chaudhary A, Singh N: Contribution of world health organization in the globalacceptance of Ayurveda. Journal of Ayurveda and integrative medicine 2011; 2(4): 179.https://doi.org/10.4103%2F0975-9476.90769
- Rehman and Samiya: "Drier climatic conditions increase withanolide content of Withania coagulans enhancing its inhibitory potential against human prostate cancer cells." Applied biochemistry and biotechnology 188 (2019): 460-480. https://doi.org/10.1007/s12010-018-02933-8
- Maurya and Rakesh: "Chemistry and pharmacology of Withania coagulans: an Ayurvedic remedy." Journal of pharmacy and pharmacology 62.2 (2010): 153-160. https://doi.org/10.1211/jpp.62.02.0001
- Langdon, Amy, Nathan Crook and Gautam Dantas: "The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation." Genome medicine 8.1 (2016): 1-16. https://doi.org/10.1186/s13073-016-0294-z
- Maurya R: Coagulanolide, a withanolide from Withania coagulans fruits and antihyperglycemic activity. Bioorg Med Chem Lett 2008; 18: 6534-6537. https://doi.org/10.1016/j.bmcl.2008.10.050
- Rahman Atta-Ur: Five new withanolides from Withania coagulans. Heterocycles 1998; 48: 1801–1811.
- Atta-ur-Rahman: Two new ergostane type steroidal lactones from Withania coagulans”, Journal of Natural Products 1998, 61, 812-814. https://doi.org/10.1021/np970478p
- Ramaiah PA: Spectroscopic studies on a withanolide from Withania coagulans. Phytochemistry 1984; 23: 143–149. https://doi.org/10.1016/0031-9422(84)83095-2
- Khan MI, Maqsood M, Saeed RA, Alam A, Sahar A, K ieliszek M, Miecznikowski A, Muzammil HS and Aadil RM: Phytochemistry, foodapplication, and therapeutic potential of the medicinal plant (Withania coagulans): Areview. Molecules 2021; 26(22): 6881.https://doi.org/10.3390/molecules26226881
- Kreis W, Müller-Uri F: Biochemistry of sterols, cardiacglycosides, brassinosteroids, phytoecdysteroids and steroids a ponins. Annual Plant Reviews Volume 40: Biochemistry of Plant Secondary Metabolism.2010:304-63.https://doi.org/10.1002/9781444320503.ch6
- Sun W, Shahrajabian MH and Cheng Q: Fenugreek cultivation with emphasis on historical aspects and its uses intraditional medicine and modern pharmaceutical science. Mini Reviews in Medicinal Chemistry 2021; 21(6): 724-30.https://doi.org/10.2174/1389557520666201127104907
- Kumar emphasisonhistorical Shubham: "Herbal medicines for diabetes management and its secondary complications." Current Diabetes Reviews 17.4 (2021): 437-456. https://doi.org/10.2174/1573399816666201103143225
- Srinivasa, Uma Maheshwari, and Madeneni Madhava Naidu. "Fenugreek (Trigonella foenum-graecum L.) seed: promising source of nutraceutical." Studies in Natural Products Chemistry 71 (2021): 141-184. https://doi.org/10.1016/B978-0-323-91095-8.00014-3
- Pareek emphasisonhistorical Sunil: "Indian gooseberry (Emblica officinalis gaertn.)." Fruit and Vegetable Phytochemicals: Chemistry and Human Health, 2nd Edition (2017): 1077-1106.https://doi.org/10.1002/9781119158042.ch54
- Variya, Bhavesh C, Anita K. Bakrania and Snehal S. Patel: "Emblica officinalis (Amla): A review for its phytochemistry, ethnomedicinal uses and medicinal potentials with respect to molecular mechanisms." Pharmacological research 111 (2016): 180-200. https://doi.org/10.1016/j.phrs.2016.06.013
- Sriwatcharakul S: Evaluation of bioactivities of Phyllanthus emblica seed. Energy Reports 2020; 6: 442-7.https://doi.org/10.1016/j.egyr.2019.08.088
- Padayatty SJ, Levine M: Vitamin C: the known and the unknown and Goldilocks. Oraldiseases 2016; 22(6): 463-93.https://doi.org/10.1111/odi.12446
- Mabasa XE: "Molecular spectroscopic (FTIR and UV-Vis) and hyphenated chromatographic (UHPLC-qTOF-MS) analysis and in-vitro bioactivities of the Momordica balsamina leaf extract." Biochemistry Research International 2021 (2021). https://doi.org/10.1155/2021/2854217
- Arathy R: "Assessment of In-vitro Antidiabetic Potential of Purified Anthocyanin Extract from Floral Petals of Wild Balsam Species." Journal of Drug Delivery and Therapeutics 10.3 (2020): 31-35. https://doi.org/10.22270/jddt.v10i3.4050
- Datatraya Raut: synthesis, molecular docking and biological evaluation of 2- (2-hydrazinyl) thiazoles as potential Antioxidant, Anti-inflammatory and significant anticancer agent, Recent advances in inflammation and allergy drug discovery 2022; 16: 96-106. https://doi.org/10.2174/2772270816666220902094019
- Parasuraman S, Thing GS emphasisonhistorical Dhanaraj SA: Polyherbal formulation: Concept of ayurveda. Pharmacognosy reviews. 2014; 8(16): 73.https://doi.org/10.4103%2F0973-7847.134229
- Gauttam VK and Kalia AN: Development of polyherbal antidiabetic formulation encapsulated in the phospholipids vesicle system. Journal of advanced pharmaceutical technology & research. 2013; 4(2): 108.https://doi.org/10.4103%2F2231-4040.111527
- Arathy R, Murugan K, Babu KD, Manoj G. Assessment of in-vitro Antidiabetic Potential of Purified Anthocyanin Extract from Floral Petals of Wild Balsam Species. Journal of Drug Delivery and Therapeutics 2020; 10(3): 31-5.
- Raut DG, Bhosale RB, Lawand AS, Hublikar MG, Kadu VD and Patil SB: Syntheses, Molecular Dockingand Biological Evaluation of 2-(2-hydrazinyl) thiazoles as Potential Antioxidant, Anti- Inflammatory and Significant Anticancer Agents. Recent Advances in Inflammation & Allergy Drug Discovery 2022; 16(2): 96-106.https://doi.org/10.2174/2772270816666220902094019
How to cite this article:
Mane SR: Exploring Ayurveda and traditional medicines: efficacy of polyherbal antidiabetic tablets and their antioxidant properties. Int J Pharmacognosy 2023; 11(1): 15-26. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(1).15-26.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
15-26
1350 KB
556
English
IJP
Shiom Ramesh Mane *, Chinmay Shivaji Patil, Priti Rajagonda Patil, Snehal Sanjay Patil, Pranali Piraji Bhosale, Poonam Nilesh Chougule and Sachinkumar Patil
Ashokrao Mane College of Pharmacy, Peth, Vadgaon, Maharashtra, India.
shiommane@gmail.com
04 November 2023
19 December 2023
27 January 2024
10.13040/IJPSR.0975-8232.IJP.11(1).15-26
31 January 2024