EVALUATION OF THE IMMUNOMODULATORY EFFECT OF PIPLARTINE AND ITS DERIVATIVE ON PERITONEAL MACROPHAGES OF SWISS MICE INFECTED WITH LEISHMANIA AMAZONENSIS
HTML Full TextEVALUATION OF THE IMMUNOMODULATORY EFFECT OF PIPLARTINE AND ITS DERIVATIVE ON PERITONEAL MACROPHAGES OF SWISS MICE INFECTED WITH LEISHMANIA AMAZONENSIS
Keline Medeiros de Araújo Vilges 1, Stefan Vilges de Oliveira * 2, Harold Hilarion Fokoue 3, Massuo Jorge Kato 3,Tatiana Karla dos Santos Borges 1, José Roberto de Souza Almeida Leite 4 and Selma Aparecida Souza Kuckelhaus 4
Laboratory of Cell Immunology 1, Laboratory of Morphology 4, Faculty of Medicine, University of Brasilia Campus Darcy Ribeiro, Brasilia-DF, Brazil.
Department of Public Health 2, Faculty of Medicine, Federal University of Uberlandia, Campus Umuarama, Uberlandia-MG, Brazil.
Institute of Chemistry 3, University of São Paulo, São Paulo-SP, Brazil.
ABSTRACT: Piplartine is an amide obtained from Piper tuberculatum, which is promising in antiparasitic tests. Therefore, it has been used as the basis for the synthesis of new molecules. This study was designed to analyze the immunomodulatory effect of piplartine (A) and its derivative (B), in-vitro, on macrophages infected with Leihmania amazonensis, as well as the production of hydrogen peroxide (H2O2), nitric oxide (NO) and lipid bodies. Were evaluated by staining with giemsa and counting the cells under optical microscopy; labeling with tetramethyl benzidine and peroxidase type II; Greiss reagent and oil red, respectively, with amides A and B concentrations ranging from 0.0 to 64 μg/mL. Modulation of the immune system was observed. There was a decrease in the infection index at the highest concentration (64 μg/mL) for A and B in infected macrophages. Amide A (4 μg/mL) reduced the H2O2 production of infected macrophages by 99.3% compared to control. Cultures treated with A and B increases the NO production at 16 μg/mL and 0.25 μg/mL, respectively. The production of lipid bodies by infected macrophages treated at 64 μg/mL of A decreased when compared to the control macrophage infected with Leishmania. When derivative B was used, the percentage of infected macrophages with lipid bodies increased at concentrations 0.25 μg/mL, 1 μg/mL, 4 μg/mL and 16 μg/mL. Compounds A and B have shown promise for the treatment of leishmaniasis and suggest the continuity of preclinical studies to assess their effects in-vivo.
| Keywords: |
Leishmania, Piperaceae, Piplartine, Cinnamides
INTRODUCTION: Leishmaniasis is a disease caused by intracellular protozoa belonging to the genus Leishmania 1, and are considered endemic in 88 countries worldwide, of which 72 are in development 2.
Pentavalent antimonials have been used as the first choice for the treatment of American Tegumentary Leishmaniasis (ATL) and Visceral Leishmaniasis (VL). However, they are expensive and toxic drugs, besides there are reports in the literature of eventual failures in therapy due to the increase of resistant forms of these parasites 1, 2, 3.
In view of this, new research is needed to find substances that are effective against these parasites and have fewer side effects. The medicinal plants have numerous constituents beneficial to the man that make them a target of great interest in the scientific field 4. The species Piper tuberculatum L. known in North-Eastern Brazil as "long pepper" or "pepper" has been widely used for medicinal purposes 5. Piplartine is an amide/alkaloid present in species of the genus Piper and found in Piper tuberculatum L. It presents different pharmacological activities, such as leishmanicidal action when tested in-vitro in L. donovani promastigotes and also when tested in-vivo, in hamster with visceral leishmaniasis 6, anxiolytic and antidepressant action, anti-inflammatory, antiplatelet, antitumor and antifungal activity 7.
In leishmania infection, macrophages are cells that play a central role. They act both for the replication of the parasite and for effector cells to kill it through the reactive species of oxygen and nitrogen 8, 9. Also, pathological studies evidenced an increase in the formation of lipid bodies in peritoneal macrophages of mice infected with L. amazonensis 10. In cells of the immune system, lipid bodies are recognized as sites for the generation of inflammatory mediators, which can modulate the immune response, inhibiting or stimulating the growth of the pathogen 11, 12. Considering that piplartine has different biological activities, it was used as a model for the synthesis of its (E)-3-(3, 4, 5-trimethoxyphenyl)-N-pentylprop-2-enamide derivative. The present study relates the effect of these amides on the modulation of the immune response in leishmaniasis, in-vitro, by the assays on the cultures of macrophages infected with L. amazonensis, as well as on the functions of these cells when infected.
MATERIAL AND METHODS:
Piplartine and Derivative: The piplartine (A) was obtained from the root of the species Piper tuberculatum Jacq (Piperaceae), collected on 4 May 4, 2015, in the garden of the Institute of Chemistry (University of São Paulo). The identification of the species P. tuberculatum was carried out by Dr. Elsie F. Guimarães and a voucher was deposited in the Herbarium of the Botanical Garden of Rio de Janeiro, Brazil (Kato-0169). The roots were dried at 60 °C for 48 h, then ground 105 g of a powder was extracted four times with dichloromethane: methanol (2:1; 400 mL). The extract was filtered and concentrated on a rota evaporator. The crude extract was submitted to re-crystallization using ethyl acetate and methanol yielding pure piplartine (150 mg) 13. The derivative amide (B) was synthesized by adding triethylamine (3 equiv.) and amine (N-pentylamine and morpholine) to the acid chloride solution (1 equiv.) in CH2Cl2.
In order to prepare the acid chloride, a solution of (E)-3, 4, 5-trimethoxycinnamic acid and (E)-3, 4-dimethoxycinnamic acid (1 equiv.), both prepared by the Knoevenagel condensation using 3, 4, 5-trimethobenzaldehyde and 3, 4-dimetbenzaldehyde and malonic acid in dry tetrahydrofuran (10 mL), kept under a nitrogen atmosphere, oxalyl chloride (5 equiv.) added dropwise and stirred at room temperature for 5-6 h. The excess oxalyl chloride was then removed under reduced pressure to afford the corresponding acid chloride 14. The reaction mixture was stirred overnight at room temperature and quenched with saturated aqueous NH4Cl, and extracted with CH2Cl2 (three times). The combined organic phases were washed with brine and dried over MgSO4. After filtration and concentration, the residue was purified by flash chromatography to provide the desired amide. Piplartine and its derivative were diluted in 1% dimethylsulfoxide (DMSO), and from that pre-dilution, the serial dilution was performed to begin the assays.
Microorganism:
Leishmania amazonensis: The MHOM/BR/pH8 strain was transferred to the NNN solid medium (Novy-MacNeal-Nicolle) for 48 h at 24 °C. Then the promastigote forms were transferred and cultured in RPMI 1640 medium supplemented with 10% of inactivated fetal calf serum (Sigma-Aldrich, St. Louis, MO) and gentamycin (40 mg/mL) (Schering-Plough, Sao Paulo, Brazil) until the parasites reached the log phase of growth.
Animals and Peritoneal Cells: To obtain peritoneal cells, six adult Swiss mice (male and female), weighing 30 ± 6 g, were used. During the experiments, the animals were kept in the Faculty of Medicine, the University of Brasilia under ambient temperature, the light/dark cycle of 12 h, fed with a balanced diet and water ad libitum. Cells were obtained by washing the peritoneal cavity with 10 mL of cold phosphate buffered saline (PBS), pH 7.2 at 4 ºC, centrifuged at 400 g for 10 min and suspended with 1 mL of RPMI 1640 medium were quantified at Neubauer chamber with 0.05% of nigrosin solution.
Evaluation of the Microbicidal Effect of Piplartine (A) and Derivative (B) on Macrophage Infected or not with Axenic Amastigotes forms of L. amazonensis: Viable cells (2×105) were added to 13 mm diameter glass coverslips placed in 24 well plastic plates and incubated in a wet chamber for 2 h at 37 ºC with 5 % CO2. The coverslips were washed with PBS; the adherent cells were incubated with 106 L. amazonensis amastigotes for 12 h at 37 ºC with 5 % CO2. After, were washed with PBS to remove non-phagocytosed leishmania and then incubated for 4 h with different concentrations of A and B (0, 25; 1; 4; 16 and 64 µg/mL).
After that were washed with PBS, RPMI, dried, fixed with methanol, dried, giemsa stained at 10% for 10 min, washed with water, dried and mounted on microscopic slides. For the infection index, 200 macrophages were considered and visualized by optical microscopy (100 ×) by a single observer. To calculate the % of infected macrophages the following formula was used: (% macrophage with leishmania × 100 / Total cells considered). For the mean of leishmania by macrophages: (Total leishmania / infected macrophages). For the index of infection: % Infected macrophages × mean of leishmania by macrophages.
Hydrogen Peroxide (H2O2) Production in Cultures of Peritoneal Macrophages of Mice Infected or not by Leishmania and Treated with Different Concentrations of Piplartine (A) and Derivative (B): Hydrogen peroxide (H2O2) production by peritoneal macrophages was assessed of pH 3, 3’, 5, 5’- tetramethylbenzidine (TMB) and peroxidase type II 15. Viable cells (2 × 105) were incubated for 2 h at 37°C with 5% CO2 in 96 wells plastic plate for adherence of the macrophages. After, the wells were washed with PBS to remove non-adherence cells and then incubated with 106 L. amazonensis amastigotes for 12 h at 37 ºC with 5% CO2. Then the wells were washed with PBS for removal of the non-phagocytized leishmania and then again incubated for 24 h with different concentrations of A and B (0.25; 1; 4; 16 and 64 μg/mL). For negative control, only macrophages and RPMI were used. For the positive control, phorbol myristate acetate (PMA; Sigma, St. Louis, MO, USA) at 40 mM.
During the incubation, a standard curve was prepared in hydrogen peroxide (0 μM, 0.39 μM, 0.78 μM, 1.56 μM, 3.12 μM, 6.25 μM, 12.5 μM, 25 μM, 50 μM and 100μM) and an 'A' solution with 20 μL of peroxidase type II (2.5 mU), 150 μL of 3,3',5', 5'-tetramethylbenzidine (TMB - 0.7 mM) and 9830 μL of PBS pH = 6,0 (0.1M). After 24 h of incubation with the amides, 10μL of the standard curve dilutions, 10 μl of the supernatant from the amide treated cultures were taken to another plate plus 200 μl of the solution 'A,' homogenized and left at room temperature for 15 min. The reaction was stopped by adding H2SO4 (0.5 M) per well. The absorbance was read at 450 nm (SpectraMax® Plus384, Molecular Devices, Sunnyvale, CA, USA). The results were expressed as μM H2O2.
Nitric Oxide Production in Cultures of Peritoneal Macrophages of Mice Infected or not by Leishmania and Treated with Different Concentrations of Piplartine (A) and Derivative (B): Nitric oxide production by peritoneal macrophages was indirectly assessed by means of nitrite (NO2-) determination, using the Greiss reagent by the nitrite quantification of these cultures, according to the technique described by Green et al. 16 Viable cells (2 ×105) were incubated for 2 h at 37 °C with 5% CO2 in 96 wells plastic plate for adherence of the macrophages. After, the wells were washed with PBS to remove non-adherence cells and then incubated with 106 L. amazonensis amastigotes for 12 h at 37 ºC with 5% CO2. Then the wells were washed with PBS for removal of the non-phagocytized leishmania and then again incubated for 24 h with different concentrations of A and B (0.25; 1; 4; 16 and 64 μg/mL).
For negative control, only macrophages and RPMI were used. For the positive control lipopolysaccharide (LPS Escherichia coli, serotype 055: b5, Sigma-Aldrich, St Louis, USA, at 50 ng/mL). After 24 h incubation, the plates were subjected to centrifugation at 400×G for 10 min, and then 100 μl of the supernatant from each well was transferred to another plate with 100 μl of the Greiss reagent. (1% sulphanilamide/0.1% N-1-naphthylethylene diamine dihydrochloride/2.5% H3PO4). The standard curve was prepared with different concentrations (0 μM, 0.39 μM, 0.78 μM, 1.56 μM, 3.12 μM, 6.25 μM, 12.5 μM, 25 μM, 50 μM and 100 μM) of sodium nitrite (NaNO2) in distilled water. The absorbance was read at 540 nm (SpectraMax® Plus384, Molecular Devices, Sunnyvale, CA, USA). The results were expressed as (mM) NO2-.
Formation of Lipid Bodies by Macrophages Infected or not by Leishmania and Treated with Different Concentrations of Piplartine (A) and Derivative (B): For quantification of the lipid bodies, the staining with oil red was used, adapted by Muniz - Junqueira et al. 17 Viable cells (2 × 105) were added to 13 mm diameter glass coverslips placed in 24 well plastic plates and incubated in a wet chamber for 2 h at 37 ºC with 5% CO2 for adherence of the macrophages. The wells were washed three times with PBS for removal of non-adherence cells and then added 106 L. amazonensis amastigotes at each well and incubated for 12 h at 37 ° C with 5 % CO2.
After, were washed with PBS to remove non-phagocytosed leishmania and then incubated for 4 h with different concentrations of A and B (0.25; 1; 4; 16 and 64 µg/mL). For the negative control, only macrophages and RPMI were used. For the positive control lipopolysaccharide (LPS Escherichia coli, serotype 055: b5, Sigma-Aldrich, St Louis, USA, at 1 µg/mL). Wells were washed twice with PBS, fixed with 4% paraformaldehyde for 30 min, washed twice with PBS and once with 60% isopropyl alcohol, stained with oil Red® for 15 min, washed once with 60% isopropyl alcohol, twice with milique water, stained with hematoxylin for 5 min, washed once with distilled water and once with milique and mounted in gelatinous medium. Light microscopy analysis revealed that positively stained macrophages presented red-stained lipid bodies in the cytoplasm. To calculate the % of macrophages with lipid bodies, the following formula was used: (% Macrophage with corpuscles × 100 / Total cells considered). For the mean of corpuscles by macrophages: (Total corpuscles/macrophages with corpuscles). For the infection index: % Macrophage with corpuscles × mean of corpuscles by macrophages.
Statistical Analysis: The normality of the variables was analyzed employing the Kolmogorov-Smirnov test and, Paired t-test or Wilcoxon were used to compare two normal or non-normal samples, respectively. The Prism® Software Package program (GraphPad, USA, 2005) was used to analyze and represent the results. Differences were considered significant at a P value <0.05.
Ethical Consideration: The study was approved by the Ethics Committee on Animal Use (CEUA) of the Institute of Biological Sciences at the University of Brasilia (Doc. no. 22199/2014).
RESULTS:
Microbicidal Effect of Piplartine (A) and Derivative (B): When the effect of piplartine (A) on the percentage of infected macrophages was evaluated, a decrease in infection was observed at concentrations 1, 16 and 64 μg/mL. Regarding the mean number of leishmania phagocytosed by macrophages, the results indicate that there was a decrease only at the concentration of 64 μg/mL. Similarly, for the infection index, the results showed that there was a decrease in that index to the highest concentration of 64 μg/mL.
TABLE 1: MICROBICIDAL EFFECT OF DIFFERENT CONCENTRATIONS OF A AND B ON MACROPHAGES INFECTED WITH L. AMAZONENSIS
| Different concentrations of A and B (μg/mL) in macrophages infected with Leishmania | |||||||
| 0 | 0.25 | 1 | 4 | 16 | 64 | ||
| A | % Infec. mac. | 91.7 | 90.5 | 83.1* | 90.0 | 74.4* | 80.2* |
| Average Leish./Mac. | 5.0 | 4.5 | 5.0 | 4.2 | 3.8 | 3.2* | |
| Infection index | 442 | 415 | 407 | 385 | 304 | 260* | |
| B | % Infec. mac. | 92.2 | 90 | 88 | 89 | 93.6 | 85.6* |
| Average Leish./Mac. | 5.3 | 5 | 5 | 5.8 | 4.8 | 3.6* | |
| Infection index | 470 | 459 | 438 | 525.3* | 455 | 315* | |
% Infec. mac.: % infected macrophages; Average Leish./Mac.: Mean of leishmania by macrophages. The results were evaluated by paired t-test. (*) Values considered significant when compared to control (P<0.05)
For the cultures treated with the derivative (B), when evaluated the percentage of infected macrophages and the average of phagocytosed leishmania by macrophages, a decrease of the infection and a decrease of the average only in the concentration of 64 μg/mL were observed. For the infection index, the results showed that there was a decrease in the highest concentration of 64 μg/mL and an increase with 4 μg/mL. All results were compared to the untreated control Table 1.
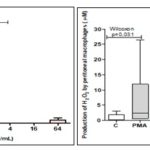
Hydrogen Peroxide (H2O2) Production: The results showed that the infected peritoneal macrophages treated with 4 μg/mL of piplartine (A) showed a reduction of 99.3% in H2O2 production about the RPMI control. For the other concentrations (0.25, 1, and 64 μg/mL) no differences were observed Fig. 1A. Similarly, for the cultures treated with different concentrations (0.25, 1, 4, 16 and 64 μg/mL) of the derivative (B) there was no difference about the untreated control Fig. 1B.
Cells that were stimulated with PMA (positive control) showed an increase of 87% in H2O2 production compared to RPMI (negative control). However, the leishmania control did not differ from the RPMI control.
FIG. 1: PRODUCTION OF HYDROGEN PEROXIDE BY PERITONEAL MACROPHAGES OF SWISS MICE (n = 6) INFECTED OR NOT WITH L. AMAZONENSIS AND TREATED WITH DIFFERENT CONCENTRATIONS (0.25, 1, 4, 16 & 64 μg/mL) OF PIPLARTINE (A) AND DERIVATIVE (B). The results were analyzed by the Wilcoxon test, considering the significant values when P<0.05. The data are represented in medians, quartiles maximum and minimum values. (*) Represents sample with variability, evaluated by the Tukey test.
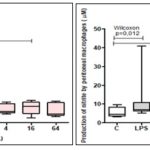
Nitric Oxide Production: The results showed that the infected peritoneal macrophages treated with 16 μg/mL of piplartine (A) there is a 46.8% increase in NO production compared to RPMI (negative control). For the other concentrations (0.25, 4 and 64 μg/mL) no differences were observed Fig. 2A. Similarly, infected peritoneal macrophages treated with derivative (B) with 0.25 μg/mL there was a 46.8% increase in NO production compared to the RPMI control. LPS stimulated cells (positive control) showed a 34 % increase in NO production compared to RPMI (negative control) Fig. 2B.
Formation of Lipid Bodies: For cultures of infected macrophages and treated with 64 μg/mL of piplartine (A), the results showed that the percentage of macrophages with lipid bodies decreased when compared to the macrophage control infected with Leishmania. Regarding the mean of lipid bodies by macrophages, the results indicate that there was an increase only for the infected macrophages treated with 0.25 μg/mL compared to the macrophage control infected with Leishmania. Similarly, for the corpuscular index, the results showed that there was a decrease of this index in the infected macrophages treated with 64 μg/mL and an increase with 0.25 μg/mL compared to the control macrophage infected with Leishmania. For macrophage cultures infected and treated with derivative (B), the percentage of macrophages with lipid bodies increased when the infected macrophage Leishmania control was compared to the infected macrophages treated with 0.25 μg/mL, 1 μg/mL, 4 μg/mL and 16 μg/mL.
About the mean lipid bodies by macrophages, the results indicate that there were no differences when compared the control macrophage infected with Leishmania to infected and treated macrophages. About the corpuscular index, the results showed that there was an increase of this index in the infected macrophages treated with 16μg/mL when compared to the macrophage control infected with Leishmania Table 2.
FIG. 2: NITRITE PRODUCTION BY PERITONEAL MACROPHAGES OF SWISS MICE (n = 6) INFECTED OR NOT WITH L. AMAZONENSIS AND TREATED WITH DIFFERENT CONCENTRATIONS (0.25, 1, 4, 16 & 64 μg/mL) OF PIPLARTINE (A) AND DERIVATIVE (B). The results were analyzed by the Wilcoxon test, considering the significant values when P<0.05. The data are represented in medians, quartiles maximum and minimum values.
TABLE 2: EFFECT OF PIPLARTINE (A) AND DERIVATIVE (B) ON THE FORMATION OF LIPID BODIES BY MACROPHAGES INFECTED OR NOT BY L. AMAZONENSIS
| Different concentrations of A and B (μg/mL) and formation of lipid bodies | ||||||||
| Mac. | Infec. mac. | 0.25 | 1 | 4 | 16 | 64 | ||
| A | % Macrophages with Lipid bodies | 3.0±3.2 | 78.7±3.7 | 83±4.6 | 79±9 | 80±10 | 76.5±8 | 31.4±21.5* |
| Average Corpuscles/ Macrophages | 4±3.1 | 6.6±1 | 10.3±2* | 8±3 | 6.2±0.9 | 5.8±0.4 | 6.8±1.6 | |
| Corpuscular Index | 16.6±15.2 | 525±105 | 859±159* | 616±153 | 494±52 | 447±38 | 217±153* | |
| B | % Macrophages with Lipid bodies | 3.0±3.2 | 78.5±4 | 87±4.3* | 89.3±4* | 90±4* | 89±4.5* | 79±7.3 |
| Average Corpuscles/ Macrophages | 4±3.1 | 6.6±1.1 | 7.3±0.6 | 6.4±0.6 | 6±0.4 | 8±1.2 | 6.4±1.4 | |
| Corpuscular Index | 16.6±15.2 | 525±105 | 641±61 | 578±69.5 | 544±46.8 | 757±153* | 545±120 | |
(*) values considered significant when compared to control of infected macrophages. Mac.: Macrophages; Infec. mac.: infected macrophages. (The results were evaluated by paired t -test, P<0.05).
DISCUSSION AND CONCLUSION: As regards the effect of piplartine (A) on macrophages infected with axenic amastigotes of L. amazonensis, a decrease in infection index was observed in 41.3% after treatment with 64 μg/mL. Likewise, derivative B decreased the infection index by 33% with 64 μg/mL. Dias 18 has shown that glucantime, the drug of the first choice for the treatment of leishmania, can decrease the infection index in macrophages infected with L. amazonensis by 55% at the concentration of 300 μg/mL. The results obtained for amides A and B were higher when compared to glucantime.
Possibly, piplartine (A) and derivative B interacted, by unknown mechanisms, with the cell membranes of the macrophages and, subsequently, with the parasite membrane. It is speculated that these amides have acted in the inhibition of adenosine (ATP) and guanosine triphosphate (GTP) 19 by blocking the fatty acid glycolytic and oxidative activity of amastigotes, so that they decrease the phosphorylation capacity of ADP to ATP leading to the decrease of intracellular ATP, and consequently, causing death of the parasite 20. It is also known that macrophages are important cells of the immune system since they play roles in both innate and adaptive immunity. These functions include phagocytosis of foreign particles, production of cytokines and chemical mediators such as H2O2 and NO, for example, 21. In the present study, amides also interfered in the production of reactive species of nitrogen, oxygen and lipid bodies in the infected macrophages, indicating that they acted as modulators of the immune response in leishmaniasis. When the macrophages were infected and treated with different concentrations of piplartine (A), there was a decrease in the production of hydrogen peroxide at a concentration of 4 μg/mL compared to production of the control. In contrast, the different concentrations of the derivative B tested did not interfere positively or negatively in this process when compared to the control.
The production of H2O2 is a natural process in the immune response, but in the presence of an infection, the cell starts to consume more oxygen, an increase in glucose oxidation occurs through hexose monophosphate with NADPH production22.
NADPH (adenine - diphosphate - nicotinamide) oxidase acts as an electron donor, promoting the reduction of O2 to O2-, and this by spontaneous dismutation or by the enzyme superoxide dismutase (SOD) is transformed into H2O2 22, 23. It is assumed that piplartine (A) may have interfered in the hexose monophosphate pathway, to inhibit the production of H2O2 at a concentration of 4 μg/mL in the infected macrophages. It is known that during the destruction of a pathogenic agent, macrophages can produce excess free radicals and, in this way, cause damage to neighboring tissues 24. In this sense, this reduction of H2O2 production at low concentrations may be good in L. amazonensis infections. The influence of piplartine (A) on NO production by peritoneal macrophages was also analyzed, and the results showed that when cultures were infected and treated with the concentration 16 μg/mL an increase in NO production occurred. Similarly, derivative B, at the lowest concentration (0.25 μg/mL), was able to increase NO production.
NO is a free, gaseous and unstable radical synthesized from the oxidation of the nitrogen atom of L-arginine by the action of nitric oxide synthase (iNOS) on macrophages 25. Ta1 cytokines, such as IFN- ϫ and TNF - α, induce the formation of iNOS. It is known that NO is effective against several microorganisms, including Trypanosoma cruzi, Toxoplasma gondii and Leishmania 26, 27, 28, 29. Studies have shown that peritoneal macrophages from mice stimulated in-vitro with IFN-ϫ in the presence of lipopolysaccharide (LPS) release NO in significant amounts capable of efficiently destroying the parasite 29. Wei et al., 30 correlate NO production with the control of leishmaniasis infection. The results of our study indicate increased NO production by peritoneal macrophages infected with L. amazonesis and treated with both piplartine (A) and derivative B, indicating that these amides may improve the action of the iNOS enzyme possibly by the increase of the inflammatory cytokines.
About the production of lipid bodies, Bozza et al., 31 report that the concentration of these organelles in resting macrophages is low in comparison to macrophages that are associated with infectious conditions, corroborating with data from this study. There are reports in the literature showing an increase in the formation of these organelles in peritoneal macrophages of mice infected with L. amazonensis, as well as with other species of Leishmania sp. which cause cutaneous leishmaniasis 10-32. It is known that the interaction of Leishmania with lipid bodies works for different purposes. These organelles can provide lipids for the biogenesis of membranes of new parasites can serve as a source of nutrients and can modulate the immune system through the production of eicosanoids, since they are sites that generate these inflammatory mediators 33, 12-32. The role of these organelles in infection also depends on the interaction of the parasite with the host 12.
Our findings show that for macrophages that were infected and treated with piplartine (A) at the highest concentration (64 μg/mL), there was a 60% reduction in the percentage of these cells with lipid bodies and also a reduction in the same percentage corpuscular index. Piplartine (A) was able to modulate the immune response by inhibiting the formation of these lipid organelles, and this inhibition could interfere with the availability of nutrients for these parasites and the availability of lipids for the biogenesis of the membranes of new parasites.
Similarly, when macrophages were infected and treated with derivative B, there was an increase in the percentage of macrophages with lipid bodies at almost all concentrations tested. This increase may be favorable to the parasite because the cell will have more nutrients for it and more lipids for its biogenesis. However, it is worth remembering that, as mentioned above, these lipid organelles are sources of inflammatory mediators, and that these can act by modulating the immune response producing leukotrienes, limiting the growth of the parasite, or producing prostaglandins, favoring this growth. This modulation will depend on the interaction of the parasite with the host 12.
The amides A and B were able to decrease L. amazonensis infection in macrophages and also to interfere in the production of reactive oxygen, nitrogen species and lipid bodies in these cells, being considered modulators of the immune response in leishmaniasis. Likewise, it will be necessary to quantify the inflammatory and anti-inflammatory cytokines produced by the macrophages infected by L. amazonensis and also which mediators are being produced, whether leukotrienes or prostaglandins to better understand the infection before treatment with these amides.
ACKNOWLEDGEMENT: We are grateful to CAPES / Ministry of Education / Brazil for financial support.
CONFLICT OF INTEREST: The authors declare no conflicts of interest.
REFERENCES:
- Esboei BR, Mohebali M, Mousavi P, Fakhar M and Akhoundi B: Potent antileishmanial activity of chitosan against Iranian strain of Leishmania major (MRHO/IR/ 75/ER): In-vitro and in-vivo J Vector Borne Dis 2018; 55(2): 111-115.
- Cavalcante IJM and Vale MR: Aspectos epidemiológicos da leishmaniose visceral (calazar) no Ceará no período de 2007 a 2011. Rev. Bras Epidemiol 2014; 17(4): 911-924.
- Pelissari DM, Cechinel MP, Sousa-Gomes ML and Júnior FEFL: Tratamento da leishmaniose visceral e leishmaniose tegumentar americana no Brasil. Epidemiol Serv Saúde 2011; 20(1): 107-110.
- Silva JG, Souza IA, Higino JS, Siqueira-Júnior JP, Pereira JV and Pereira MSV: Atividade antimicrobiana do extrato de Anacardium occidentale em amostras multi-resistentes de Staphylococcus aureus. Rev. Bras. Farmacogn 2007; 17(4): 572-577.
- Ferreira MGPR, Kayano AM, Silva-Jardim I, Silva TO, Zuliani JP, Facundo VA, Calderon LA, Almeida-e-SilvaA, Ciancaglini P and Stábeli RG: Antileishmanial activity of 3-(3,4,5-trimethoxyphenyl) propanoic acid purified from Amazonian Piper tuberculatum, Piperaceae, fruits. Rev Bras Farmacogn 2010; 20(6): 1003-1006.
- Bodiwala HS, Singh G, Singh R, Dey CS, Sharma SS, Bhutani KK and Singh IP: Antileishmanial amides and lignans from Piper cubeba and Piper retrofractum. J Nat Med 2007; 61(4): 418-421.
- Bezerra DP, Moura DJ, Rosa RM, De Vasconcelos MC, Silva AC, Moraes MO, Silveira ER, Lima MA, Henriques JA, Costa-Lotufo LV and Saffi J: Evaluation of the genotoxicity of piplartine, analkamide of tuberculatum, in yeast and mammalian V79 cells. Mutat Res 2008; 652(2): 164-174.
- Kedzierski L and Evans KJ: Immune responses during cutaneous and visceral leishmaniasis. Parasitology 2014; 141(12): 1544-1562.
- Peters NC, Egen JG, Secundino N, Debrabant A, Kimblin N, Kamhawi S, Lawyer P, Fay MP, Germain RN and Sacks D: In-vivo imaging reveals an essential role for neutrophils in Leishmaniasis transmitted by sand flies. Sci 2008; 321(5891): 970-974.
- Melo RCN and Dvorak AM: Lipidic body-phagosome interaction in macrophages during infections diseases: Host defense or pathogen survival strategy? PLoS Pathog 2012; 8(7): e1002729.
- Weller PF: Leukocyte lipid bodies - structure and function as “eicosasomes’. Trans Am Clin Climatol Assoc 2016; 127: 328-340.
- Toledo DAM, D‟ávila H and Melo RCN” Host lipid bodies as platforms for intracellular survival of protozoan parasites. Front Immunol 2016; 7(174):10.3389/fimmu. 2016.00174.
- Continguiba F, Regasini LO, Bolzani VS, Debonsi HM, Passerini GD, Cicarelli RMB, Kato MJ and Furlan M: Piperamides and their derivatives as potential anti-trypanosomal agents. Med Chem Res 2009; 18(9): 703-711.
- Fokoue HH: Síntese, atividades biológicas e estudo de relação de estrutura-atividade de piperamidas. Universidade de São Paulo 2015.
- Rhee SG, Chang TS, Jeong W and Kang D: Methods for detection and measurement of hydrogen peroxide inside and outside of cells. Molcells 2010; 29(6): 539-549.
- Green LC, Wagner DA, Glogowski J, Skipper PL, Wishhnok JS and Tannenbaum SR: Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 1982; 126(1): 131-138.
- Muniz-Junqueira MI, Peçanha LMF, Silva-Filho VL, Cardoso MCA and Tosta CE: Novel microtechnique for assessment of postnatal maturation of the phagocytic function of neutrophils and monocytes. Clin Diagn Lab Immunol 2003; 10(6): 1096-1102.
- Dias CNS: Avaliação da atividade antileishmania de produtos naturais obtidos de Phyllanthus acuminatuse de Hyptismacrostachys. Universidade Federal da Paraíba 2014.
- Berman JD: Chemotherapy for leishmaniasis: biochemical mechanisms, clinical efficacy, and future strategies. Rev Infect Dis 1988; 10(3): 560-86.
- Koff AB and Rosen T: Treatment of cutaneous leishmaniasis. J Amer Acad Dermato 1994; 31(5): 693-08.
- Elomaa O, Sankala M and Pikkarainen T: Structure of the human macrophage MARCO receptor and characterization of its bacteria-binding region. J Biol Chem 1998; 273(8): 4530-4538.
- Janeway CJ: Imunobiologia: o sistema imunológico na saúde e na doença. Editora Artes Médicas Sul Ltda, Edition 4th, 1999.
- Pick E and Keisari YA: Simple colorimetric method for the measurement of hydrogen peroxide produced by cells in culture. J Immunol Methods 1980; 38(1): 161-170.
- Pereira B: Radicais livres de oxigênio e sua importância para a funcionalidade imunológica. Motriz 1996; 2(2): 71-79.
- Queiroz SL and Batista AA: Funções biológicas do óxido nítrico. Quím Nova 1999; 22(4): 584-590.
- Gazzinelli RT, Oswald IP, Hieny S, James SL and Sher A: The microbicidal activity of interferon-gamma-treated macrophages against Trypanosoma cruzi involves a larginine-dependent, nitrogen oxide-mediated mechanism inhibitable by interleukin-10 and transforming growth factor beta. Eur J Immunol 1992; 22(10): 2501-2506.
- Adams LB, Hibbs JR, Taintor RR and Krahenbuhl JL: Microbiostatic effect of murine activated macrophages for gondii. Role for the synthesis of inorganic nitrogen oxides from L-arginine. J Immunol 1990; 144(7): 2725-9.
- Green SJ, Meltzer MS, Hibbs JB and Nacy CA: Activated macrophages destroy intracellular Leishmania major amastigotes by an L-arginine dependent killing mechanism. J Immunol 1990; 144 (1): 278-283.
- Liew FY, Millott S, Parkinson C, Palmer RMJr and Moncada S: Macrophage killing of Leishmania parasite in- vivo is mediated by nitric oxide from L-arginine. J Immunol 1990; 144(12): 4794-4797.
- Wei XQ, Charles IG, Smith A, Ure J, Feng GJ, Huang FP, Xu D, Muller W, Moncada S and Liew FY: Altered immune response in mice lacking inducible nitric oxide synthase. Nature 1995; 375(6530): 408-411.
- Bozza PT, Magalhães KG and Weller PF. Leukocyte lipid bodies-biogenesis and functions in inflammation. Biochim Biophys Acta. 2009; 1791(6): 540-551.
- Rodríguez NE, Lockard RD, Turcotte EA, Araújo-Santos EA., Bozza PT, Borges VM and Wilson ME: Lipid bodies accumulation in infantun infected C57BL/6 macrophages. Paras Immunol 2017; 39(8): 10.1111/pim. 12443.
- Rabhi S, Rabhi I, Trentin B, Piquemal D, Regnault D, Goyard S, Lang T, Descoteaux A, Enninga J and Guizani-Tabbane L: Lipid droplet formation, their localization and dynamics during Leishmania major macrophage infection. PLoS One. 2016; 11(2): e0148640.
How to cite this article:
de Araújo Vilges KM, de Oliveira SV, Fokoue HH, Kato MJ, dos Santos Borges TK, de Souza Almeida Leite JR and Kuckelhaus SAS: Evaluation of the immunomodulatory effect of piplartine and its derivative on peritoneal macrophages of Swiss mice infected with Leishmania amazonensis. Int J Pharmacognosy 2019; 6(3): 99-07. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(3).99-07.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
99-107
712
1159
English
IJP
K. M. de Araújo Vilges, S. V. de Oliveira, H. H. Fokoue, M. J. Kato, T. K. dos Santos Borges, J. R. de Souza Almeida Leite and S. A. S. Kuckelhaus
Department of Collective Health, Faculty of Medicine, Federal University of Uberlandia, Uberlândia, Brazil.
stefan@ufu.br
01 March 2019
26 March 2019
28 March 2019
10.13040/IJPSR.0975-8232.IJP.6(3).99-07
31 March 2019