EVALUATION OF PHYTOCHEMICAL SCREENING AND ACUTE TOXICITY OF SWEET POTATO LEAF MACERATE (IPOMOEA BATATAS) ON FEMALE RATS
HTML Full TextEVALUATION OF PHYTOCHEMICAL SCREENING AND ACUTE TOXICITY OF SWEET POTATO LEAF MACERATE (IPOMOEA BATATAS) ON FEMALE RATS
E. J. Ano *, Y. M. Siapo, E. J. B.ONSIYOR , D. D. S. Achi and Y. A. Tahiri
Department of Sciences and Technology, Alassane OUATTARA University, BPV 18 Bouaké 01, Ivory Coast.
ABSTRACT: In Ivory Coast, the leaves of Ipomoea batatas are consumed like spinach without any prior study of toxicity. This study aims to evaluate the toxicity of Ipomoea batatas leaf macerate on female rats. Thus, the phytochemical screening, the acute oral toxicity test of the leaf macerate of this plant was carried out on female rats. The results show that Ipomoea batatas leaves contain sterols and polyterpenes, polyphenols, flavonoids, alkaloids and saponosides. The study also reveals that the macerate of I. batatas leaves is not toxic orally. The rats showed no signs of toxicity. The weight of vital organs of female rats treated with the macerate showed no significant difference from that of control female rats. Concerning the reproductive organs, a significant increase in the weight of the ovary was observed in females’ rats treated with the extract compared to control rats. The study revealed a decrease in the level of white blood cells in the test female rats. At level of biochemical parameters, a significant decrease in AST and ALT levels was observed in rats treated with the extract. The histopathological study carried out on the heart, liver, spleen and kidney after 14 days of observation showed no damage to the vital and reproductive organs. However, the leaves of Ipomoea batatas could have an impact on the level of red blood cells and VGM by lowering them if they are used in high concentration. The leaves could also have a very positive impact on fertility by increasing the weight of the ovaries.
Keywords: Ipomoea batatas, Acute toxicity, Phytochemical screening, Female rats
INTRODUCTION: The sweet potato, Ipomoea batatas, is a perennial herbaceous plant cultivated by vegetative propagation. It is composed of more than 50 genera and 1000 species 1. It is one of the most important root and tuber plants with domestic and industrial uses. It is consumed by more than two billion people around the world 2.
Apart from the food side of the sweet potato, different parts of the plant are used in traditional medicine. In India, for example, sweet potato leaves are used in the treatment of mouth and throat tumors 3.
A decoction of the leaves is believed to be active as an aphrodisiac, astringent, demulcent, laxative, energizing, bactericidal and fungicidal agent 3. Sweet potato has also been found to be beneficial in treating various ailments such as asthma, insect bites, burns, catarrh, diarrhea, fever, nausea, stomach disorders and tumors. The leaves of Ipomoea batatas are used to treat dengue fever. In fact, they increase the number of blood platelets. In Ghana, the leaves to treat type 2 diabetes 3. In Brazil, they are used to treat infectious or inflammatory mouth and throat conditions while in Japan, raw tubers of a certain white-fleshed variety are consumed to treat anemia, high blood pressure and diabetes 3. Sweet potato fiber may be useful in combination with other therapeutic agents for skin wound therapy 4. Concerning the toxicity of sweet potato leaves, few studies mention it. While in Ivory Coast, sweet potato leaves are eaten like spinach. They are also used for animal feed. Also, young sweet potato leaves are pounded and blanched to be served with rice and meat or fish. It is for this reason that we set ourselves the objective in the present study of evaluating the toxicity of Ipomoea batatas leaves on female rats, which are even more sensitive than male rats.
MATERIALS AND METHODS :
Material: The material consists of plant material and animal material.
Vegetal Material: The vegetal material consists of leaves of Ipomoea batatas collected at the Felix Houphouet-Boigny University of Cocody. They were harvested during the months of March and April, in the morning between 8 and 10 a.m. The average temperature during harvest time is 26°C. The leaves were identified at the National Floristic Center (CNF).
Animal Material: The animal material used consists of Rattus norvegicus female rats (muridae) of Wistar strains. They come from the vivarium of the Ecole Normale Supérieur (ENS) in Abidjan. They were raised at room temperature and had ad libitum access to water and food. The rats were acclimated to laboratory conditions before the start of the experiment. The choice of this animal species is based on the fact that it is the animal model for toxicology and pharmacology studies.
Methods :
Preparation of Ipomoea batatas Leaf Macerate: The preparation of the macerate of Ipomoea batatas leaves is carried out according to the method described by 5. The fresh leaves are harvested, washed in tap water and dried in the open air. The dry leaves are crushed using a RETSCH GM 300 brand electronic grinder to obtain a fine powder. Depending on the mass of the animal, 5000 mg/kg of PC of this powder is macerated in ten (10) ml of distilled water for 24 hours. After filtration, six (6) milliliters of the macerate were given by gavage (oral route) to each animal.
Determination of Phytochemical Compounds from Ipomoea batatas Leaf Macerate: Phytochemical screening is a qualitative study based on the characterization of large chemical groups such as sterols, polyterpenes, flavonoids, tannins, phenolic compounds, quinones, saponosides, and alkaloids using standard processes 6. This study was carried out using the macerate of Ipomoea batatas leaf powder (MFIB).
Toxicological Study of the Macerate of Ipomoea batatas Leaves (MFIB): Acute toxicity which was conducted according to OCDE guideline 423 on adult female rats weighing between 180 and 200g 7. In this study, the limit test method was adopted. Thus, two batches of 5 female rats were formed. The first batch (control) received distilled water and the second was treated with a single dose of 5000 mg/kg of PC of MFIB by gavage. The animals were previously fasted for 24 hours before administration of the doses 7.
After treatment, the animals were observed regularly for the first 24 hours with particular attention for the first 4 hours then daily until the 14th day to determine possible signs of toxicity. The observation focused on various manifestations of tremor, convulsion, salivation, diarrhea, lethargy, sleep and coma. In addition, vital organs (kidney, liver, heart, spleen, lungs and adrenal glands) and reproductive organs (ovaries, tubes, uterus and vagina) were removed, weighed and preserved in formalin.
The relative masses of the removed organs were determined and histological sections of the heart, liver, spleen and kidney were made to determine the effect of MFIB on these organs. Finally, biochemical and hematological analyzes were carried out to know the effect of MFIB (5000 mg/kg of BW) on the formed elements of the blood and on the organs taken. The maximum dose of 5000 mg/kg of CP was chosen because the leaves of I. batatas are widely consumed.
The administration of I. batatas leaf macerate (MFIB) was done in three (3) doses by gavage (oral route) using a syringe according to a sequential process in which five animals are used at each stage.
Determination of the Effect of Macerate Leaves of Ipomoea batatas on the Relative Masses of Vital Organs and Reproduction : At the end of the experiment, the rats were dissected and the vital and reproductive organs were removed. The removed vital organs were rinsed in 9% NaCl and dried on paper towels. These organs were subsequently weighed. The relative mass of each organ was determined according to the following formula :
Relative mass of the organ = (Mass of the organ (mg) / (Body mass (mg)
Determination of the Effect of Macerate of Ipomoea batatas Leaves on Hematological and Biochemical Parameters: Concerning the hematological parameters, blood sampling was done by caudal puncture after inhalation anesthesia with an ethyl ether solution. Samples are collected in tubes containing EDTA anticoagulant. Hematological parameters are determined by an automatic counter. These are the level of white blood cells (WBC), the level of red blood cells (RBC), the level of hemoglobin (Hb), the hematocrit (HCT), the mean corpuscular volume (MCV), the mean corpuscular content of hemoglobin (TCMH), mean corpuscular hemoglobin concentration (CCMH).
For biochemical parameters, the animals are fasted before blood collection. The collection was done by decapitation. The blood samples collected in the dry tubes were centrifuged in a centrifuge at 4000 rpm for 10 min in order to collect the sera. The biochemical parameters that were measured are urea, creatinine, AST and ALT.
Determination of the Effect Mecerate of Ipomoea batatas Leaves on Vital Organs (Heart, Liver, Kidney and Spleen): Histology follows several stages 8. Thus, the organs after having been fixed in 10% formalin and dehydrated in increasing alcohol baths (80, 90, 96 and 100⁰C) for one hour (80⁰C) and two hours (90 ; 96 and 100°C) have was cleared in three successive baths of toluene for one hour (first bath) and two hours (second and third bath) in order to eliminate traces of alcohol in the organs 9. The organs were then impregnated in two baths of liquid paraffin in an oven at 50°C and embedded in paraffin using cassettes and molds. To facilitate unmolding, the organs were hardened in the freezer. After hardening of the organs, the sections were made using a microtome. They were then spread out on slides. The organs underwent deparaffinization through three successive toluene baths lasting 15 min each and rehydrated for 5 min in three baths of decreasing degrees of alcohol (96, 90 and 80°C). They were carefully rinsed in distilled water and then stained in a hematoxylin-eosin bath. Rinsed again with distilled water, the organs were rehydrated in an increasing bath of alcohol (80, 90 and 96) for 5 min each in order to eliminate excess eosin. The organs were cleared one last time in a toluene bath for 15 min 10. The coverslips were mounted on the slides immediately after staining the organ sections. A few drops of Eukitt inclusion were put on the organ section which was immediately covered with a coverslip. The observation and measurements were made using an OLYMPUS CX31 type microscope connected to a computer equipped with AmScope software. The magnifications (GX40 and GX100) made it possible to appreciate the tissue abnormalities of the organs.
Data Analysis : Statistical analyzes were carried out using STATISTICA software. Values are presented as mean ± standard deviation. The analysis of the results of the gains in body mass of the rats and the organs as well as the hematological and biochemical parameters of the toxicity test was carried out using the Kruskal-Wallis analysis of variance followed by the multiple comparison test of means. The signifiance threshold is set at 5%. If p<0.05, the difference is considered significant. On the contrary, the difference is not significant if p>0.05.
RESULTS :
Phytochemical Composition : Phytochemical screening showed that the leaf macerate of Ipomoea batatas contains sterols and polyterpenes, polyphenols, flavonoids, alkaloids and saponosides. On the other hand, no tannin and quinone were noted Table 1.
TABLE 1: PHYTOCHEMICAL SCREENING OF I. BATATAS LEAVE SMACE RATE
| Chemical Constituent | Macerate leaves of Ipomoea batatas MFIB | |
| Sterols and polyterpenes | + | |
| Polyphenols | + | |
| Flavonoids | + | |
| Tannins | Catechics | - |
| Gallic | - | |
| Quinones | - | |
| Alkaloids | Bouchardat | + |
| Dragendorff | + | |
| Saponosides | + | |
+ : Presence; - : Absence
Clinical Signs and Lethal Dose 50 (LD50) of Ipomea batatas Leaf Macerate (MFIB): Oral administration of 5000 mg/kg BW of MFIB did not cause any mortality in treated rats. No clinical signs of toxicity, namely tremor, convulsion, choking, salivation, diarrhea, lethargy, sleep, and coma were observed during the experiment.
Effect of Macerate Leaves of Ipomoea batatas on the Relative Mass of Vital Organs and Reproduction : The relative masses of the vital organs removed (heart, liver, spleen, kidneys and lungs) from the rats were compared to those of control rats (distilled water). The statistical analysis revealed that the relative masses of the vital organs of the rats treated with macerate of I. batatas leaves did not vary significantly (p>0.05) from those of the control rats whatever the organ studied Table 2. Concerning the reproductive organs (ovary, tube, uterus and vagina), except the ovaries where the statistical analysis showed a significant difference between the relative mass of the ovaries of the female rats treated with the extract and the control female rats (p<0. 05), at the level of the other organs no significant variation was observed in the relative masses between the rats treated with the extract and the control rats Table 2.
TABLE 2: EFFECT OF MACERATE LEAVES OF I. BATATAS ON THE RELATIVE MASS OF VITAL AND REPRODUCTIVE ORGANS OF FEMALE RATS
| Types of organs | Organs | Relative mass Control (mg) | Relative mass Treated (mg) |
|
Vital organs |
Heart | 3.838 ± 0.001 a | 3.620 ± 0.001 a |
| Liver | 31.823 ± 0.001 a | 33.693 ± 0.005 a | |
| Kidneys | 5.438 ± 0.001 a | 5.310 ± 0.001 a | |
| Spleen | 3.984 ± 0.001 a | 5.544 ± 0.003 a | |
| Lungs | 10.786 ± 0.003 a | 10.479 ± 0.004 a | |
| Adrenal glands | 0.245 ± 0.001 a | 0.199 ± 0.001 a | |
| Organs of
Reproduction |
Ovaries | 0.316 ± 0.001 a | 0.604 ± 0.001 b |
| Trompes | 1.570 ± 0.001 a | 2.260 ± 0.001 a | |
| Uterus | 0.996 ± 0.001 a | 1.625 ± 0.001 a | |
| Vagina | 1.799 ± 0.001 a | 1.682 ± 0.001 a |
On the same line, the means ± standard deviation assigned the same letter are not significantly different according to the Mann-Whitney test.
Effect of Ipomoea batatas Leaf Macerate on the Hematological Parameters of the Toxicity Test: Analysis of data for hematological parameters such as white blood cells and mean corpuscular volume showed that the rate of these parameters are significantly different in treated rats and in control rats (p<0.05). Concerning white blood cells, the analysis revealed a significant decrease at the treated rats while at the level of mean blood cell volume, it revealed an increase compared to the control. In terms of other hematological parameters studied such as red blood cells, hemoglobin, hematocrit and platelets, the analysis showed that the macerate leaves of Ipomoea batatas did not cause any significant modification compared to the female rats having received water distilled (control rats) Table 3 (p>0.05).
TABLE 3: EFFECT OF MACERATE IPOMOEA BATATAS LEAVES ON THE HEMATOLOGICAL PARAMETERS OF FEMALE RATS
| Control (0 mg/kg of BW of MFIB) | Treated (5000 mg/kg of BW OF MFIB) | |
| WBC (109/L) | 16.16 ± 1.44 b | 11.77 ± 3.65 a |
| GR (1012/L) | 6.24 ± 0.02 a | 6.31 ± 0.79 a |
| HGB (g/dL) | 11.87 ± 0.06 a | 12.07 ± 1.54 a |
| HCT (%) | 34 ± 0.27 a | 37.40 ± 4.81a |
| MCV (fl) | 54.43 ± 0.49 a | 59.33 ± 1.01 b |
| PLT (109/L) | 685 ± 2.5 a | 686 ± 2.16 a |
WBC: White blood cells; GR: Red blood cells; HGB: Hemoglobin; HCT: Hematocrit; MCV: Mean corpuscular volume; PLT: Blood platelets. On the same line, the means ± standard deviation affected the same letter are not significantly different according to the Mann-Whitney test.
Effect of Ipomoea batatas Leaf Macerate on Biochemical Parameters: The measurement of parameters such as AST and ALT revealed a difference in the level of these two parameters. Indeed, the statistical analysis showed a significant drop in these parameters in treated female rats compared to control female rats (p>0.05). Unlike these two parameters, at the level of creatinine and urea levels, the statistical analysis showed no significant difference (p<0.05) between the treated female rats and the control female rats Table 4.
TABLE 4: EFFET DU MACÉRÂT DE FEUILLES D' IPOMOEA BATATAS SUR LES PARAMÈTRES BIOCHIMIQUES
| Lots | Control (0 mg/kg of BW of MFIB) | Treated (5000 mg/kg of BW of MFIB) |
| Creatinine (mg/l) | 6.67 ± 2.08 a | 6.37 ± 0.45 a |
| Urea (mg/l) | 0.18 ± 0.04 a | 0.16 ± 0.01 a |
| AST (UI/l) | 351.67 ± 66.67 b | 136.03 ± 10.10 a |
| ALT (UI/l) | 28± 3.78 b | 27.10 ± 3.55 b |
Sur the same line, the means ± standard deviation affected the same letter are not significantly different according to the Mann-Whitney test.
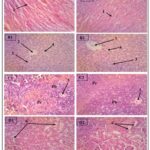
Histology of Collected Vital Organs: Histology performed on the heart, liver, spleen and kidney after 14 days of observation showed no damage to the organs. Indeed, no cellular necrosis was observed on histological sections of the heart, liver, spleen and kidney. There was no vacuolation of hepatocytes in the liver or damage to kidney cells. The spleen does not present any structural abnormalities Fig. 1.
FIG. 1: HISTOLOGICAL SECTIONS OF THE HEART, LIVER, SPLEEN AND KIDNEY FOR THE TOXICITY TEST (GX100). A1: Witness heart; A2: Heart treated; B1: Control liver; B2: Treated liver; C1: Control spleen; C2: Treated spleen; D1: Control kidney; D2: Treated kidney; 1: Cardiac muscle fibers; 2: Centrilobular vein; 3: Kupffer cell; 4: Radiated sinusoidal capillary; 5: Malpighian corpuscle; 6: Contoured tubes; Pb: White pulp; Pr Red pulp.
DISCUSSION: The phytochemical study of macerate of Ipomoea batatas leaves revealed the presence of sterols, polyterpenes, polyphenols, flavonoids, alkaloids and saponosides. The absence of tannins and quinones in the potato leaf extract was noted. These results are similar to those obtained by 11. But, 12 obtained different results. Indeed, these authors did not find alkaloids, quinones, sterols and polyterpenes in the leaf extract of I. batatas. On the other hand, they highlighted the presence of tannins in extract of the sweet potato leaves. These results differ from those obtained in this study. This could be explained by the place where the leaves were harvested and the extraction method used. Indeed, 12 collected the leaves of Ipomoea batatas in Lubumbashi (DRC) where the average temperature was 20°C. These authors dried the harvested leaves for 15 days. In our work, the leaves were harvested at an ambient temperature of 25°C and then dried for 7 day.
Administration of Ipomoea batatas leaf macerate in a single dose of 5000 mg/kg body weight caused no mortality and no signs of toxicity. This suggests that the LD50 of this extract is greater than 5000 mg/kg. This leaf extract is a substance classified as non-toxic according to the OECD.
Despite the absence of signs of toxicity and mortality, the hematological and biochemical analyzes made it possible to assess the effect of the dose of 5000 mg/kg of PC of leaves of Ipomoea batatas on the figured elements of the blood and on the tissues of heart, liver, spleen and kidneys. In terms of hematological parameters, a drop in the level of leukocytes and an increase in the MCV of the treated rats was noted. Indeed, leukocytes are involved in the body's defense and their level increases in the event of infection or in the presence of a foreign element in the body. This decline is not a negative indicator of the state of health of the rats. The VGM reflects the average size of red blood cells.
It allows the diagnosis of anemia. The increase in MCV is linked to anemia caused by a deficiency of vitamins B12 and B9 13. Thus, this extract in high doses can cause small vitamin deficiencies. As for biochemical parameters, a decrease in AST and ALT levels was noted. In fact, AST and ALT are enzymes whose dosage makes it possible to detect cellular damage in the liver, heart, kidneys or muscles. AST is specific to the liver, muscles, kidneys, brain and pancreas. ALT is specific to the liver. The drop in these biochemical parameters is linked to a vitamin B6 deficiency 13. Despite these variations, MFIB does not have a negative effect on the heart, liver, spleen and kidneys.
The administration of MFIB led to an increase in the relative mass of the ovaries of treated rats compared to those of controls. The significant increase in ovarian weight gain could be attributed to steroids or gonadotropic hormones. Indeed, the ovary can be considered as an aggregate of three endocrine tissues: the stroma, the follicle and the corpus luteum. The weights of these tissues constitute the net weight of the ovary. During the estrous cycle, the weight of ovarian tissue increases under the influence of gonadotropic and steroid hormones 14. The classic target organs of estrogens are: the mammary gland, the uterus, the ovaries, the vagina. In these organs, thanks to its anabolic and mitotic effects, estrogens stimulate cell proliferation and the biosynthesis of progesterone receptors 14.
The significant increase in the weight of the ovaries of rats treated with 5000 mg/Kg bw of MFIB compared to that of the control could indicate an increase in the activity of the stroma, follicle and corpus luteum of the ovary. This observation suggests that the macerate of Ipomoea batatas resulted in a high availability of gonadotropic or steroid hormones or both 15. Likewise, steroid genesis is one of the causes of increased weight of sexual organs. The increase in these parameters could be considered as a biological indicator of the effectiveness of the plant extract in improving the genesis of steroid hormones 16. Thus, the follicles gradually increase in size, by a progressive increase in the cell layers of the granulosa, the formation of the internal and external theca and cavities filled with fluid or antrum whose volume gradually increases up to the De Graaf follicles. These phenomena reach their maximum in the proestrus phase where the gonadotropin level is maximum 17, resulting in an increase in the weight of the ovaries 18. This phenomenon, which promotes a strong accumulation of protein in the ovary, could also explain the increase in dry weight of the ovaries.
CONCLUSION: This study shows that the leaves of Ipomoea batatas can be used in the treatment of anemia instead of standard anti-anemic drugs without any danger. The leaves of I. batatas are non-toxic substances in mammals. They contain sterols, polyterpenes, polyphenols, flavonoids, alkaloids and saponosides. The LD50 is greater than 5000 mg/kg of PC. Macerate of I. batatas leaves has no impact on vital organs. In addition, the leaves could stimulate fertility and therefore have a positive effect on reproduction. Macerate of I. batatas leaves increases the relative mass of rat ovaries. On the other hand, the extract lowered the level of white blood cells and the level of Mean Globular Volume.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Scott GJ and Ewell PE: Sweet potato in African food systems, at the IITA, Ibadan, Nigeria, International Potato Center, Lima; Peru 1992; 170.
- Lebot V, Champagne A, Malapa R and Shiley D: NIR Determination of Major Constituents in Tropical Root and Tuber Crop Flours. Journal of Agricultural and Food Chemistry 2009; 57(22): 10539–10547.
- Remya M and Subha S: Sweet Potato (Ipomoea batatas [L.] Lam) A Valuable Medicinal Food. Journal of Medicinal Food 2014; 17(7): 733–741.
- Suzuki T, Tada H, Sato E and Sagae Y: Application of sweet potato fiber to skin wound in rat. Biological and Pharmaceutical Bulletin 1996; 19: 977–983.
- Sukdev SH, Suman PSK, Gennaro L and Dev DR: Extraction technologies for medicinal and aromatic plants, International Centre for Science and High Technology 2008; 1.
- Tidiane K, Ouattara GA, Monon K, Abdoulaye T and Karamoko O: Etude phytochimique et activité antioxydante des extraits d’écorces de tiges de Vitellaria paradoxa C.F. Gaertn, une plante médicinale utilisée au Nord de la Côte d’Ivoire. European Scientific Journal 2021; 17(34): 241-251.
- OCDE: Guidelines for the testing of chemicals, revised draft guidelines 423; acute oral toxicity-acute toxic class method, revised document 2001; 14.
- Hould R: Techniques d’histologie et de cytopathologie. Editor: Centre collégial de développement de matériel didactique. Québec (Province) 1984; 399.
- Alturkistani HA, Tashkandi FM and Mohammedsaleh ZM: Histological Stains: A literature review and case study. Global journal of Health Science 2016; 8(3): 72-79.
- Kierszenbaum LA: Histologie et biologie cellulaire, une introduction à l'anatomie pathologique. Bruxelles. De Boeck 2006; 1-618.
- Milind P and Milind M: Sweetpotato as a super-food, International Journal of Research in Ayurveda and Pharmacy 2015; 6.
- Ndibualonji BBV, Pemba MG, Maryabo KG and Kaputo M: Évaluation de la valeur nutritive et recherche des substances bioactives des feuilles de deux variétés de patate douce consommées à Lubumbashi (RDC). Journal of Applied Biosciences 2016; 102: 9771-9776.
- Cardenas J: Transaminases: definition, dosage dans le sang, signification des résultats (en ligne) Consulté le 20 Août 2023 sur www.doctissimo.fr
- Curtis PS, Hanson PJ, Bolstad P, Barford JC, Randolph HP and Wilson KB: Biometric and eddy-covariance based estimates of annual carbon storage in five eastern North American deciduous forests. Agricultural and Forest Meteorology 2002; 113: 3-19.
- Shivalingappa H, Satyanarayan ND, Purohit MG, Sharanabasappa A and Patil SB: Effect of ethanol extract of Rivea hypocrateriformis on the estrous cycle of the rat. Journal of Ethnopharmacology 2002; 82: 11-17.
- Thakur S, Bawara B, Dubey A, Nandini D, Chauhan NS and Saraf DK: Effect of Carum carvi and Curcuma longa on hormonal and reproductive parameter of female rats. International Journal of Phytomedicine 2009; 1: 31-38.
- Freemann FE: Neuroendocrine control of the ovarian cycle of the rat. In “Physiology of reproduction”, knobil E. and Jimmy D. Neil (eds), 3nd edition, Raven Press. New York (USA) 2008; 613-658.
- Bleu GM: Etude phytochimique, toxicologique et pharmacologique de Passiflora foetida Linn. (Passifloraceae), une plante utilisée dans le traitement de l’infertilité féminine. Thèse de Doctorat, Université Félix Houphouët-Boigny Abidjan (Côte d’Ivoire) 2013; 188.
How to cite this article:
Ano EJ, Siapo YM, Onsyior EB, Achi DDS and Tahiri YA: Evaluation of phytochemical screening and acute toxicity of sweet potato leaf macerate (Ipomoea batatas) on female rats. Int J Pharmacognosy 2024; 11(8): 438-44. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(8).438-44.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
438-444
1228 KB
586
English
IJP
E. J. Ano *, Y. M. Siapo, E. J. B.ONSIYOR , D. D. S. Achi and Y. A. Tahiri
Department of Sciences and Technology, Alassane OUATTARA University, BPV 18 Bouaké 01, Ivory Coast.
anoehuijoachim@gmail.com
04 August 2024
23 August 2024
28 August 2024
10.13040/IJPSR.0975-8232.IJP.11(8).438-44
31 August 2024