EVALUATION OF IN VITRO ANTI CANCER ACTIVITY OF HYDROALCOHOLIC FLOWER EXTRACT OF BUTEA MONOSPERMA VAR. LUTEA
HTML Full TextEVALUATION OF IN-VITRO ANTI-CANCER ACTIVITY OF HYDROALCOHOLIC FLOWER EXTRACT OF BUTEA MONOSPERMA VAR. LUTEA
M. A. Kamble * 1, D. M. Dhabarde 1, A. R. Ingole 1 and A. P. Sant 2
Kamla Nehru College of Pharmacy 1, Butibori, Nagpur - 441108, Maharashtra, India.
University Department of Pharmaceutical Sciences 2, RTM Nagpur University, Nagpur - 440003, Maharashtra, India.
ABSTRACT: Butea monosperma var. lutea, a native of India, is commonly known as “Palash” and popularly known as “Flame of Forest.” Traditionally it has been found that flowers have antimicrobial, wound healing, antifungal, antidiarrhoeal, hypoglycemic, hepatoprotective, antioxidant, anthelmintic, anti-convulsive, antistress, antidiabetic, anti-inflammatory activity. In the present study, crude hydroalcoholic flower extract was examined for anticancer activity. To determine in-vitro anticancer activity, different concentrations of crude extract were tested on MCF-7 breast cancer cell line by 3-(4,5-dimethyl thiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The extract showed significant antiproliferative activity and a dose-dependent effect. Minimum inhibition 16.8% was shown by extract at concentration 62.5 µg/ml and maximum inhibition (46.89%) observed at 500 µg/ml. The flower extract showed activity in the potential range for further investigation of cancer cells.
| Keywords: |
Butea monosperma, MCF-7 breast cancer cell line, MTT assay, Anticancer activity, IC50
INTRODUCTION: Cancer, one of the leading causes of death worldwide, is a group of more than 100 diseases that can affect any part of the body, characterized by uncontrolled cellular growth. It is multifactorial, multifaceted and multi-mechanistic disease requiring a multidimensional approach for its control, treatment, and prevention 1. It is the third leading cause of death worldwide following cardiovascular and infectious diseases 2. The major cause of cancer is smoking, dietary imbalances, hormones and chronic infections leading to chronic inflammation 2. Although, chemotherapy is now being used as a standard treatment method 3, search for anticancer agents from natural product has increased.
To annotate the mechanism of prevention of cancer and to identify new anticancer activities some plants have been explored. The utility of these plants is increasing day by day. Naturally obtained compounds are considered safer and easily biodegradable than synthetic compounds and the problem of drug resistance observed in synthetic drugs is also reduced 4.
Butea monosperma under the family Fabaceae grows throughout the Indian subcontinent, especially in Indo-Gangetic plains. This tree grows up to 50 ft high, with clusters of flowers. It loses its leaves as the flowers develop in the month of January-march 5. The flowers are 2 cm to 4 cm in diameter; these tend to be densely crowded on leafless branches. Flowers are large, rigid racemes 15 cm long with 3 flowers together form the tumid node of the dark olive green velvety rhachis. The leaves are trifoliate. The plant parts used are bark, leaf, flower, seed and gum 6. It is mainly useful as antihelmentic, appetizer, aphrodisiac, laxative etc. 7 Moreover, it is used for ethnoveterinary medicine and as a traditional medicine for many ailments in the various parts of India and South Asia 8-11. Traditionally it has been found that flowers have anti-microbial 12, wound healing 13, anti-fungal 14, anti-diarrhoeal 15, hypoglycaemic 16, hepato-protective 17, anti-convulsive 18, anti-stress 19, anti-diabetic, antioxidant and anti-inflammatory activity 20. Moreover, they also have the property of ‘Kapha” and ‘Vata’ (Ayurveda) 21.
MATERIALS AND METHODS:
Plant Collection and Identification: Flowers were collected in January at morning from MIDC area, Butibori. The species for the proposed study was identified and authenticated as Butea monosperma var. lutea belonging to family Fabaceae at Department of Botany, RTM Nagpur University, Nagpur. The herbarium is kept in the department.
Extraction: The flowers’ petals were dried in the shade and powdered, and 100 gm of dried powder was subjected to continuous hot Soxhlet extraction with water and alcohol (ethanol 90%) at a temperature range of 55 to 65 °C. The solvent was removed under reduced pressure and controlled temperature by using rotary vacuum evaporator. Phytochemical screening of the extract revealed the presence of tannins, flavonoids, alkaloids, sterols, and terpenes.
Cell Line: Breast cancer MCF-7 cell lines 22 was obtained from NCCS, Pune, India. The cells were maintained in dulbecco’s modified eagle’s medium (DMEM) 50 µg/ml gentamicin sulphate supplemented with 10% heat-inactivated fetal bovine serum (FBS), in a humidified atmosphere (incubator) of 50 µg/ml CO2 at 37 °C. The media were changed frequently.
Reagents: DMEM, FBS and 3-(4, 5-dimethyl thiazole-2-yl) -2, 5-diphenyltetrazolium bromide (MTT) were purchased from Sigma Chemical Co. (St.Louis, MO, USA).
An in-vitro Assay for Cytotoxicity Activity (MTT Assay): The cytotoxicity of the sample on MCF-7 was determined by the MTT assay 23. Cells (1×104/well) were plated in 100 µl of medium/well in 96 well plates (Costar Corning, Rochester, NY). After 24 h when confluent growth was observed, the medium was removed, and the drug at various concentrations dissolved in maintenance medium (DMEM containing 2.5% FBS) containing 0.1% DMSO was added to each of the wells. The plates were incubated for 48 h.
After incubation, the medium was removed, and 50µl of freshly prepared MTT (2mg/ml in PBS) was added to each of the wells and incubated for 4h at 37 °C. The Formazan crystals formed were solubilized in 50 µl DMSO. The absorbance was measured at 540 nm using microtitre plate reader (Elisa reader, Bio-Tek XL-800) and percentage viability was calculated. Measurements were performed, and the concentration required for inhibition (IC50) was determined graphically. The effect of the sample on the proliferation of MCF-7 was expressed as the %cell viability & cell death using the following formulas:
% Cell death= (Control OD – Sample OD)/ Control OD × 100
RESULTS AND DISCUSSION:
In-vitro Anticancer Activity: From MTT assay, after treatment with a various concentration of the extract, the parameters like cell viability, cell death were compared with untreated (control) cells. The results for cell growth inhibition by the extract against MCF-7 cell lines for various concentrations are shown in Table 1. As the concentration increases, there is an increase in cell growth inhibition. Plants are a storehouse of “pre-synthesized” molecules that act as lead structures, which can be optimized for new drug development. In practice, a large number of chemotherapeutic agents that are currently available can be traced back to their plant source.
Some of the plant-derived compounds gained importance in anticancer therapy include paclitaxel, vincristine, podophyllotoxin, camptothecin, etc. Although there are some new approaches to drug discovery, like combinatorial chemistry and computer-based molecular modeling and design none of them can replace the importance of natural products in drug discovery and development 8-9.
Literature data proved that flavonoid and triterpenes are biologically active against different strains of bacteria as well as many human cancer cell lines 24. Flavonoids may alter hormone production and inhibits aromatase to prevent the development of cancer cells 25.
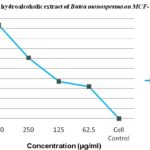
TABLE 1: IN-VITRO ANTICANCER EFFECT OF BUTEA MONOSPERMA EXTRACT ON MCF-7 CELL LINE
| S. no. | Concentration (µg/ml) | % Cell death |
| 1 | 500 | 46.89 |
| 2 | 250 | 30.42 |
| 3 | 125 | 18.62 |
| 4 | 62.5 | 16.08 |
| 5 | Cell Control | 0 |
FIG. 1: EFFECT OF HYDROALCOHOLIC EXTRACT OF BUTEA MONOSPERMA ON MCF-7 CELL LINE
CONCLUSION: The present study showed the in-vitro anticancer activity of flower extract of Butea monosperma on human breast cancer cell line (MCF-7) at increasing concentrations. Inhibitory concentration (IC50) was found to be 683.80 µg/ml.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Jemal A, Murray T, Samual A, Gafoor A, Ward E and Thun M: Cancer statistic. Cancer J Clin 2003; 53: 5-26.
- Kelloff GJ: Perspective on cancer chemoprevention research and drug development. Adv Cancer Res 2008; 78: 199-334.
- Ames BN, Gold LS and Willett WC: The cause and prevention of Cancer, Proc. Natl. Acad Sci. The USA. 1995; 92: 5258-5265.
- Uma Devi P, Selvi S, Devipriya D, Murugan S and Suja S: Antitumor and antimicrobial activities and inhibition of in-vitro lipid peroxidation by Dendrobium nobile. African Journal of Biotechnology 2009; 8(10): 2289-2293.
- Raj CN and Balasubramaniam A: Pharmacognostic and antimicrobial studies of the leaves of Tabernaemontana divaricatabr. Pharmacologyonline 2011; 2: 1171-1177.
- Kirtikar KR and Basu BD: Indian Medicinal Plants, Allahaba, India, Edition 2nd, Vol. 1, 1935: 785-788.
- Burli DA and Khade AB: A comprehensive review on Butea monosperma (Lam) Kuntze. Phcog Rev 2007; 1(2): 333-337.
- Prasad PV, Subhaktha PK, Narayana A and Rao MM: Palasa (Butea monosperma) and its medico-historical study. Bull Indian Inst History of Med Hyderabad 2006; 36: 117-28.
- Mridula BR, Sonawane AC and Thorat SR: Study of medicinal plants in North Maharashtra, University Campus of Jalgaon city in Khandesh area of Maharastra, India. Biosci Biotechnol Res Asia 2008; 5: 741-6.
- Sikarwar RLS and Kumar V: Ethnoveterinary knowledge and Practice prevalent among the tribals of Central India. J Nat Rem 2005; 5: 147-52.
- Tambekar DH and Saratkar KR: Antibacterial properties of the traditionally used medicinal plant for enteric infections by adivasi (Bumaka) in Melghat of Amravati District. Asian J Microbiol Biotechnol Environ Sci 2005; 7: 873-8.
- Mehta BK, Dubey A, Bokadia MM and Mehta SC: Isolation and in-vitro antimicrobial efficiency of Butea monosperma seed oil on human pathogenic bacteria and phytopathogenic fungi. Acta Microbiol Hungarica 1983; 30: 75-7.
- Sumitra M, Manikandan P and Suguna L: Efficacy of Butea monosperma on dermal wound healing in rats. Int J Biochem Cell Biol 2005; 37: 566-73.
- Ratnayake BM, Savitri Kumar N and Swarna S: An antifungal constitution from the stem bark of Butea monosperma. J. Ethnopharmacol 1989; 25: 73-5.
- Gunakkunru A, Padmanaban K, Thirumal P and Pritila J. Antidiarrhoel activity of Butea monosperma in experimental animals. J Ethnophamacol 2005; 98: 241-4.
- Bavarva JH and Narasimhachrya AVRL: Preliminary study on antihyperglycemic and antihyperglycemic effects of Butea monosperma in NIDDM rats. Fitoterapia 2008; 79: 328-31.
- Wagner H, Geyer B and Fiebig M. Isobutrin and butrin, the antihepatotoxic principles of Butea monosperma flowers. Planta Medica. 198; 2:77-9.
- Kasture VS, Kasture SB and Chopde CT: The anti-covulsive activity of Butea monosperma flowers in laboratory animals. Pharmacol Biochem Behav. 2002; 72: 965-72.
- Bhargavan B, Gautam AK, Singh D, Kumar A and Tyagi M: Methoxylated isoflavones, cajanin & isoformononetin, have non-estrogenic bone forming effect via differential mitogen-activated protein kinase (MAPK) signaling. J Cell Biochem 2009; 108: 388-99.
- Shahavi VM and Desai SK: Anti-inflammatory activity of Butea monosperma Fitoterapia 2008; 79: 82-5.
- Srivastava M, Srivastava SK, Khatoon S, Rawat AKS and Mehrotra S: Pharmacognostical evaluation of seed of Butea monosperma Nat Prod Sci 2002; 8: 83-9.
- Levenson AS and Jordan VC: MCF-7; the first hormone responsible for breast cancer cell line. Cancer Research 1997; 57(15): 3071-3078.
- Mosmann T: Rapid colorimetric assay for cellular grow and survival: application to proliferation and cytotoxicity assay. J Immunol Meth 1983; 65: 55-63.
- Choedon T, Shukla SK and Kumar V: Chemopreventive and anti cancer properties of the aqueous extract of flowers of Butea monosperma. J Ethnopharmacol 2010; 129(2): 208-13.
- Zhao M, Yang B, Wang J, Liu Y, Yu L and Jiang Y: Studies in Natural Product Chemistry, Nwenes 2013; 40: 306.
How to cite this article:
Kamble MA, Dhabarde DM, Ingole AR and Sant AP: Evaluation of in-vitro anti-cancer activity of hydroalcoholic flower extract of Butea monosperma var. lutea. Int J Pharmacognosy 2015; 2(4): 186-89. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(4).186-89.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
186-89
492
2429
English
IJP
M. A. Kamble *, D. M. Dhabarde , A. R. Ingole and A. P. Sant
Kamla Nehru College of Pharmacy, Butibori, Nagpur, Maharashtra, India.
manish.kamble21@gmail.com
02 February 2015
23 March 2015
29 March 2015
10.13040/IJPSR.0975-8232.IJP.2(4).186-89
01 April 2015