EVALUATION OF FICUS RACEMOSA LEAF FOR WOUND HEALING ACTIVITY
HTML Full TextEVALUATION OF FICUS RACEMOSA LEAF FOR WOUND HEALING ACTIVITY
A. Lavanya *, B. Madhava Reddy and V. Neeharika
Department of Phytochemistry and Pharmacognosy, G. Pullareddy College of Pharmacy, Mehdipatnam, Hyderabad - 500028, Telangana, India.
ABSTRACT: The wound healing activity of leaves of Ficus racemosa Linn. alcoholic, chloroform and ethyl acetate extract ointments were evaluated on Wistar rats by excision wound model for 12 days and incision wound model for 10 days. In incision model the parameter carried out was tensile strength of wounded skin. In excision model percentage wound contractions were determined for all the groups. Reference standard drug was curcumin ointment. Animals treated with extracts of Ficus racemosa Linn. showed a marked reduction in wound area in comparison to the control group from 4th day onwards. From the observations, it is found that in both the models, an alcoholic extract of Ficus racemosa and enriched diet had greater wound healing activity in terms of tensile strength in incision model and percentage wound closure, in excision model than that of other groups. Enriched diet also showed good wound healing activity which may be due to high protein content and fatty acids present in the oil.
| Keywords: |
Ficus racemosa, Alcoholic, Chloroform, Ethyl acetate and enriched diet by excision and incision models
INTRODUCTION: Ficus genus includes about 900 species. It is a well-known moderate-sized to large spreading tree distributed throughout the greater part of India in most localities, also found on rocky slopes, often cultivated in the villages of West Bengal, India, for its edible fruits. The specific identification of species is difficult, but figs as a group are relatively easy to recognize. Many have aerial roots, and a distinctive shape or habit and their fruits distinguish them from other plants. The fig fruit is an enclosed inflorescence. All figs possess a white to yellowish sap (latex). Species in this genus commonly found in India are F. altissima, F. auriculata, F. bengalensis, F. cacor, F. carica, F. cunia, F. elastica, F. hispida, F. palmata, F. pumila, F. religiosa, and F. rumphi1.
Statement of Problem: Wound can be defined as cut or break in the continuity of any tissue which may arise due to physical, chemical or microbial agents.
Wound Healing Process: 2, 3 The wound healing process is a complex series of events that starts with an injury and can continue for days to months.The entire process is a dynamic one, which can be divided into four phases. They are hemostasis, inflammatory, proliferative, maturation and remodeling. The wound-healing process is not linear and can progress forward and backward through the phases depending on various intrinsic and extrinsic factors.
Phases of Wound Healing:
- Hemostasis: When tissue is first wounded, blood comes in contact with collagen, triggering blood platelets to begin secreting inflammatory factors.
- Inflammatory Phase: The inflammatory phase is characterized by its cardinal signs: rubor (redness), calor (warmth), tumor (swelling), dolor (pain), and functionless (loss of function).
- Proliferative Phase: Fibroblasts start migrating inward from wound margins over the fibrinous matrix. They are stimulated by basic fibroblast growth factor (bFGF), and tumor growth factor-b from macrophages and platelet-derived growth factor (PDGF) from platelets.
In the first week, fibroblasts produce glycosaminoglycans (hyaluronic acid), proteoglycans, and collagen; these products are the main extracellular substances of granulation tissue.
- Maturation Phase or Remodeling: The newly synthesized collagen, which is deposited randomly in the granulation tissue, is typical for the newly formed granulation tissue (described earlier). Subsequently, the collagen is remodeled into a more organized structure with increased tensile strength.
Nutrition and Wound Healing: 4 Wound healing is a complex process of cellular and biochemical events that are dependent on the nutritional substrates available. The wound-healing phase is extremely energy demanding: The strong increase in cell proliferation, protein synthesis, and enzyme activity during the healing process requires energy and building substrates.
Review of Literature: 5-8
Taxonomy:
Kingdom: Plantae
Subkingdom: Virdaeplantae
Phylum: Tracheophyta
Sub-phylum: Euphyllophytina
Infra-phylum: Radiatopses
Class: Magnoliopsida
Subclass: Dilenidae
Super order: Urticanae
Order: Urticales
Family: Moraceae
Genus: Ficus
Species: racemosa
Vernacular Names:
- English: Cluster fig, Gular fig
- Hindi: Gular
- Telugu: Udamabaramu, bramhamedi, medi
- Sanskrit: Udamabarah
- Tamil: Atti, anjiram
- Malayalam: Atti, audamabaram
- Kannada: Attimara
Uses: Bark is used as an astringent, anti-diuretic galactagogue, refrigerant, effective in threatened abortions, employed to remove the poison from wounds made by a tiger or a cat and Useful in asthma, piles, wash for wounds and used by lactating mothers to increase the secretion of milk.
Fruits are useful in miscarriage, menorrhagia, leucoderma diseases of kidney and spleen biliousness (nausea and vomiting) and burning sensations, treatment of chronic bronchitis, dry cough, loss of voice, leprosy, diarrhea, and cancer. The unripe fruit possesses acrid, astringent, styptic, tonic properties and allays thirst is used to give tone and vitality of the body.
Leaves are used to improve complexion, treatment of bronchitis, dysentery, diarrhea, good wash for wounds and ulcers. The leaf powder mixed with honey is given in bilious affections. Latex is administered in the treatment of hemorrhoids, diarrhea, diabetes, and boils. Latex is aphrodisiac and alleviates enema in traumatic swelling and toothache.
MATERIALS AND METHODS: The leaves of Ficus racemosa Linn. were collected from G. Pulla Reddy Educational Campus, botanical garden, Mehdipatnam, Hyderabad in the month of January 2013. The collected leaves were made free from extraneous matter and air-dried. The dried leaves were powdered and stored in well-closed container.
Successive Solvent Extraction: The method is based on the extraction of active constituents present in the drug, using various solvents ranging from non-polar to polar. The solvents used were toluene, chloroform, ethyl acetate, and alcohol.
Procedure: 500g of dried powdered material of Ficus racemosa leaves was taken in a round bottom flask (RBF) and added 1.5 liters of toluene and kept for cold maceration for 2 days. The drug to solvent ratio 1:3 was maintained for maceration. The container was shaken from time to time to ensure better extraction. After 2 days the contents were filtered through Whatman filter paper no.1.
Before filtration, the container was heated to a temperature of (40 ± 2 ºC) for 30 min for better extraction (Digestion). Each time before extraction with the next solvent the powdered material was air-dried. The filtrate was concentrated under reduced pressure by rotary flash evaporator [model: PBU-6D-SUPER FIT]. The concentrated extracts were dried in desiccator.
The marc left over was extracted successively with chloroform, ethyl acetate, and alcohol (95% v/v) and processed similarly to get the respective extracts. Then the extracts were weighed, and the % of extractive values was calculated in terms of air-dried weight of the plant Material.
Enriched Diet Composition: 500g of normal feed powder + 50g of egg powder + 25g of rice bran oil.
Procedure: The eggs were boiled, and the shell was removed. Boiled eggs were chopped into small pieces, dried and powdered. To it feed powder, rice bran oil and water added and mixed to form a solid mass, and made into sticks and dried.
Preparation of Ointment: 9 The extracts were prepared in ointment form. For the preparation of extract ointment, the aqueous base was selected.
The Formula for the Aqueous Base:
| Ingredient | Quantity |
| Polyethylene glycol (PEG) 400 | 70g (62.8ml) |
| Polyethylene glycol (PEG) 6000 | 20g |
| Propylene glycol | 10ml |
Procedure: Aqueous base was prepared by adopting the fusion method in which the ingredients were added in the decreasing of their melting point. Polyethylene glycol (PEG) 6000 was taken in a china dish, melted on a water bath and to this PEG 400 and propylene glycol were added slowly with continuous stirring to avoid aeration, segregation of ingredients. Stirring was continued until the molten base was cooled to room temperature.
Incorporation of Extract into Ointment Base: Ointment base 95 parts. Extract [chloroform/ ethyl acetate/ alcohol] - 5 parts.
The extract was incorporated into the base in geometrical progression on an ointment slab with continuous mixing by using ointment spatula. It was continued until a homogenous product was formed.
Wound Healing Activity: 10, 13
Materials: Wistar rats (150-200g), curcumin ointment 2%, polyethylene glycol 400, poly-ethylene glycol 6000, propylene glycol, extracts (chloroform, ethyl-acetate and alcoholic extracts), Enriched diet.
Qualitative Phytochemical Screening: 16-17 The toluene, chloroform, ethyl acetate and alcoholic extracts of Ficus racemosa were subjected to qualitative phytochemical screening to detect the presence of various phytoconstituents present in it.
Test Animals: Wistar rats weighing between 150-200g used in the experiment are obtained from National Institute of Nutrition, Tarnaka, Hyderabad. They were maintained at standard environmental conditions of temperature, dark/light cycles and had free access to feed (nutrient animal feed) and water ad libitum during the quarantine period. The animal was fasted for 12hrs before experimentation but had been allowed free access to water.
Models: Wound healing activity was evaluated by:
- Excision wound model 11
- Incision wound model 12
1. Excision Wound Model: Wistar rats weighing between 150-200 g were selected. They were divided into groups.
Group-I: Control
Group-II: Ointment base
Group-III: Standard (Curcumin ointment-2%)
Group-IV: 5% w/w chloroform extract ointment
Group-V: 5% w/w ethyl acetate extract ointment
Group-VI: 5% w/w alcoholic extract ointment
Group-VII: Enriched diet.
The method of Mortan and Malone was adopted. The hair on the skin of the back surface of animals was removed by using a depilatory agent (Veet hair remover). Circular wounds of 10mm diameter were inflicted on the cleared skin by cutting under mild ether anesthesia.
The areas of the wounds were measured (sq mm) immediately by placing a transparent polythene graph paper over the wound and then tracing the area of wound on it. This was taken as the initial wound area reading. The wound was left undressed to open environment.
The ointments were applied topically with a fine brush twice in a day, starting from the day of operation till the complete epithelisation occurs. The parameters studied were percentage closure of excision wound and time of epithelisation.
The wounds were traced on sq mm graph paper on 0, 2nd, 4th, 6th, 8th, 10th and 12th days until healing was completed. The period of epithelisation was calculated as the number of days required for falling of scar without any residual raw wound. The degree of wound healing was calculated as percentage closure in wound area from the original wound area using the formula:
Percentage wound closure = Wound area on day 0 – Wound area on specific day/ Wound area on day 0 ×100
The mean, S.E.M values were calculated.
2. Incision Wound Model: Wistar rats weighing between 150-200gm were selected. The rats were divided into 7 groups same as excision model. The method of Ehrlich and Hunt was adopted. Each animal was secured to the operation table in its natural position under light ether anesthesia. Two para-vertebral straight incisions of 6 cm each are made on the depilated back of the animal by cutting through the entire skin with a sterilized sharp blade. Care was taken to see that the incision was at least 1 cm lateral to the vertebral column.
FIG. 1: SPECIAL FABRICATED INSTRUMENT TO MEASURE TENSILE STRENGTH
After complete hemostasis, the wounds were closed using interrupted sutures placed at equidistant points about 1cm apart, using 4-0 silk thread and curved needle (no-00). The animals were caged separately. The animals were divided into 7 groups. The grouping was similar to that of the excision wound model. Ointments were applied topically with a fine brush once daily for 7 days. On the 8th post wounding, day sutures were removed.
Tensile strength was measured on 10th post-wound day using locally prepared instrument by increasing the weight on the instrument. The breaking point of tissue was determined. Anesthetized animals were secured on to the operation table in its normal position, and allies forceps were firmly applied on either side of the incision wound 3 mm away from the wound margin on adjacent normal skin.
The forceps on one side was hooked to a fixed metal rod, while the other forceps was attached to a thread suspended by weight running over a pulley. As soon as gapping of the wound occurred, the addition of weights was stopped and simultaneously the weights were lifted so as to avoid opening of the entire wound. The weights required to produce gapping was noted. The tensile strength of the wound is expressed in minimum weight required to cause gapping of the wound.
Statistical Analysis: The results are expressed as mean ± S.E.M.
Biochemical Estimation in Incision Model: 14-15 The granulation tissue formed on day 8 of wound creation was used for further biochemical studies. About 50 mg tissue was washed in physiological saline and cut into pieces, defatted with chloroform: methanol (2:1) and lyophilized with 5ml of 6N Hydrochloric acid for 20hours in sealed tubes. After hydrolysis, the sample was evaporated to dryness to get the residue. The residue obtained was dissolved in water and made up to 3ml which was used for the estimation of hydroxyproline and total protein.
Determination of hydroxyproline:
Reagents: 0.01M copper sulphate solution, 2.5N sodium hydroxide, 6% hydrogen peroxide, 3N sulphuric acid, 5% p-dimethylaminobenzaldehyde in n-propanol.
Procedure: For the assay of an unknown solution, nine dry test-tubes were used. Standard hydroxyproline 5,10,15 and 20 µg/ml and distilled water(blank)and unknown solution are taken in different test tubes (in duplicate) and added 1ml of 0.001M copper sulphate solution, 1ml of 2.5N sodium hydroxide and 1 ml of 6% hydrogen peroxide were added successively.
The solutions were mixed and shaken occasionally for 5 minutes and then placed in a water bath at 80ºc for 5 min with frequent vigorous shaking. The heating and shaking destroy excess of peroxide. Traces of peroxide which remain will decreases color formation and produce an Orange-red blue.
The tubes were chilled in an ice and water bath, and 4ml of 3N sulphuric acid was added with agitation, then 2 ml of 5% p-dimethyl-aminobenzaldehyde solution was added with thorough mixing. The tubes were placed in a water bath at 70 ºC for 16 min and then cooled in tap water. The contents were transferred to selected absorption tubes, and light transmission was read at 540 nm with an Elico double beam spectro-photometer.
The amount of hydroxyproline (measured in micrograms) in 1 ml of an unknown solution was established by finding the point corresponding to its optical density on the standard curve prepared at the same site.
Estimation of Total Protein: Godkar et al., 1994 modified biuret endpoint assay method was used for the estimation of total proteins and analyzed in auto-analyzer (STAR 21 PLUS, manufactured by Aspen diagnostics Pvt. Ltd).
The peptide bonds of proteins react with cupric ions in alkaline solution to form a colored chelate, the absorbance of which was measured at 578nm. The biuret reagent contains sodium potassium tartrate, which helps in maintain solubility of this complex at alkaline pH. The absorbance of the final colour is proportional to the concentration of total protein in the sample. All the above solutions were mixed well and Incubated at 37ºC for 5 min and analysed in an auto analyser.
Total protein concentration (g/dl) = Absorbance of test × 6.5
Standard concentration of protein = 6.5
RESULTS AND DISCUSSION:
Extraction: 500g of leaf powder was successively extracted by cold maceration process with toluene, chloroform, ethyl acetate and alcohol, and the percentage yields were found to be:
- Chloroform extract - 98g (0.59%)
- Ethyl acetate extract - 14g (0.62%)
- Alcoholic extract - 53g (2.70%)
Qualitative Phytochemical Screening: The extracts are subjected to phytochemical screening to detect the presence of various phytochemical constituents as per the standard reported test procedures.
Wound Healing Activity:
Excision Wound Model: 18 The chloroform, ethyl acetate, alcoholic extracts and enriched diet were tested for excision wound healing activity. Quantitative measurements of wound size are routinely used to assess initial wound size before and after debridement, as well as progress toward wound closure. The wound contraction rate was measured as the percentage reduction in wound size on day 2, 4, 6, 8 and 12.
TABLE 1: QUALITATIVE PHYTOCHEMICAL SCREENING OF FICUS RACEMOSA LEAF EXTRACTS
| S. no | Chemical test | Chloroform extract | Ethyl acetate extract | Alcoholic extract |
| 1 | Alkaloids
Dragendroff’s test |
+++ | ++
|
+++
|
| 2 | Glycosides
Borntrager’s test Legal test Killer-killani test |
- - + |
- - + |
- - + |
| 3 | Carbohydrates
Molish’s test Benedict’s test Fehling’s test |
+++ - - |
+++ - - |
+++ + + |
| 4 | Steroids
Libermann-Burchard test Liebermann’s test Salkowski test |
+++ +++ +++ |
+++ +++ +++ |
+++ +++ +++ |
| 5 | Phenols and tannins
Ferric chloride test Shinoda test Lead acetate test Gelatin test |
- - + - |
- ++ + - |
+ +++ +++ - |
| 6 | Saponins (foam test) | + | + | + |
| 7 | Proteins and Amino acids
Ninhydrin test Biuret test |
- - |
- - |
- - |
‘+’ Presence of chemical constituents ’-‘Absence of chemical constituents ‘++/+++’ Presence of chemical constituents more intensely.
TABLE 2: EFFECT OF FICUS RACEMOSA LINN. EXTRACTS ON CLOSURE OF WOUND AREA ON POST WOUNDING DAYS
| Post Wounding days | Wound area -mm2 ± S.E.M (% closure of wound area) | ||||||
| Group-1 | Group-2 | Group-3 | Group-4 | Group-5 | Group-6 | Group-7 | |
| Control | Ointment
base |
Standard curcumin 2% w/w | Alcoholic extract 5%
w/w ointment |
Chloroform extract 5% w/w Ointment | Ethyl acetate extract 5%
w/w Ointment |
Enriched diet animals | |
| Day 0 | 78.50±2.6
(0%) |
79.65±3.2
(0%) |
78.95±2.4
(0%) |
78.35±2.9 (0%) | 79.85±3.1
(0%) |
80.05±3.4
(0%) |
80.35±2.7 (0%) |
| Day 2 | 74.25±2.9 (5.4%) | 71.66±3.6 (10%) | 69.37±2.8 (12.1%) | 67.90±3.4 (13.3%) | 72.36±3.1 (9.38%) | 73.87±3.4 (7.72%) | 68.39±2.9 (14.8%) |
| Day 4 | 68.32±3.2 (12.9%) | 65.25±2.7 (18%) | 52.39±3.5 (33.6%) | 49.15±3.6 (37.2%) | 61.27±2.8 (23.2%) | 66.21±2.9 (17.2%) | 51.12±2.4 (36.37%) |
| Day 6 | 65.56±2.7 (16.48%) | 63.86±2.5 (19.82%) | 35.79±3.1 (54.6%) | 34.54±2.2 (55.9%) | 48.19±2.8 (39.6%) | 55.40±3.4 (30.7%) | 34.90±3.5 (56.5%) |
| Day 8 | 62.47±3.1 (20.42%) | 59.37±2.2 (25.4%) | 24.53±2.98 (68.9%) | 23.90±2.4 (69.4%) | 36.19±3.4 (54.6%) | 45.28±3.2 (43.4%) | 24.51±2.2 (69.4%) |
| Day 10 | 59.54±2.3 (24.15%) | 56.49±3.1 (29%) | 15.13±2.4 (80.8%) | 15.07±2.8 (80.7%) | 26.73±3.2 (66.5%) | 35.82±2.6 (55.2%) | 16.25±2.4 (79.7%) |
| Day 12 | 56.84±2.56 (27.5%) | 54.37±2.9 (31.7%) | 3.31±2.6 (95%) | 3.78±3.4 (95.1%) | 18.35±2.8 (77%) | 26.41±3.1 (67%) | 5.03±2.2 (93%) |
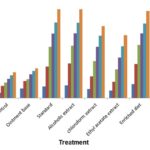
FIG. 2: PERCENTAGE CLOSURE OF EXCISION WOUND AREA
Significant progress in the percentage of wound contraction was observed in the treated excision wounds compared with the untreated control. On days 6, 8, 10, and 12, the alcoholic extract treated and enriched diet animals had significantly increased the percentage of wound contraction compared to chloroform, ethyl acetate extracts. Results were comparable to that of the standard. The percentage wound contraction for alcoholic extract, enriched diet and standard are 95.1%, 93%, and 95% respectively. Results are shown in Table 2, Fig. 2.
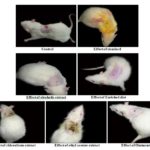
Photographs Showing the Progress of the Incision Wound Model:
FIG. 3: PHOTOGRAPHS SHOWING THE PROGRESS OF EXCISION WOUND MODEL
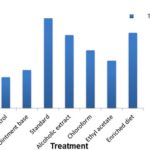
Incision Wound Model: 10 In incision model, the tensile strengths of the wounds were measured on the 10th post-wounding day by using a specially fabricated instrument. The tensile strength is determined by the weights required to break the stitch. The smaller weight which can break the stitch indicates poor wound healing. As the weights increase to break, the stitch indicate perfect and complete wound healing. The tensile strength was tested for alcoholic, chloroform, ethyl acetate extracts, standard (curcumin 2%) and enriched diet. Among which alcohol extract and enriched diet were significantly increased the tensile strength and the results are comparable to that of standard curcumin. Chloroform and ethyl acetate extract treated animals showed less effect compared to alcoholic extract. The tensile strength of curcumin-treated animal (899gm), enriched diet animal (754gm), alcoholic extract treated animal (702gm), chloroform extract treated animal (584gm) was more than ethyl acetate extract treated animal (469gm), ointment base (394gm) and control (322gm). The detailed results are shown in Table 3, Fig. 4.
TABLE 3: EFFECT OF FICUS RACEMOSA LINN LEAF EXTRACTS ON TENSILE STRENGTH OF RESUTURED INCISION WOUND ON 10th POST WOUNDING DAY
| Group | Treatment | Tensile strength (Grams ± S.E.M) |
| I | Control | 302±6.08 |
| II | Ointment base | 375±5.72 |
| III | Standard (Curcumin 2%w/w) ointment | 882±8.27 |
| IV | Alcoholic extract ointment (5%w/w) | 718±12.2 |
| V | Chloroform extract ointment (5%w/w) | 566±8.03 |
| VI | Ethyl acetate extract ointment (5%w/w) | 465±5.34 |
| VII | Enriched diet animals | 741±7.82 |
Values are mean ± S.E.M, n= 6.
FIG. 4: TENSILE STRENGTH OF INCISION WOUNDS ON 10TH POST WOUNDING DAY
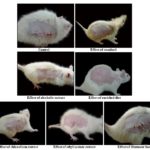
Photographs Showing the Progress of the Incision Wound Model:
FIG. 5: PHOTOGRAPHS SHOWING THE PROGRESS OF INCISION WOUND MODEL
Biochemical Estimation in the Incision Model: Biochemical estimations like hydroxyproline and protein content in the incision model further supports the wound healing activity.
Estimation of Hydroxyproline: Hydroxyproline is one of the biochemical parameter implicated in the wound healing process. Hydroxyproline stimulates collagen formation. Therefore, estimation of hydroxyproline is yet another evidence for the wound healing effect of the extracts. Hydroxyproline is estimated in the granulation tissue of the wound as per the literature reported procedure. Hydroxyproline was higher in animals treated with alcohol extract, enriched diet and curcumin comparable to chloroform and ethyl acetate extracts. Results are shown in Table 4, Fig. 5.
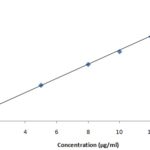
A representative calibration graph of absorbance Vs. concentration resulted in the regression equation. (Y=mx).
Y = 0.006x, R2 = 0.998
Where, Y = Absorbance, m = slope = 0.006, x = Concentration.
The calibration curve was found to be linear from 5 - 15µg/ml with coefficient of 0.998.
FIG. 6: STANDARD CALIBRATION CURVE OF HYDROXYPROLINE
TABLE 4: ESTIMATION OF HYDROXYPROLINE CONTENT IN GRANULATION TISSUE
| Group | Hydroxyproline in µg/50mg tissue |
| Control
Ointment base standard alcohol chloroform Ethyl acetate Enriched diet |
3.38 µg/ml
3.70 µg/ml 13.38 µg/ml 12.74 µg/ml 9.64 µg/ml 8.38 µg/ml 13.06 µg/ml |
Estimation of Total Protein: Total protein is another biochemical parameter implicated in the wound healing process. There is an increase in protein content, which was predominantly due to enhanced collagen synthesis. Total protein content was more in animals treated with alcoholic extract, enriched diet and standard (curcumin) compared to chloroform and ethyl acetate extracts. Results are shown in Table 5.
TABLE 5: EFFECT OF FICUS RACEMOSA EXTRACTS ON TOTAL PROTEIN
| Group | Total protein concentration(g/l) |
| Control
Ointment base standard alcohol chloroform Ethyl acetate Enriched diet |
16.50
17.20 48.18 47.20 36.38 30.48 47.69 |
DISCUSSION: 19-21 Wound healing is a stepwise process, which consists of different phases such as hemostasis, inflammation, proliferative and remodeling or maturation.
The genetic response regulating the body’s cellular resistance mechanisms contributes to the wound and its repair. Therefore, in this study, two different models were used to establish the healing potential of alcohol, ethyl acetate and chloroform extracts of Ficus racemosa and by using an enriched diet.
In the excision wound healing model the alcohol extract of F. racemosa and enriched diet showed a significant increase in percentage wound closure by enhanced epithelialization. This enhanced epithelialization may be due to the effect of F. racemosa extracts on enhanced collagen synthesis.
In incision wound, the increase in tensile strength of treated wounds may be due to the increase in collagen concentration and stabilization of the fibers. A healing tissue synthesizes collagen, which is a constituent of the growing cell. Increase in blood vessels and the role of antioxidants were experimentally proved. The higher tensile strength indicates better healing of wounds. Thus it supports the wound healing activity of Ficus racemosa.
From the observations, it was evident that Ficus racemosa possesses a definite potential healing action. The tensile strength of the incision wounds was increased in alcohol extract and enriched diet groups. The wound healing activity of extracts is further evidenced by the estimation of hydroxyproline, and total protein content which was found to be at higher levels in alcoholic extract and enriched diet treated animals compared to control.
Recent studies with other plant extracts have shown that phytochemical constituents like flavonoids, alkaloids, saponins, and tannins are known to promote the wound-healing process.
The study reveals that alcohol extract possesses good wound healing activity which may be attributed to the individual or combined action of phytoconstituents like, flavonoids, alkaloids, saponins, and tannins present in it.
Enriched diet also showed good wound healing activity which may be due to high protein content and fatty acids present in the oil.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kirtikar KR and Basu BD: Indian medicinal plants, International Book Distributors, Dehradun 1996: 3, 2327.
- Torre De la JI and Chambers JA: Wound healing chronic wounds. Emedicine.com 2008.
- Midwood KS, Williams LV and Schwarzbauer JE: Tissue repair and the dynamics of the extracellular matrix. The International Journal of Biochemistry and Cell Biology 2004: 36: 1031.
- Seiler WO and Regeniter A: Dekubitusprophylaxe and the rapieausernahrungsmedizinischer Sicht, Erna hrung in der Chirurgie 2005: 25: 5.
- longman O: Indian Medicinal Plants, A Compendium of 500 species, Orient longman Ltd., Madras 1996; 4, 34.
- Anonymous: The Wealth of India raw materials, CSIR, New Delhi, 1956: 4, 35.
- K.M. Dr. Nadakarni, The India Materia Medica (Popular Prakashan Pvt. Ltd.,), 1976: 1, 548.
- http,//bsienvis.nic.in/medi.htm.
- Carter SJ: Ointments, Pastes and Jellies in cooper and Gunns, Dispensing for pharmaceutical studies, 12th edition (CBS Publishers and Distributors, New Delhi), 2000: 192.
- Murti K and Kumar U: Antimicrobial Activity of Ficus benghalensis and Ficus racemosa roots. American Journal of Microbiology 2011; 2: 21.
- Morton JJ and Malone MH: Evaluation of vulnerary activity by open wound procedure in rats. Archives Internationales de Pharmacodynamie et de Thérapie. 1972; 196: 117.
- Ehrlich HP, Tarver H and Hunt TK: Inhibitory effects of vitamin E on collagen synthesis and wound repair, Ann Surg 1972; 175: 235.
- Wolfgang Vogel H, Bernward A, Sandow SJ, Müller G and Wolfgang F: Vogel Drug Discovery and Evaluation Pharmacological Assays: Healing of skin wounds 2002; 1360.
- Robert Neuman E and Milan A and Logan: The determination of Hydroxyproline, University of Cincinnati 1949: 299.
- Marium A and Abu-al-basal: The Healing potential of Rosmarium officinalis Linn. on full thickness excision cutaneous wounds in alloxan-induced diabetic BALB/C mice, Journal of Ethno-pharmacology, 2010; 131: 443.
- Khandeiwal KR: Practical pharmacognosy techniques and experiments, Nirali Prakashan, 2004: 19, 149.
- Kokate CK: Practical pharmacognosy, Vallabh Prakashan, New Delhi, Edition 4th, 1994: 107.
- Vadivelarul and Reenakartha: A therapeutic approach for diabetic wound healing using biotinylated GHK incorporated collagen matrices, Journal of Life Sciences, 2007; 80: 275.
- Pattanayak SP and Sunita P: Wound healing anti-microbial and antioxidant potential of Dendrophthoe falcate (L.f) Ettingsh, J Ethnopharmacol 2008; 120: 241.
- Allen LV, Popovich NG and Ansel HC: Ansel’s pharmaceutical dosage form and drug delivery system, Baltimore: Lippincott Williams and Wilkins, 2005: 278.
- Murti K, Lambole V, Panchal M, Shah M and Gajera V: Evaluation of wound healing activity of polyherbal formulation in rats, Res J Pharmacognosy Phytochem 2011: 3: 112.
How to cite this article:
Lavanya A, Reddy BM and Neeharika V: Evaluation of Ficus racemosa leaf for wound healing activity: Herbal remedy for wound management: a review. Int J Pharmacognosy 2016; 3(12): 501-12. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(12).501-12.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
501-512
720
2018
English
IJP
A. Lavanya *, B. Madhava Reddy and V. Neeharika
Department of Phytochemistry and Pharmacognosy, G. Pullareddy College of Pharmacy, Mehdipatnam, Hyderabad, Telangana, India
lavanya.svkk@gmail.com
28 October 2016
21 November 2016
26 December 2016
10.13040/IJPSR.0975-8232.IJP.3(12).501-12
31 December 2016