EVALUATION OF ANTIMICROBIAL ACTIVITY AND BIOACTIVE PHYTOCHEMICAL PROPERTIES OF MANGO (MANGIFERA INDICA) STEM-BARK EXTRACTS
HTML Full TextEVALUATION OF ANTIMICROBIAL ACTIVITY AND BIOACTIVE PHYTOCHEMICAL PROPERTIES OF MANGO (MANGIFERA INDICA) STEM-BARK EXTRACTS
Odangowei I. Ogidi * 1, Chioma C. Okore 2, Udeme M. Akpan 3, Mike N. Ayebabogha 4 and Chidinma J. Onukwufo 1
Department of Biochemistry 1, Department of Science Laboratory Technology 3, Federal Polytechnic, Ekowe, Bayelsa State.
Department of Environmental Biology 2, Federal Polytechnic Nekede Owerri, Imo State.
Department of Microbiology 4, Niger Delta University, Wilberforce Island, Bayelsa State.
ABSTRACT: The spread of infectious diseases in the world is a major challenge for health institutions and pharmaceutical industries. The current trends of multi-drug resistance to the available synthetic drugs or antibiotics among emerging and re-emerging bacterial pathogens lead to serious risks. It is therefore necessary to search for new antimicrobial agents that are better, cheaper and without side effects for treating these infectious diseases, especially in developing countries. In this study, phytochemical composition and antimicrobial activities of aqueous and methanol extracts of stem-barks of Mangifera indica were investigated. Standard methods were employed to screen for the presence of phytochemicals. Agar well diffusion method was used to determine the antimicrobial effects of aqueous and methanolic extracts of M. indica stem-bark against selected bacterial (Shigella sp, Staphylococcus sp, Escherichia coli, Vibrio sp) and fungi (Penicillium sp, Yeast, Mould) isolates. Phytochemical results showed the presence of active pharmacological components such as tannins, saponins, glycoside, flavonoid, terpenoid, alkaloids, and steroid. Methanol extract demonstrated the highest activity of bacterial (Staphylococcus sp with 15.4 ± 0.36 mm zone of inhibition) and fungi (Penicillium sp with 9.3 ± 0.2 mm zone of inhibition). Meanwhile, in aqueous extracts, Escherichia coli (10.6 ± 0.2 mm) and Penicillium sp (10.3 ± 0.3 mm) were observed to have a higher zone of inhibition. M. indica stem-bark exhibited significant antimicrobial activity; this, therefore, suggests that the extracts are as good as other commercially sold antibiotics in inhibiting these microorganisms and could possibly serve as an alternative.
| Keywords: |
Antimicrobial, Phytochemicals, Mangifera indica, Stem- bark, Extracts, Medicinal
INTRODUCTION: Continuous spread of infectious diseases is a major apprehension for health institutions and pharmaceutical companies all over the world.
Failure of treatment, particularly with the current escalating trends of multi-drug resistance (MDR) to the available modern drugs or antibiotics among emerging and re-emerging bacterial pathogens, leads to serious risks 1.
Serious attention is being given to medicinal plants, as evidenced by the recommendation given by the World Health Organization (WHO) in 1970. It emphasized on the need to include traditional remedies within national drug policies as these plants serve as the best sources of a variety of drugs. It is important to study plants so that a better understanding of their properties, safety, and efficacy is derived for improved benefit. Investigation of African medicinal plants for their antimicrobial activity rank highest among biological tests carried out on the plants and their isolates 2. In view of this, medical practitioners, whether allopath (medical doctors), homeopaths, naturopaths, herbalists, or shamans, had to know the plants in their areas and how to use them since many of their drugs were derived from plants 3-6.
Plant-derived products like gums, oils, and extracts have been used for therapeutic purpose before the introduction of modern drugs 7 and continue to provide health coverage for over 80% of the world’s population 8, however, in the decades that followed, the development of synthetic drugs from petroleum products caused a sharp decline in the pre-eminence of drugs from live plant sources 9-11. Until the recent trend of high percentage resistance of microorganisms to the present day antibiotics, efforts have been intensified by researchers towards the search for more sources of antimicrobial agents 1, 12.
Mangifera indica, commonly called mango in English, locally known as mangoro (Yoruba), mangolo (Igbo), mangwaro (Hausa), and Ogboin (Izon) belongs to the family Anacardiaceae which consists of about sixty genera and six hundred species, which are mainly tropical trees shrubs. It is widely used as a source of food, medicines and timber. Mango stem bark is the major by product of any mango processing industry. This waste product causes tremendous investment of capital to decompose safely and to prevent any environmental pollution. Therefore, its conversion to produce bioactive compounds becomes eminent to save the food processing industries a huge sum of capital. In Nigeria, different parts of the plant are commonly used as a dentifrice, antiseptic, astringent, diaphoretic, stomachic, vermifuge, tonic, laxative, and diuretic. It is also used as herbal preparations in the treatment of diarrhea, dysentery, anaemia, asthma, bronchitis, cough, gastrointestinal disorders, hypertension, insomnia, rheumatism, toothache, gastrointestinal tract infections, respiratory and urinary tract infections, sore-gums, sore throats, leucorrhoea, haemorrhage, and pile. The mango stem-bark contains Mangiferine and is astringent and employed against rheumatism and diphtheria in India. The leaves are used for floral decoration at Hindu marriages and religious ceremonies 13.
The objective of this study was to investigate the antimicrobial activity and bioactive phytochemical properties of mango (Mangifera indica) stem-bark medicinal plants.
MATERIALS AND METHODS: Samples of stem-bark were obtained from Mangifera indica from Ekowe community in Southern Ijaw Local Government Area of Bayelsa State and were identified in the School of Agricultural Technology, Federal Polytechnic, Ekowe, Bayelsa State, Nigeria.
Preparation of Plant Materials: Freshly collected stem-bark of M. indica were washed with distilled water and dried under the shade at normal room temperature for 15 days. After drying, the plant material was pounded using mortar and pestle into smaller particles and then blended to powder using an electric blender. 200 g of the powdered samples were stored in airtight containers and kept under normal room temperature for further screening.
Mango stem-bark Sample Extraction: The extraction reagents were methanol and aqueous. About 10 g of the pulverized mango stem-bark sample was placed in a beaker, and 25 ml of methanol added and mixed by vortexing. It was centrifuged at 3000 rmp for 10 min. The supernatant was collected and transferred to a stoppered test tube by filtration. The resulting supernatant was evaporated to dryness with a gentle stream of nitrogen and reconstituted in 10 ml dimethyl sulphoxide and was mixed by vortexing. The same procedure was repeated for that of aqueous.
Preparation of Dried Filter Paper Discs: Whatman filter paper no. 102 was used to prepare discs. Approximately 5 mm in diameter was perforated using a perforator. These were placed in a petri dish after sterilization in an autoclave.
Mango stem-bark Extract Disc Placement: Mango stem-bark disc containing 3 ml (3 µl) concentration, as well as mango bark, were made using filter paper and then placed on the plates using sterile forcep. One sterile antibiotic disc was placed on the surface of an agar plate using a forceps. The forceps was sterilized by immersing in alcohol each time before placing another antibiotic disc. The disc was then gently pressed with the forceps to ensure complete contact with the agar surface and placed away from the edge of the plates so that it is easily measured. Once all discs were in place, the plates were inverted and placed in a 37 °C incubator for 24 h.
Bacteria/ Fungi Suspension Preparation: Media used: Nutrient agar, buffered peptone water, shigella agar, McConkey agar, and cetrimide agar. These media were prepared according to manufacturer’s instruction. Using a sterile inoculating loop and needle for bacteria and fungi, respectively, through aseptic techniques the test organisms of each colony were taken from the subculture plate. The organism was suspended in 4 ml of normal saline and vortexed for overall suspension. Mcfarland standard solution was used as a reference to adjust the turbidity of individual bacterium isolate in the suspension (1 × 108). And 10 fold serial dilutions were made and plated for the antimicrobial sensitivity test.
Inoculation of Isolates on the Nutrient Agar Plate Proper: A sterile swab stick was dipped into the bacterial/ fungi suspension, and the test organisms were suspended in 4 ml of buffered peptone water.
The swab was rotated against the side of the tube using firm pressure to remove excess fluid, but the swab was not dipped wet. The dried surface of the nutrient agar plate was inoculated by streaking the swab over the entire agar surface by rotating the plate at 60 degrees each time to ensure an even distribution of the inoculum.
Phytochemical Screening: Phytochemical tests for the screening and identifying bioactive chemical constituents in the plant under study were carried out using the standard procedures as previously described 14.
Qualitative analysis of phytochemical constituents: Tannins: The powdered mango bark sample (0.5 g) was boiled in 20 ml of distilled water in a test tube and filtered, 0.1% FeCl3 was added to the filtered samples and observed for brownish green or a blue-black colouration which shows the presence of tannins.
Saponins: The powdered mango stem-bark sample (2.0 g) was boiled in 20 ml of distilled water in a water bath and filtered off; the filtrate was mixed with 5ml of distilled water in a test tube and shaken vigorously to obtain a persistent table froth. The frothing is then mixed with 3 drops of olive oil and for the formation of emulsion, which indicates the presence of saponins.
Flavonoids: A few drop of 1% NH3 solution was added to the aqueous extract of the plant sample in a test tube. Yellow coloration is observed if flavonoids compound is present.
Glycosides: Concentrated H2SO4(1ml) was prepared in a test tube, 5 ml of aqueous extract from the powdered mango stem-bark sample was mixed with 2 ml of glacial CH3COOH containing 1 drop of FeCl3. The above mixture was carefully added to 1ml of concentrated H2SO4 so that the concentrated H2SO4 settled beneath the mixture. The presence of cardiac glycoside constituent was indicated by appearance of a brown ring.
Alkaloids: The plant sample (5.0 g) was prepared in a beaker and 200 ml of10% CH3COOH in C2H5OH was added to the plant sample nearly 0.5g.
Terpenoid: Mango stem-bark sample (1 g) was marcarated with 50 ml of ethanol and filtered. To the filtrate (2.5 ml), 2.5 ml of 5% aqueous phosphomolybdic acid solution was added and 2.5 ml of concentrated H2SO4 was gradually added and mixed. The mixture was left to stand for 30 min and then made up to 12.5 ml with ethanol. The absorbance was taken at 700 nm.
Total Steroid: The steroid content of the plant sample was determined using the method described by Trease and Evans 15. A portion of 2 ml was taken from a solution of 2.5 g of powdered plant material prepared in 50 ml of distilled water after vigorous shaking for 1 hour. The extract solution was washed with 3 ml of 0.1M NaOH (pH 9) and later mixed with 2 ml of chloroform and 3 ml of ice cold acetic anhydride followed by the cautious addition of two drops of concentrated H2SO4. The absorbance of both sample and blank were measured using a spectrophotometer at 420 nm.
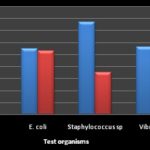
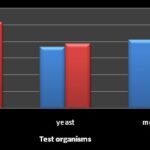
RESULTS AND DISCUSSION: The characteristics of the biochemical test of Shigella sp, E. coli, Staphylococcus sp and Vibrio sp and the cultural morphology of fungi isolates are presented in Table 1 and 2. The antimicrobial activities of aqueous and methanol mango bark extracts on both bacterial and fungi isolates are shown in Table 3 and 4. Methanol extract gave the best result in bacterial and fungi with highest zone of inhibition against Staphylococcus aureus (15.4±0.36 mm) and Penicillium sp (9.3±0.2 mm) respectively. Meanwhile, for aqueous extract, the plant samples gave the best results in bacterial and fungi with highest zones of inhibition of 10.6±0.2 mm and 10.3 ± 0.3 mm against Escherichia coli and Penicillium sp respectively. This result shows that mango bark extract was active against gram positive and gram negative bacteria as well as fungi. These findings are in conformity with the works of Chukwudebe et al., 16 and Mada et al., 17.
TABLE 1: BIOCHEMICAL CHARACTERISTICS OF BACTERIA ISOLATES
| Microorganisms | Shigellasp | Escherichia coli | Staphylococcus sp. | Vibrio sp | |
| Cell morphology (cell shape) | Rod | Rod | Coccus | Comma | |
| Colony (cell shape) | Round | Spindle | Circular | Curved | |
| Gram reaction | Negative | Negative | Positive | Negative | |
| Biochemical Test | Nitrate reductive | Positive | Positive | Positive | Positive |
| Oxidase | Negative | Negative | Negative | Positive | |
| Catalase | Positive | Positive | Positive | Negative | |
| Methyl red | Positive | Positive | Positive | Negative | |
| V.P. | Negative | Negative | Positive | Positive | |
| Indole | Negative | Positive | Negative | Positive | |
| Citrate | Negative | Negative | Positive | Positive | |
| H2S reduction | Negative | Negative | Negative | Negative | |
| Ureas activity | Negative | Negative | Positive | Negative | |
Note: VP = Voges Proskauer; H2S = Hydrogen sulfide
TABLE 2: IDENTIFICATION OF FUNGI WITH CULTURAL MORPHOLOGY
| Organisms | Microscopic observation (Medium) | Microscopic observation (gram reaction) |
| Yeast | White colour, creamy growth on the media surface | Pink colour large cells obtained by gram’s staining, oval, budding cells obtained by LPCB staining. |
| Penicillium sp. | Greyish-green colour colonies, smooth colonies. | Brush like conidiophores and branched mycelium spores arranged on conidiophores |
| Mould | Black huge colonial growth | Heavy mycelial growth arranged in filamentous form. |
TABLE 3: ANTIBACTERIAL ACTIVITY OF MANGO STEM-BARK EXTRACTS
| Name of organism | Methanol extract | Aqueous extract |
| Shigellasp | 9.7±0.2 | 6.7±0.2 |
| E. coli | 11.0±1.0 | 10.6±0.2 |
| Staphylococcus sp | 15.4±0.36 | 7.0±1.0 |
| Vibrio sp | 11.3±0.3 | 8.0±1.0 |
(Zone of inhibition in mm) (Means ± SD)
TABLE 4: ANTIFUNGAL ACTIVITY OF MANGO STEM-BARK EXTRACTS
| Name of organism | Methanol extracts | Aqueous extracts |
| Penicilliumsp | 9.3±0.2 | 10.3±0.3 |
| Yeast | 7.3±0.3 | 7.7±0.1 |
| Mould | 8.2±0.3 | 6.3±0.3 |
(Zone of inhibition in mm) (Means ± SD)
Zones of inhibition ≤7 mm indicated that the microorganism was resistant, 8-10 mm indicated an intermediate sensitivity, while ≥ 11 mm indicated sensitivity. Sensitivity implied that the plant extract could inhibit the growth of that particular microorganism at the given level of concentration. Comparatively, in bacterial isolates methanol extract was observed to be more effective than aqueous extract as shown in Fig. 1. While in fungi, the aqueous extract was observed to be more effective in Penicillium sp and yeast isolates, and methanol extract was only effective in mould as presented in Fig. 2. These findings are synonymous with the reports of Odangowei et al. 18, 19.
FIG. 1: COMPARISON OF METHANOL AND AQUEOUS EXTRACTS OF BACTERIA ISOLATES
FIG. 2: COMPARISON OF METHANOL AND AQUEOUS EXTRACTS OF FUNGI ISOLATES
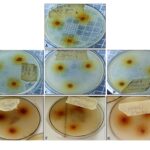
FIG. 3: ANTIMICROBIAL ACTIVITY OF MANGO STEM-BARK SAMPLES ON TEST ORGANISMS (A) E. coli (B) Vibrio sp (C) Staphylococcus sp (D) Shigella sp (E) Yeast (F) Penicillium sp (G) Mould
The qualitative and quantitative phytochemical results obtained from this study reveal the presence of saponin, alkaloid, tannin, flavonoid, glycosides, terpenoid, and steroid as presented in Table 5 and 6. Similar findings were made by Olasehinde et al., 20 who in their findings on the phytochemical properties of mango bark, showed the presence of saponins, cardiac glycosides, and alkaloids. It is also in agreement with the findings of Diso et al., 21 and Mada et al., 17. Alkaloids and saponin compounds were observed to be more active compounds in the plant samples, and according to Chukwudebe et al., 16, they indicate the cytotoxic effects of the plants.
The presence of these bioactive components of mango bark could be said to be the reason for its effectiveness against microorganisms.
TABLE 5: QUALITATIVE PHYTOCHEMICAL RESULTS OF MANGO BARK
| Sample code | Alkaloid | Tannin | Flavonoid | Saponin | Glycosides | Terpenoid | Steroid |
| Mango stem-bark | +++ | + | ++ | +++ | + | ++ | + |
Note; + (presence), ++ (abundance), +++ (more abundance)
TABLE 6: QUANTITATIVE PHYTOCHEMICAL RESULTS OF MANGO BARK
| Sample Code | Phytochemicals % | |||||
| Alkaloid | Tannin | Flavonoid | Saponin | Terpenoid | Steroid | |
| Mango stem-bark | 8.8±0.01 | 1.2±0.02 | 6.5±0.01 | 7.8±0.02 | 0.7±0.01 | 0.68±0.02 |
CONCLUSION: Based on this study, it was observed that the plant samples contained several bioactive phytochemicals such as alkaloid, flavonoid, tannins, saponins, steroid, terpenoid, and glycosides, which accounted for the activities of the plant against microorganisms.
The methanol extract was observed to have the highest zone of inhibition for bacterial, while aqueous extracts show a higher zone of inhibition for moulds.
In conclusion, the antibiotic sensitivity testing in this study had zones of inhibition equivalent to that of other plant part extracts.
This therefore suggests that mango bark extracts are as good as other commercially sold antibiotics in inhibiting these microorganisms and therefore could possibly serve as an alternative.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Olasehinde GI, Okolie ZV, Oniha MI, Adekeye BT and Ajayi AA: In-vitro antibacterial and antifungal activities of Chrysophyllum albidum and Diospyrosmon buttensis J Pharm. Phytothera 2016; 8 (1): 1-7.
- Cowan MM: Plant Products as Antimicrobial Agents. Clinical Microbiology Review 1999; 12(4):564-82.
- Aderibigbe AO, Emudianughe TS and Lawal BA: Evaluation of the antidiabetic action of Mangifera indica in mice. Phytothera. Res 2016; 15(5): 456-58.
- Aderibigbe AO, Emudianughe TS and Lawal BA: Antihyperglycaemic effect of Mangifera indica in rat. Phyto. Res 1999; 13(6): 504-7.
- Zheng MS and Lu ZY: Antiviral effect of mangiferin and isomangiferin on herpes simplex virus. Chi Med J 1999; 103(2): 160-65.
- Zhu XM, Song JX, Huang ZZ, Wu YM and Yu MJ: Antiviral activity of mangiferin against herpes simplex virus type 2 in-vitro. Zhongguoyao li xuebao 1999; 14(5): 452-54.
- Lima ME, Cordeiro M, Claudia MY, Marcos EG, Sobra M and Moreno PR: Antimicrobial activity of the essential oil from the specimens of Pimenta pseudocaryophyllus (Gomes) L. R. Landrum (Myrtaceae) native from Sao Paulo State. Brazil Pharmacol 2006; 3: 589-93.
- Kafaru E: Immense help formative workshop. In: Khan R, Islam B, Akram M, Shakil S, Ahmad A, Ali MS, Sadiqui M and Khan AU: Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules. 2008; 1994: 13
- Singh M, Khatoon S, Singh S, Kumar V, Rawat AKS and Mehrotra S: Antimicrobial screening of ethnobotanically important stem bark of medicinal plants. Pharma Res 2010; 2(4): 254–57.
- Scartezzini P and Speroni E: Review on some plants of Indian traditional medicine with antioxidant activity. J Ethnopharma 2000; 71(1–2): 23-43.
- Sofowora AO: Medicinal plants and traditional medicine in Africa. University of Ife Press 2nd edition1993; pp 320.
- Barie PS: Multidrug- Resistant Organisms and Antibiotic Management. Surg Clin N Amer 2012; 92(2): 345-91.
- Khan MR and Omotoso A: Antimicrobial activities of medicinal plants used in folklore remedies in southwestern. Fitoterapia 2003; 74(7-8): 695-8.
- Odangowei IO, Okiemute O and Precious AE: Ethno pharmacologically active Components of Brassica juncea (Brown Mustard) Seeds. International Journal of Pharmaceutical Research and Development 2019; 1(1): 09-13.
- Trease GE and Evans MC: Pharmacognosy, fourteenth edition. Elsevier, New Delhi, India 2005.
- Chukwudebe EP, Bankole SO, Asonibare AO and Adedokun SA: Antimicrobial and phytochemical properties of crude extracts of some selected plants against some microbial isolates. International Journal of Agriculture, Environment and Bioresearch 2019; 4(5): 30-42.
- Mada SB, Garba A, Muhammad A, Mohammed A and Adekunle DO: Phytochemical Screening and Anti-microbial Efficacy of Aqueous and Methanolic Extract of Mangifera indica (Mango stem bark). World Journal of Life Sciences and Medical Research 2012; 2(2): 81-5
- Odangowei IO, Patrick CA, Uchechi EE and Mike NA: Determination of antimicrobial susceptibility of ethanol, methanol, and acetate extracts of processed honey. Journal of Apitherapy 2019; 6(1): 1-5.
- Odangowei IO, Dienize GG, Uchechi EE, Ngozi GE and Udeme MA: Efficacy evaluation of extracts of Brassica juncea (Brown mustard) seeds as potential antimicrobial agent against pathogenic microbes. Journal of Medicinal Plants Studies 2019; 7(4): 263-65.
- Olasehinde GI, Sholotan KJ, Openibo JO, Taiwo OS, Bello OA, Ajayi JB, Ayepola OO and Ajayi AA: Phytochemical and Antimicrobial Properties of Mangifera indica Leaf Extracts. Covenant Journal of Physical & Life Sciences 2018; 6(1): 55-63.
- Diso SU, Ali M, Mukhtar SI and Garba M: Antibacterial activity and Phytochemical screening of Mangifera indica (Mango) stem and leaf extracts on clinical isolates of methicillin Resistant Staphylococcus aureus. J of Adv in Medical and Pharmaceutical Sci 2017; 13(1): 1-6.
How to cite this article:
Ogidi OI, Okore CC, Akpan UM, Ayebabogha MN and Onukwufo CJ: Evaluation of antimicrobial activity and bioactive phytochemical properties of mango (Mangifera indica) stem-bark extracts. Int J Pharmacognosy 2021; 8(5): 189-95. doi link: http://dx.doi.org/10.13040/ IJPSR.0975-8232.IJP.8(5).189-95.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
189-195
663
1323
English
IJP
O. I. Ogidi *, C. C. Okore, U. M. Akpan, M. N. Ayebabogha and C. J. Onukwufo
Department of Biochemistry, Federal Polytechnic, Ekowe, Bayelsa State.
ogidiodangowei@gmail.com
30 April 2021
25 May 2021
28 May 2021
10.13040/IJPSR.0975-8232.IJP.8(5).189-95
31 May 2021