EVALUATION OF ANTI-NOCICEPTIVE ACTIVITY OF ETHANOL EXTRACT OF AMARANTHUS SPINOSUS LINN. (ROOTS) IN SWISS ALBINO MICE
HTML Full TextEVALUATION OF ANTI-NOCICEPTIVE ACTIVITY OF ETHANOL EXTRACT OF AMARANTHUS SPINOSUS LINN. (ROOTS) IN SWISS ALBINO MICE
Pritesh Ranjan Dash * 1, Shishir Ahmed Sikta 2, Farjana Akter 3 and Farjana Islam 4
Department of Pharmacy 1, Jahangirnagar University, Savar, Dhaka - 1342, Bangladesh.
Department of Pharmaceutical Sciences 2, North South University, Dhaka, Bangladesh.
Department of Chemistry 3, University of Dhaka, Dhaka, Bangladesh.
Department of Pharmacy 4, East West University, Dhaka, Bangladesh.
ABSTRACT: Amaranthus spinosus, belongs to Amaranthaceae family, is an annual or perennial herb grows annually; native to tropical America and found throughout India as a weed in cultivated as well as fallow lands. Though the whole plant is used as a laxative, the roots are regarded as highly specific for colitis, and it is also used as antipyretic, febrifuge, diuretic. Other than its culinary value, it is utilized in the treatment of bronchitis, digestible, appetizer, biliousness, haematinic, galactagogue, stomachic, flatulence, nausea, anorexia, blood diseases, leucorrhoea, burning sensation, leprosy, and piles. This study aimed to investigate the anti-nociceptive activity of the ethanol extract of the root of Amaranthus spinosus Linn. The anti-nociceptive activity was investigated for its peripheral and central pharmacological action using acetic acid-induced writhing, hot plate, and tail immersion methods in Swiss albino mice at the doses of 200 and 400 mg/kg body weight. Ethanol extract of the root (200 and 400 mg/kg) showed the most significant anti-nociceptive activity in acetic acid induced writhing method with writhing inhibition of 48.83% and 67.44% accordingly, where the standard drug diclofenac-Na (25 mg/kg) showed 72.09% inhibition. In the hot plate test, ethanol extract of the root at the doses of 200 and 400 mg/kg showed maximum 61.50% and 62.80% nociception inhibition of thermal stimulus respectively, whereas the standard drug morphine (5 mg/kg) displayed maximum 64.18% nociception inhibition. In the tail immersion test, maximum 67.81% and 75.93% nociception inhibition of thermal stimulus was exhibited with ethanol extract of the root (200 and 400 mg/kg, body weight respectively). The present investigation revealed that ethanol root extracts of Amaranthus spinosus possess potent and dose-dependent central and peripheral anti-nociceptive activity justifying its traditional use in treating painful conditions.
| Keywords: |
Antinociceptive activity, Writhing test, Hot plate test, Tail immersion test
INTRODUCTION: Analgesics alleviate pain as an indicator and it does not affect the cause of that pain.
Analgesic drugs like opiates and NSAIDs, which are currently available, are not of much use because of their unfavorable effects. These drawbacks urged for the emergence of seeking for a new compound with superior pain management capacity and lesser side effects 1.
Amaranthus spinosus Linn. a glabrous herb (Amaranthaceae) abundant in tropical and sub-tropical regions of Bangladesh and India, which was reportedly known as the bearer of anti-malarial, anti-diarrheic, stimulation of proliferation of b-lymphocytes and hematological properties; its root is used as a diuretic and febrifuge 2, 3. Traditionally boiled leaves and roots of Amaranthus spinosus were given to children as a laxative. The plant is also used traditionally as anti-diabetic, antipyretic, anti-leprotic, anti-snake venom and anti-gonorrheal 4. In Malaysia, Amaranthus spinosus is used as an expectorant and in acute bronchitis. Some tribes in India use Amaranthus spinosus to induce abortion 5. The Amaranthus spinosus is noted for its effects on hematology, anti-inflammatory properties, immunomodulatory activity, anti-androgenic activity, and anthelmintic properties 6, 7, 8, 9, 10. Amaranthus spinosus L. is traditional medicine in China where it is prescribed to treat diabetes.
The seeds can be applied on broken bones as a poultice. It is also useful in several internal treatments such as internal bleeding, diarrhea, and excessive menstruation. A decoction of the root is a great cure for gonorrhea and is also used as an emmenagogue and antipyretic in South-East Asia. Inducing abortion using A. spinosus is a very common phenomenon among the Nepalese and several tribes in India. Amaranthus spinosus contains 7-p-coumaroyl apigenin 4-O-beta-D-glucopyranoside, a new coumaroyl flavone glycoside (spinoside), xylofuranosyl uracil, beta-D-ribofuranosyl adenine, hydroxycinnamates, quercetin, beta-sitosterol-glucoside and kaempferol glycosides, betalains; betaxanthin, betacyanin; amaranthine and is amaranthine, gomphrenin, stigmasterol, b-sitosterol,betanin, linoleic acid, 0.15% rutin and beta-carotene 11, 12, 13, 14.
The aim of our present investigation was envisaged to ascertain anti-nociceptive activity of ethanol extracts from the roots of Amaranthus spinosus and to evaluate its possible mechanism of action utilizing various pain models such as acetic acid-induced writhing test, hot-plate test and tail-immersion tests. The analgesic activity of the roots of Amaranthus spinosus Linn. is reported in this paper as a part of our current investigation 15, 16, 17 on local medical plants of Bangladesh.
MATERIALS AND METHODS:
Chemicals: Diclofenac sodium and aspirin were bought from Square Pharmaceuticals Ltd., Bangladesh; Acetic acid was brought from Merck, Germany; morphine from Gonoshasthaya Pharmaceuticals Ltd., Bangladesh. 0.9% sodium chloride solution (Normal saline) was bought from Orion Infusion Ltd., Bangladesh, and other reagents were of analytical grade.
Plant Material: The plant (Amaranthus spinosus Linn.) was collected from the local area of Savar, Dhaka in November 2018. The collected plant was then identified by Bushra Khan, Principal Scientific Officer, Bangladesh National Herbarium, Mirpur, Dhaka, and a voucher specimen has been deposited (DACB: 46,794) for further reference.
Extraction: The dried leaves were powdered coarsely and then extracted with ethanol by a cold extraction method at room temperature. By using rotary evaporator, the solvent was completely removed, and the obtained dried crude extract is used for investigation.
Animal: For the experiment Swiss albino mice of either sex, 3-4 weeks of age, were collected from Animal Production Unit of Animal House at the Department of Pharmacy, Jahangirnagar University which weights are between 20-25 gm. Animals were maintained under standard environmental conditions (temperature: (24.0 ± 1.0º), relative humidity: 55-65% and 12 h light/12 h dark cycle) and had free access to feed and water ad libitum. The animals were acclimatized to laboratory condition for one week before experimentation 18.
All studies performed following the approval by the Institutional Animal Ethical Committee (IAEC) of Jahangirnagar University, Savar, Dhaka, Bangladesh.
Acute Toxicity Study: Mice were divided into control and test groups (n=6). In the test groups, the extract was given orally at the doses of 500, 1000, 1500, and 2000 mg/kg. Then the animals were kept in separate cages, and they were allowed to food and ad libitum. The control group received the water. The animals were observed for possible behavioral changes, allergic reactions, and mortality for the next 72 h 19.
Anti-nociceptive Activity:
Acetic Acid-Induced Writhing Test: The method explained by 20, adopted to study the effect of the Amaranthus spinosus Linn. extract on the acetic acid-induced writing test. Test samples and control (n = 6) were administered orally 30 min before intraperitoneal administration of 0.7% acetic acid, but diclofenac-sodium (i.p) and aspirin (p.o) were administered 15 min before injection of acetic acid. After 5 min interval, the mice were observed (abdominal contraction, elongation of the body, and extension of the hind limb were referred to as writhing) for the next 10 min. Percentage inhibition of writhing was calculated using the following formula:
Writhing inhibition (%) = Mean no. of writhings (control) - Mean no. of writhings (test) / Mean no. of writhings (control) ×100
Hot Plate Test: The animals which are in the control group received water (5 ml/kg, p.o.) while the standard groups were treated with diclofenac sodium (25 mg/kg, i.p.). The animals in the test groups were treated with 100 and 200 mg/kg, per oral of Amaranthus spinosus. Then the animals were placed on Eddy’s hot plate kept at 52 ± 0.5 °C temperature. A cut off period of 28s 21, was observed to avoid damage to the paw. Reaction time was recorded when animals licked their fore or hind paws or jumped before and 0, 30, 60, and 90 min after the administration of the standard and test drugs (n=6). Percentage of elongation was calculated using the following formula:
Elongation (%) = Latency (test) – Latency (control) / Latency (test) × 100
Tail Immersion Test: The technique depends on the perception that morphine-like drugs selectively prolongs the reaction time of the typical tail withdrawal reflex in mice 22. The animals were treated as described above. One to two cm of the tail of mice was immersed in warm water, and it was kept constant at 55 ± 1 °C. The time is taken by the mice to deflect their tails is referred to as reaction time. The first reading was discarded. Then the next three readings were recorded, and the reaction time was calculated as a mean of these three readings.
A latency period of 28 s was characterized as complete analgesia, and then the measurement was stopped to avoid injury to mice. After the administration of standard and test drugs (n=6), the latent period of the tail-immersion response was determined at 0, 30, 60, and 90 min. Percentage of elongation was calculated using the following formula:
Elongation (%) = Latency (test) – Latency (control) / Latency (test) × 100
RESULTS:
Acute Toxicity: Oral administration of the roots Amaranthus spinosus at the doses of 500-2000 mg/kg did not produce any mortality or noticeable behavioral changes in mice within 72 h observation period. Therefore, it can be suggested that roots of Amaranthus spinosus have low toxicity profile with LD50 higher than 2000 mg/kg.
Analgesic Activity:
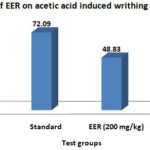
Acetic Acid-Induced Writhing Test: In this test, EER (200 mg/kg) inhibited maximum 67.44% writhing, whereas the writhing inhibition of the standard drug diclofenac-Na (25 mg/kg) showed 72.09%.
FIG. 1: EFFECTS OF EER ON MICE. EER = Ethanolic extract of roots, the control group received water 5 ml/kg body weight (p.o.), standard received diclofenac-Na 25 mg/kg body weight, EER were treated with 200 and 400 mg/kg body weight (p.o.) of the ethanolic root extract of Amaranthus spinosus. Values are mean ± SEM, (n = 6).
TABLE 1: EFFECTS OF THE EER ON ACETIC ACID-INDUCED WRITHING TEST IN MICE
| Groups | Dose (mg/kg) | No. of writhing | % of writhing | % of writhing inhibition |
| Control | 5 ml/kg | 43±0.57 | 100 | - |
| Standard | 25 | 12±0.39 | 27.90 | 72.09 |
| EER | 200 | 22±0.55 | 51.16 | 48.83 |
| 400 | 14±0.63 | 32.55 | 67.44 |
EER = Ethanolic extract of roots, Control group received water 5 ml/kg body weight (p.o.), standard received diclofenac-Na 25 mg/kg body weight, EER were treated with 200 and 400 mg/kg body weight (p.o.) of the ethanolic root extract of Amaranthus spinosus. Values are mean ±SEM, (n = 6).
Tail Immersion Test: In tail immersion test, maximum 67.81% and 75.93% nociception inhibition of thermal stimulus were exhibited with the EER at the doses of 200 and 400 mg/kg, bw respectively. In this study, morphine 5 mg/kg (66.55% inhibition) was used as standard Table 2.
Hot Plate Method: In the hot plate test, EER (200 and 400mg/kg) showed maximum 61.50 and 62.80% nociception inhibition of thermal stimulus respectively, whereas the standard drug morphine (5 mg/kg) displayed maximum 64.18% nociception inhibition.
TABLE 2: EFFECTS OF THE EER ON TAIL WITHDRAWAL REFLEX OF MICE INDUCED BY TAIL IMMERSION METHOD
| Groups | Dose
(mg/kg) |
Mean reaction time (s) before and after drug administration |
% of
elongation |
|||||
| 0 min | 30 min | 60 min | 90 min | 30 min | 60 min | 90 min | ||
| Control | 5 ml/kg | 1.67±0.13 | 1.92±0.26 | 1.88±0.39 | 1.97±0.24 | - | - | - |
| Standard | 5 | 2.69±0.33 | 4.85±0.95 | 5.66±0.93 | 5.89±0.39 | 60.41 | 66.78 | 66.55 |
| EER | 200 | 2.19±0.29 | 3.39±0.29* | 4.55±0.93* | 6.12±0.39* | 43.36 | 58.68 | 67.81 |
| 400 | 2.44±0.19 | 4.45±0.21* | 5.99±0.23* | 8.25±0.23* | 56.85 | 68.61 | 75.93 | |
EER = Ethanolic extract of roots, the control group received water 5 ml/kg body weight (p.o.), standard groups received morphine 5 mg/kg body weight (i.p.), EER were treated with 200 and 400 mg/kg body weight (p.o.) of the crude extract of Amaranthus spinosus . Values are mean ± SEM, (n = 6).
TABLE 3: EFFECTS OF THE EER ON HOT PLATE METHOD
| Groups | Dose
(mg/kg) |
Mean reaction time (s) before and after drug administration |
% of
elongation |
|||||
| 0 min | 30 min | 60 min | 90 min | 30 min | 60 min | 90 min | ||
| Control | 5 ml/kg | 9.93±0.63 | 8.95±0.69 | 9.39±0.75 | 9.67±0.36 | - | - | - |
| Standard | 5 | 9.95±1.29 | 15±3.20** | 18±1.18** | 27±2.36** | 40.33 | 47.83 | 64.18 |
| EER | 200 | 9±0.85 | 15.23±2.36* | 19.19±2.63* | 25.12±2.69* | 41.23 | 51.06 | 61.50 |
| 400 | 10.19±1.19 | 16.19±2.41* | 21.23±1.47* | 26.19±1.63* | 44.71 | 55.77 | 62.80 | |
EER = Ethanolic extract of roots, the control group received water 5 ml/kg body weight (p.o.), standard groups received morphine 5 mg/kg body weight (i.p.), EER were treated with 200 and 400 mg/kg body weight (p.o.) of the ethanolic root extract of Amaranthus spinosus. Values are mean ± SEM, (n = 6).
EER = Ethanolic extract of roots, the control group received water 5ml/kg body weight (p.o.), standard groups received morphine 5 mg/kg body weight (i.p.), EER were treated with 200 and 400 mg/kg body weight (p.o.) of the crude extract of Amaranthus spinosus. Values are mean ± SEM, (n = 6).
DISCUSSION: The methods which are used for investigating anti-nociception had been selected for both peripherally and centrally mediated effects. While the hot plate and the tail immersion test (thermal stimuli) investigated the central activity only, the acetic acid (chemical stimuli) elucidated peripherally and the central activity. A chemical stimulus is used in the acetic acid-induced writhing assay to screen both peripherally and centrally acting analgesic activity. Intraperitoneal injection of 0.7% acetic acid which irritates serous membranes; provokes a stereotypical behavior in rodents that is characterized by whole-body movements, contractions of the abdominal muscle; reduced motor activity and incoordination. The effective analgesic doses for agents that can be used in humans can be predicted by the writhing test 23, 24. Both doses of the extract significantly reduced the number of writhing in mice, which were induced by 0.7% acetic acid.
From the result, it could be demonstrated that Amaranthus spinosus had the effect to decrease visceral pain through a peripheral and central mechanism. Acetic acid-induced writhing can increase the peritoneal fluid levels of PGE2 and PGF2α 25. The analgesic activity of NSAIDs was exerted both peripherally and centrally. Their analgesic activity is mainly connected with their anti-inflammatory activity, which was resulted from the inhibition of prostaglandin synthesis via cyclo-oxygenase. Prostaglandin produces little pain by themselves but can increase the pain caused by other mediators (e.g., histamine and bradykinin) 26.
It was revealed from the results of acetic acid-induced writhing test that Amaranthus spinosus extracts reduced the writhing responses in mice which suggested that the mechanism of action of antinociceptive activity of the extracts is linked partially to the cyclo-oxygenase enzymes in the peripheral tissue via the inhibition of prostaglandin in synthesis and probably due to interference with the synthesis or release of endogenous substances which excite pain nerve endings 27. In the hot plate and tail immersion tests, all the doses of the extracts prolong latency of nociceptive response, especially the higher dose of 400 mg/kg body weight. Both the doses of the extracts had the more potent analgesic activity on the tail immersion test than that in the hot plate test.
So, it can be demonstrated that the Amaranthus spinosus extract had an analgesic activity-like morphine via central mechanism through the opioid receptor at the spinal level more than the supraspinal level. It has been revealed from the findings mentioned above that the extract effectiveness in inhibiting the centrally and thermally-induced nociception. Furthermore, the ability of the ethanolic root extract of A. spinosus to affect both the chemically and thermally-induced nociceptive response is characterized as strong analgesics like opioid agonists 28.
CONCLUSION: Medicinal plants have attracted considerable global interest in recent years. Investigation of traditional medicine is very important for the welfare of rural and tribal communities for the treatment of conventional illness. From the result of this study, it can be concluded that Amaranthus spinosus Linn. ethanol extracts possess significant peripheral and central anti-nociceptive effects in laboratory animals. The results provided experimental evidence for the traditional use of this plant in some painful conditions and also suggested the presence of biologically active principles, which may be worth of further investigation and elucidation.
ACKNOWLEDGEMENT: Authors are grateful to Jahangirnagar University, Department of Pharmacy, Savar, and Dhaka, Bangladesh, for providing the necessary facility to carry out the study.
CONFLICT OF INTEREST: Authors declare no conflict of interest.
REFERENCES:
- Tripathi KD: Essentials of Medical Pharmacology. Jaypee Brothers Medical Publishers (P) Ltd. New Delhi, India, Edition 4th, 1999: 432.
- Ghani A: Medicinal Plants of Bangladesh with Chemical Constituents and Uses. Dhaka, Bangladesh, Asiatic Society of Bangladesh, 2003: 81.
- Kirtikar KR and Basu BD: Indian Medicinal Plants. Part II. International Book Distributors, Dehradun, India 1980: 937.
- Kirtikar RR and Basu BD: Indian Medicinal Plants. International book distributors. Dehadran, India, 1987; 3: 205-59.
- Grubben GJH and Denton OA: Plant resources of tropical africa and vegetables 2004.
- Olumayokun A, Olajid, Babatude R, Ogunleya, Temitope, O and Ernile: Anti-inflammatory properties of Amaranthus spinosus. Pharmaceutical Biology, 2004; 521-25.
- Olufemi BE, Assiak IE, Ayoade GO and Onigemo MA: Studies on the effects of Amaranthus spinosus leaf extract on the hematology of growing pigs. African Journal of Biomedical Research 2003; 6: 149-50.
- Tatiya AU, Surana SJ, Khope SD, Gokhale SB and Sutar MP: Phytochemical investigation and immunomodulatory activity of Amaranthus spinosus Indian Journal of Pharm Educa Res 2007; 44(4): 337-41.
- Assiak IE, Olufemi BE, Ayonde GO and Onigemo MA: Preliminary studies on the effects of Amaranthus spinosus leaf extract as an anthelmintic in growing pigs. Tropical Veterinarian, 2002; 20(2): 126-29.
- Murugan K, Vanithakumari G and Sampathraj R: Biochemical changes in epididymis following treatment with combined extracts of Amaranthus spinosus roots and Dolichos biflorus Ancient Science of Life 1993; 13: 154-59.
- Jamaluddin ATM, Qais N, Ali MA, Howlader MA, Shams-Ud-Doha KM and Sarker AA: Analgesic activity of extracts of the whole plant of Amaranthus spinosus Int J of Drug Dev and Res 2011; 3(4): 189-93.
- Chaudhary MA, Imran I, Bashir S, Mehmood MH, Rehman N and Gilani AHI: Evaluation of gut modulatory and bronchodilator activities of Amaranthus spinosus BMC Comp and Alter Med 2012; 12: 166-78.
- Jhade D, Ahirwar D, Jain R, Sharma NK and Gupta S: A Pharmacological Review: Amaranthus spinosus Res J Pharmacognosy and Phytochemistry 2009; 1(3): 169-72.
- Hussain Z, Amresh G, Singh S and Rao CV: Anti-diarrhoeal and antiulcer activity of Amaranthus spinosus in experimental animals. Pharmaceutical Biology 2009; 47(10): 932-39.
- Dash PR, Nasrin M, Raihan SZ and Ali MS: Study of anti-diarrhoeal activity of two medicinal plants of Bangladesh in castor-oil induced diarrhoea. Int J Pharm Sci Res 2015; 5(9): 3864-8.
- Dash PR, Rana MS and Sohrab MH: Biological activities of Polygonum flaccidum. Int J Pharmacognosy 2016; 3(5): 100-04.
- Mou KW and Dash PR: A comprehensive review on Gynura procumbens Int J Pharmacognosy 2016; 3(4): 167-74.
- Chatterjee TK: Handbook on Laboratory Mice and Rats. Department of Pharmaceutical Technology, Jadavpur University, India, Edition 1st, 1993: 157.
- Walker CI, Trevisan G, Rossato MF, Franciscato C, Pereira ME and Ferreira J: Antinociceptive activity of jalapain mice. J Ethnopharmacol 2008; 120: 169-75.
- Dash PR, Nasrin M and Saha MR: Evaluation of analgesic and neuropharmacological activities of methanolic rhizome extract of Hedychium Coronarium. IJPSR 2011; 2(4): 979-84.
- Franzotti EM, Santos CVF, Rodrigues HMSL, Mourao RHV, Andrade MR and Antoniolli AR: Anti-inflammatory, analgesic activity and acute toxicity of Sida cordifolia (Malva-branca). J Ethnopharmacol 2000; 72: 273-8.
- Toma W, Graciosa JS, Hiruma-Lima CA, Andrane FDP, Vilegas W and Brita S: Evaluation of the analgesic and antiedemetogenic activities of Quassia amara bark extract. J Ethnopharmacol 2003; 85: 19-23.
- Dubinsky B, Gebre-Mariam S, Capetola RJ and Rosenthale ME: The antalgesic drugs: human therapeutic correlates of their potency in laboratory animal models of hyperalgesia. Agents Actions 1987; 20: 50-60.
- Eaton M: Common animal models of spasticity and pain. JRRD 2003; 40(4): 41-54.
- Derardt R, Jougney S, Delevalcee F and Falhout M: Release of prostaglandins E and F in an algogenic reaction and its inhibition. Eur J Pharmacol 1980; 51: 17-24.
- Neal MJ: Non-steroidal anti-inflammatory drugs (NSAIDs).In: Neal MJ. (ed.). Medical Pharmacology at a Glance Oxford, USA. Blackwell Science, Edition 2nd, 1992: 62-67.
- Collier HO, Dinneen LC, Johnson CA and Schneider C: The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br J Pharmacol 1968; 32: 295-10.
- Zakaria ZA, Sulaiman MR, Gopalan HK, Abdul Ghani ZD, Raden Mohd Nor RN, Mat Jais AM and Abdullah FC: Antinociceptive and anti-inflammatory properties of Corchorus capsularis leaves chloroform extract in experimental animal models. Yakugaku Zasshi 2007; 127(2): 359-65.
How to cite this article:
Dash PR, Sikta SA, Akter F and Islam F: Evaluation of antinociceptive activity of ethanol extract of Amaranthus spinosus Linn. (roots) in Swiss albino mice. Int J Pharmacognosy 2019; 6(5): 181-86. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(5).181-86.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.