ESTIMATION OF SCOPOLETIN CONTENT IN COMMERCIALLY EXTRACTED LEAVES OF MEDICINAL HERB ARTEMISIA ANNUA L. USING HPTLC
HTML Full TextESTIMATION OF SCOPOLETIN CONTENT IN COMMERCIALLY EXTRACTED LEAVES OF MEDICINAL HERB ARTEMISIA ANNUA L. USING HPTLC
B. Sharma 1, A. Dangash 2 and N. Pandya 2
M. K. Amin Arts & Science College and College of Commerce 1, The Maharaja Sayajirao University of Baroda, Padra - 391440, Gujarat, India.
Department of Botany, Faculty of Science 2, The Maharaja Sayajirao University of Baroda, Vadodara - 390002, Gujarat, India.
ABSTRACT: Plants have a long history as therapeutic tools in the treatment of human diseases and have been used as a source of medicines for ages. In search of new biologically active natural products, many plants and herbs used in traditional medicine are screened for natural products with pharmacological activity. Qinghao is a traditional Chinese medicine prepared from the aerial parts of Artemisia annua L. which belongs to Compositae family. It is used as an anti-parasitic and fever relieving agent. Artemisinin was isolated from Qinghao as a major bioactive constituent and has been used to treat malaria. Besides artemisinin, the chemical constituents of Qinghao include volatile oil, terpenes, flavonoids, and coumarins. Scopoletin-derived sesquiterpene ethers Sesquiterpene derivatives of 7-hydroxy-6-methoxycoumarin (Scopoletin) are not so common. The aim of the study was to investigate chemical constituents present in the leaves of Artemisia annua after the commercial extraction process of the plant for artemisinin. From the preliminary studies, the compound identified was found to be a coumarin- Scopoletin. The residual leaves were used for the extraction of Scopoletin. The study estimated Scopoletin using thin-layer chromatography (TLC). The Scopoletin content of the leaves was calculated from the area calibration curve by this method was found to be 0.04477% w/w (plant dry weight basis). This HP-TLC procedure may be used effectively for identity, quality evaluation as well as quantitative determination for this plant or its derived products.
| Keywords: |
Sesquiterpenenes, Scopoletin, Artemisinin, HP-TLC
INTRODUCTION: Scopoletin (6-methoxy-7-hydroxycoumarin) is a coumarin compound with antifungal properties that have been isolated from several plant species. It is a biomarker widespread in plant kingdom, especially in the phytotherapic and nutraceutical ones.
Scopoletin is derived from the phenylpropanoid pathway with strong blue fluorescence under UV light 1, can be isolated from many plant species 2, and was proposed as an important phytoalexin against microbial pathogens 3. Scopoletin increases considerably after fungal infection exhibiting fungitoxicity in-vitro 4 - 8. Scopoletin possesses antioxidant property, scavenged superoxide anion in the xanthine/xanthine oxidase reaction system in a concentration-dependent manner 9 Scopoletin obtained from fruits of Tetrapleura tetraptera (Mimosaceae) has hypertensive effect 10. It also shows antidepressant activity, angiogenic activity and antifungal activity 11, 12, 13. Lee (2013) isolated Scopoletin from Artemisia iwayomogi, 14Hofer & Greger (1984) found scopoletin farnesyl ether scopofarnol and the new scopoletin drimenyl ether scopodrimol A from the roots of Artemisia persica 15. Silva et al., (2002) reported, Scopoletin inhibited the conidial germination of Corynespora cassiicola isolated from the uninfected mature leaves of Hevea brasiliensis.
The present study deals with the investigation of the compounds which are so tightly bound that even after the rigorous extraction process, they remain in the plant. Such compound should be extracted as they hold important medicinal value. With this view, the estimation was done in the commercial sample of Artemisia annua L. which were previously extracted (extracted for Artemisinin), again with solvent. This was an effort to extract such important compounds from the residue so that no compound goes wasted.
MATERIALS AND METHODS:
Thin Layer Chromatographic Study: Thin-layer chromatography (TLC) is an important analytical tool in separation, identification, and estimation of different classes of natural products. Comparative TLC (co-TLC) with marker compound/s can be used for identification of chemical constituents and to standardize the herbal raw materials.
High-Performance Thin Layer Chromatography: Camag TLC systems equipped with Camag Linomat V, an automatic TLC sample spotter, Camag glass twin trough chamber (20 × 10 cm) were used for the analysis. Chromatography was performed using pre-activated (60 °C for 5 min) silica gel 60F254 TLC plates (20 × 10 cm; layer thickness 250 μm) (Merck, Darmstadt, Germany). Samples and standards were applied on the plate as 6 mm wide bands with an automatic TLC sampler under a flow of N2 gas, 10 mm from the bottom and 10 mm from the side and the space between two spots was 15 mm of the plate. The linear ascending development was carried out in a camag twin trough chamber saturated with 20 ml mobile phase (chloroform: methanol: toluene, 8:1:1, v/v/v) for 20 min at room temperature (25 ± 2 °C and 40% relative humidity). The plates were developed up to 8 cm under chamber saturation conditions. Subsequent to the development, TLC plates were dried in current air with the help of a hairdryer. Evaluations of the plates were performed with Camag scanner 3 (win CATS 4.0 integration software). Densitometric scanning was performed at 360 nm (absorption-reflection mode), using a slit width of 6 × 0.45 mm, data resolution 100 μm step and scanning speed 20 mm/s with a computerized Camag TLC scanner. Peak areas were recorded and the amount of scopoletin was calculated using the calibration curve. Quantification was done by using external standard method.
Preparation of Extract: Accurately weighed 5 g of previously extracted Artemisia annua sample was again extracted with methanol (4 × 50 ml) under reflux (30 min each time). The combined extracts were filtered, concentrated and transferred to a 50 ml volumetric flask and the volume was made up with the same solvent.
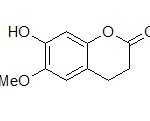
Preparation of Standard Scopoletin Solution: A stock solution of scopoletin Fig. 1 (Himedia Laboratories Pvt. Ltd., Mumbai, India) (5 mg/50 ml) was prepared in methanol. Working solutions were prepared by appropriate dilution of the stock solution with the same mixture of solvents.
Calibration Curve for Scopoletin: Standard scopoletin solution in the range of 200 to 700 ng spot-1 was applied on TLC plate for preparation of the calibration curve of peak area versus concentration.
RESULT AND DISCUSSION: Plants use an intricate defense system against pests and pathogens, including the production of low molecular mass secondary metabolites with antimicrobial activity, which are synthesized de novo after stress and are collectively known as phytoalexins. Phytoalexins are antimicrobial secondary metabolites produced de novo by plants in response to stress, including microbial attack. Scopoletin, a derivative of coumarin, is a benzopyrone in nature and found in the root of plants in the genus Scopolia like Scopolia japonica, Artemisia scoparia, Kleinhovia hospita, Stevia, Agle marmelos etc. 16 Coumarins, flavonol glycosides and a group of unidentified aglycones have been reported from the inflorescence of Artemisa scoparia 17. Several species of Artemisia annua recorded the presence of Scopoletin like Artemisia capillaries 18, Artemisia campestris 19, Artemisia feddei 20, Artemisia argyi 21, Artemisia annua 22 and Artemisia iwayomogi 13. The sample plant taken for the study was examined for Scopoletin and the results were satisfactory.
TLC Separation Optimization: The A. annua extract (5 µl), when subjected to TLC showed the presence of scopoletin Fig. 2. A comparison of the spectral characteristics of the peak for standard compound and that of the sample further confirmed the presence of scopoletin in the sample Fig. 3. Good resolution with a symmetrical and reproducible peak was obtained.
FIG. 1: CHEMICAL STRUCTURE OF SCOPOLETIN
Linearity: The peak area versus concentration plot was found to be linear in the range of 200-700 ng spot-1 for scopoletin Fig. 4. The regression equation and correlation coefficient for scopoletin indicated good linearity Table 1.
Sample Analysis: The scopoletin content of the leaves calculated from the area calibration curve Table 2 by this method was found to be 0.04477% w/w (plant dry weight basis).
TABLE 1: VALIDATION PARAMETERS FOR QUANTIFICATION OF SCOPOLETIN
| S. no. | Parameter | Results |
| 1 | Rf | 0.62 |
| 2 | Dynamic range
(ng spot-1) |
200 - 700 |
| 3 | Equation | Y=8549.709+20.799x |
| 4 | Slope | 20.799 |
| 5 | Intercept | 8549.709 |
| 6 | Linearity
(correlation coefficient) |
0.99236 |
TABLE 2: CALIBRATION CURVE PARAMETERS FOR QUANTIFICATION OF SCOPOLETIN IN A. ANNUA EXTRACT
| Rf | Amount/Fraction | Area | X(calc) |
| 0.63 | 200 ng | 12091.80 | - |
| 0.62 | 300 ng | 15005.72 | - |
| 0.61 | 400 ng | 17225.91 | - |
| 0.62 | 500 ng | 19514.45 | - |
| 0.61 | 600 ng | 21051.78 | - |
| 0.63 | 700 ng | 22565.71 | |
| 0.62 | - | 13205.57 | 223.85 ng |
CONCLUSION: From the above spectroscopic studies, it was concluded that this TLC procedure may be used effectively for identity, quality evaluation as well as quantitative determination for this plant or its derived products. Also, the residual leaves can be reused for the isolation of Scopoletin.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kai K, Mizutani M, Kawamura N, Yamamoto R, Tamai M, Yamaguchi H, Sakata K and Shimizu BI: Scopoletin is biosynthesized via ortho‐hydroxylation of feruloyl CoA by a 2‐oxoglutarate‐dependent dioxygenase in Arabidopsis thaliana. The Plant Journal 2008; 55(6): 989-99.
- Murray RDH: The natural coumarins: Occurrence, Chemistry and Biochemistry 1982
- Gnonlonfin GB, Sanni A and Brimer L: Review scopoletin–a coumarin phytoalexin with medicinal properties. Critical Rev in Plant Sci 2012; 31(1): 47-56.
- Goy PA, Signer H, Reist R, Aichholz R, Blum W, Schmidt E and Kessmann H: Accumulation of scopoletin is associated with the high disease resistance of the hybrid Nicotiana glutinosa x Nicotiana debneyi. Planta 1993; 191(2): 200-6.
- Churngchow N and Rattarasarn M: Biosynthesis of scopoletin in Hevea brasiliensis leaves inoculated with Phytophthora palmivora. Journal of Plant Physiology 2001; 158(7): 875-82.
- Carpinella MC, Ferrayoli CG and Palacios SM: Anti-fungal synergistic effect of scopoletin, a hydroxycoumarin isolated from Melia azedarach L. fruits. Journal of Agricultural and Food Chemistry 2005; 53(8): 2922-7.
- El Oirdi M, Trapani A and Bouarab K: The nature of tobacco resistance against Botrytis cinerea depends on the infection structures of the pathogen. Environmental Microbiology 2010; 12(1): 239-53.
- Gnonlonfin GB, Sanni A and Brimer L: Review scopoletin–a coumarin phytoalexin with medicinal properties. Critical Rev in Plant Sci 2012; 31(1): 47-56.
- Shaw CY, Chen CH, Hsu CC, Chen CC and Tsai YC: Antioxidant properties of scopoletin isolated from S. acutum. Phytotherapy Res 2003; 17(7): 823-5.
- Ojewole JA and Adesina SK: Mechanism of the hypotensive effect of scopoletin isolated from the fruit of Tetrapleura tetraptera. Planta Medica 1983; 49(09): 46-50.
- Capra JC, Cunha MP, Machado DG, Zomkowski AD, Mendes BG, Santos AR, Pizzolatti MG and Rodrigues AL: Anti-depressant-like effect of scopoletin, a coumarin isolated from polygala sabulosa (Polygalaceae) in mice: evidence for the involvement of monoaminergic systems. European Journal of Pharmacology 2010; 643(2-3): 232-8.
- Pan R, Dai Y, Yang J, Li Y, Yao X and Xia Y: Anti‐angiogenic potential of scopoletin is associated with the inhibition of ERK1/2 activation. Drug Development Research 2009; 70(3): 214-9.
- Lee SH, Ding Y, Yan XT, Kim YH and Jang HD: Scopoletin and scopolin isolated from artemisia iwayomogi suppress differentiation of osteoclastic macrophage RAW 264.7 cells by scavenging reactive oxygen species. Journal of Natural Products 2013; 76(4): 615-20.
- Hofer O and Greger H: Scopoletin sesquiterpene ethers from Artemisia persica. Phyto Chem 1984; 23(1): 181-2.
- Silva WP, Deraniyagala SA, Wijesundera RL, Karunanayake EH and Priyanka UM: Isolation of scopoletin from leaves of Hevea brasiliensis and the effect of scopoletin on pathogens of H. brasiliensis. Mycopathologia 2002; 153(4): 199-02.
- Verma A, Dewangan P, Kesharwani D and Kela SP: Hypoglycemic and hypolipidemic activity of scopoletin (coumarin derivative) in streptozotocin induced diabetic rats. Int J Pharm Sci Rev Res 2013; 22(1): 79-83.
- Thakur RS, Jain MP and Rao PR: Scoparone: isolation from Artemisia scoparia Waldst & Kit and synthesis. Research and Industry 1975.
- Okuno I, Uchida K, Nakamura M and SAKURAI K: Studies on Choleretic Constituents in Artemisia capillaris THUNB. Chemical and Pharmaceutical Bulletin 1988; 36(2): 769-75.
- Vasconcelos JM, Silva AM and Cavaleiro JA: Chromones and flavanones from Artemisia campestris subsp. maritima. Phytochemistry 1998; 49(5): 1421-4.
- Kang TH, Pae HO, Jeong SJ, Yoo JC, Choi BM, Jun CD, Chung HT, Miyamoto T, Higuchi R and Kim YC: Scopoletin: an inducible nitric oxide synthesis inhibitory active constituent from Artemisia feddei. Planta Medica 1999; 65(05): 400-3.
- Adams M, Efferth T and Bauer R: Activity-guided isolation of scopoletin and isoscopoletin, the inhibitory active principles towards CCRF-CEM leukaemia cells and multi-drug resistant CEM/ADR5000 cells, from Artemisia argyi. Planta Medica 2006; 72(09): 862-4.
- Tzeng TC, Lin YL, Jong TT and Chang CM: Ethanol modified supercritical fluids extraction of scopoletin and artemisinin from Artemisia annua L. Separation and Purification Technology. 2007; 56(1): 18-24.
How to cite this article:
Sharma B, Dangash A and Pandya N: Estimation of scopoletin content in commercially extracted leaves of medicinal herb artemisia annua L. using hptlc. Int J Pharmacognosy 2019; 6(8): 273-76. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(8).273-76.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
273-276
673
1212
English
IJP
B. Sharma, A. Dangash and N. Pandya
M. K. Amin Arts & Science College and College of Commerce, The Maharaja Sayajirao University of Baroda, Padra, Gujarat, India.
bs1384@gmail.com
11 August 2019
25 August 2019
27 August 2019
10.13040/IJPSR.0975-8232.IJP.6(8).273-76
31 August 2019