EFFICACY OF TOPICAL NEPAFENAC IN THE TREATMENT OF DIABETIC MACULAR EDEMA- A CLINICAL STUDY
HTML Full TextEFFICACY OF TOPICAL NEPAFENAC IN THE TREATMENT OF DIABETIC MACULAR EDEMA- A CLINICAL STUDY
Mridula V. Amarnath
Chinmaya Mission Hospital, Bangalore - 560038, Karnataka, India.
ABSTRACT: To analyze the efficacy of topical nepafenac, 0.1% eye drops in the treatment of diabetic macular edema (DME). Method: a prospective interventional study in 14 eyes having center involving DME were taken into consideration. They were administered topical nepafenac eye drops 0.1% thrice daily for 6 months. Their vision and foveal thickness were recorded systematically at the start of the treatment and then at 2nd, 4th, and 6th month follow up. Results: The mean baseline and final Log MAR visual acuity were 0.33 and 0.15, respectively and the mean baseline and final foveal thickness were 450 µm and 290 µm respectively. Conclusion: A statistically significant improvement was noticed in the visual acuity and foveal thickness after the administration of 0.1% of topical nepafenac eye drops.
| Keywords: |
Center involving diabetic macular edema, Foveal thickness
INTRODUCTION: Diabetic macular edema is one of the common causes of decreased vision in patients having diabetic retinopathy 1. The focal laser was the initial modality of treatment for clinically significant macula edema. Early treatment diabetic retinopathy study (ETDRS) showed that focal laser reduced the risk of visual loss by 50%. But however focal laser treatment is associated with various side effects like scotoma, reduced color vision, vision loss due to the formation of choroidal neovascular membrane, subretinal fibrosis formation, migration of hard exudates into the foveal center, enlargement of the laser scars towards the foveal center. Hence, various other treatment modalities were looked in to and evaluated, which included intravitreal injections of steroids, agents that target vascular endothelial growth factors (VEGF).
Intravitreal injection of triamcinolone for DME was effective for a short period and again was associated with adverse effects like the development of cataract, increase in the intraocular pressure as was seen in nearly 50% of the patients. Anti VGEF agents were effective in DME 2. But it required repeated injections, which carried an increased risk of endophthalmitis. Hence, a safer alternative modality of treatment became necessary. It is known that inflammation plays a key role in the pathogenesis of diabetic retinopathy.
Cyclo oxygenase-2 (COX-2) is up-regulated in the retina in diabetic individuals, which in turn leads to increased production of prostaglandins E2 (PGE2), which is an important mediator of inflammation. An elevated PGE2 leads to increased VEGF in diabetic retinopathy and also leads to retinal endothelial cell degeneration. Nepafenac is a non-steroidal anti-inflammatory agent. It is a pro-drug and gets converted to Amfenac by intraocular hydrolases, which inhibits both Cox 1 and 2. In a study conducted on a rat model, it was shown that topical nepafenac inhibits diabetes induced retinal microvascular abnormalities. Topical nepafenac reaches its bioactive concentration in the posterior segment.
METHODS: A prospective interventional case study was done on patients who came to the ophthalmology OPD and was confirmed to have a center involving DME. Written consent was taken from the patients prior to the procedure. The patients were administered topical nepafenac eye drops 0.1% three times a day.
Inclusion Criteria:
- Diabetic retinopathy patients with center involving DME, as demonstrated by optical coherence tomography (OCT).
- Patients who are not willing for anti VGEF or triamcinolone intravitreal injections.
- Patients who were willing for a follow up of 6 months.
Exclusion Criteria:
- Any patient who was subjected to intravitreal steroid or anti VGEF injection within 6 months period.
- Patients who had undergone focal / grid laser treatment in the previous 6 months.
- Vision loss not solely attributed to diabetic retinopathy.
The patient's vision was assessed with the help of Snellen’ s chart, and the anterior segment was evaluated by a slit lamp biomicroscope, and the posterior segment was assessed by a slit lamp biomicroscope with 90 D lens and indirect ophthalmoscope. OCT was done for all the selected patients, and the central macular thickness was evaluated.
The patients were followed up on the 2nd, 4th, and 6th month and during each follow-up up, their visual acuity and CMT were recorded. Snellen’s chart was converted into Log MAR for analysis. Wilcox on signed-rank test for visual acuity analysis and paired T-test for CMT were used. The software used was state 13.
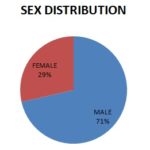
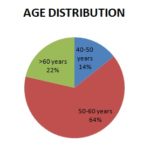
RESULTS: In our study, 14 eyes were taken into consideration. Fig. 1 shows the sex distribution of the patients that had 10 male and 4 female patients having an average age distribution of 56 years, as seen in Fig 2.
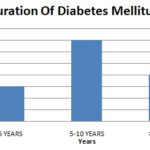
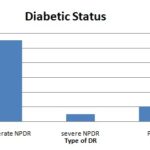
The mean duration of diabetes among the patients was 7.4 years, Fig. 3. Nine eyes were phakic, and five were pseudophakic, who had undergone a cataract surgery at least 1 year ago. Eleven eyes had moderate non-proliferative diabetic retinopathy, one eye had severe non-proliferative diabetic retinopathy, and two eyes had stable proliferative diabetic retinopathy post-pan-retinal photocoagulation Fig. 4. Eight eyes had previously received focal laser treatment, and two eyes had received intravitreal triamcinolone injection 8 months before the study.
The baseline means Log MAR visual acuity was 0.34 and was 0.25 at 2nd month, 0.22 at 4th month, and 0.18 at 6th month follow up. There was a statistically significant improvement between the baseline and the final visual outcome. (p = 0.025) Table 1. The mean baseline CMT was 465.78 µ, and it decreased to 353.18 µ at the 2nd month follow up and to 303.07 µ at the 4th month and to 290 µ at the 6th month. There was a statistically significant improvement in the CMT from the baseline to the final follow up visit (p = 0.0014) Table 1.
FIG. 1: SHOWS THE SEX DISTRIBUTION OF PATIENTS
FIG. 2: SHOWS THE AGE DISTRIBUTION OF PATIENT
FIG. 3: SHOWS THE MEAN DURATION OF DIABETES
FIG. 4: SHOWS THE DIABETIC STATUS OF THE PATIENTS
TABLE 1: SHOWING A COMPARATIVE STUDY BETWEEN THE INITIAL AND FINAL VISUAL ACUITY (VA) AND CENTRAL MACULAR THICKNESS (CMT)
| Baseline VA | Baseline CMT | Final VA | Final CMT |
| 6/12 | 380 | 6/6 | 303 |
| 6/6 | 493 | 6/6 | 240 |
| 6/6 | 635 | 6/6 | 321 |
| 6/6 | 420 | 6/6 | 236 |
| 6/6 | 483 | 6/6 | 412 |
| 6/12 | 289 | 6/6 | 297 |
| 6/18 | 315 | 6/18 | 276 |
| 6/12 | 553 | 6/12 | 593 |
| 6/12 | 340 | 6/9 | 312 |
| 6/6 | 404 | 6/9 | 236 |
| 6/24 | 456 | 6/12 | 187 |
| 6/60 | 478 | 6/18 | 254 |
| 6/36 | 567 | 6/12 | 189 |
| 6/36 | 578 | 6/12 | 197 |
DISCUSSION: It is seen that in diabetic retinopathy, there is an increase in the production of prostaglandins. Inhibition of PG production helps to prevent diabetic retinal microvascular abnormalities 3.
In a study done by Baudoin et al., he said that aspirin, which is an anti-inflammatory agent, tends to inhibit the mean yearly increase in the number of microaneurysms, but it was contrary to the ETDRS study which revealed that aspirin did not have any beneficial effect on diabetic retinopathy. Another study showed that Celecoxib, which is a Cox-2 inhibitor, only helps to decrease the fluorescein leakage and has no drastic effect on the visual outcome in DME 4. Nepafenac inhibits both Cox1 and COX-2 5 and has an overall effect on prostaglandin production, which plays a key role in the pathogenesis of diabetic retinopathy. Since there is most of the time a drop in the vision in patients with DME and the necessity to undergo repeated anti VGEF injections for the treatment of DME, topical nepafenac was considered as a therapeutic measurement. In our study, the use of topical nepafenac showed a positive result in the visual outcome.
The study showed that 7 eyes had an improvement in the vision, 6 eyes maintained the same vision, and 1 eye had a drop despite having a reduction in the CMT. 11 eyes had a decrease in the CMT, and 3 eyes had an increase in the CMT during the final follow up. It was noted that none of the patients had any side effects due to topical nepafenac.
Limitation: Our study had a limitation as the sample size was small, and the systemic control status of the patients was not adequate. Considering the beneficial effect of topical nepafenac in the treatment of center DME, a larger study group could also be taken and assessed.
CONCLUSION: Topical nepafenac is a safe and cost-effective option in the treatment of DME. It can be used as a therapeutic optic in cases of DME with good vision and in patients who have multiple systemic Co-morbidities such as uncontrolled hypertension, diabetes, recent myocardial infarction, and stroke. It is also helpful in conditions where the usage of anti-VGEF needs to be contraindicated or used with extra caution.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Bhagat N and Grigorian RA: Diabetic macular edema: pathogenesis and treatment. Surv Ophthalmol 2009; (54): 1-32.
- Salam A and Da-Costa J: Antivascular endothelial growth factor agents for diabetic maculopthy. Br J Ophthalmol 2010; (94): 821-26.
- Sennlaub F and Valamnsesh F: Cyclooxygenase-2 in human and ischemic proliferative retinopathy. Ophthalmology 2003; (10): 198-04.
- Du Y and Sarthy T: Interaction between now and cyclooxygenase pathways in retinal cells. Am J Physiol 2004; (67): 23-39.
- Chew KT and Kim J: Preliminary assessment of celecoxib in diabetic macular. Edema 2010; (30): 456-57.
How to cite this article:
Amarnath VM: Efficacy of topical nepafenac in the treatment of diabetic macular edema- a clinical study. Int J Pharmacognosy 2019; 6(12): 407-10. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(12).407-10.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.