EFFECT OF PHENANTHRENE EXPOSURE ON THE TOTAL ANTIOXIDANT CAPACITY, CATALASE, SUPEROXIDE DISMUTASE AND XANTHINE OXIDASE, OF THE LIVER
HTML Full TextEFFECT OF PHENANTHRENE EXPOSURE ON THE TOTAL ANTIOXIDANT CAPACITY, CATALASE, SUPEROXIDE DISMUTASE AND XANTHINE OXIDASE, OF THE LIVER
Enemchukwu Onyinye Victory, C. Enemuo Ijeoma and H. Enemuo Emeka *
Department of Medicine, Nnamdi Azikiwe University, Nnewi Campus.
ABSTRACT: Background and Aim: Phenanthrene is an aromatic compound with three benzene rings forming the main structure. It represents the majority of polycyclic aromatic hydrocarbons found in the environment and is found in a variety of areas including, burnt smoky coal particles soil and sediment, diesel emissions, tobacco and food smoke. The aim of the study was to determine the effect of phenanthrene on oxidative stress markers in male Wistar rats. Methodology: Fifty healthy rats weighing between 100 and 180g were used for this study. The animals were randomly assigned into five groups of ten rats per group. Group A served as control and the animals were administered with 1ml of DMSO oil only orally for three weeks. Groups B animals were administered orally with 5mg/kg of phenanthrene dissolved in 9ml of DMSO oil and Group C animals were administered orally with 10mg/kg of phenanthrene dissolved in 13ml of DMSO oil for three weeks. Group D animals, had 180mg/kg of phenanthrene dissolved in 1ml of DMSO oil orally, once, while Group E animals were also, administered orally with 900mg/kg of phenanthrene dissolved in 1ml of DMSO oil, given once. Results: Results of the experiment showed significant increase in Superoxide dismutase, Xanthine oxidase levels and in Total antioxidant capacity. Conclusion: Phenanthrene increased oxidative stress, in male Wistar rats.
Keywords: Phenanthrene; Polycyclic aromatic hydrocarbons; Oxidative stress; Wistar rats; Superoxide dismutase
INTRODUCTION: Phenanthrene is an aromatic compound with three benzene rings forming the main structure. It was discovered in coal tar by E. Ostermayer in 1872. Phenanthrene derivatives are compounds obtained by transformation and derivation of new compounds from the already existing structure of phenanthrene. These derivatives consist of: mono-substituted phenanthrene, diametrical phenanthrene, poly substituted phenanthrene, phenanthrequinone and dihydrophenanthrene 36.
Phenanthrene represents the majority of polycyclic aromatic hydrocarbons found in the environment. It is found in a variety of areas including: burnt smoky coal particles 37 soil and sediment, diesel emissions 63, tobacco and food smoke 43. Phenanthrene has a low molecular weight and is not normally carcinogenic 41. It is one of the most commonly found polycyclic aromatic hydrocarbons in the environment, largely due to the abundant amounts found in petroleum, creosote and coal tar in sediments 58.
Phenanthrene is a very dangerous chemical commonly found in air, soil and water 21. It is a type of polycyclic aromatic hydrocarbon that poses a serious threat to the lives of people exposed to it 35. Exposure to phenanthrene for a long period of time makes an animal highly susceptible to hepatotoxicity 23. The liver, the largest internal organ in the body, plays a crucial role in secretion, digestion, blood detoxification and nutrient storage 32. It is extremely vulnerable to toxicants because constant exposure of hepatocytes to environmental toxins can significantly impair liver function and ultimately cause liver damage 65. The effect of phenanthrene on the liver is not completely known, as most research done on the effects of phenanthrene in the liver has focused on aquatic animals 64, which do not share the same phenotype as humans and as such cannot be used to understand how phenanthrene will affect the human internal structure. For this reason, it is important to investigate the effect of phenanthrene on male wistar rats, which share similar internal structure to that of humans. The aim of the study was to determine the effect of phenanthrene exposure on oxidative stress markers of male Wistar rats.
FIG. 1: STRUCTURE OF PHENANTHRENE (HINTZEET AL., 2010).
MATERIALS AND METHODS: Fifty healthy rats weighing between 100 and 180g were used for this study. The animals were obtained from the animal house of Igbinedion University, Okada, Edo State and they were distributed randomly into five well ventilated plastic cages with wood beddings with 10 rats each, where they were kept for two weeks before commencement of the experiment for acclimatization. They were fed with pelletized commercially prepared growers mash purchased in a local store in Okada, Edo State and had access to water. They were housed at standard laboratory conditions under 12h light/12h dark cycle. The cages were continuously kept clean every day to ensure good hygiene and prevent the animals from disease.
The animals were randomly assigned into five groups of ten rats per group.
Group A: Animals were administered with 1ml of DMSO oil only orally for three weeks. (DMSO only), and had free access to the pelletized commercially prepared growers mash and water ad libitum. This group served as the control group.
Group B: Animals were administered with 5mg/kg of phenanthrene that was weighed using Mettler balance and dissolved in 9ml of DMSO oil orally for three weeks. (PHE + DMSO)
Group C: Animals were administered with 10mg/kg of phenanthrene that was weighed using Mettler balance and dissolved in 13ml of DMSO oil orally for three weeks.
Group D: Animals were administered with 180mg/kg of phenanthrene that was weighed using Mettler balance and dissolved in 1ml of DMSO oil orally, once.
Group E: Animals were administered with 900mg/kg of phenanthrene that was weighed using Mettler balance and dissolved in 1ml of DMSO oil orally, once.
TABLE 1:
| Groups | Treatments |
| A | DMSO oil only |
| B | 5mg/kg Phenanthrene dissolved in DMSO oil |
| C | 10mg/kg Phenanthrene dissolved in DMSO oil |
| D | 180mg/kg Phenanthrene dissolved in DMSO oil |
| E | 900mg/kg Phenanthrene dissolved in DMSO oil |
Analytical Procedure: The Phenanthrene administration was done for 21 days. At the end of the experiment, the rats were weighed and blood samples collected through retro-orbital puncture for hematological analysis. Biochemical analysis was carried out using spectrophotometric method.
Catalase (CAT): Catalase is a common enzyme found in nearly all living organisms exposed to sunlight, such as bacteria, plants and animals, which catalyzes the decomposition of hydrogen peroxide to water and oxygen. It is highly essential in the protection of cells from oxidative damage by reactive oxygen species (ROS) 17.
Principles: The enzymatic reaction of catalase (CAT) in decomposing peroxide (H2O2) can be quickly stopped by ammonium molybdate. The residual H2O2 reacts with ammonium molybdate to generate a yellowish complex. CAT activity can be calculated by production of the yellowish complex at 405 nm against Blank.
Assay Procedure: Heparinized whole blood samples was diluted between 5 – 10 fold and tissue samples were homogenized in 10 vol. of 50 mmol/l phosphate buffer, pH 7.4 and centrifuged at 3000rpm for 20 minutes and the supernatant collected before starting the procedure. 200ul sample was added into a cuvette. 1ml of Substrate solution was added into the cuvette, mixed and allowed to stand for 1 minute. 1ml of molybdate solution was added into the cuvette and mixed for 2 minutes. The absorbance was read at a wavelength of 405nm and recorded. For the blank measurement, the samples were replaced with 200ul distilled water and the procedure was repeated.
Calculation:
Catalase Activity in U/ml (Serum or Plasma) = Abs Blank – Abs Sample × 162.5
Catalase Activity in U/mg Protein (Tissue) = Abs Blank – Abs Sample / Sample Protein Conc. in mg/ml × 162.5
Superoxide Dismutase: Superoxide dismutase (SOD), which catalyzes the dismutation of the superoxide anion (O2.-) into hydrogen peroxide and molecular oxygen, is one of the most important antioxidative enzymes 39.
0.5ml of whole blood was centrifuged for 10 minutes at 3500 rpm and then aspirated off the plasma. Red cells were washed 4 times with 5 ml of 0.9% saline and centrifuged for 10 minutes at 3500 rpm after each run. The washed centrifuged erythrocytes were then made up to 2.0 ml with cold distilled water, mixed and left to stand at 4°C for 15 minutes. Tissue samples were homogenized in 10 vol. of 50 mmol/l phosphate buffer, pH 7.4, then centrifuged for 20 min at 3000 rpm. The supernatant was collected and used in the assay.
Principles: The enzyme Superoxide dismutase has the ability to inhibit the autoxidation of pyrogallol. The autoxidation of pyrogallol in the presence of EDTA in the pH 8.2 is 50%. The principle of this method is based on the competition between the pyrogallol autoxidation by O2•¯ and the dismutation of this radical by SOD.
Assay Procedure: 50ul sample was added into a clean cuvette, 1ml of SOD assay buffer was added to the cuvette, 1ml of SOD chromogen solution was added to the cuvette and mixed. The absorbance was read immediately at 420nm, then read again after 1 minute. For Blank measurement, the sample was replaced with 50ul distilled water and the procedure was repeated.
Calculations: The change in absorbance of samples and blank was determined using the following equation:
Change in absorbance of Sample (ΔS) = Absorbance of sample (1 minute) – Absorbance of sample (Initial)
Change in absorbance of blank (ΔC) = Absorbance of blank (1 minute) – Absorbance of blank (Initial)
% inhibition of Pyrogallol autoxidation = ΔS / ΔC × 100%
SOD Activity In (U/ml) = % inhibition of Pyrogallol autoxidation / 50%
Total Antioxidant Capacity (TAC) Assay: Oxidative stress has been implicated in a number of diseases such as atherosclerosis, chronic inflammatory disease, chronic renal failure, and cancer. It is a condition where an imbalance exists between the production of reactive oxidizing species and the body’s ability to neutralize these intermediates, resulting in cellular damage. The body has designed several physiological responses to oxidative stress including counterbalances such as enzymes and variously functionalized molecules that effectively neutralize these damaging species. These antioxidants can be either water or lipid soluble, and are localized transiently throughout various tissues, cells and cell types.
Principles of Procedure: The reduction potential of the sample or standard effectively converts Cu+2 to Cu+1, thus changing the ion’s absorption characteristics. This reduced form of copper will selectively form a stable 2:1 complex with the chromogenic reagent with an absorption maximum at ca. 450 nm. A known concentration of Trolox is used to create a calibration curve, with the data being expressed as mMTrolox equivalents or in μM copper reducing equivalents.
Assay Procedure: 50ul of samples and each standard were placed into the cuvette supplied. Blank contained 50ul of Dilution buffer in place of Standard or Sample. 1ml Assay Buffer was added to the cuvette containing samples, standards and blank. Background absorbance was read at 450nm. 100 μL of Chromogen was added and incubated for 5 minutes at room temperature. The cuvette was read a second time at 450 nm.
Standard Preparation: The trolox powder was diluted with 1 ml of TAC Dilution Buffer to make a 2mM Trolox solution. Serial dilutions were carried out to prepare the following concentration; 2mM, 1mM, 0.5mM, 0,25mM, 0.125mM, 0mM.
Calculations:
- The change in absorbance of Sample and standard was calculated by subtracting the first absorbance readings from the second.
- A calibration curve was plotted by plotting the Absorbance of the Standards (Y-axis) against the given concentration of the Standards (X-Axis).
- The Total antioxidant Capacity of the samples was extrapolated from the calibration curve
- The Total antioxidant capacity of the samples is expressed in mMTrolox equivalents
Xanthine Oxidase (XO) Assay: The rate of formation of urate from hypoxanthine was determined by measuring increased absorbance at 290 nm. A unit of activity is that forming one micromole of urate per minute at 25°C 62.
Procedure: Into cuvettes the following was pipetted:
TABLE 2:
| Test | Control | |
| Buffer | 1.9 ml | 1.9 ml |
| Reagent grade water | ---- | 1.0 ml |
| Enzyme | 0.1 ml | 0.1 ml |
| Substrate (at zero time) | 1.0 ml | ---- |
Increase in absorbance was recorded and ΔA290 was determined from the linear portion of the curve. The rate was proportional to enzyme concentration within limits of 0.01 to 0.02 units per test.
Calculations:
Units/ mgP = ΔA/ min × 1000/ 1.22 × 104 ×mg/ ml rection mixture
Units/ ml = ΔA/ min × 1000 × 3 ml × dilution / 1.22 × 104 × 0.1 ml
The molar absorbancy of uric acid = 1.22 X 104 cm-1 62.
PICTURE ABOVE SHOWED THE FEED GIVEN TO THE RATS, THE RAT CAGE THE TOXICANT ADMINISTERED TO THE RATS (PHENANTHRENE)
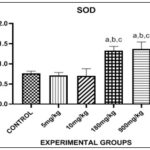
RESULTS: Fig. 1 showed the effect of Phenanthrene on Superoxide Dismutase (SOD) in the liver. There was a significant increase in the levels of SOD in the 180mg and 900mg groups when compared with control. There was significant increase in levels of SOD in the 180 and 900mg groups when compared with 5mg group. There was significant increase in levels of SOD in the 180 and 900mg groups when compared with 10mg group. Meanwhile, there was no statistically significance difference between 900mg group when compared to 180 group. There was no significance difference between 10mg group when compared to 5mg group and control. There was also no significant difference between 5mg group when compared with control.
FIG. 1: EFFECT OF PHENANTHRENE (PHE) ON SUPEROXIDE DISMUTASE (SOD) IN THE LIVER. Bars are expressed in (1.327 ± 0.05302), (1.364 ± 0.07305). a, b, c represent significant differences when compared with control, 5mg and 10mg groups respectively (p<0.05).
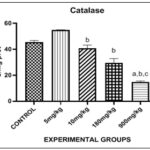
FIG. 2: EFFECT OF PHENANTHRENE (PHE) ON CATALASE (CAT) IN THE LIVER. Bars are expressed in (64.71 ± 10.22), (14.69 ± 0.4072). a, b, c, represent significant differences when compared with control group, 5mg group and 10mg group respectively (p <0.05).
Fig. 2, showed the effect of Phenanthrene on catalase in the liver. There was a significant increase in the level of catalase in the 5mg group when compared with control. There was significant decrease in the level of catalase in 900mg group when compared with control. There was significant decrease in 10mg, 180mg and 900mg group when compared with 5mg group. There was a significant decrease in 900mg group when compared with 10mg group. Meanwhile, there was no significance in 10mg and 180mg group when compared with control group.
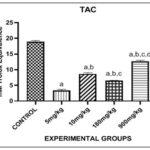
FIG. 3: EFFECT OF PHENANTHRENE (PHE) ON TOTAL ANTIOXIDANT CAPACITY ASSAY (TAC) IN THE LIVER. Bars are expressed in (3.450 ± 0.1559), (8.685 ± 0.1381), (6.537 ± 0.01827), (12.66 ± 0.1795). a, b, c, d represent significant differences when compared with 5mg/kg, 10mg/kg, 180mg/kg and 900mg/kg groups respectively (p<0.05).
Fig. 3 showed the effect of Phenanthrene on Total Antioxidant Capacity assay (TAC) in the liver. There was significant decrease in levels of TAC in 5mg, 10mg, 180mg and 900mg groups when compared to control. There was significant increase in levels of TAC in 10mg, 180mg and 900mg groups when compared with 5mg group. There was significant decrease in levels of TAC in 180mg group when compared with 10mg group. There was significant increase in levels of TAC in 900mg group when compared with 10mg group. There was significant increase in levels if TAC in 900mg group when compared with 180mg group.
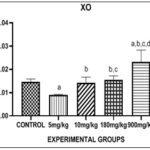
FIG. 4: EFFECT OF PHENANTHRENE (PHE) ON XANTHINE OXIDASE (XO) IN THE LIVER. Bars are expressed in (0.009 ± 9.364), (0.02321 ± 0.002071). a, b, c, d represent significant differences when compared to control groups, 5mg, 10mg and 180mg respectively (p<0.05).
Fig. 4, showed the effect of Phenanthrene on Xanthine Oxidase (XO) in the liver. There was a significant decrease in the level of XO in the 5mg group when compared with control. There was a significant increase in the level of XO in 900mg group when compared with control. There was a significant increase in level of XO in 10mg, 180mg and 900mg group when compared to 5mg group. There was a significant increase in level of XO in 180mg and 900mg groups when compared to 10mg group. There was a significant increase in level of XO in 900mg group when compared to 180mg group. There was no significant difference between 10mg and 180mg group when compared with control group.
DISCUSSION: Oxidative stress is known to be an important mechanism of PAH-induced toxicity 60. PAHs have been shown to be associated with a variety of toxic effects including oxidative stress, inflammation, and immunological disorders after they are metabolically activated to electrophilic intermediates. These reactive intermediates are able to covalently bind to DNA or participate in redox cycles. This process leads to an overproduction of reactive oxygen species, also known as ROS 47. Antioxidant defenses include enzymes such as SOD and CAT. In a normal physiological situation, antioxidant defense systems are thought to prevent the production of ROS and other reactive oxygen species 33.
Antioxidant defenses can be induced by a mild oxidative stress as a compensatory response; however, a severe oxidative stress suppresses the activities of these enzymes and can lead to oxidative damage 64. When OH is produced in excess or the cellular antioxidant defense is deficient, it can stimulate free radical chain reactions with proteins, lipids and nucleic acids, causing cellular damage and even disease 12. Phenanthrene can accumulate in the liver and induce OH production, leading to oxidative stress that alters the activities of antioxidant enzymes such as catalase and superoxide dismutase 64.
Superoxide dismutase and catalase are antioxidant enzymes that play not only a fundamental but also an indispensable role in the antioxidant protective capacity of biological systems against the attack of free radicals. Superoxide dismutase (SOD) is the first detoxifying enzyme and the most powerful antioxidant in the cell. It acts as a first-line of defense system against reactive oxygen species (ROS) and catalyzes the dismutation of two superoxide anion molecules into hydrogen peroxide and molecular hydrogen, making the potentially harmful superoxide anion less dangerous 24. Catalase is a widely distributed antioxidant enzyme that is found in almost all living tissues that consume oxygen. It utilizes iron (Fe) or manganese (Mn) as a cofactor and catalyzes the breakdown or reduction of hydrogen peroxide (H2O2) to water and molecular oxygen, thus completing the detoxification process initiated by SOD. It is abundant in cells, where it continuously searches for and breaks down hydrogen peroxide molecules, to produce oxygen and water 13.
The ability of CAT to effectively limit the concentration of hydrogen peroxide in cells makes it a more reliable tool in regulating the above physiological processes and, in addition, it is a first defense antioxidant enzyme 24. Catalase deficiency in the cells has been linked to various diseases and abnormalities 55.
The decrease in catalase activity and total antioxidant capacity is a sure indicator of oxidative stress 49. SOD deficiency is associated with a number of pathologies including neurodegeneration, myocardial damage and perinatal death in mice 34. Unfortunately, SOD deficiency is quite common as it significantly promotes cellular health and protects the body’s cells from excess oxygen radicals, free radicals and harmful agents that promote cell death 28.
Effect of Phenanthrene on Superoxide Dismutase: Superoxide dismutase (SOD) is the first detoxification enzyme and the most powerful antioxidant in the cell. It acts as a first line of defense against reactive oxygen species and helps neutralize the superoxide ion of the free radical 24. In this study, the results show a significant increase in superoxide dismutase levels in the 180mg and 900mg groups in comparison to control.
This is possibly due to oxidative stress in the liver of these groups, as increased superoxide ion in the cells stimulates the first-order antioxidant enzyme superoxide dismutase to begin oxidizing superoxide anion to hydrogen peroxide and molecular oxygen 24. However, this may cause the accumulation of hydrogen peroxide in the cells. This accumulation will lead to significantly increased amounts of hydrogen peroxide in the cells. This significant increase in the amount of Hydrogen Peroxide in the cells is also known as high levels of hydrogen peroxide in the cells, which is extremely toxic 24. This finding is similar to that in the study carried out by (Ma et al., 2020) 42, where administration of phenanthrene led to increased superoxide dismutase levels.
Effect of Phenanthrene on Catalase: Catalase is a common antioxidant enzyme that is found in almost all living tissues that utilize oxygen. Its function is to convert hydrogen peroxide to water and oxygen, thereby neutralizing it 24.
This is because Hydrogen Peroxide accumulated at high levels in the body can cause cell damage and oxidative stress 24. Oxidative stress is a phenomenon that occurs when the number of reactive oxygen species in the cells is significantly greater than the number of antioxidants needed to neutralize them 59. Catalase levels were measured using spectrophotometry. In this study, the results show a decrease in catalase in the 900mg group in comparison to the control group, which may have been as a result of oxidative stress 49. This decrease in Catalase could have been as a result of the increase in Superoxide dismutase levels. Because the superoxide dismutase is “working overtime” to oxidize the ever increasing levels of superoxide ion in the cells, there becomes excess hydrogen peroxide for the catalase to “clean up”. The more that catalase is being utilized to oxidize the hydrogen peroxide into oxygen and water, with an ever increasing amount of hydrogen peroxide levels, the less catalase there is to perform any antioxidant activity. Eventually, the number of hydrogen peroxide levels significantly increases more than the levels of catalase needed to oxidize it. This leads to oxidative stress 24.
This is similar to the work of (Ma et al., 2020) 42 where administration of environmental toxicant Phenanthrene also led to decrease in catalase. It is also similar to the work of Yin et al., 2007, whose catalase also reduced upon exposure to phenanthrene. The results also show a statistically significant increase in catalase in the 5mg group, which may have occurred as a result of an antioxidant defense being induced by slight oxidative stress in the liver of the 5mg/kg group. Little concentration of the toxicant can trigger slight increase in catalase levels 30.
Effect of Phenanthrene on Total Antioxidant Capacity: Total Antioxidant capacity is a measure of the ability of a biological system to neutralize oxidants and free radicals 59. It is an important biochemical in medical and nutritional studies, as it provides insight into the overall antioxidant status and oxidative stress in the body 27. In this study, there was a significant decrease in total antioxidant capacity levels in the 5mg, 10mg, 180mg and 900mg groups in comparison to the control group. This might have been as a result of oxidative stress in the livers of these groups 49, impairing the ability of their antioxidants to combat against the reactive oxygen species over produced as a result of the environmental toxicant phenanthrene in their livers 42. Upon entry of environmental toxicant phenanthrene into the liver of the experimental groups, there might have been an increase in reactive oxygen species specifically to combat the effects of this strange toxicant 42.
However, upon unwillingness of this toxicant to leave the cells, the naturally occurring reactive oxygen species may have continued to multiply, in an effort to eradicate environmental toxicant phenanthrene. This leads to significant increase in amount of reactive oxygen species in the cells. This significant increase triggered the total antioxidant capacity to neutralize the suddenly excessive reactive oxygen species 59. Unfortunately, the continued presence of phenanthrene in the cells kept triggering the reactive oxygen species to try to combat its effect, which continued to trigger antioxidants to neutralize these excessive reactive oxygen species until finally there is a significant decrease in the total antioxidant capacity 49.
Effect of Phenanthrene on Xanthine Oxidase: Xanthine oxidase is a form of xanthine oxidoreductase, a type of enzyme that generates reactive oxygen species. These enzymes catalyze the oxidation of hypoxanthine to xanthine and further catalyze xanthine to uric acid 7. During severe liver damage, xanthine oxidase is released into the blood, so a blood assay for xanthine oxidase is a way to determine if severe liver damage occurred 52.
In this study there was a significant increase in xanthine oxidase levels in 900mg/kg group of the rats in comparison to control. This might have occurred as a result of severe liver damage 52, and might have resulted in accumulation of uric acid in the joints of the 900mg/kg group, causing various diseases 7. There was a significant decrease in the 5mg/kg group in comparison to control. This might have occurred as a result of superoxide anion suppression by the increase in superoxide dismutase levels in the study, and might have resulted in inhibition of uric acid formation by xanthine oxidase itself 5.
CONCLUSION: Phenanthrene increased oxidative stress markers in male Wistar rats.
ACKNOWLEDGEMENT: Authors acknowledge the Department of Physiology Igbenedion University Okada for providing encouragement and enabling environment for this study.
Funding: The research was self- funded and did not receive any external funding.
CONFLICT OF INTEREST: Authors declare no conflict of interest.
REFERENCES:
- Agarwal T, Banerjee D, Konwarh R, Esworthy T, Kumari J, Onesto V, Das P, Lee BH, Wagener F, Makvandi P, Mattoli V, Ghosh SK, Maiti TK, Zhang LG and Ozbolat IT: Recent Advances in bioprinting technologies for engineering hepatic tissue. Materials Science and Engineering 2021; 123: 11-2013.
- Alturkistani HA, Tashkandi FM and Mohammedsaleh ZM: Histological stains: a literature review and case study. Global Journal of Health Science 2016; 8: 72.
- Apak R, Guclu K, Ozyurek M and Karademir SE: Novel Total Antioxidant Capacity index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine: CUPRAC method. Journal of Agricultural Food Chemistry 2004; 52: 7970-7981
- Allard JP, Aghadassi E, Chau J, Salit I and Walmsley S: The American Journal of Clinical Nutrition 1998; 67: 143-147
- Brentnall M, Rodriguez-Menocal L, Ladron de Guevara R, Cepero E and Boise LH: Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biology 2013; 14(1): 32.
- Brook R: Cardiovascular effects of air pollution Clinical Science. (London) 2008; 115: 175-187.
- Battelli MG, Polito L and Bolognesi A: Xanthine oxidoreductase in atherosclerosis pathogenesis: Not only Oxidative Stress Atherosclerosis 2014; 237: 562-567.
- Brumbarova T and Ivanov R: Perl’s staining for histochemical detection of iron in plant samples. Bio-protocol 2014; 4: 12-45.
- Cerutti P and Trump BF: Inflammation and oxidative stress in carcinogenesis. Cancer Cells 1991; 3: 1-7.
- Cottone S, Lorito MC, Riccobene R, Nardi E, Mule G, Buscemi S, Geraci C, Guaneri M, Arsena R and Cerasola G: Oxidative Stress, inflammation and cardiovascular disease in chronic renal failure. Journal of Nephrology 2008; 21: 175-179.
- Ciencewicki J, Trivedi S and Kleeberger SR: Oxidants and the pathogenesis of lung diseases. Journal of Allergy and Clinical Immunology 2008; 122: 456-468.
- Cheng YM, Lim PS and Wei YH: Increase in oxidative damage to lipids and proteins in skeletal muscle of uremic patients. Free Radical Research 2002; 36(3): 295-302.
- Chelikani P, Fita I and Loewen PC: Diversity of structures and properties among catalases. Cell Mol Life Sci 2004; 61(2): 192-208.
- Dennis RA, Trappe TA, Simpson P, Carroll C, Huang BE, Nagarajan R, Bearden E, Gurley C, Duff GW, Evans WJ, Kornman K and Peterson CA: Interleukin‐1 polymorphisms are associated with the inflammatory response in human muscle to acute resistance exercise. The Journal of Physiology 2004; 560: 617-626.
- Fatokun A, Stone T and Smith R: Front. Biosci 2008; 13: 3288-3311.
- Frei B: Small molecule antioxidant defenses in human extracellular fluids. Molecular Biology of Free Radical Scavenging System 1992; 23-45.
- Goth L: A simple method for determination of serum catalase activity and revision of reference range. Clinical Chemistry 1991; 143-152
- Goth L, Meszaros I and Nkmeth H: Serum catalase enzyme activity in acute pancreatitis. Clinical Chemistry 1982; 28: 1999-2000.
- Guicciardi ME, Malhi H, Mott JL and Gores GJ: Apoptosis and Necrosis in the liver. Comprehensive Physiology 2013; 3: 2.
- Goth L, Nemeth H and Meszaros I: Serum catalase activity for detection of hemolytic diseases. Clinical Chemistry 1983; 29: 741-742.
- Guo Z, Kang Y, Wu H, Li M, Hu Z and Zhang J: Enhanced removal of phenanthrene and nutrients in Wetland sediment with metallic bio char: performance and mechanisms. Chemosphere 2023; 327: 138-523.
- Hintze PE, Buhler CR, Schuerger AC, Calle LM and Calle CI: Alteration of five organic compounds by glow discharge plasma and UV light under simulated Mars conditions Elsevier 2010; 208: 749-757.
- Hong X, Qin J, Chen R, Yuan L, Zha J, Huang C, Li N, Ji X and Wang Z: Phenanthrene-induced apoptosis and its underlying mechanism. Environmental Science and Technology 2017; 51: 14397-14405.
- Ighodaro OM and Akinloye OA: First-line defence antioxidants- superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx): Their fundamental role in the entire antioxidant defense grid. Alexandria Journal of Medicine 2018; 54: 287-293.
- Julien O and Wells JA: Caspases and their substrates. Cell Death and Differentiation 2017; 24: 1380-1389.
- Jaeschke H, Gujral JS and Bajt ML: Apoptosis and Necrosis in Liver disease Liver International 2004; 24: 85-89.
- Kusano C and Ferrari B: Total antioxidant capacity: a biomarker in biochemical and nutritional studies Journal of Molecular Cell Biology 2008; 7: 1-15.
- Krishnamurthy and Wadhwani: Antioxidant enzymes and human health Antioxidant Enzyme 2012; 1: 3-18.
- Keith M, Geranmayegan A, Sole JM, Kurian R, Robinson A, Omran AS and Jeejeebhoy KN: Increased oxidative stress in patients with congestive heart failure. Journal of the American College of Cardiology 1998; 31: 1352-1362
- Kenney MC, Chwa M, Atilano SR, Tran A, Carballo M, Saghizadeh M, Vasiliou V, Adachi W and Brown DJ: Increased levels of Catalase and cathepsin V/L2 but decreased TIMP-1 in keratoconus corneas: evidence that oxidative stress plays a role in this disorder. Investigative Ophthalmology and Visual Science 2005; 46: 823-832.
- Katz NR: Metabolic heterogeneity of hepatocytes across the liver acinus. The Journal of Nutrition 1992; 122: 843-849.
- Kubes P and Jenne C: Immune Responses in the Liver Annual Review of Immunology 2018; 36: 247-277.
- Livingstone DR: Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Marine Pollution Bulletin 2001; 42: 656-666.
- Lebovitz RM: Quantitative examination of dynamic interneuronal coupling via single-spike extracellular potassium ion transients. Journal of Theoretical Biology 1996; 180: 11-25.
- Li J, Peng W, Yin X, Wang X, Liu Z, Liu Q, Deng Z, Lin S and Llang R: Identification of an efficient phenanthrene-degrading pseudarthrobacter sp. L1AW and characterization of its metabolites and catabolic pathway Journal of Hazardous Materials 2024; 465: 133-138.
- Li H, Li L, Xu H, Du H, Wang L: Insights into phenanthrene attenuation by hydroxyl radicals from reduced iron-bearing mineral oxygenation Journal of Hazardous Materials 2022; 439: 129-658.
- Mumford JL, Li X, Hu F, Lu XB and Chuang JC: Human exposure and dosimetry of polycyclic aromatic hydrocarbons in urine from Xuan Wei, China with high lung cancer mortality associated with exposure to unvented coal smoke Carcinogenesis 1995; 16: 3031-3036.
- Manco R, Leclerq IA and Clerbaux LA: Liver regeneration: different sub-populations of parenchymal cells at play choreographed by an injury specific microenvironment. International Journal of Molecular Sciences 2018; 19: 4115.
- Magnani L, Gaydou M and Jean CH: Spectrophotometric measurement of antioxidant properties of flavones and flavonols against superoxide anion. Anal Chim Acta 2000; 411: 209-216.
- Marklund S and Marklund G: Involvement of the superoxide anion radical in the autooxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 1974; 47: 469-474.
- Martins M, Costa PM, Ferreira AM and Costa MH: Comparative DNA damage and oxidative effects of carcinogenic sediment-bound PAHs in the gills of a bivalve. Aquatic Toxicology 2013; 142: 85-95.
- Ma H, Wang H, Zhang H, Guo H, Zhang W, Hu F, Yao Y, Wang D, Li C and Wang J: Effects of phenanthrene on oxidative stress and inflammation in lung and liver of female rats. Environmental toxicology 2020; 35: 37-46.
- Marti-Cid R, Llobet JM, Castell V and Domingo JL: Evolution of the dietary exposure to polycyclic aromatic hydrocarbons in Catalonia. Spain Food and Chemical Toxicology 2008; 46: 3163-3171.
- Norata GD, Pirillo A and Catapano L: Modified HDL: Biological and physiopathological consequences Nutrition. Metabolism and Cardiovascular Diseases 2006; 16: 371-386.
- Nassir F, Rector SR, Hammoud GM and Ibdah JA: Pathogenesis and prevention of hepatic steatosis. Journal of Gastroenterology and Hepathology 2015; 11: 3.
- Nicholson DW and Thornberry NA: Caspases: Killer proteases Trends in biochemical Sciences 1997; 22: 299-306.
- Omayma EA, Sawsan MA and El Nady MM: Application of polycyclic aromatic hydrocarbons in identification of organic pollution in seawater around Alexandria coastal Area, Egypt Journal of Environmental Life Science 2016; 1: 39-55.
- Owumi SE, Otunla MT, Elerewe OO and Arunsi UO: Co-exposure to aflatoxin B1 and therapeutic coartem worsens hepatic and renal function through enhanced oxido-inflammatory responses and apoptosis in rats. Toxicon 2023; 222: 106-988.
- Pal S, Singh A, Chattopadhyay A and Goswami A: Oxidative Stress and its implications in various diseases: A comprehensive review. Current Issues in Molecular Biology 2023; 45: 6651-6666.
- Porter AG and Janicke RU: Emerging roles of caspase-3 in apoptosis. Cell Death and Differentiation 1999; 6: 99-104.
- Pawlowski J and Kraft A: Bax induced Apoptotic cell death. Proceedings of the National Academy of Sciences 2000; 97: 529-531.
- Pacher P, Nivorozhkin A and Szabo C: Therapeutic effects of xanthine oxidase inhibitors: renaissance half a century after the discovery of allopurinol. Pharmacological Reviews 2006; 58(1) 87-114.
- Rishi P, Arora S, Kaur UJ, Chopra K and Kaur IP: Better Management of Alcohol Liver Disease Using a ‘Microstructured Synbox’ System Comprising plantarum and EGCG. Journals PLOS One 2017; 12: 780-910.
- Reiter EB, Escher BI, Siebert U and Jahnke A: Environment. International 2022; 165: 107-337.
- Rass P, Goth L and Pay A: Catalase enzyme mutations and their association with diseases. Molecular Diagnosis 2004; 8(3): 141- 149.
- Rodriguez SA, Murray AP and Leiro JM: Xanthine Oxidase Inhibition by Aqueous Extract of Limonium brasiliense (Plumbaginaceae) 2020.
- Sasse D, Spornitz UM and Maly IP: Liver Architecture Enzyme 1992; 46: 8-32.
- Santana JL, Massone CG, Valdes M, Vazquez R, Lima LA and Olivares-Rieumont S: Occurrence and source appraisal of polycyclic aromatic hydrocarbons (PAHs) in surface waters of the Almendares River, Cuba. Archives of Environmental Contamination and Toxicology 2015; 69: 143-152.
- Silvestrini A, Meucci E, Ricerca BM and Mancini A: Total antioxidant capacity: Biochemical aspects and clinical significance. International Journal of Molecular Sciences 2023; 24: 10-978.
- Shi Z, Tao S, Pan B, Fan W, He XC, Zuo Q, Wu SP, BG Li, Cao J, Liu WX, Xu FL, Wang XG, Shen WR and Wong PK: Contamination of rivers in Tianjin, China by polycyclic aromatic hydrocarbons. Environmental Pollution 2005; 134(1): 97-111.
- Wang L, Du H, Xu H, Li H and Li L: Insights into phenanthrene attenuation by hydroxyl radicals from iron-bearing mineral oxygenation. Journal of Hazardous Materials 2022; 439: 129-658.
- Westerfield PE: The development of an electronic device to measure the velocity, acceleration and jerk of shaft rotation. University of Wyoming 1959; 12: 157-159.
- Westerholm R, Christensen A, Tornqvist M, Ehrenberg L, Rannug U, Sjogren M, Rafter J, Soontjens C, Almen J and Gragg K: Comparison of exhaust emissions from Swedish environmental classified diesel fuel (MK1) and European Program on Emissions, Fuels and Engine Technologies (EPEFE) Reference Fuel: A chemical and Biological Characterization, with Viewpoints on Cancer Risk Environmental Science and Technology 2001; 35: 1748-1754.
- Yin Y, Jia H, Sun Y, Yu H, Wang X, Wu J and Xue Y: Bioaccumulation and ROS generation in liver of Carassiusauratus, exposed to phenanthrene Comparative Biochemistry and Physiology Part C: Toxicology and Pharmacology 2007; 145: 288-293.
- Zheng JL, Parfett C, Williams A, Yagminas A, Zhou G, Douglas GR and Yauk CL: Assessment of subclinical, toxicant-induced hepatic gene expression profiles after low-dose, short term exposures in mice. Regulatory Toxicology and Pharmacology 2011; 60: 54-72.
How to cite this article:
Victory EO, Ijeoma CE and Emeka HE: Effect of phenanthrene exposure on the total antioxidant capacity, catalase, superoxide dismutase and xanthine oxidase, of the liver. Int J Pharmacognosy 2026; 13(2): 88-98. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(2).88-98.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
88-98
782 KB
82
English
IJP
Enemchukwu Onyinye Victory, C. Enemuo Ijeoma and H. Enemuo Emeka *
Department of Medicine, Nnamdi Azikiwe University, Nnewi Campus.
emekaenemuo875@yahoo.com
31 January 2026
25 February 2026
26 February 2026
10.13040/IJPSR.0975-8232.IJP.13(2).88-98
28 February 2026