DEVELOPMENT AND CHARACTERIZATION OF SOYA PHOSPHATIDYLCHOLINE COMPLEX OF COUMESTANS FROM ECLIPTA ALBA FOR THE EFFECTIVE MANAGEMENT OF HEPATOTOXICITY
HTML Full TextDEVELOPMENT AND CHARACTERIZATION OF SOYA PHOSPHATIDYLCHOLINE COMPLEX OF COUMESTANS FROM ECLIPTA ALBA FOR THE EFFECTIVE MANAGEMENT OF HEPATOTOXICITY
Abhishek Kumar Jain 1, Anki Jain 1 and Ashish Jain * 2
Sagar Institute of Pharmaceutical Sciences 1, Sagar - 470228, Madhya Pradesh, India.
Department of Pharmaceutical Sciences 2, Dr. Harisingh Gour Central University, Sagar - 470003, Madhya Pradesh, India.
ABSTRACT: It is known that Eclipta alba Hassk. (Family: Compositae) contains coumestans (Wedelolactone and dimethyl wedelolactone) are used in liver disorders. The objective of the present investigation was to develop a novel formulation of these coumestans in combination with the soya phosphatidylcholine (PC), to overcome the limitation of absorption and to investigate the protective effect of the coumestans-phosphatidylcholine complex (C-PC) on carbon tetrachloride-induced acute liver damage in rats. Methanolic extract (ME) of the whole plant of Eclipta alba was fractionated with water and then with ethyl acetate. The ethyl acetate fraction of methanolic extract (EFME) contains coumestans which were characterized by chemical tests. EFME and PC prepared the C-PC. FT-IR and DSC spectroscopy characterized the C-PC. In-vitro drug release from EFME and C-PC through egg membrane was performed using UV-Visible spectrophotometer. The hepatoprotective activity of C-PC (Equivalent to 5.35 and 10.7 mg/kg body weight of EFME), ME 250 mg/kg and EFME 5.35 mg/kg were evaluated on various enzymes level. C-PC significantly provided better protection to the liver by restoring the enzyme levels of SGPT (Serum glutamate pyruvic transaminase), SGOT (Serum glutamate oxaloacetate transaminase), ALP (Alkaline phosphatase) and total bilirubin on carbon tetrachloride (CCl4) induced liver damaged animals (P<0.001). Histopathological studies were performed, and the results showed that the C-PC provided better protection to rat liver than ME and EFME at similar doses as well as shown significant regeneration of hepatocytes, central vein, intact cytoplasm and nucleus.
| Keywords: |
Soya phosphatidylcholine, Coumestans, Eclipta Alba, Hepatotoxicity, Wedelolactone
INTRODUCTION: Eclipta alba (L.) Hassk. (Syn. Eclipta prostrata L.), is a plant belonging to the family Asteraceae. It grows commonly in moist places as a weed all over the world.
It is widely distributed throughout India, China, Thailand, and Brazil. E. alba is widely used in India as a cholagogue and deobstruent in hepatic enlargement, for jaundice and other ailments of the liver and gallbladder 1. It is also reported to improve hair growth and color 2. The herb Eclipta alba contains mainly coumestans, i.e. wedelolactone and demethyl-wedelolactone, β-amyrin, ecliptine, ecliptal, α-trimethylmethanol, stigmasterol, 2- formyl- αterthenylandloteolin-7 glucoside 3, 4, 5.
Coumestans are being used in phytopharmaceutical formulations of medicines prescribed for the treatment of cirrhosis of the liver and infectious hepatitis 6. Wedelolactone and demethyl wedelolactone both constituents exhibit antihepatotoxic activity 7. Wedelolactone is also used for direct inhibition of IKK (Inhibitor of Kappa beta kinase) complex resulting in suppression of LPS (Lipopolysaccharide) -induced caspase-11 expression 8, anti-venom 9 and antioxidant 10. Most of the bioactive constituents of herbal drugs are water soluble molecules. However, water-soluble phytoconstituents like flavonoids, i.e., coumestans are poorly absorbed either due to their multiple-ring large size molecules which cannot be absorbed by simple diffusion or due to their poor miscibility with oils and other lipids, severely limiting their ability to pass across the lipid-rich outer membranes of the enterocytes of the small intestine 11. Wedelolactone has lesser hydrophobicity 12, so it may be assumed that it will have poor absorption profile.
Keeping in view, the promise of coumestan (Wedelolactone) as a therapeutically active agent and its poor oral absorption, it is pertinent to develop a new formulation of coumestan, which can increase its oral absorption and enhance its therapeutic activity. Several studies have indicated the beneficial role of phospholipids in enhancing the therapeutic efficacy of some molecules having poor oral absorption. Silybin is one such molecule having poor oral bioavailability. Efforts have been given to prepare phospholipids complex of silybin and to increase its oral bioavailability, therefore, therapeutic efficacy. It was observed that the silybin-phospholipids complex has the significant upper hand over the pure molecule in protecting the liver and exerting antioxidant activities 13, 14, 15, 16, 17. A recent study with quercetin-phospholipid complex showed that the formulation exerted better therapeutic efficacy than the molecule in rat liver injury induced by carbon tetrachloride 18.
Aim of the present study was to prepare phospholipids complex of coumestans (i.e. wedelolactone) to determining its physicochemical properties and its hepatoprotective activity in carbon tetrachloride intoxicated rats in comparison to ME and its ethyl acetate fraction (EFME), to substantiate the claim that complexation between coumestan (Wedelolactone) and phospholipid can enhance the therapeutic efficacy of the parent molecule by increasing its absorption.
EXPERIMENTAL:
Materials: Eclipta alba was collected from the area of Shahgarh District, Sagar (M.P.), India and authenticated by Dr. Amarjeet Bazaz, MVM College (references number 01/2008), Bhopal, India. Phospholipid hydrogenated soy phosphatidylcholine (PC) was purchased from Lipoid, Ludwigshafen, Germany. Wedelolactone was received as a gift sample from Laila Impex Vijayawada, India. Dichloromethane, n-hexane, and chloroform were purchased from Rankem (Pvt.) Ltd. Assay kits for serum alanine amino-transferase (ALT) and aspartate amino-transferase (AST), total bilirubin and alkaline phosphatase (ALP) were purchased from vital diagnostics Ltd., Mumbai, India. Animals used in the study were obtained from animal house of VNS Institute of Pharmacy, Bhopal, India and were kept under pathogen-free conditions. All other reagents used were of analytical grade and were used as received.
Extraction and Fractionation: The dried powdered plant material (463 g) was extracted with methanol (3.0 L) by maceration with agitation at room temperature for 24 h. The solvent was removed with reduced pressure obtaining a dark green residue (90.3 g). Part of the residue (70 g) was suspended in 770 ml of water and warmed in a water bath at <75 °C for 30 min, the aqueous phase was filtered and successively extracted four times with 250 ml of ethyl acetate; the ethyl acetate fraction (brown) was dried under vacuum giving 1.50 g of brown residue. Legal test and Keddie's test were performed to confirm the presence of lactones in the EFME. TLC studies on EFME and standard drug wedelolactone were performed using stationary phase silica gel 60 F254 using mobile phase, CHCl3 + Methanol (70:30), FeCl3 0.5N was used as detecting agent 7.
Preparation of Coumestan - Phospholipid Complex: The complex was prepared with EFME and PC at a ratio of 1:1. A weighed amount of EFME and PC were taken in a 100 ml round bottom flask and 50 ml of dichloromethane were added. The mixture was agitated continuously on a magnetic stirrer at a temperature not exceeding 40°C for 2 h. The resultant clear solution was concentrated in vacuum to a 1/3 of its volume, and 25 ml of n-hexane was added to it with continuous stirring. The C-PC was precipitated, and the precipitate was filtered and dried under vacuum to remove traces of solvents.
Solubility Studies: EFME, phosphatidylcholine and the prepared C-PC 10 mg were dissolved in 2 ml chloroform separately to check the solubility profile.
Microscopic View of the Complex: The complex was suspended in distilled water, and a drop was placed on a slide and covered with a cover slip. Microscopic view of the complex was observed at a magnification of 400X (Labomed microscope type CX R3).
FT-IR Spectroscopy: FT-IR spectra for the various powders and semisolids of EFME, PC, PM, and C-PC were obtained on an 8400 Shimadzu FTIR spectrometer (Shimadzu, Tokyo, Japan) in the transmission mode with the wave number region 500-4,000 cm-1. KBr pellets were prepared by gently mixing 1 mg sample powder with 100 mg KBr.
Differential Scanning Calorimetry (DSC): Thermograms of EFME, PC, C-PC and PM (1:1 ratio) of EFME and PC, were recorded using Mettler Toledo Star* system (UK). The samples were sealed in the aluminum crimp cell and heated at the speed of 10 °C/min from 10 to 350 °C in the nitrogen atmosphere (60 ml/min).
Dissolution Study: The dissolution study was performed to determine the effect of complex formation on the release profile of wedelolactone. For that purpose egg membrane was selected because egg membrane mimics the biological membrane and in-vivo performance based on in-vitro results can be predicted more accurately. The release through egg membrane was measured for 2 h. Samples were withdrawn and replaced with fresh media to maintain a constant volume of dissolution media. Concentration measurement was done spectrophotometrically using UV-Visible spectrophotometer at 347 nm.
In-vivo Studies: All experiments were performed due permission from the Institutional Animal Ethical Committee (Reg. No. 778/03/ c/CPCSEA). Adult Albino rats (80-110 g) of either sex were chosen for hepatoprotective activity. All the tests were carried out in a quiet laboratory setting with ambient illumination and at temperature 37 ± 2 °C. Animals were allowed to acclimatize to the laboratory for 7 days before the experiment begins. Animals were divided into six groups of six in each. Group 1 served as control and received vehicle 1% w/v gum tragacanth at a dose of 1 ml/ kg p.o. for 4 days and coconut oil (1 ml/kg i.p.) on day 2 and 3. Groups 2, 3, 4, 5 and 6 received 2 ml/kg i.p. of CCl4: coconut oil (1:1) on second and third day 30 min after treatment with ME, EFME and C-PC. Animals from CCl4 group received vehicle 1% w/v gum tragacanth at a dose of 1 ml/kg p.o. for 4 days. The ME of Eclipta alba was administered to Group 3 at the dose of 250 mg/kg p.o. for 4 days. The EFME of Eclipta alba was administered to Group 4 at the dose of 5.35 mg/kg p.o. for 4 days. Groups 5 and 6 were treated with C-PC at doses equivalent to 5.35 mg/kg p.o. and 10.7 mg/kg p.o. of EFME for 4 days, respectively. The CCl4, ME, EFME and C-PC were administered concomitantly to the respective groups of animals. All the animals were sacrificed on day 5 under light ether anesthesia. The blood samples were collected separately by carotid bleeding into sterilized dry centrifuge tubes and allowed to coagulate for 30 min at 37 °C. The clear serum was separated at 2500 rpm for 10 min, and biochemical parameters were evaluated to assess liver functions viz., total bilirubin 19, SGPT 20, SGOT 21 and serum ALP 22 using star 21plus autoanalyzer. The results were summarized in Table 1.
Statistical Analysis: All the experiments were conducted using at least six animals in each group, and the biochemical estimations have been reported as mean ± SEM and calculated for each parameter. The statistical significance between groups was analyzed using one way ANOVA followed by Dunnett's Multiple Comparison Test. A P-value (<0.001) was considered significant.
Histopathology: A portion of liver tissue in each group was fixed in 10% formalin then after paraffin embedding and block making, serial sections of 5m thicknesses were made, stained with hematoxylin and eosin and examined under Labomed microscope CX R3.
RESULTS AND DISCUSSION:
Extraction and Fractionation: For fractionation, the residue of the ME of the whole plant of Eclipta alba was first treated with water, and the aqueous phase was fractionated with ethyl acetate. Legal test and Keddie's test were found positive showing presence of lactones in the EFME. TLC studies have shown the presence of wedelolactone in the EFME (Rf value 0.61).
The yield of Prepared Complex: C-PC was prepared by a simple and reproducible method, yielding 70.0 ± 2.26% of the product. The resultant C-PC was kept in an amber colored glass bottle, stored at room temperature.
Solubility Profile: Solubility test revealed that EFME was not soluble in chloroform, while PC and the prepared C-PC were soluble indicating that a reaction occurred between the EFME and PC resulting in producing a lipophilic envelope which allows the complexed coumestans to dissolve in low polarity solvents.
Microscopic Observations: The microscopic view Fig. 1 shows the presence of spherical structures of the C-PC. The vesicles consisted of PC, and coumestans were intercalated in the lipid layer.
FIG. 1: PHOTOMICROGRAPH OF C-PC AT 400X
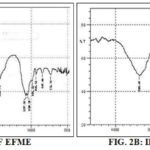
FT-IR Spectroscopy: FT-IR studies were done to detect the possible interaction between EFME and PC in the C-PC Fig 2A, 2B and 2C.
FIG. 2C: IR SPECTRA OF C-PC
Hydroxyl (–OH) stretching band at 3345.48 cm-1, -C–H stretching band of saturated long fatty acid chain at 2930.26 and 2860.77 cm-1, carbonyl stretching band at 1728.49 cm-1 in the fatty acid ester, –P=O stretching band at 1240.56 cm-1 and –P–O–C stretching band at 1060.87 cm-1 in the PC structure, whereas vibration peaks of –OH stretching (nOH, 3300.36 cm-1), –C=O (nC=O, 1708.30 cm-1), –C=C vibration in the benzene-ring (nC=C, 1610.12 1572.78, 1511.82, 1449.71 cm-1), –C– O–R (nC–O, 1049.38 cm-1) and the –C–C single bond (nC–C(benzene), 945.51, 847.44, 735.78 cm-1) were detected in the IR spectra of EFME. The spectra of the C-PC shows shifts, disappearances or attenuation of the characteristic EFME bands revealed a modification of its environment, hence providing evidence for the possible interaction occurring between the EFME and PC. The C-PC spectra exhibited a shift in the benzene-ring framework stretching bands from 1610.12 and 1572.78 cm-1 to a new band at 1593.25 cm-1, and –C–C vibrations of benzene-ring moved to lower wave numbers of 932.56, 833.2 and 728.59 cm-1, all suggesting a possible change in the environment of benzene-ring framework of coumestans present in EFME.
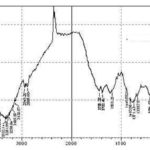
Differential Scanning Calorimetry (DSC): Differential scanning calorimetry (DSC) represents a thermoanalytical method that measures the difference in the thermal energy required to increase the temperature of a sample and a reference with well-defined heat capacity as a function of the temperature. Ideally, both the sample and the reference are kept at an identical temperature, while the temperature increases linearly as a function of time. As soon as the sample undergoes a physical transformation; either exothermic or endothermic, the heat flow to the sample will be decreased or increased relative to the reference, thus, resulting in a DSC signal 23.
Fig. 3A, 3B, 3C and 3D shows the DSC thermograms of EFME (3A), PC (3B), C-PC (3C) and physical mixture (PM) of EFME and PC (3D). The thermogram of EFME shows three peaks first broad at 142.36 °C, second at 191.16 °C and third sharp at 220.03 °C. Thermogram of PC exhibit two different peaks; the first peak (80.92 °C) is mild, which appears because of the hot movement of PC polar head group. The second peak (152.19 °C) is very sharp, and it appears due to phase transition from gel to the liquid crystalline state. The non-polar hydrocarbon tail of PC may be melted during this phase, yielding a sharp peak. Two peaks were appeared in the DSC thermogram of C-PC first broad peak, with an onset at 61.68 °C and maximum occurrence at 106.07 °C, which is different from the peaks of the individual components of the complex and second sharp at 220.03 °C also present in the DSC curve of EFME. PM of EFME and PC shows four peaks, first at 81.60 °C, second at 142.36 °C third at 191.16 °C and fourth at 220.03 °C. The first has the same onset temperature of 61.68 °C of the C-PC, and the later three have same onset temperatures 118.76, 182.07 and 207.21 °C of EFME. It may be assumed that with the rise in temperature the PC become melted and EFME gets dissolved in PC, partly forming the complex. The thermogram of the C-PC exhibits a peak which differs from the peak of EFME and PC. It is evident that the original peaks of EFME and PC disappear from the thermogram of C-PC and the phase transition temperature is lower than that of PC.
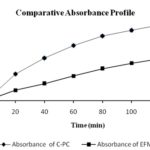
Dissolution Study: Comparative dissolution study was performed between EFME of Eclipta alba and C-PC. The data are shown in Fig. 4. The less quantity of drug detected in case of EFME which may be attributed to hydrophilic nature of the wedelolactone, which prevent it from crossing through the egg membrane while higher absorbance was detected in case of C-PC. The higher concentration in case of C-PC may be attributed due to the presence of a PC, which provides lipophilicity to the resulting complex. The lipophilicity makes it possible to easily cross through the egg membrane. From the data, it may be hypothesized that the C-PC will show better bioavailability in comparison to EFME.
FIG. 4: COMPARATIVE ABSORBANCE PROFILE OF C-PC AND EFME
In-vivo Studies: Since, the changes associated with CCl4-induced liver damage are similar to that of acute viral hepatitis 24. The hepatotoxicity induced by CCl4 is due to its metabolite CCl3• carbon trichloride free radical that alkylates cellular proteins and other macromolecules with a simultaneous attack on lipids, in the presence of oxygen, to produce lipid peroxides, leading to liver damage 25.
TABLE: 2 EFFECT OF ME, EFME AND C-PC OF ECLIPTA ALBA ON CCl4-INDUCED HEPATOTOXICITY IN RAT
| Groups (n) | Dose | SGPT (U/l) | SGOT (U/l) | Bilirubin (mg/dl) | ALP (U/l) |
| Group 1
(Control) |
1 % w/v Tragacanth suspension 1 ml/kg p.o. | 44.62±0.91 | 75.60±1.25 | 0.54±0.01 | 178.40±3.75 |
| Group 2
(CCl4 control) |
CCl4
2 ml/kg i.p. |
186.60±2.02a* | 138.50±1.66a* | 2.17±0.02a* | 412.20±3.18a* |
| Group 3
(ME + CCl4 treated) |
250 mg/kg p.o.+ CCl4
2 ml/kg i.p. |
122.40±0.94b* | 109.70±1.27b* | 1.42±0.01b* | 289.90±2.27b* |
| Group 4 (EFME + CCl4 treated) | 5.35 mg/kg p.o.+ CCl4
2 ml/kg i.p. |
95.08±1.15b* | 100.60±1.3b* | 1.22±0.02b* | 247.90±2.35b* |
| Group 5 (C-PC 1 + CCl4 treated) | 5.35 mg/kg p.o.+ CCl4
2 ml/kg i.p. |
82.86±1.37b* | 94.06±1.04b* | 1.07±0.02b* | 217.80±1.79b* |
| Group 6 (C-PC 2 + CCl4 treated) | 10.7 mg/kg p.o.+ CCl4
2 ml/kg i.p. |
69.77±0.83b* | 83.47±0.95b* | 0.95±0.01b* | 196.30±2.13b* |
Values are Mean ± SEM of 6 animals each in a group. A significant statistical test for comparison was done by ANOVA, followed by Dunnet’s `t’ test (n = 6). Comparison between: a–Group 1 vs. Group 2, b–Group 2 vs. Group 3, 4, 5 and 6. *P<0.0001
Aminotransferases are present in high concentration in liver, an important class of enzymes linking carbohydrate and amino acid metabolism. Alanine aminotransferase and aspartate aminotransferase are well known diagnostic indicators of liver disease. In cases of liver damage with hepatocellular lesions and parenchymal cell necrosis, these marker enzymes are released from the damaged tissues into the bloodstream 26. Alkaline phosphatase is a membrane-bound enzyme, and its elevations in plasma indicate membrane disruption in the organ.
Alkaline phosphatases, although not a liver-specific enzyme, the liver is the major source of this enzyme. The level of this enzyme increases in cholestasis 27. Hepatotoxicity is characterized by a cirrhotic liver condition which in turn increased the bilirubin release 28. Serum SGPT, SGOT, total bilirubin, and ALP levels were selected as parameters to measure the hepatoprotective effect of C-PC. Table 1 compares the effect of ME, EFME and C-PC at different dose levels on the CCl4-induced rise in serum SGPT, SGOT, total bilirubin and ALP levels of the rat. From the results, it can be observed that group 2 was found to possess highly significant increases in (P<0.001) levels of serum SGPT, SGOT, total bilirubin, and ALP when compared with group 1.
The treatment of animals with ME at a dose of 250 mg/kg (group-3) and EFME at 5.35 mg/kg (group-4), afforded a highly significant (P<0.001) protection against the CCl4 induced enhanced serum SGPT, SGOT, total bilirubin, and ALP levels and the results were comparably better in EFME. Treatment of animals with C-PC equivalent to 5.35 mg/kg of EFME (group 5) provided highly significant protection (P<0.001) against CCl4-induced rise in serum SGPT, SGOT, total bilirubin, and ALP levels. The results were comparably better in comparison with group 3 and group 4.
On comparing the SGPT, SGOT, total bilirubin, and ALP levels, it was observed that C-PC at the dose equivalent to 10.7 mg/kg of EFME (group 6) revealed significantly greater protection against CCl4-induced serum SGPT, SGOT, total bilirubin, and ALP levels. The results were better as compared to C-PC at the dose equivalent to 5.35 mg/kg of EFME (group 5). Thus, a dose-dependent hepatoprotective activity of C-PC was observed.
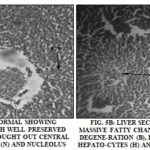
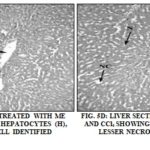
Histopathological Observations: Histopathological studies of liver were observed for a visual assessment of hepatic architecture. Fig. 5A, 5B, 5C, 5D, 5E, and 5F show the representative photomicrographs of a liver section of rats. The rat treated with vehicle control (group 1) Fig. 5A showed a normal hepatic architecture as hepatocytes, central vein, nucleus, cytoplasm, and visible portal tract.
Treatment with CCl4 (2 ml/kg) (group 2) resulted in acute hepatotoxicity as observed from massive fatty changes, necrosis, ballooning degeneration, degeneration of hepatocytes and loss of cellular boundaries Fig. 5B. Treatment of animals with ME (250 mg/kg, group 3) resulted in hepatoprotection, as can be observed by the recovery of hepatocytes, lesser necrosis and a well-identified nucleus which indicate normal hepatic architecture Fig. 5C. The similar results were observed on pretreatment of animals with EFME at 5.35 mg/kg (group 4) Fig. 5d.
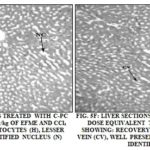
C-PC at a dose equivalent to 5.35 mg/kg and 10.7 mg/kg of EFME (group 5 and 6) showed better protection then EFME as observed by the presence of normal hepatocytes, central vein, intact cytoplasm and nucleus Fig. 5E and 5E.
CONCLUSION: The hepatoprotective activity of ME 250 mg/kg, EFME 5.35 mg/kg and C-PC (equivalent to 5.35 and 10.7 mg/kg body weight of EFME) was evaluated by measuring SGPT, SGOT, Bilirubin, and ALP enzymes level. From the results, it can be observed that animals of CCl4 control were found to possess highly significant increases in levels of serum SGPT, SGOT, total bilirubin, and ALP when compared with normal. The treatment of animals with ME at a dose of 250 mg/kg and EFME at 5.35 mg/kg and C-PC equivalent to 5.35 mg/kg of EFME afforded highly significant protection against the CCl4 induced enhanced serum SGPT, SGOT, total bilirubin, and ALP levels.
The results were comparably better in C-PC than EFME and ME and ME was found least active. The treatment of animals with C-PC at the dose equivalent to 10.7 mg/kg of EFME revealed significantly greater protection against CCl4-induced serum SGPT, SGOT, total bilirubin and ALP levels. The results were better as compared to C-PC at the dose equivalent to 5.35 mg/kg of EFME. Thus, a dose dependent hepatoprotective activity of C-PC was observed. Histopathological studies were also performed. The rat treated with a vehicle control showed a normal hepatic architecture as hepatocytes, central vein, nucleus, cytoplasm and visible portal tract.
Treatment with CCl4 (2 ml/kg) resulted in acute hepatotoxicity as observed from massive fatty changes, necrosis, ballooning degeneration, degeneration of hepatocytes and loss of cellular boundaries. Treatment of animals with ME 250 mg/kg resulted in hepatoprotection, as can be observed by the recovery of hepatocytes, lesser necrosis and a well identified nucleus which indicate normal hepatic architecture. The similar results were observed on pre treatment of animals with EFME at 5.35 mg/kg. C-PC at a dose equivalent to 5.35 mg/kg and 10.7 mg/kg of showed better protection then EFME as observed by the presence of normal hepatocytes, central vein, intact cytoplasm and nucleus. These findings suggested that ME, EFME and C-PC administration has significantly neutralized the toxic effect of CCl4 and helped in the regeneration of hepatocytes. The investigations also confirm that the phosphatidylcholine complex is another break-through in the development of herbal products as the complex formation process enhances the efficacy of herbal compounds by improving their absorption, bioavailability, and delivery of therapeutic substances of herbal origin to the specific tissue or organ involved.
ACKNOWLEDGEMENT: The authors are thankful to Dr. Zia Ul Hasan for identification of plants and to Laila Impex Vijayawada to provide Wedelolactone.
CONFLICT OF INTEREST: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
REFERENCES:
- Orning L, Hammarstrom S and Samuelsson B: Leukotriene D: a slow-reacting substance from rat basophilic leukemia cells. Proc Natl Acad Sci 1980; 77: 2014-2017.
- Kirtikar KR and Basu BD: Indian Medicinal plants, International Book Distributor, Dehradun, Ed 2nd, 1998.
- Govindachari TR, Nagrajan K and Pai SR: Wedelolactone from Eclipta alba. Journal of Scientific and Industrial Research 1956; 15: 664-665.
- Krishnaswamy NR and Prasanna S: Occurrence of demethylwedelolactone and 2-formyl-α-triethyl in Eclipta alba and the fecile oxidation of α-terthienyl Indian Journal of Chemistry 1970; 8: 761-762.
- Krishnaswamy NR, Sheshadri TR and Sharma BR: The structure of a new polythienyl from Eclipta alba. Tetrahedron Letters 1966; 35: 4227-4230.
- Murphy RC, Hammarstrom S and Samuelsson B: Leukotriene C: A slow-reacting substance from murine mastocytoma cells. Proceedings of the National Academy of Sciences of the United States of America 1979; 76: 4275- 4279.
- Wagner H, Geyer B, Kiso Y, Hikino H and Rao G: Coumestans as the main active principles of the liver drugs Eclipta alba and Wedelia calendulacea-1. Planta Medica 1986; 5: 370-374.
- Kobori M, Yang Z, Gong D, Heissmeyer V, Zhu H, Jung Y-K, Angelica M, Gakidis M, Rao A, Sekine T, Ikegami F, Yuan C and Yuan J: Wedelolactone suppresses LPSinduced caspase-11 expression by directly inhibiting the IKK Complex. Cell Death and Differentiation 2004; 11: 123-130.
- Mors WB, Nascimento MC, Parente JP, Silva MH, Melo PA and Suarez-Kurtz G: Neutralization of lethal and myotoxic activities of South American rattlesnakes venom by extracts and constituents of the plant Eclipta prostata (Asteraceae). Toxicon 1989; 27: 1003-1009.
- Unnikrishnan KP, Fathima A, Hashim KM and Balachandran I: Antioxidant studies and determination of wedelolactone in alba. J Plant Sci 2007; 2(4): 459- 464.
- Manach C, Scalbert A and Morand CC: Polyphenols: food sources and bioavailability. Am J Cl Nut 2004; 79: 727-47.
- Basu NK, Waffo AB, Talele TT, Basu A, Costa PRR, Silva AJM, Sarafianos SG and Noel F: Identification and characterization of coumestans as novel HCV NS5B polymerase inhibitors. Nucleic Acid Res 2008; 36(5): 1482-1496.
- Carini R, Comoglio A, Albano E and Poli G: Lipid peroxidation and irreversible damage in the rat hepatocyte model: protection by the silybin-phospholipid complex. Biochem Pharmacol 1992; 43: 2111-2115.
- Conti M, Malandrino S and Magistretti MJ: Protective activity of silybin-phosphatidylcholine complex on liver damage in rodents. Jpn J Pharmacol 1992; 60: 315-321.
- Comoglio A, Tomasi A, Malandrino S, Poli G and Albani E: Scavenging effect of silipide, a new silybinphospholipid complex, on ethanol-derived free radicals. Biochem Pharmacol 1995; 50: 1313-1316.
- Morazzoni P, Montalbetti A, Malandrino S and Pifferi G: Comparative pharmacokinetics of silybin-phosphatidyl choline complex and silymarin in rats. Eur J Drug Metab Pharmacokinet 1993; 18: 289-297.
- Yanyu X, Yunmei S, Zhipeng C and Qineng P: The preparation of silybin-phospholipid complex and the study on its pharmacokinetics in rats. Int J Pharm 2006; 307: 77-82.
- Maiti K, Mukherjee K, Gantait A, Ahamed HN, Saha BP and Mukherjee PK: Enhanced therapeutic benefit of quercetin–phospholipid complex in carbon tetrachloride– induced acute liver injury in rats: a comparative study. Iran J Pharmacol Ther 2005; 4: 84-90.
- Pearlman PC and Lee RT: Detection of measurement of total bilirubin in serum, with use of surfactants as solubilizing Clin Chem 1974; 20: 447-448.
- Bradley DW, Maynard JE, Emeny E and Webster H: Transaminase activity in serum of long term haemolysis Clin Chem 2003; 18: 1442-1443.
- Rej R, Fasce CF and Vanderlinde RE: Increased aspartate aminotransferase activity of serum after in-vitro supplementation with pyridoxil Clin Chem 1973; 19: 92-93.
- McComb RB and Boweres GN: Study of optimum buffer conditions for measuring alkaline phosphatase activity in human serum. Clin Chem 1972; 18: 97-98.
- Jain A and Jain SK: L-Valine appended PLGA nanoparticles for oral insulin delivery. Acta Diabetol 2015; 52(4): 663-76.
- Suja SR, Latha PG, Pushpangadan P and Rajasekharan S: Evaluation of hepatoprotective effects of zeylanica (L.) Hook against carbon tetrachloride induced liver damage in Wistar rats. J Ethnopharmacol 2004; 92: 61-66.
- Bishayee A, Sarkar A and Chatterjee M: The hepatoprotective activity of carrot (Daucus carota) against carbon tetrachloride intoxication in mouse liver. J Ethnopharmacol 1995; 47: 69-74.
- Sethumadhavan S, Theruvathil KS, Rangasamy A and Paruthapara TM: Hepatoprotective activity of chitosan against isoniazid and rifampicin-induced toxicity in experimental rats. Eur J Pharmacol 2007; 572(1): 69-73.
- Sanmugopriya E and Venkataraman S: Studies on hepatoprotective and antioxidant actions of Strychnos potatorum seeds on CCl4 -induced acute hepatic injury in experimental rats. J Ethnopharmacol 2006; 105: 154-160.
- Man-Fung Y, Takanobu K, Masashi M, Annie On-On C, John CHY and He-Jun Y: Clinical outcome and virologic profiles of severe hepatitis B exacerbation due to YMDD mutations. J Hepatol 2003; 39: 850-855.
How to cite this article:
Jain AK, Jain A and Jain A: Development and characterization of soya phosphatidylcholine complex of coumestans from Eclipta alba for the effective management of hepatotoxicity. Int J Pharmacognosy 2019; 6(1): 6-14. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(1).6-14.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
6-14
1,015
1397
English
IJP
A. K. Jain, A. Jain and A. Jain *
Department of Pharmaceutical Sciences, Dr. Harisingh Gour Central University, Sagar, Madhya Pradesh, India.
ashish.g.jain@gmail.com
08 December 2018
09 January 2019
18 January 2019
10.13040/IJPSR.0975-8232.IJP.6(1).6-14
31 January 2019