DEVELOPMENT AND ASSESSMENT OF LABETALOL RAPID DISSOLVING TABLETS
HTML Full TextDEVELOPMENT AND ASSESSMENT OF LABETALOL RAPID DISSOLVING TABLETS
Pushpendra *, Tulsi Bisht and Ankit Goel
Kunti Naman Institute of Pharma Technology & Science, Roorkee, Haridwar, Uttarakhand, India.
ABSTRACT: Fast dissolving or mouth disintegrating tablets are oral delivery systems intended for a rapid breakdown of the dosage form in the mouth or oral cavity, in contrast to other conventional dose forms. Labetalol, an alpha-1-adrenergic antagonist of the third generation, has vasodilatory and antihypertensive properties. It is used to treat hypertension and angina. Using novel co-processed super disintegrants and physical combinations combining croscarmellose and crospovidone in 1:1, 1:2, 1:3, 2:1, and 3:1 ratios, this study attempts to produce and evaluate labetalol fast dissolving tablets. The direct compression method was used to make the tablets. The tablets work instantly since they dissolve quickly when used orally. Like liquid dosage forms, orally disintegrating tablets are easier to take and offer greater convenience. Important excipients called disintegrants are added to solid dosage forms in order to break or disintegrate tablets in aqueous media. The disintegrants enhance the formulation's surface area, or tablet's surface area, and counteract the cohesive forces that hold the particles together.
Keywords: Direct compression, Co-processed super disintegrants, Crospovidone, Croscarmellose and fast-dissolving tablets
INTRODUCTION: Any medication needs to be packaged into a variety of dose forms in order to demonstrate its therapeutic action. Different dosage forms, including solid, semi-solid, and liquid dosage forms with various drug delivery mechanisms, are available to help the medicine demonstrate its therapeutic effect. A medication must be administered to its specific site of action at a predetermined rate and concentration in order to have the least amount of side effects and the greatest amount of therapeutic efficacy 1. A medicine should be formulated using a variety of physicochemical principles in order to generate an appropriate dosage form.
To overcome problems in the conventional tablets, new drug delivery systems called mouth disintegrating tablets or mouth dissolving tablets (MDTs) were developed. These tablets contain ingredients known as super disintegrants which include croscarmellose sodium, crospovidone 2, 3, 4. Sodium starch glycolate which disintegrate inside the mouth or oral cavity in a very short span, on contact with the oral fluid or saliva it shows its therapeutic action.
In order to improve patient acceptance, start of action, rate and amount of absorption, and stability, quick dissolving tablet preparation and evaluation are the goals of this research project 5, 6. Because saliva is present in the mouth and distributes the medicine as it travels to the stomach, FDTs also enhance drug absorption and decrease first-pass metabolism more than regular tablets. In certain formulations where the medicine dissolves quickly, pre-gastric absorption occurs in the oral cavity for drug dispersion.
Consequently, it aids in enhancing the safety profiles of medications that cause the liver to first pass through a certain level of metabolite production 7, 8, 9. A portion of the drug's metabolism is absorbed orally and in the GIT's pre-gastric regions 10.
MATERIALS AND METHODS:
Materials: Labetalol, MCC (microcrystalline cellulose), sodium starch glycolate, mannitol, crospovidone, magnesium stearate, sodium saccharin, talc.
Equipments:
- Electronic balance.
- Tablet compression machine.
- Hardness tester.
- Thickness tester – Screw Gauge.
- Dissolution test apparatus.
- Sieves – Techno instrument Co.
- UV/Visible spectrophotometer.
Methods:
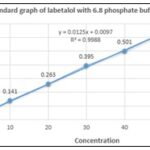
Using Phosphate Buffer pH (68.8), the Standard Calibration Curve for Labetalol is Prepared: Labetalol (100 mg) was precisely weighed, dissolved in a small amount of phosphate buffer (6.8 pH), and the entire volume known as the stock-1 solution was leveled using the same. In a dry, clean volumetric flask, 10 milliliters of the previously mentioned stock solution were diluted to create 100 milliliters, or the "stock-II Solution." From this stock-II solution, serial dilutions were made by pipetting out 1 ml, 2 ml, 4 ml, 6 ml, 8 ml, and 100 ml, respectively, to achieve concentrations ranging from 10, 20, 40, 60, 80, and 100 µg/ml. A UV-visible spectrophotometer was used to measure the total absorbance of the prepared solutions at 303 nm. A graph showing the relationship between concentration and absorbance was created using the measured absorbance values.
Preparation of Standard Calibration Curve of Labetalol in Methanol: In a volumetric flask, 100 ml of methanol and 100 milligrams of labetalol were combined after being weighed. The medication solution was taken in one milliliter and diluted to make one hundred milliliters. The concentrations were made by diluting 0.5, 1, 1.5, 2, 2.5, 3, 3.5, and 4 milliliters of the stock solution up to 10 milliliters in order to create solutions with concentrations of 5 mg/ml, 10 mg/ml, 15 mg/ml, 20 mg/ml, 25 mg/ml, 30 mg/ml, and 40 mg/ml. Using a Shimadzu UV/visible spectrophotometer, absorbance was measured at 320 nm for each solution, and a graph showing absorbance versus labetalol concentration was created.
TABLE 1: ABSORBANCE OF LABETALOL OBSERVED AGAINST PH 6.8 PHOSPHATE BUFFER
| Concentration (µg/ml) | Absorbance |
| 0 | 0 |
| 10 | 0.141 |
| 20 | 0.263 |
| 30 | 0.395 |
| 40 | 0.501 |
| 50 | 0.632 |
FIG. 1: STANDARD CALIBRATION GRAPH OF LABETALOL USING PHOSPHATE BUFFER PH 6.8
Preparation of Super Disintegrant Mixture: Step 1 assembling the super disintegrant combination. Two procedures were used to prepare the super disintegrant combination.
Method 1: Preparation of Super Disintegrant by Co-processed Technique:
- The solvent evaporation method was used to formulate these super disintegrates.
- Ten milliliters of ethanol were mixed with a combination of croscarmellose and crospovidone (in varying ratios of 1:1, 1:2, and 1:3).
- The contents of the beaker were well combined, and stirring was done until the majority of the ethanol had evaporated.
- The wet mass of the super disintegrant combination was passed through sieve no. 44 to create the granules. For twenty minutes, the granules were left to dry in the hot air oven at 60ºC.
- The granules were placed in an airtight container and left to dry before being sieved through sieve number 44.
Method 2: Making a Super Disintegrant using a Physical Mixing Method: Using this procedure, the super disintegrants were added straight to the mortar and pestle in the appropriate ratios, completely mixed, and then added to the dry components.
TABLE 2: FORMULATION OF LABETALOL FAST DISSOLVING TABLETS
| Formula code | Co-processed super disintegrates | Physical mixture | |||||||||
| F0 | F1 | F2 | F3 | F4 | F4 | F6 | F7 | F8 | F9 | F10 | |
| CF | (1:1) | (1:2) | (1:3) | (2:1) | (3:1) | (1:1) | (1:2) | (1:3) | (2:1) | (3:1) | |
| Labetalol | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Co-processed super disintegrates | - - - | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| MCC | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 |
| Manitol | 30 | 30 | 30 | 30 | 30 | 30 | 30 | 30 | 30 | 30 | 30 |
| Sodium saccharin | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 |
| Magnesium stearate | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| Talc | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| Total weight | 198 | 198 | 198 | 198 | 198 | 198 | 198 | 198 | 198 | 198 | 198 |
Labetalol Fast-dissolving Tablet Preparation using Direct Compression Method: The fast-dissolving labetalol tablets were formulated using the direct compression method with co-processed super disintegrants such as sodium starch glycolate and mannitol. Crospovidone. Microcrystalline cellulose is utilized as the diluent, while sodium saccharin is added as a sweetener. Talc and magnesium stearate were utilized as glidant and lubricant. All ingredients except those employed in the granule formulation were sieved through sieve 60. Ultimately, using rotary tablet compression machines with 6 mm round and flat punches, materials were sorted according to weight, combined, and then crushed again to create tablets with a total weight of 198 mg.
Evaluation:
Preformulation Studies:
Pre-compression Parameters: (a) Angle of repose (b) Bulk density (c) Tapped density (d) Hausner ratio (e) Compressibility index (%).
Angle of Repose (θ): Angle of repose is the maximam angle possible between the surface of the powder and horizontal plane. The frictional force of the loose powder or granules can be measured by angle of repose.
Tan θ =h/r, θ = tan-1 (h/r)
Where, θ is the angle of repose, h is height of powder, r is the radius of the base of powder.
TABLE 3: DIFFERENT RANGES OF FLOWABILITY IN TERMS OF ANGLE OF REPOSE
| Angle of repose (θ) (degrees) | Flow |
| <25 | Excellent |
| 25-30 | Good |
| 30-40 | Passable |
| >40 | Very poor |
Method: A funnel that was completely filled with the test sample was utilized for this experiment. Subsequently, the test sample was forced through the opening. The test sample's area was calculated using the cone that was drawn on the graph paper. Thus, measurements were made of the granules' height and flow characteristics.
Bulk Density: It is calculated by dividing the sample's total weight by its bulk volume. The distribution of particle sizes, the shape of the particles, and how firmly they attach to one another all affect the powder's bulk density.
Method: Powder was accurately weighed from each formula and was taken into a measuring cylinder to measure its volume.
Bulk density = Mass / volume
Tapped Density: By grabbing the grains and tapping them for a predetermined amount of time, the taped density was computed. Both the mass and volume of the granules within the cylinder were measured. The following formula was used to get the tapped density.
Tapped density (ρt) = M/ V
Hausner Ratio: Hausner ratio is the measure of ease of powder flow.
Hausner ratio = ρt/pd
Where ρt = tapped density ρd = bulk density.
Lower the hausner ratio i.e. (<1.25), better is the flow properties.
Carr’s Compressibility Index: Carr’s compressibility index determine granule’s compressibility
Carr’s compressibility index (%) = Tapped density- Bulk density x 100/ Tapped density
Evaluation of Tablets:
Post Compression Parameters:
- Hardness
- Weight variation
- Uniformity of thickness
- Wetting time
- In-vitro dissolution studies
Hardness: It measures the tablet's ability to withstand mechanical shocks during handling, manufacturing, and packing. A Monsanto hardness tester was used to measure the hardness of tablets. Kg/cm2 is the unit of hardness. From each formulation, five tablets were chosen at random, and the mean and standard deviation were computed.
Weight Variation Test: To measure weight variance, pills from each formulation were chosen at random and weighed separately.
TABLE 4: PERCENTAGE DEVIATION IN WEIGHT VARIATION ACCORDING TO IP
| Average weight of a tablet | Percentage deviation |
| 130mg or less | 10 |
| More than 130 mg and less than 324mg | 7.5 |
| 324 mg or more | 5 |
Since, the weight of the tablet was found to be greater than 350 mg for all formulations, a maximum variation of 5% was permitted.
Uniformity of Thickness: A micrometer can be used to measure the thickness of each tablet individually, providing precise variation values between tablets. Alternative techniques involve arranging five or ten tablets on a tray such that a sliding caliper scale can be used to measure the crown's overall thickness. A screw gauge was used to measure the tablet's thickness.
Wetting Time: The measurement involves folding a tissue paper piece twice and placing it in a petri dish containing 10 milliliters of 6.8 pH phosphate buffer solution. The time it took for the pill to get entirely wet was recorded when it was placed on the tissue paper. Three trials were conducted for every batch, and the standard deviation was computed.
In-vitro Dissolution Studies: Using a USP type-II apparatus, the substance was dissolved in 900 milliliters of phosphate buffer (6.8). The constant temperature was maintained at 37±0.5 °C. Every minute, 5 ml of aliquots were taken and filtered. Using a UV spectrophotometer set to detect the absorbance of the filtered solution at 320 nm, the drug concentration was calculated using a standard calibration curve.
RESULTS AND DISCUSSION:
Results:
TABLE 5: PHYSICAL APPEARANCE OF LABETALOL TABLETS
| Formulation code | Colour | Appearance |
| F0 | White | Good |
| F1 | White | Good |
| F2 | White | Good |
| F3 | White | Good |
| F4 | White | Good |
| F5 | White | Good |
| F6 | White | Good |
| F7 | White | Good |
| F8 | White | Good |
| F9 | White | Good |
| F10 | White | Good |
Pre-compression Parameters: The drug powder's loose bulk density, tapped bulk density, angle of repose, Carr's index, and Hausner's ratio were assessed, as were the excipients. The evaluations' findings are displayed below.
TABLE 6: PRE- COMPRESSION STUDIES OF LABETALOL
| Formulation code | Bulk density (g/cc) | Tapped
Density (g/cc) |
Angle of repose
(degree) |
Carr’s index (%) | Hausner’s ratio |
| F0 | 0.47 | 0.59 | 36.2 | 17.6 | 1.23 |
| F1 | 0.49 | 0.60 | 36.1 | 16.7 | 1.21 |
| F2 | 0.49 | 0.59 | 36.3 | 15.3 | 1.18 |
| F3 | 0.51 | 0.60 | 35.4 | 13.5 | 1.15 |
| F4 | 0.50 | 0.61 | 36.2 | 16.4 | 1.20 |
| F5 | 0.51 | 0.63 | 35.2 | 17.4 | 1.17 |
| F6 | 0.49 | 0.58 | 35.4 | 15.6 | 1.29 |
| F7 | 0.50 | 0.60 | 35.6 | 15.3 | 1.19 |
| F8 | 0.50 | 0.63 | 35.1 | 17.7 | 1.22 |
| F9 | 0.50 | 0.61 | 35.3 | 16.7 | 1.20 |
| F10 | 0.51 | 0.62 | 34.9 | 18.0 | 1.22 |
TABLE 7: POST-COMPRESSIONAL EVALUATION PARAMETERS
| Formulation code | Weight variation | Hardness ( kg/cm2 ) | Thickness (mm) | Wetting time (sec) |
| F0 | 193±0.22 | 4.1±0.40 | 5.1±0.15 | 71 |
| F1 | 194±0.46 | 4±0.61 | 5±0.71 | 68 |
| F2 | 198±0.47 | 4.2±0.30 | 5.2±0.75 | 53 |
| F3 | 194±0.84 | 4.1±0.75 | 5.1±0.22 | 48 |
| F4 | 197±0.95 | 4.1±0.22 | 5.1±0.55 | 66 |
| F5 | 196±0.71 | 4±0.40 | 5.2±0.10 | 55 |
| F6 | 194±0.75 | 4±0.65 | 5±0.90 | 64 |
| F7 | 197±0.31 | 4.2±0.34 | 5±0.70 | 61 |
| F8 | 192±0.71 | 4±0.55 | 5±0.21 | 51 |
| F9 | 196±0.95 | 4±0.66 | 5±0.52 | 58 |
| F10 | 194±0.08 | 4±0.22 | 5±0.40 | 50 |
Optimization of F3 and F10 Formulations: Two formulations, F3 and F10, were chosen for additional assessment based on pre- and post-compressional properties, particularly based on the wetting time. F3 (a 1:3 ratio of croscarmellose to crospovidone, respectively, made using the co-processed super disintegrants method). F10, which uses a physically combined super disintegrants technology to create croscarmellose and crospovidone in a 3:1 ratio, respectively.
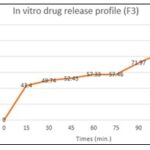
TABLE 8: IN-VITRO DISSOLUTION OF FORMULATION F3
| Time | % Drug dissolved |
| 0 | 0 |
| 15 | 43.40 |
| 30 | 49.74 |
| 45 | 52.43 |
| 60 | 57.33 |
| 75 | 57.46 |
| 90 | 71.97 |
| 105 | 84.53 |
| 120 | 96.01 |
FIG. 2: IN-VITRO DISSOLUTION PROFILE OF FORMULATION F3
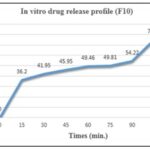
TABLE 9: IN-VITRO DRUG RELEASE PROFILE OF F10
| S. no. | Time | % Drug dissolved |
| 1 | 0 | 0 |
| 2 | 15 | 36.2 |
| 3 | 30 | 41.95 |
| 4 | 45 | 45.95 |
| 5 | 60 | 49.46 |
| 6 | 75 | 49.81 |
| 7 | 90 | 54.22 |
| 8 | 105 | 74.91 |
| 9 | 120 | 77.05 |
FIG. 3: IN-VITRO DRUG RELEASE PROFILE OF FORMULATION F10
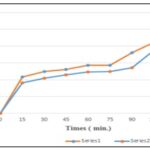
TABLE 10: COMPARISON OF DISSOLUTION PROFILES OF FORMULATIONS F3 AND F10
| Time (min) | % Drug release (F3) | % Drug release (F10) |
| 0 | 0 | 0 |
| 15 | 43.40 | 36.2 |
| 30 | 49.74 | 41.95 |
| 45 | 52.43 | 45.95 |
| 60 | 57.33 | 49.46 |
| 75 | 57.46 | 49.81 |
| 90 | 71.97 | 54.22 |
| 105 | 84.53 | 74.91 |
| 120 | 96.01 | 77.05 |
FIG. 4: COMPARISON OF DISSOLUTION PROFILES OF FORMULATION F3 AND F10
DISCUSSION: After completing the labetalol calibration curve, concentrations between 0 and 40 µg/ml were measured, and the linear relationship in Fig. 1 indicated that the R2 value of the curve was 0.9988. It therefore adhered to Beer Lambert's law.
Various Evaluation Parameters of Labetalol Tablets:
- Angle of repose: The drug's flow characteristic is acceptable because the angle of repose is between 30 and 40.
- Hardness: Based on the I.P. restrictions, the tablet hardness was approximately 5 kg/cm².
- Thickness: Based on the I.P. restrictions, the tablet's thickness is approximately 6 mm.
- Wetting time: The I.P limitations state that the tablets should be wetted for a duration of 50 to 75 seconds.
Dissolution Studies of Labetalol Tablets: As illustrated in Fig. 4, the formulation F3 utilizing the co-processed approach demonstrated 97.02% drug release in 120 minutes, while the formulation F10 utilizing a physical combination demonstrated 78.06% drug release in the same amount of time. We infer that the co-processed super disintegrant technique yields better outcomes than the physical mixing technique based on the aforementioned data. Therefore, based on the current work, it can be said that co-processed super disintegrants of croscarmellose and crospovidone exhibit improved dissolving rate and better release than their physical mixture.
CONCLUSION: The co-processed super disintegrants crospovidone and croscarmellose, which were used in the formulations F0, F1, F2, F3, F4, and F5, respectively, were used in the formulation and preparation of the beta-lactone tablets. The physical mixture consisted of 1:1, 1:2, 1:3, 2:1, and 3:1 of the formulations F6, F7, F8, F9, and F10. Evaluation tests were carried out before and after compression, and the results revealed that the formulation F3 (2:1), which uses co-processed super disintegrants, had a wetting time of 49 seconds, while F10 (3:1), which uses a physical mixture, had a wetting time of 51 seconds.
In-vitro dissolution studies were conducted on optimized formulations of F3 and F10. Of these, formulation F3 (2:1) demonstrated 97.02% drug release in 120 minutes, whereas formulation F10 (3:1) shown 78.06% drug release. The results of this investigation indicate that, in comparison to the physical mixture, the drug release from the formulation was improved by the use of co-processed super disintegrants such as crospovidone and croscarmellose.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Sharma D: Formulation development and evaluation of fast disintegrating tablets of salbutamol sulphate for respiratory disorders. ISRN Pharm [Internet]. 2013; Available from:http://dx.doi.org/10.1155/2013/674507
- Srilatha KS, Senthil Kumar, Likhith and Gupta BP: Formulation and in-vitro evaluation of fast dissolving tablets of anti-ulcer drugs. Am J Pharm Tech Res [Internet]. 2020;8(2):49– 59. Available from: http://dx.doi.org/10.46624/ajphr.2020.v8.i2.004
- Mehra P, Kapoor V, Gupta N, Singh Rajpoot D and Sharma N: Formulation evaluation and characterization of fast dissolving tablets of Rofecoxib. Res J Top Cosmet Sci [Internet]. 2021; 59–63. Available from: http://dx.doi.org/10.52711/2321-5844.2021.00009
- Madan JR, Kamate VJ, Awasthi R and Dua K: Formulation, characterization and in-vitro evaluation of fast dissolving tablets containing Gliclazide hydrotropic solid dispersions. Recent Pat Drug Deliv Formul [Internet]. 2017; 11(2): 147–54. Available from: http://dx.doi.org/10.2174/1872211311666170427100213
- Siddiqui MN, Garg G and Sharma K: Fast dissolving tablets: Preparation, characterization and evaluation: An overview [Internet]. Globalresearchonline.net. [cited 2022 Nov 16]. Available from:https://www.globalresearchonline.net/journalcontents/volume4issue2/Article%20015.pdf
- com. [cited 2022 Nov 16]. Available from: https://www.ijpsonline.com/articles/oral-dispersible-system-a-new-approach-in-drugdelivery-system.html?view=mobile
- A short review on fast dissolving tablets - a novel drug delivery system [Internet]. PharmaTutor. [cited 2022 Nov 16]. Available from: https://www.pharmatutor.org/articles/short-review-fast-dissolving-tablets-novel-drugdelivery-system
- Nagar P, Singh K, Chauhan I, Verma M, Yasir M and Khan A: Orally disintegrating tablets: formulation, preparation techniques and evaluation [Internet]. Japsonline.com. [cited 2022 Nov 16].
- Sharma S, Sharma N, Gupta GD. Formulation of fast-dissolving tablets of promethazine theoclate. Trop J Pharm Res [Internet]. 2010 [cited 2022 Nov 16]; 9(5). Available from: https://www.ajol.info/index.php/tjpr/article/view/61063/49259
- Shailesh Sharma, Sudhir Bharadwaj, G. D. Gupta, editor. Fast Dissolving Tablets of. Promethazine Theoclate by Using Natural Super disintegrants. Research J Pharm and Tec 2008; 1.
How to cite this article:
Pushpendra, Bisht T and Goel A: Development and assessment of labetalol rapid dissolving tablets. Int J Pharmacognosy 2024; 11(3): 100-06. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(3).100-06.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
100-106
671 KB
1083
English
IJP
Pushpendra *, Tulsi Bisht and Ankit Goel
Kunti Naman Institute of Pharma Technology & Science, Roorkee, Haridwar, Uttarakhand, India.
pusp92744@gmail.com
22 February 2024
12 March 2024
27 March 2024
10.13040/IJPSR.0975-8232.IJP.11(3).100-06
31 March 2024