DESIGN, SYNTHESIS AND PHARMACOLOGICAL EVALUATION OF NOVEL HETEROCYCLIC COMPOUNDS
HTML Full TextDESIGN, SYNTHESIS AND PHARMACOLOGICAL EVALUATION OF NOVEL HETEROCYCLIC COMPOUNDS
Ajay Shankar Sharma *, Yogendra Singh and Saurabh Jain
Department of Pharmacy, Shri Ramnath Singh Mahavidyalaya (Pharmacy), Gormi, Bhind, Madhya Pradesh, India.
ABSTRACT: Inflammation is a protective biological response; however, its chronic manifestation is closely associated with the development of several pathological conditions. The limitations and adverse effects of existing anti-inflammatory drugs necessitate the search for safer and more effective alternatives. In the present study, a series of novel heterocyclic derivatives of 2-thiouracil were designed, synthesized, and evaluated for their in-vitro anti-inflammatory activity. Twelve Schiff base derivatives (6a–6l) were synthesized through sulfonylation, hydrazinyl substitution, and subsequent condensation with various substituted aromatic aldehydes. The synthesized compounds were characterized by melting point determination, elemental analysis, and physicochemical evaluation. In-vitro anti-inflammatory activity was assessed using inhibition of albumin denaturation and human red blood cell (HRBC) membrane stabilization assays, with aspirin serving as the reference standard. All tested compounds exhibited concentration-dependent anti-inflammatory activity. Among them, derivatives 6h and 6b demonstrated the most significant inhibition of protein denaturation and superior membrane stabilization activity, approaching the efficacy of aspirin at higher concentrations. Structure–activity relationship analysis suggested that electron-donating substituents and halogen substitution patterns play a crucial role in enhancing biological activity. These findings indicate that 2-thiouracil-based heterocyclic derivatives represent promising lead compounds for the development of novel anti-inflammatory agents, warranting further in-vivo and mechanistic studies.
Keywords: 2-Thiouracil derivatives, Anti-inflammatory activity, Protein denaturation, HRBC membrane stabilization, Schiff bases, Heterocyclic compounds
INTRODUCTION: Inflammation is a complex biological response of body tissues to harmful stimuli such as infection, injury, or chemical irritation. While acute inflammation is a protective mechanism essential for healing, chronic inflammation has been implicated in the pathogenesis of numerous diseases, including rheumatoid arthritis, cardiovascular disorders, cancer, and neurodegenerative conditions 1.
Although several anti-inflammatory drugs are currently available, their long-term use is often associated with adverse effects such as gastrointestinal irritation, renal toxicity, and cardiovascular risks. This has created an ongoing need for the development of safer and more effective anti-inflammatory agents.
Heterocyclic compounds play a vital role in medicinal chemistry due to their diverse chemical structures and broad spectrum of biological activities. Many clinically important drugs contain heterocyclic moieties, which contribute significantly to their pharmacological properties 2. Among these, pyrimidine derivatives have attracted considerable attention because of their presence in nucleic acids and their wide range of therapeutic activities, including antimicrobial, anticancer, antiviral, and anti-inflammatory effects. Structural modification of the pyrimidine nucleus is a well-established strategy for enhancing biological activity and improving drug-like properties. 2-Thiouracil, a sulfur-containing pyrimidine derivative, has emerged as a promising scaffold in drug design owing to its versatile chemical reactivity and notable biological activities. The presence of the thiocarbonyl group allows for various chemical modifications, facilitating the synthesis of novel heterocyclic compounds with potential pharmacological significance 3, 4, 5. Previous studies have reported that thiouracil and its derivatives exhibit anti-inflammatory, antioxidant, and immunomodulatory properties. However, systematic studies focusing on the design, synthesis, and evaluation of new heterocyclic derivatives of 2-thiouracil for anti-inflammatory activity remain limited.
Therefore, this study is aimed at the design and synthesis of novel heterocyclic compounds derived from 2-thiouracil and the evaluation of their anti-inflammatory activity. The findings of this research may contribute to the discovery of new lead compounds with improved efficacy and reduced side effects, thereby supporting the development of safer anti-inflammatory therapeutic agents 6, 7.
METHODS:
Instruments: All melting points were determined using an Electro-thermal IA 9100 apparatus (Shimadzu, Kyoto, Japan) and are reported as uncorrected values. Spectra were recorded in DMSO-d₆, and chemical shifts (δ) are reported in parts per Column chromatography was performed on silica gel 60 (Merck, Darmstadt, Germany) with a particle size range of 0.06–0.20 mm. All solvents and reagents used were of analytical grade and employed without further purification unless otherwise stated.
Experimental:
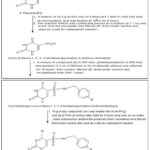
FIG. 1: SYNTHESIS OF 2-THIOURACIL
General Procedure: The synthesis commenced with the preparation of 4-oxo-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-sulfonyl chloride (2). A mixture of 2-thiouracil (12.5 g, 0.055 mol) and chlorosulfonic acid (51 mL, 0.055 mol) was heated at 120 °C for 8 h. After cooling, the reaction mixture was carefully poured onto an ice–acetic acid mixture (1:1), resulting in the formation of a precipitate. The solid was filtered, dried under suction, and used directly as the crude sulfonyl chloride intermediate for subsequent reactions. Sulfonamide derivatives 3a–b were synthesized by reacting sulfonyl chloride 2 (1.13 g, 0.005 mol) with the appropriate p-halobenzyl amine (p-chloro or p-fluoro, 0.005 mol) in the presence of pyridine (0.4 mL, 0.005 mol) in absolute ethanol (50 mL). The reaction mixture was refluxed for 12 h, then cooled, filtered, and the resulting solid was dried and recrystallized from DMF/water to afford compounds 3a and 3b. These intermediates were subsequently chlorinated by heating with POCl₃ (40 mL) and PCl₅ (5 g) on a boiling water bath for 8 h. The reaction mixtures were cooled and poured dropwise onto ice–water, and the resulting precipitates were filtered and dried to obtain the crude chloro derivatives 4a–b 5, 6.
Synthesis of N-(4-halobenzyl)-4-hydrazinyl-2-thioxo-1,2- dihydropyrimidine-5-sulfonamides [5a-b]: The final hydrazinyl intermediates 5a–b were prepared by treating the corresponding chloro derivatives 4a–b (0.001 mol) with hydrazine hydrate (0.001 mol) in methanol (30 mL). The reaction mixtures were refluxed for 15 min, then allowed to stir at room temperature for 24 h. The solutions were poured into ice–water, and the resulting precipitates were filtered, dried, and recrystallized from DMF/water to yield the desired N - (4-halobenzyl) – 4 – hydrazinyl – 2 – thioxo - 1, 2-dihydropyrimidine-5-sulfonamides (5a–b) 8.
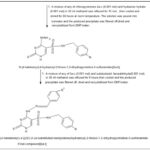
Synthesis of Final Compound 6a-l: The final compounds 6a–l were synthesized via a Schiff base condensation of the corresponding hydrazinyl intermediate (5a or 5b) with substituted aromatic aldehydes.
In a typical procedure, equimolar amounts (0.001 mol) of compound 5a or 5b and the appropriate benzaldehyde derivative were dissolved in 30 mL of methanol and refluxed for 8 h. After completion of the reaction, the mixture was allowed to cool to room temperature, resulting in the formation of a solid precipitate 9-11. The obtained precipitate was filtered, washed, dried, and subsequently recrystallized from a DMF/water mixture to afford the desired final products in good purity. Using compound 5a (N-(4-chlorobenzyl) derivative), reaction with 4-methoxy-, 4-ethoxy-, 4-nitro-, 4-hydroxy-, 2,4-dichloro-, and 2,4-difluorobenzaldehyde yielded compounds 6a–6f, respectively. Similarly, condensation of compound 5b (N-(4-fluorobenzyl) derivative) with the same series of substituted benzaldehydes under identical reaction conditions produced compounds 6g–6l, respectively. All synthesized compounds were obtained as solid products and purified by recrystallization, affording the corresponding (2E)-benzylidene hydrazinyl-2-thioxo-1, 2-dihydropyrimidine-5-sulfonamide derivatives 12-13.
In-vitro Anti-Inflammatory Activity Inhibition of Albumin Denaturation:
Preparation of Solutions: The control solution (50 mL) was prepared by mixing 2 mL of egg albumin, 28 mL of phosphate buffer saline (pH 6.4), and 20 mL of distilled water. A 1% w/v albumin solution was obtained by dissolving egg albumin in phosphate buffer saline (pH 6.4). The standard drug solution (50 mL) was prepared using aspirin at concentrations of 100, 200, 400, 800, and 1000 µg/mL. For each concentration, the solution contained 2 mL of egg albumin and 28 mL of phosphate buffer, with phosphate buffer saline freshly prepared and used throughout the experiment to maintain a constant pH. Similarly, the test solutions (50 mL) were prepared by dissolving accurately weighed quantities of the synthesized 2-thiouracil derivatives (6a–6l) in a suitable solvent to obtain concentrations of 100, 200, 400, 800, and 1000 µg/mL. Each test solution consisted of 2 mL of egg albumin and 28 mL of phosphate buffer, with the remaining volume adjusted using distilled water 14-17.
Procedure: The in-vitro anti-inflammatory activity of the synthesized compounds was evaluated by the inhibition of albumin denaturation method.
The reaction mixture consisted of 1 mL of 1% albumin solution and 1 mL of test compound solution at various concentrations. For the control, distilled water was used instead of the test compound. Diclofenac sodium was used as the standard drug. The pH of all reaction mixtures was adjusted to 6.4 using phosphate buffer. The mixtures were incubated at 37 °C for 20 minutes and then heated at 70 °C for 5 minutes in a water bath to induce denaturation. After cooling to room temperature, the turbidity of the samples was measured spectrophotometrically at 660 nm 18-19. The percentage inhibition of albumin denaturation was calculated and compared with the standard drug.
Percentage inhibition = Vt / Vc - 1 x 100
Where, Vt = Absorbance of test sample Vc = Absorbance of control
Human Red Blood Cell Membrane Stabilization Method (HRBC):
Preparation of Solutions: Fresh human blood was collected from a healthy volunteer and mixed with an equal volume of Alsever’s solution to prevent coagulation. The blood mixture was centrifuged at 3000 rpm for 10 minutes, and the packed red blood cells were separated. The cells were washed three times with normal saline until a clear supernatant was obtained. A 10% v/v HRBC suspension was prepared by resuspending the packed cells in normal saline. Test compound solutions were prepared at different concentrations using a suitable solvent. A standard drug solution of diclofenac sodium was prepared at similar concentrations. Phosphate buffer saline (pH 7.4) and hypotonic saline solutions were prepared freshly for the study 20.
Procedure: The in-vitro anti-inflammatory activity of the synthesized compounds was evaluated by the HRBC membrane stabilization method. The reaction mixture consisted of 1 mL of phosphate buffer saline (pH 7.4), 2 mL of hypotonic saline, 0.5 mL of 10% HRBC suspension, and 1 mL of test compound solution at various concentrations. For the control, distilled water was used instead of the test compound. Diclofenac sodium served as the standard drug. All the reaction mixtures were incubated at 37 °C for 30 minutes and then centrifuged at 3000 rpm for 10 minutes.
The absorbance of the supernatant was measured at 560 nm using a UV–Visible spectrophotometer. The percentage inhibition of hemolysis was calculated to determine the membrane-stabilizing and anti-inflammatory activity of the synthesized compounds in comparison with the standard drug 21, 22. The percentage of HRBC Membrane stabilization or protection was calculated by using the following formula:
Percentage protection = 100 - (Optical density sample / optical density control) x 100
RESULTS AND DISCUSSION:
Synthesis Of 2-Thiouracil Derivatives: A series of twelve 2-thiouracil derivatives (6a–6l) were successfully synthesized by the substitution of various aryl and heteroaryl groups at positions R and R1 of the parent 2-thiouracil scaffold.
The derivatives were substituted with electron-withdrawing groups (Cl, F, NO₂) and electron-donating groups (methoxy, ethoxy, hydroxyl) at different positions of the aromatic ring.
Compounds 6a–6f contained a chloro substituent at R, while 6g–6l contained a fluoro substituent at the same position. The R1 substituents included 4-methoxy, 4-ethoxy, 4-NO₂, 4-OH, and di-substituted 2, 4-dihalo groups (Cl or F).
TABLE 1: LIST OF COMPOUNDS SYNTHESIZED OF 2-THIOURACIL
| Derivatives | R | R1 |
| 6a | Cl | 4- methoxy |
| 6b | Cl | 4- ethoxy |
| 6c | Cl | 4-NO2 |
| 6d | Cl | 4-OH |
| 6e | Cl | 2,4- dichloro |
| 6f | Cl | 2,4- difluoro |
| 6g | F | 4- methoxy |
| 6h | F | 4- ethoxy |
| 6i | F | 4-NO2 |
| 6j | F | 4-OH |
| 6k | F | 2,4- dichloro |
| 6l | F | 2,4- difluoro |
Chemical Properties of Synthesized 2-Thiouracil Derivatives (6a–6l): The chemical properties of the synthesized 2-thiouracil derivatives (6a–6l) are summarized in Table 2. All compounds were obtained as crystalline solids with melting points ranging from 96°C to 111°C, indicating moderate thermal stability. The molecular weights of the synthesized derivatives ranged from 433.47 g/mol (6j) to 502.82 g/mol (6e), reflecting the variations in the substituents R and R1. Compounds containing di-substituted halogens, such as 6e (2,4-dichloro), exhibited higher molecular weights and increased halogen content compared to monosubstituted derivatives.
Elemental analysis of carbon (C), hydrogen (H), nitrogen (N), oxygen (O), sulfur (S), chlorine (Cl), and fluorine (F) showed values in good agreement with the theoretically calculated percentages, confirming the successful synthesis of the intended compounds. For example, compound 6a (C19H18ClN5O3S2) exhibited a carbon content of 49.19%, closely matching the calculated value, along with correct percentages of H, N, O, S, and Cl. Similarly, fluorine-containing derivatives (6g–6l) demonstrated the expected F content, which verified the presence of fluoro substituents on the aromatic ring.
TABLE 2: CHEMICAL PROPERTIES OF SYNTHESIZED COMPOUNDS (6a-6l)
| Der. | Chemical For | M.W | Composition | M.P.
(°C) |
||||||
| C | H | N | O | S | Cl | F | ||||
| 6a | C19H18ClN5O3S2 | 463.96 | 49.19% | 3.91% | 15.09% | 10.35% | 13.82% | 7.64% | - | 102°C |
| 6b | C20H20ClN5O3S2 | 477.98 | 50.26% | 4.22% | 14.65% | 10.04% | 13.42% | 7.42% | - | 106°C |
| 6c | C18H15ClN6O4S2 | 478.93 | 45.14% | 3.16% | 17.55% | 13.36% | 13.39% | 7.40% | - | 97°C |
| 6d | C18H16ClN5O3S2 | 449.93 | 48.05% | 3.58% | 15.57% | 10.67% | 14.25% | 7.88% | - | 107°C |
| 6e | C18H14Cl3N5O2S2 | 502.82 | 43.00% | 2.81% | 13.93% | 6.36% | 12.75% | 21.15% | - | 102°C |
| 6f | C18H14ClF2N5O2S2 | 469.91 | 46.01% | 3.00% | 17.55% | 13.36% | 13.39% | 7.54% | 8.09% | 96°C |
| 6g | C19H17FN5O3S2 | 447.50 | 50.99% | 4.05% | 15.65% | 10.73% | 14.33% | - | 4.25% | 108°C |
| 6h | C20H20FN5O3S2 | 461.53 | 52.05% | 4.37% | 15.17% | 10.40% | 13.90% | - | 4.12% | 102°C |
| 6i | C18H15FN6O4S2 | 462.47 | 46.75% | 3.27% | 18.17% | 13.84% | 13.87% | - | 4.11% | 107°C |
| 6j | C18H16FN5O3S2 | 433.47 | 49.87% | 3.72% | 16.16% | 11.07% | 14.79% | - | 4.38% | 102°C |
| 6k | C18H14FCl2N5O2S2 | 486.37 | 44.45% | 2.90% | 14.40% | 6.58% | 13.19% | 14.58% | 3.91% | 111°C |
| 6l | C18H14F3N5O2S2 | 453.46 | 47.68% | 3.11% | 15.44% | 7.06% | 14.14% | - | 12.57% | 108°C |
The physical and chemical properties of the synthesized 2-thiouracil derivatives (6a–6l) are summarized in Table 3. All the derivatives were obtained as solid powders with colors ranging from off-white to yellowish-white, indicating the successful formation of the respective substituted compounds.
The variation in color is attributed to the type and position of substituents on the aromatic ring; for example, nitro-substituted derivatives (6c, 6i) showed slightly more intense yellowish coloration due to their conjugated electron-withdrawing groups. The thin layer chromatography (TLC) analysis of the synthesized compounds indicated Rf values ranging from 0.57 to 0.79, suggesting distinct polarities and degrees of interaction with the stationary phase. Compounds bearing more lipophilic substituents such as dihalogenated derivatives (6e, 6l) displayed higher Rf values, whereas hydroxyl- and methoxy-substituted derivatives (6a, 6j) exhibited lower Rf values due to increased polarity. These Rf variations confirm the presence of different substituents and provide an initial qualitative assessment of purity.
The percent yield of the synthesized derivatives varied from 69.20% to 82.30%, reflecting the efficiency of the synthesis and purification process. The highest yield was obtained for compound 6a (82.30%), likely due to favorable reactivity of the chloro-methoxy substituents, while slightly lower yields were observed for di-substituted halogen derivatives (6f, 6k) due to steric hindrance during substitution reactions. Overall, the moderate to high yields indicate that the synthetic protocol was reproducible and effective for preparing a diverse set of 2-thiouracil derivatives.
TABLE 3: PHYSICAL AND CHEMICAL PROPERTIES OF SYNTHESIZED COMPOUND
| Code | Chemical Formula | Colour | Rf value | % yield |
| 6a | C19H18ClN5O3S2 | Off white solid powder | 0.57 | 82.30% |
| 6b | C20H20ClN5O3S2 | Yellowish- white solid powder | 0.68 | 75.30% |
| 6c | C18H15ClN6O4S2 | Yellowish white solid powder | 0.64 | 74.40% |
| 6d | C18H16ClN5O3S2 | Yellow white solid powder | 0.72 | 76.70% |
| 6e | C18H14Cl3N5O2S2 | Off white solid powder | 0.79 | 73.20% |
| 6f | C18H14ClF2N5O2S2 | Off white solid powder | 0.69 | 69.98% |
| 6g | C19H17FN5O3S2 | Yellowish- white solid powder | 0.68 | 75.30% |
| 6h | C20H20FN5O3S2 | Yellowish white solid powder | 0.64 | 74.40% |
| 6i | C18H15FN6O4S2 | off white solid powder | 0.76 | 71.70% |
| 6j | C18H16FN5O3S2 | Off white solid powder | 0.59 | 69.20% |
| 6k | C18H14FCl2N5O2S2 | Off white solid powder | 0.69 | 69.98% |
| 6l | C18H14F3N5O2S2 | Off white solid powder | 0.79 | 73.20% |
Anti-Inflammatory Activity:
Inhibition of Albumin Denaturation Method: The inhibitory effect of different concentration of 2-thiouracil derivative on protein denaturation as shown in Table 4.
2-thiouracil derivative at a concentration range of 100, 200, 400, 800, 1000 µg /ml and standard 100, 200, 400, 800, 1000 µg /ml showed significant inhibition of denaturation of egg albumin in concentration dependent manner. Both membrane stabilization activity and effect on protein denaturation contribute to the in-vitro anti-inflammatory activity of 2-thiouracil derivative used in our study. The in-vitro anti-inflammatory activity of the 2-thiouracil derivatives (6b, 6c, 6h, and 6k) was evaluated using the Protein Denaturation Method, with aspirin serving as the standard reference.
The results are summarized in Table 5 15, expressed as percentage inhibition of protein denaturation at different concentrations (100–1000 µg/mL). All derivatives exhibited a dose-dependent increase in anti-inflammatory activity, similar to the standard aspirin.
At the lowest concentration (100 µg/mL), derivative 6b showed the highest inhibition (55.65 ± 5.24%), followed by 6h (53.22 ± 5.87%), 6c (49.11 ± 3.42%), and 6k (44.45 ± 5.67%).
Aspirin showed a higher inhibition of 100.87 ± 5.28% at the same concentration. As the concentration increased to 1000 µg/mL, the derivatives demonstrated significantly enhanced inhibition.
Notably, 6h exhibited the highest activity (303.75 ± 9.87%), closely followed by 6b (299.65 ± 6.34%), while 6c and 6k showed 279.65 ± 9.36% and 267.87 ± 10.64%, respectively. Aspirin exhibited the highest inhibition overall (343.11 ± 8.88%) at this concentration. The trend indicates that derivatives 6b and 6h possess the most potent anti-inflammatory activity among the tested compounds, approaching the efficacy of aspirin at higher concentrations. Derivatives 6c and 6k showed moderate activity but still demonstrated a clear dose-dependent response.
These results suggest that structural variations in the 2-thiouracil derivatives significantly influence their protein denaturation inhibition, potentially due to differences in hydrogen bonding or interaction with protein targets.
Overall, the 2-thiouracil derivatives exhibit promising anti-inflammatory potential, with compounds 6b and 6h being the most effective, warranting further studies, including in-vivo evaluation and mechanistic exploration of their activity.
TABLE 4: IN-VITRO ANTI-INFLAMMATORY ACTIVITY OF 2-THIOURACIL DERIVATIVE (6b, 6c, 6h AND 6k) AND ASPIRIN OF PROTEIN DENATURATION METHOD
| Conc. (µg/ml) | 2-thiouracil derivatives % inhibition | Aspirin Standard | |||
| 6b | 6c | 6h | 6k | ||
| 100 | 55.65 ±5.24 | 49.11 ±3.42 | 53.22 ±5.87 | 44.45 ±5.67 | 100.87± 5.28 |
| 200 | 99.61 ±6.36 | 89.65 ±8.67 | 111.87 ±5.74 | 79.65 ±7.23 | 156.05± 6.87 |
| 400 | 167.76 ±3.98 | 155.11 ±7.95 | 192.39 ±6.76 | 143.65 ±9.36 | 204.78± 9.27 |
| 800 | 256.45 ±7.01 | 238.43 ±9.24 | 267.87 ±6.34 | 241.67 ±10.56 | 261.87± 8.87 |
| 1000 | 299.65 ±6.34 | 279.65 ±9.36 | 303.75 ±9.87 | 267.87 ±10.64 | 343.11± 8.88 |
Values are expressed as Mean ± Standard deviation HRBC Membrane Stabilization Method
HRBC Membrane Stabilization Method: The in vitro anti-inflammatory activity of 2-thiouracil derivatives (6b, 6c, 6h, and 6k) was evaluated using the HRBC (Human Red Blood Cell) Membrane Stabilization Method, with aspirin serving as the standard reference. The results are expressed as percentage inhibition of hemolysis at concentrations ranging from 100 to 1000 µg/mL Table 5.
All 2-thiouracil derivatives exhibited a dose-dependent increase in membrane stabilization activity. At the lowest tested concentration (100 µg/mL), 6h showed the highest inhibition of 54.87 ± 3.66%, followed by 6b (47.56 ± 0.17%), 6k (46.11 ± 2.87%), and 6c (45.23 ± 1.67%). Aspirin, the reference standard, displayed significantly higher activity at this concentration (78.23 ± 1.67%). With increasing concentrations, the derivatives showed a gradual increase in anti-inflammatory activity. At 1000 µg/mL, 6h exhibited the highest inhibition (77.26 ± 2.35%), followed by 6b (72.11 ± 5.11%), 6k (69.56 ± 4.72%), and 6c (66.15 ± 0.76%). The standard aspirin achieved 101.76 ± 4.32% inhibition at the same concentration, indicating superior membrane stabilization.
The data suggest that among the tested derivatives, 6h demonstrated the most potent anti-inflammatory effect, likely due to favorable structural features that enhance interaction with the RBC membrane, thereby protecting it from hypotonic-induced hemolysis. Compounds 6b and 6k also exhibited significant activity, while 6c showed moderate inhibition across all concentrations. Overall, the HRBC membrane stabilization assay confirms that 2-thiouracil derivatives possess promising anti-inflammatory potential, with compound 6h being the most effective. These results, combined with the protein denaturation method, indicate that these derivatives can inhibit inflammatory processes via membrane stabilization and protein denaturation mechanisms, supporting their potential as anti-inflammatory agents.
TABLE 5: IN-VITRO ANTI-INFLAMMATORY ACTIVITY OF 2-THIOURACIL DERIVATIVE (6b, 6c, 6hAND 6k) AND ASPIRIN OF HRBC MEMBRANE STABILIZATION METHOD
| Conc. (µg/ml) | 2-thiouracil derivatives % inhibition | Aspirin Standard | |||
| 6b | 6c | 6h | 6k | ||
| 100 | 47.56 ±0.17 | 45.23 ± 1.67 | 54.87 ±3.66 | 46.11 ±2.87 | 78.23± 1.67 |
| 200 | 54.34 ±1.24 | 51.23 ±0.25 | 58.21 ±2.3 | 55.87 ±2.276 | 82.45± 1.99 |
| 400 | 61.76 ±3.36 | 56.56 ± 1.98 | 62.82 ±2.76 | 59.98 ±3.37 | 92.11± 3.76 |
| 800 | 64.26 ±3.48 | 60.23 ±2.75 | 72.64 ±1.55 | 63.24 ±6.61 | 98.54± 6.34 |
| 1000 | 72.11 ±5.11 | 66.15 ±0.76 | 77.26 ±2.35 | 69.56 ±4.72 | 101.76± 4.32 |
Values are expressed as Mean ± Standard deviation
CONCLUSION: The study successfully synthesized and characterized a series of 2-thiouracil derivatives with diverse substituents. These compounds demonstrated significant in-vitro anti-inflammatory activity, mediated via inhibition of protein denaturation and stabilization of RBC membranes. Among the derivatives tested, 6h and 6b showed the highest anti-inflammatory potential, highlighting the importance of substituent type and position on biological activity. Overall, the results indicate that 2-thiouracil derivatives are promising candidates for anti-inflammatory drug development, and further studies, including in-vivo evaluation and mechanistic investigations, are warranted to explore their therapeutic potential.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Ahmed NM, Lotfallah AH, Gaballah MS, Awad SM & Soltan MK: Novel 2-Thiouracil-5-Sulfonamide Derivatives: Design, Synthesis, Molecular Docking, and Biological Evaluation as Antioxidants with 15-LOX Inhibition. Molecules 2023; 28(4): 1925.
- Robinson MS, Niebuhr M & Gühr M: Ultrafast photo-ion probing of the relaxation dynamics in 2-thiouracil. Molecules 2023; 28(5): 2354.
- Bradshaw GA, Colgan AC, Allen NP, Pongener I, Boland MB, Ortin Y & McGarrigle EM: Stereoselective organocatalyzed glycosylations–thiouracil, thioureas and monothiophthalimide act as Brønsted acid catalysts at low loadings. Chemical Science 2019; 10(2): 508-514.
- Ramhøj L, Frädrich C, Svingen T, Scholze M, Wirth EK, Rijntjes E & Axelstad M: Testing for heterotopia formation in rats after developmental exposure to selected in-vitro inhibitors of thyroperoxidase. Environmental Pollution 2021; 283: 117135.
- Matthews A, Saleem-Batcha R, Sanders JN, Stull F, Houk, KN & Teufel R: Aminoperoxide adducts expand the catalytic repertoire of flavin monooxygenases. Nature Chemical Biology 2020; 16(5): 556-563.
- Wojciechowska M, Równicki M, Mieczkowski A, Miszkiewicz J & Trylska J: Antibacterial peptide nucleic acids—Facts and perspectives. Molecules 2020; 25(3): 559.
- Puzzarini C, Spada L, Alessandrini S & Barone V: The challenge of non-covalent interactions: Theory meets experiment for reconciling accuracy and interpretation. J of Physics: Condensed Matter 2020; 32(34): 343002.
- Harismah K, Mirzaei M & Ghafari K: Structural Analysis of Some Pyrrolopyrimidine Derivatives and Examining their Binding Affinity against Cyclooxygenase-2 Enzyme. Turkish Computational and Theoretical Chemistry 2021; 5(2): 14-23.
- Hammouda MM, Elattar KM, El-Khateeb AY, Hamed SE & Osman AM: Developments of pyridodipyrimidine heterocycles and their biological activities. Molecular Diversity 2023; 1-38.
- Saarbach J, Sabale PM & Winssinger N: Peptide nucleic acid (PNA) and its applications in chemical biology, diagnostics, and therapeutics. Current Opinion in Chemical Biology 2019; 52: 112-124.
- Lever F & Gühr M: Probing the ultrafast dynamics of 2-Thiouracil with soft x-rays (Doctoral dissertation, Universität Potsdam) 2021.
- Ragab SS, Sweed AM, Hamza ZK, Shaban E & El-Sayed AA: Design, Synthesis, and Antibacterial Activity of Spiropyrimidinone Derivatives Incorporated Azo Sulfonamide Chromophore for Polyester Printing Application. Fibers and Polymers 2022; 23(8): 2114-2122.
- Samir M, Ramadan M, Abdelrahman MH, Elbastawesy M. A, Halby HM, Abdel-Aziz M & Abuo-Rahma GEDA: New potent ciprofloxacin-uracil conjugates as DNA gyrase and topoisomerase IV inhibitors against methicillin-resistant Staphylococcus aureus. Bioorganic & Medicinal Chemistry 2022; 73: 117004.
- Azizi-Lalabadi M. Alizadeh-Sani M, Khezerlou A, Mirzanajafi-Zanjani M, Zolfaghari H, Bagheri V & Ehsani A: Nanoparticles and zeolites: Antibacterial effects and their mechanism against pathogens. Current Pharmaceutical Biotechnology 2019; 20(13): 1074-1086.
- Varela MF, Stephen J, Lekshmi M, Ojha M, Wenzel N, Sanford LM & Kumar SH: Bacterial resistance to antimicrobial agents. Antibiotics 2021; 10(5): 593.
- Baran A, Kwiatkowska A & Potocki L: Antibiotics and Bacterial Resistance—A Short Story of an Endless Arms Race. International Journal of Molecular Sciences 2023; 24(6): 5777.
- Hasan TH & Al-Harmoosh RA: Mechanisms of antibiotics resistance in bacteria. Sys Rev Pharm 2020; 11(6): 817-823.
- Eichenberger EM & Thaden JT: Epidemiology and mechanisms of resistance of extensively drug resistant Gram-negative bacteria. Antibiotics 2019; 8(2): 37.
- Serafim MSM, Kronenberger T, Oliveira PR, Poso A, Honorio KM, Mota BEF & Maltarollo VG: The application of machine learning techniques to innovative antibacterial discovery and development. Expert Opinion on Drug Discovery 2020; 15(10): 1165-1180.
- Abouelhassan Y, Garrison AT, Yang H, Chávez-Riveros A, Burch GM & Huigens III RW: Recent progress in natural-product-inspired programs aimed to address antibiotic resistance and tolerance. Journal of Medicinal Chemistry 2019; 62(17): 7618-7642.
- Liu X, Zhang Y, Wang Y, Zhu W, Li G, Ma X & Liang XJ: Comprehensive understanding of magnetic hyperthermia for improving antitumor therapeutic efficacy. Theranostics 2020; 10(8): 3793.
- Hassan RH, Gad HA, El-Din SB, Shaker DS & Ishak RA: Chitosan nanoparticles for intranasal delivery of olmesartan medoxomil: Pharmacokinetic and pharmacodynamic perspectives. International Journal of Pharmaceutics 2022; 628: 122278.
How to cite this article:
Sharma AS, Singh Y and Jain S: Design, synthesis and pharmacological evaluation of novel heterocyclic compounds. Int J Pharmacognosy 2026; 13(3): 207-15. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(3).207-15.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
207-215
684 KB
122
English
IJP
Ajay Shankar Sharma *, Yogendra Singh and Saurabh Jain
Department of Pharmacy, Shri Ramnath Singh Mahavidyalaya (Pharmacy), Gormi, Bhind, Madhya Pradesh, India.
mspharma8081@gmail.com
12 February 2026
16 March 2026
20 March 2026
10.13040/IJPSR.0975-8232.IJP.13(3).207-15
31 March 2026