COMPREHENSIVE STUDY ON PHYTOCHEMICAL ANALYSIS OF MEDICINAL PLANTS
HTML Full TextCOMPREHENSIVE STUDY ON PHYTOCHEMICAL ANALYSIS OF MEDICINAL PLANTS
Asna Haseen, M. L. Lal Prasanth and A. Anu Jagajith *
Dr. Moopen’s College of Pharmacy, Wayanad, Kerala, India.
ABSTRACT: The biologically active compounds present in plants are called phytochemicals. Phytochemical analysis unlocks the secrets within these plants, revealing the identity of these valuable molecules. This review helps in study of exciting world of plant chemistry, utilizing diverse techniques to unveil the phytochemicals of various medicinal species. Analysing the profile of these phytochemicals in medicinal plants is crucial for understanding their potential therapeutic effects, identifying novel drug leads, and ensuring product quality control. The goal of phytochemical analysis is to evaluate the medicinal potential of plant by various methods like extraction, chromatographic techniques, spectroscopic methods for identifying and isolating their phytoconstituents. Extraction techniques are the main one. Digestion, maceration, and percolation are the examples of extraction techniques. Similarly phytochemical screening of Alkaloids, Flavonoids etc. also included in this study. Spectroscopic methods play a pivotal role in this process, offering a powerful tool to qualitatively identify and quantitatively analyse these fascinating molecules. Phytochemical study is very useful for us because of many applications are occurred especially in disease prevention or enhances the immunity.
Keywords: Phytochemical, Drug discovery, Chromatography, Carotenoids
INTRODUCTION: Phytochemicals are biologically active, naturally occurring chemical compounds found in plants, which provide health benefits for humans as medicinal ingredients and nutrients. They protect plants from disease and damage and contribute to the plant’s color, aroma and flavor. The plant chemicals that protect plant cells from environmental hazards such as pollution, stress, drought, UV exposure and pathogenic attack are called as phytochemicals. Phytochemical analysis is a quickly developing and relatively new chemical discipline that investigates the structure, biosynthesis, metabolism, and biological function of organic compounds in plants 1.
How do phytochemicals helps in prevent disease?
- Stimulate the immune system, the body’s defence against viruses, bacteria and other disease‐causing agents.
- Block the potential for carcinogens (cancer causing substances) to be formed in the body from substances we eat, drink and absorb from the environment.
- Prevent DNA damage and help with DNA repair mechanisms.
- Reduce oxidation, the damage to cells that occurs with aging and exposure to pollution.
- Slow the growth rate of cancer cells.
- Help to regulate hormones, such as oestrogen and insulin. Excess levels of these hormones are linked with increased risk for breast and colon cancer.
FIG. 1: PHYTOCHEMICALS PRESENT IN VARIOUS FOODS
General Mechanism of Action of Phytochemicals:
Alkaloids: Inhibit the release of Autocoid and prostaglandins.
Terpenoids: Membrane disruption. Which inhibit the release of autocoids and prostaglandin release 2.
Flavonoids: Complex with cell wall, binds to adhesins inhibits the release of autocoids and prostaglandins.
Saponins: Leads to vacuolization and disintegration of teguments 3.
Steroids: Enhance intestinal absorption of sodium and water
Extraction: Extraction is the process of efficiently dissolving and separating the desired constituents from the crude drug with the use of solvents. The choice of solvent depends on the characteristics of the secondary metabolites like polarity, PH and thermal stability. The solvents should be non-inflammable, inert, non-toxic, easy to remove and should dissolve the maximum amount of desired phytoconstituents 12.
Factors affecting extraction of crude drugs:
- Moisture content of drugs
- Quantity and chemical nature of drug
- Size of powder of crude drug
- Nature and volume of solvent
- Temperature of extraction process
- Lipophilicity of the solvent mixture and sample
- pH of extracting solvent
Extraction Techniques:
Digestion: This is a form of maceration in which gentle heat is used during the process of extraction. It is used when moderately elevated temperature is not objectionable. The solvent efficiency of the menstruum is thereby increased 12.
Maceration: In this process, the whole or coarsely powdered crude drug is placed in a stoppered container with the solvent and allowed to stand at room temperature for a period of at least 3 days with frequent agitation until the soluble matter has dissolved. The mixture then is strained, the marc (the damp solid material) is pressed, and the combined liquids are clarified by filtration or decantation after standing 17.
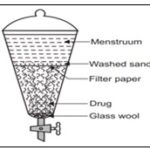
Percolation: Percolationimplies a slow passage of the menstruum under the influence of gravity through a column of drug powder and during this movement it goes on extracting the drug molecules layer wise. This is the procedure used most frequently to extract active ingredients in the preparation of tinctures and fluid extracts. A percolator (a narrow, cone-shaped vessel open at both ends) is generally used. The solid ingredients are moistened with an appropriate amount of the specified menstruum and allowed to stand for approximately 4 h in a well closed container, after which the mass is packed and the top of the percolator is closed. Additional menstruum is added to form a shallow layer above the mass, and the mixture is allowed to macerate in the closed percolator for 24 h. The outlet of the percolator then is opened and the liquid contained therein is allowed to drip slowly. Additional menstruum is added as required, until the percolate measures about three-quarters of the required volume of the finished product. The marc is then pressed and the expressed liquid is added to the percolate. Sufficient menstruum is added to produce the required volume, and the mixed liquid is clarified by filtration or by standing followed by decanting 18.
FIG. 2: PERCOLATOR
Infusion: It is a very simple method of extraction used for vitamins, volatile ingredients and soft ingredients in which powdered drug is extracted with hot or cold water. In this method, the powdered drug is soaked in hot water for the specified period with or without stirring and then filtered. Once the powdered drug is added to hot water no further heating is done and kept aside. If necessary, press the marc and extract again with fresh hot water.
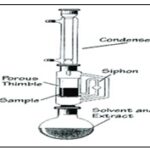
Soxhlet Extraction: This approach involves placing a porous bag, or "thimble," composed of sturdy filter paper within chamber E of the Soxhlet apparatus containing the finely ground crude drug. After being heated in flask A, the extracting solvent's vapours condense in condenser D 12. By dripping into the thimble holding the crude medication, the condensed extractant extracts it through contact. Chamber E's liquid contents syphon into flask A when the liquid level reaches the top of syphon tube C. Until a drop of solvent from the syphon tube evaporates without leaving any residue behind the process continued.
FIG. 3: SOXHLET APPARATUS
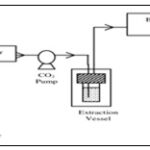
Super Critical Fluid Extraction: The sample is placed in an extraction vessel and pressurized with SCF. Carbon dioxide to dissolve the sample 8.
After extraction the extract is transferred to the fraction chamber and depressurized due to which carbon dioxide loses its solvating power causing entire material to precipitate. Now the carbon dioxide gets recycled. Precipitated material is extracted with addition of small amount of solvent 26.
FIG. 4: SUPERCRITICAL FLUID EXTRACTOR
Ultrasound Extraction (Sonication): The procedure involves the use of ultrasound with frequencies ranging from 20 kHz to 2000 kHz; this increases the permeability of cell walls and produces cavitation. Although the process is useful in some cases, like extraction of rauwolfia root, its large-scale application is limited due to the higher costs. One disadvantage of the procedure is the occasional but known deleterious effect of ultrasound energy (more than 20 kHz) on the active constituents of medicinal plants through formation of free radicals and consequent Digestion.
FIG. 5: SONICATOR
Microwave Assisted Extraction: Microwave-assisted extraction (MAE) is a process of using microwave energy to heat solvents in contact with a sample in order to partition analytes from the sample matrix into the solvent 12. The ability to rapidly heat the sample solvent mixture is inherent to MAE and the main advantage of this technique.
FIG. 6: MICROWAVE ASSISTED EXTRACTOR
Expression Extraction: It is a purely mechanical technique, which involve sponge, ecuelle and mechanical methods to extract essential oils. volatile oil absorption on sponge by rupturing the oil glands from citrous peel by squeezing is the best example of sponge method. In ecuelle method the sharp projection containing vessel is used to rupture the glands from the citrous peels.
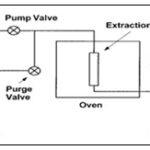
Accelerated Solvent Extraction: Accelerated solvent extraction is a technique for extracting organic compounds from solid and semisolid samples with liquid solvents. The extraction cell is filled with the solid sample to be examined and placed in a temperature-controllable oven. After adding the solvent, the cell is heated at constant pressure (adjustable between 0.3 and 20 MPa) up to a maximum temperature of 200°C and kept at constant conditions for a while so that equilibrium can be established.
The extract is then transferred to a sample tube. A sample often goes through several extraction cycles. Finally, the extraction cell is rinsed with solvent, the rinsing valve is opened and the cell and all lines are rinsed with nitrogen and the apparatus is prepared for further extractions 12.
FIG. 7: ACCELERATED SOLVENT EXTRACTOR
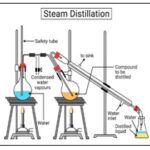
Steam Distillation: This is the most suitable method for extraction of volatile oils. The steam can be generated and passed through the plant material suspended in water. The steam vapourizes which is then condensed and separated. In direct steam distillation, the material to be extracted is spread on a mesh as thin layer and the generated steam is passed through the material.
FIG. 8: DISTILLATION APPARATUS
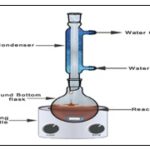
Reflux Extraction: Reflux extraction is a solid–liquid extraction process at a constant temperature with repeatable solvent evaporation and condensation for a particular period of time without the loss of solvent. The system is widely used in herbal industries as it is efficient, easy to operate and cost effective. Reaction mixture can be heated without losing volatile substances, they condense and run back down into the flask. If a bung in the top there could be pressure build up and an explosion. Water does not flow counter current to heat as the need to sweep out air bubbles is more important.
FIG. 9: REFLUX APPARATUS
Enflurage Method: In the extraction method called enfleurage, petals are placed between layers of purified animal fat, which become saturated with flower oil, and alcohol is then used to obtain the absolute. The expression method, used to recover citrus oils from fruit peels, ranges from a traditional procedure of pressing with sponges 15.
FIG. 10: ENFLEURAGE METHOD
Phytochemical Screening: The process of detection of various constituents in a plant extract is known as phytochemical screening. Plant contains numerous chemical constituents that are responsible for eliciting various physiological and therapeutic responses. Therefore, plants are generally tested for the presence of biologically active and medicinally useful phytochemical constituents responsible for a particular biological activity. Some of the examples of phytoconstituents include alkaloids, carbohydrates, saponins, tannins, flavonoids etc.
Detection of Alkaloids:
Stock Solution: About 50 mg of solvent free extract is stirred with little quantity of dilute hydrochloric acid and filtered 21. The filtrate is tested with various alkaloidal reagents as follows:
TABLE 1: DETECTION OF ALKALOIDS
| Test | Procedure | Observation |
| Mayer’s Test | To a few ml of filtrate, two drops of mayer’s reagent is added along with the sides of the test tube. | White or creamy precipitate. |
| Wagner’s Test | To a few ml of the filtrate few drops of wagner’s reagent were added along with the sides of the test tube. | Reddish brown precipitate |
| Hager’s Test | To a few ml of filtrate 1or 2 ml Hager’s reagent is added. | Yellow precipitate |
| Dragendorff’s Test | To a few ml of filtrate, 1 or 2 ml of Dragendorff’s reagent is added. | Reddish brown precipitate |
Detection of Flavonoids: 0.5 ml of aqueous solution of extract is added to 2ml of furfuraldehyde in a test tube- Red colour indicate the presence of flavonoid.
TABLE 2: DETECTION OF FLAVONOIDS
| Test | Procedure | Observation |
| Alkaline reagent test | Extract is treated with 10 % NaOH solution. | Intense yellow colour |
| Ammonium hydroxide test | 3ml of extract is 10% NH4OH solution. | Yellow fluorescent |
| Mg turning test | Extract were treated with Mg turning and add conc. HCl to this solution add 5ml of 95% ethanol | Crimson red colour |
| Zn test | 2ml Extract were treated with Zn and add conc. HCl[36]. | Red colour |
TABLE 3: DETECTION OF TANNINS
| Test | Procedure | Observation |
| Ferric chloride test | About 50 ml of extract is dissolved in distilled water and to this few drops of neutral 5% ferric chloride solution is added. | Blue, green and violet colour |
| Gelatine test | A little quantity of extract is dissolved in distilled water and 2 ml of 1% solution of Gelatine containing 10% sodium chloride is added to it. | White precipitate |
| Lead acetate test | A small quantity of extract is dissolved in distilled water and to this; 3 ml of 10% lead acetate solution is added. | Bulky white precipitate |
| Alkaline reagent test | Aqueous solution of extract is treated with 10%ammonium hydroxide solution. | Yellow fluorescence 22 |
Detection of Glycosides: For detection of glycosides, about 50 mf of extract is hydrolysed with concentrated hydrochloric acid for 2 hrs on a water bath and filtered.
TABLE 4: DETECTION OF GLYCOSIDES
| Test | Procedure | Observation |
| Borntrager’s Test | To 2 ml of filtrate, 3 ml of ethyl acetate is added and shaken, ethyl acetate layer is separated and 10% ammonia solution is added to it. | Pink colour |
| Legal’s Test | About 20 mg of the extract is dissolved in pyridine. sodium nitroprusside solution is added and make alkaline using 10% sodium hydroxide solution | Pink colour |
TABLE 5: DETECTION OF TERPENOIDS
| Test | Procedure | Observation |
| Liebermann-Buchard’s test | The extract is dissolved in acetic anhydride, heated to boiling cooled and then 1 ml of conc. sulphuric acid is added along the side of the test tube. | Red, pink or violet |
| Salkowski test | Few drops of conc. sulphuric acid is added to the extract, shaken on standing 36. | Golden yellow colour |
TABLE 6: DETECTION OF SAPONINS
| Test | Procedure | Observation |
| Foam or froth test | A small quantity of the extract is diluted with Distilled water to 20ml.The suspension is shaken in a graduated cylinder for 15 minutes. | Foam or froth which stable for 10 minutes. |
TABLE 7: DETECTION OF RESINS
| Test | Procedure | Observation |
| HCl test | Drug powder is treated with hydrochloric acid | Pink colour |
| Ferric chloride test | Drug is treated with ferric chloride solution | Greenish blue |
TABLE 8: DETECTION OF VOLATILE OILS
| Test | Procedure | Observation |
| Fluorescence test | Fluorescence test l0 mL of extract, filtered till saturation, exposed to UV light | light Bright pinkish fluorescence |
Isolation and Purification of Phytochemicals:
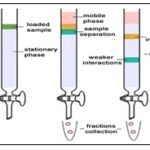
Chromatographic Techniques: Chromatography is a separation method where the analyte is combined within a liquid or gaseous mobile phase, which is pumped through a stationary phase. Usually one phase is hydrophilic and the other is lipophilic. The components of the analyte interact differently with these two phases. Depending on their polarity they spend more or less time interacting with the stationary phase and are thus retarded to a greater or lesser extent. This leads to the separation of the different components present in the sample. As the components pass through the detector their signal is recorded and plotted in the form of a chromatogram.
Paper Chromatography: In paper chromatography support material consists of a layer of cellulose highly saturated with water. In this method a thick filter paper comprised the support, and water drops settled in its pores made up the stationary “liquid phase.” Mobile phase consists of an appropriate fluid placed in a developing tank. Paper chromatography is a “liquid-liquid” chromatography 29.
FIG. 11: PAPER CHROMATOGRAPHY
Thin Layer Chromatography: In the process of thin-layer chromatography (TLC), the mixture of substances is separated into its components with the help of a glass plate coated with a very thin layer of adsorbent, such as silica gel and alumina. The plate used for this process is known as chrome plate 26. The solution of the mixture to be separated is applied as a small spot at a distance of 2 cm above one end of the plate. The plate is then placed in a closed jar containing a fluid termed as an eluant, which then rises up the plate carrying different components of the mixture to different heights.
FIG. 12: THIN LAYER CHROMATOGRAPHY
High Performance Thin Layer Chromatography (HPTLC): HPTLC is a chromatographic technology that can be utilised for many purposes such as constituent identification, impurity identification and determination, and active substance quantitative determination. Compared to conventional TLC, HPTLC offers improved accuracy, reproducibility, and record-keeping capabilities, making it one of the best TLC methods for analytical applications. A type of thin-layer chromatography (TLC) known as high-performance thin-layer chromatography (HPTLC) uses an optimised coating material, automated processes for feeding the mobile phase, layer preconditioning, precise sample application, scanning of the chromatogram development, and photo documentation to provide superior separation power. It encourages more effective data collection and processing, reduced volumes of mobile phase, quicker analysis times, and improved separation efficiency 31.
FIG. 13: HIGH PERFORMANCE THIN LAYER CHROMATOGRAPHY
Column Chromatography: Column chromatography is a technique in which the substances to be separated are introduced onto the top of a column packed with an adsorbent, passed through the column at different rates that depend on the affinity of each substance for the adsorbent and for the solvent or solvent mixture, and are usually collected in solution as they pass from the column at different times. It is a solid-liquid technique in which the stationary phase is a solid & the mobile phase is a liquid or gas 31.
FIG. 14: COLUMN CHROMATOGRAPHY
High Performance Liquid Chromatography (HPLC): Using this chromatographic technique it is possible to perform structural, and functional analysis, and purification of many molecules within a short time, This technique yields perfect results in the separation, and identification of amino acids, carbohydrates, lipids, nucleic acids, proteins, steroids, and other biologically active molecules 27. In this technique, use of small particles, and application of high pressure on the rate of solvent flow increases separation power, of HPLC and the analysis is completed within a short time 16.
FIG. 15: HIGH PERFORMANCE LIQUID CHROMATOGRAPHY
Characterization of Phytochemicals:
UV-Visible Spectroscopy: Ultraviolet (UV) spectroscopy is a valuable analytical technique used in pharmacognosy, the study of medicinal plants and natural products.
UV spectroscopy involves the measurement of the absorption of ultraviolet light by a substance, providing information about its electronic structure and the presence of certain functional groups 26. In pharmacognosy UV spectroscopy is employed for various purposes:
Identification of Compounds: UV spectroscopy helps in the identification of specific compounds based on their characteristic absorption patterns. Many natural products, such as alkaloids, flavonoids, and polyphenols, exhibit distinctive UV absorption spectra. Comparing the obtained spectra with reference standards can aid in the identification of compounds present in medicinal plants.
Quantitative Analysis: UV spectroscopy is often utilized for quantitative analysis of specific constituents in herbal extracts. By establishing a calibration curve relating concentration to absorbance at a specific wavelength, the amount of a particular compound in a sample can be determined. This is especially useful in quality control and standardization of herbal products.
Purity Assessment: The purity of herbal extracts or isolated compounds can be assessed using UV spectroscopy. Impurities or contaminants may have different UV absorption characteristics compared to the main compound, allowing for their detection and quantification.
Mass Spectrometry: The method has been used to peptide analysis and is effective with nearly all low molecular weight plant constituents. Mass spectrometry (MS) is a powerful analytical technique widely used in pharmacognosy, the branch of pharmacology that deals with the study of medicinal plants and natural products.
Compound Identification: Mass spectrometry is used to identify and confirm the presence of specific compounds within complex mixtures derived from medicinal plants. By measuring the mass-to-charge ratio (m/z) of ions, MS helps in determining the molecular weight and structural information of various phytochemicals.
Structural Elucidation: MS, particularly tandem mass spectrometry (MS/MS), aids in the structural elucidation of complex molecules. The fragmentation patterns generated during MS/MS experiments provide information about the connectivity of atoms within a compound, assisting in the determination of its structure.
Quantitative Analysis: Mass spectrometry is employed for quantitative analysis of bioactive compounds in herbal extracts. Techniques such as liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS) allow for the accurate measurement of the concentration of specific compounds, contributing to dosage determination and quality control 26.
Nuclear Magnetic Resonance Spectroscopy (NMR): The technique known as nuclear magnetic resonance (NMR For the purpose of identifying a medicine or an excipient, assessing the amount of contaminants (and clarifying their structure), tracking the breakdown process, assessing residual solvents, and figuring out isomeric compositions, NMR spectroscopy is crucial 28.
Structural Elucidation: NMR spectroscopy is widely used to determine the structures of natural products isolated from medicinal plants. By analysing the NMR spectra, researchers can deduce the connectivity of atoms, identify functional groups, and elucidate the overall molecular structure of complex compounds.
Identification of Chemical Constituents: NMR is employed to identify and characterize specific chemical constituents present in herbal extracts. It provides detailed information about the types of molecules present, helping in the identification of known and novel compounds in complex mixtures.
Quality Control: NMR can be employed for quality control purposes to ensure the consistency and authenticity of herbal products. It helps in detecting variations in the chemical composition of herbal preparations, ensuring adherence to quality standards.
X-Ray spectroscopy: X-ray spectroscopy is not a commonly used technique in pharmacognosy, which focuses on the study of medicinal plants and natural products.
However, X-ray techniques, such as X-ray crystallography and X-ray fluorescence spectroscopy, can have applications in related areas, including the structural analysis of crystalline compounds and elemental analysis 30.
X-ray Crystallography: X-ray crystallography is a powerful technique used for determining the three-dimensional atomic structure of a crystalline compound. While this method is more commonly associated with the field of structural biology, it can be applied in pharmacognosy when dealing with purified crystalline compounds derived from natural products.
By analysing X-ray diffraction patterns obtained from a crystal, researchers can determine the arrangement of atoms within the crystal lattice, providing detailed structural information.
Elemental Analysis - X-ray Fluorescence (XRF): X-ray fluorescence spectroscopy is a technique used for elemental analysis. In pharmacognosy, XRF can be employed to determine the elemental composition of plant samples, extracts, or herbal formulations. This information can be useful for quality control and ensuring that the expected elements are present within specified limits.
Application of Phytochemicals:
Health and Medicine: Disease prevention and treatment: Phytochemicals like flavonoids, carotenoids, and terpenes have been linked to reduced risk of chronic diseases like cancer, diabetes, heart disease, and neurodegenerative disorders 36.
Food and Beverage: Food preservation and quality enhancement: Certain phytochemicals act as natural antioxidants and preservatives, extending the shelf life of food products.
Food and Agriculture: Natural food preservatives: Some phytochemicals, such as rosemary extract and tocopherols, have antimicrobial properties that can help prevent food spoilage, extending the shelf life of food products without the need for synthetic preservatives 36.
Cosmetics and Skincare: Phytochemicals are incorporated into cosmetic and skincare products for their antioxidant and anti-aging properties 37.
Flavouring Agents: Phytochemicals contribute to the flavour and aroma of foods and beverages. Herbs and spices, such as turmeric, ginger, and cinnamon, are rich in phytochemicals and are used to enhance the taste of dishes.
Natural Pesticides and Herbicides: Neem oil, for example, is derived from the neem tree and is used as a naturalpesticide.
Research and Development: Phytochemicals are subjects of ongoing research to uncover new compounds with potential health benefits.
Phytochemicals in Marketed Formulations:
Antioxidant-rich Skincare Products:
Green Tea Extract: Contains polyphenols such as catechins, which have antioxidant and anti-inflammatory properties.
Lavender Oil: Contains compounds like linalool and linalyl acetate, known for their
Calmingand skin-soothing properties.
- Turmeric extract: Contains curcumin, known for its anti-inflammatory and antioxidant properties.
- Fruit extracts: citrus extract-Rich in vitamin c 40
CONCLUSION: The comprehensive study on the phytochemical analysis of medicinal plants aimed to elucidate the chemical composition of various plant extracts and explore their potential therapeutic applications. The investigation encompassed a diverse range of medicinal plants known for their traditional uses in folk medicine. The study employed rigorous extraction and analytical techniques to identify and quantify key phytochemicals present in these plants. The comprehensive phytochemical analysis of medicinal plants has yielded valuable insights into their chemical composition and potential therapeutic applications.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Harsharan Pal Singh, Sanchit Sharma, Shikha Baghel Chauhan and Ishpreet Kaur: Clinical trials of traditional herbal medicines in India: current status and challenges. IJP 2014; 1(7): 415-421.
- Chanda & Ramachandra: A review on some Therapeutic aspects of Phytochemical present. In Medicinal Plants. PInternational Journal of Pharmacy & Life Sciences 2019; 10(1): 6052-6058.
- Abdul Wadood, Mehreen Ghufran, Syed Babar Jamal, Muhammad Naeem, Ajmal Khan, Rukhsana Ghaffar and Asnad: Phytochemical Analysis of Medicinal Plants Occurring in Local Area of Mardan. Biochemistry & Analytical biochemistry Research Article 2013;
- Wendy L. Applequist & James S. Miller: Selection and authentication of botanical materials for the development of analytical methods 2012; DOI 10.1007/s00216-012-6595-1
- Harsharan Pal Singh, Sanchit Sharma, Shikha Baghel Chauhan and Ishpreet Kaur: AN Overview of major classes of phytochemicals: Their types and role in disease prevention TH Hislopia Journal 9 (1/2) 2016; ISSN: 0976-212
- Guyon I and Elisseeff A: An introduction to feature extraction. In Feature extraction: foundations and applications 2006; 1-25). Berlin, Heidelberg: Springer Berlin Heidelberg.
- Appelt DE: Introduction to information extraction. Ai Communications 1999; 12(3): 161-72.
- Andersen MR, Swanson JT, Porter NL and Richter BE: Supercritical fluid extraction as a sample introduction method for chromatography. Journal of Chromatographic Science 1989; 27(7): 371-7.
- Leblanc J, Akbarzadeh A, Andrews J, Lu H and Golding P: Heat extraction methods from salinity-gradient solar ponds and introduction of a novel system of heat extraction for improved efficiency. Solar Energy 2011; 85(12): 3103-42.
- Bart HJ: Extraction of natural products from plants–An introduction. Industrial Scale Natural Products Extraction 2011; 1-25.
- Galanos C, Lüderitz O and Westphal O: A new method for the extraction of R lipopolysaccharides. European Journal of Biochemistry 1969; 9(2): 245-9.
- Azwanida NN: A review on the extraction methods use in medicinal plants, principle, strength and limitation. Med Aromat Plants 2015; 4(196): 2167-0412.
- Azmir J, Zaidul IS, Rahman MM, Sharif KM, Mohamed A, Sahena F, Jahurul MH, Ghafoor K, Norulaini NA and Omar AK: Techniques for extraction of bioactive compounds from plant materials: A review. Journal of Food Engineering 2013; 117(4): 426-36.
- Trier ØD, Jain AK and Taxt T: Feature extraction methods for character recognition-a survey. Pattern Recognition 1996; 29(4): 641-62.
- Pensuk W, Padumanonda T and Pichaensoonthon C: Comparison of the chemical constituents in Michelia alba flower oil extracted by steam distillation, hexane extraction and enfleurage method. Journal of Thai Traditional & Alternative Medicine.
- Dean JR: Extraction methods for environmental analysis. Chichester: John Wiley 1998; 14.
- Raynie DE: Modern extraction techniques. Analytical Chemistry 2006; 78(12): 3997-4004.
- Raynie DE: Modern extraction techniques. Analytical Chemistry 2010; 82(12): 4911-6.
- Ong ES: Extraction methods and chemical standardization of botanicals and herbal preparations. Journal of Chromatography B 2004; 812(1-2): 23-33.
- Salomé-Abarca LF, Soto-Hernández RM, Cruz-Huerta N and González-Hernández VA: Chemical composition of scented extracts obtained from Calendula officinalis by three extraction methods. Botanical Sciences 2015; 93(3): 633-8.
- Farnsworth NR. Biological and phytochemical screening of plants. Journal of Pharmaceutical Sciences 1966; 55(3): 225-76.
- Shaikh JR and Patil M: Qualitative tests for preliminary phytochemical screening: An overview. International Journal of Chemical Studies 2020; 8(2): 603-8.
- Chhabra SC, Uiso FC and Mshiu EN: Phytochemical screening of Tanzanian medicinal plants. I. Journal of Ethnopharmacology 1984; 11(2): 157-79.
- Konieczka P and Namieśnik J: Estimating uncertainty in analytical procedures based on chromatographic techniques. Journal of Chromatography A 2010; 1217(6): 88291.
- Marston A: Role of advances in chromatographic techniques in phytochemistry. Phytochemistry 2007; 68(22-24): 2786-98.
- Barman BN, Cebolla VL and Membrado L: Chromatographic techniques for petroleum and related products. Critical Reviews in Analytical Chemistry 2000; 30(2-3): 75-120.
- Swartz M: HPLC detectors: a brief review. Journal of Liquid Chromatography & Related Technologies 2010; 33(9-12): 1130-50.
- Wilson ID, Plumb R, Granger J, Major H, Williams R and Lenz EM: HPLC-MS-based methods for the study of metabonomics. Journal of Chromatography B 2005; 817(1): 67-76.
- Clegg DL: Paper chromatography. Analytical Chemistry. 1950; 22(1): 48-59.
- Agarwal BK: X-ray spectroscopy: an introduction. Springer 2013; 29.
- Coskun O: Separation techniques: chromatography. Northern clinics of Istanbul 2016; 3(2): 156.
- Parikh H and Khanna A: Pharmacognosy and Phytochemical Analysis of Brassica juncea Seeds. Pharmacognosy Journal 2014; 6(5).
- Shinde V and Dhalwal K: Pharmacognosy: The changing scenario. Pharmacognosy Reviews 2007; 1(1): 1-6.
- Yang YN, Zhu H, Chen Z, Liu F, An YW, Feng ZM, Jiang JS and Zhang PC: NMR spectroscopic method for the assignment of 3, 5-dioxygenated aromatic rings in natural products. Journal of Natural Products 2015; 78(4): 705-11.
- Chakraborty SB, Horn P and Hancz C: Application of phytochemicals as growth‐promoters and endocrine modulators in fish culture. Reviews in Aquaculture 2014; 6(1): 1-9.
- Jeevanandam J, Aing YS, Chan YS, Pan S and Danquah MK: Nanoformulation and application of phytochemicals as antimicrobial agents. In Antimicrobial Nnanoarchitectonics Elsevier 2017; 61-82.
- Raj R, Lata KS, Raj V and Kumar DM: Phytochemicals and its application. The Pharma Innovation 2020; 9(10): 386-90.
- Barani M, Sangiovanni E, Angarano M, Rajizadeh MA, Mehrabani M, Piazza S, Gangadharappa HV, Pardakhty A, Mehrbani M, Dell’Agli M and Nematollahi MH: Phytosomes as innovative delivery systems for phytochemicals: A comprehensive review of literature. International Journal of Nanomedicine 2021; 6983-7022.
- Tkachenko NV: Optical spectroscopy: methods and instrumentations. Elsevier 2006.
- Krochmal R, Hardy M, Bowerman S, Lu QY, Wang HJ, Elashoff RM and Heber D: Phytochemical assays of commercial botanical dietary supplements. Evidence-based Complementary and Alternative Medicine 2004; 1: 305-13.
How to cite this article:
Haseen A, Prasanth LML and Jagajith AA: Comprehensive study on phytochemical analysis of medicinal plants. Int J Pharmacognosy 2024; 11(3): 53-64. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(3).53-64.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
53-64
1391 KB
1466
English
IJP
Asna Haseen, M. L. Lal Prasanth and A. Anu Jagajith *
Dr. Moopen’s College of Pharmacy, Wayanad, Kerala, India.
asnahaseen2001@gmail.com
20 February 2024
19 March 2024
27 March 2024
10.13040/IJPSR.0975-8232.IJP.11(3).53-64
31 March 2024