COMPARISON OF VITAMIN C CONTENT IN FRESH AND PACKED JUICES OF ORANGE AND MANGO
HTML Full TextCOMPARISON OF VITAMIN C CONTENT IN FRESH AND PACKED JUICES OF ORANGE AND MANGO
Amanuel Yebio, Mebrahtom Gebrelibanos *, Aman Karim, Gereziher Gebremedhin, Biruk Sintayehu and Gomathi Periasamy
Department of Pharmacy, Course and Research Unit of Pharmacognosy, College of Health Sciences, Mekelle University, Ethiopia.
ABSTRACT: Vitamin C, also known as ascorbic acid, is a water-soluble antioxidant vitamin having important nutritional and health benefits. Adequate consumption of fruits, vegetables and their juices with high vitamin C content and antioxidant capacity result in improved health, reduced risk of chronic diseases, optimal nutrition and general well-being. Thus, it is essential that vitamin C content in food items, intended to provide its supplement, is determined and its amount stated on labels of packages. The objective of the present study was to determine and compare the vitamin C content of fresh and commercial juices made from mango and orange. Fruits and packed juices were purchased from the local market of Mekelle city, Ethiopia. Iodine titration method was employed to determine the vitamin C content of test sample juices. Results showed that the amount of vitamin C was more in fresh juice than the corresponding packed juice in both mango and orange. For the fresh juices, the amount of vitamin C was more in orange than in mango while for the packed juices the amount in mango was more than in orange; although the manufacturing dates and hence storage times of both packed juices were similar. Therefore, it was concluded that in case of mango juices, both fresh and packed may be considered as vitamin C supplements whereas in case of orange juices, the fresh one is far more preferred to the packed one because the packed one may not be sufficient source of vitamin C, especially if supplied in liquid packed form.
| Keywords: |
Vitamin C, Mango, Orange, Juice
INTRODUCTION: Vitamin C, also known as ascorbic acid, is a water-soluble, antioxidant vitamin, which is important in forming collagen, a protein that gives structure to bones, cartilages, muscles, and blood vessels.
It helps maintain capillaries, bones, and teeth. It also helps the body in iron absorption from plant-based foods (i.e., non-haem iron), wound healing, red blood cell formation, and in fighting infections as it helps the immune system work properly to protect the body from disease. It has been stated that large doses of vitamin C (one gram or more) may prevent or cure a common cold or influenza.
Also, some studies claim that vitamin C prevents cancer. Lack of vitamin C cause scurvy, iron deficiency, and poor wound healing; indicating that a healthy diet should include adequate amounts of vitamin C. Moreover, low vitamin C levels have been associated with high risk of blood pressure, heart attack, cataracts, and certain types of cancer, stunted growth, tooth decay, improper bone development, loss of appetite, weakened cartilages, poor collagen production, skin hemorrhages, and impaired digestion. Fruits and vegetables are rich sources of vitamin C 1, 2, 3 and it is claimed that adequate consumption of fruits and vegetables with high vitamin C content result in improved health, reduced risk of chronic diseases, optimal nutrition and general well-being 4, 5. On the other hand, high levels of ascorbic acid in the human body can cause adverse effects: it may lead to gastric irritation, and its metabolic product (oxalic acid) can cause renal problems.
Thus, the ascorbic acid content of foodstuffs and beverages represents a relevant indicator of quality which has to be carefully monitored, regarding its variation during manufacturing and storage. Numerous methods have been employed to quantify vitamin C in different matrices including chemiluminescence, chromatographic, electro-chemical, fluorometry, redox titrimetric, and spectrophotometric 3, 6, 7. For reasons of its simplicity, low cost, and easily availability, the redox iodine titrimetric method has been employed in the present study; and this study aimed to determine and compare the vitamin C content of fresh and packed mango and orange juices.
MATERIALS AND METHODS:
Materials: Fruits: Mango (Mangifera indica) and orange (Citrus sinensis); commercially packed juices: Mango juice and Orange juice; chemicals and solvents: standard ascorbic acid, soluble starch, potassium iodide (KI), potassium iodate (KIO3), sulfuric acid, distilled water. Equipment: burette and stand, volumetric flasks, pipette, measuring cylinders, conical flasks, juice blender, and muslin cloth. Fruits and packed juices were purchased from the local market of Mekelle city, Ethiopia; and all reagents and chemicals used in this experiment were analytical grades.
Methods:
Determination of Vitamin C Content by Iodine Titration: Vitamin C content of fresh and commercial juices was determined using iodine titration method as per the procedure described by Njoku et al., (2011) 8 and Helmenstine, (2014) 9 with slight modifications.
Preparation of Solutions: 1% starch indicator solution was prepared by adding 0.50 gm soluble starch to 50 ml near-boiling distilled water, mixed well and allowed to cool before use. To prepare the iodine solution, 5.00 g potassium iodide (KI) and 0.268 gm potassium iodate (KIO3) were dissolved in 200 ml of distilled water. 30 ml of 3 M sulphuric acid was added to this solution and diluted to a final volume of 500 ml with distilled water. Vitamin C standard solution was prepared by dissolving 0.250 gm vitamin C in 100 ml distilled water and diluted to 250 ml with distilled water.
To prepare fresh fruit juice, 100 gm fruit sample, cut into small pieces, was blended using juice blender with 50 ml of distilled water; pulps strained through muslin cloth, washed with 10 ml portions of distilled water, and extracted solution was made up to 100 ml. Packaged juices were opened and strained through muslin cloth immediately before titration.
Standardizing of Vitamin C and Titration: 25 ml of vitamin C standard solution and juice samples were added to 125 ml Erlenmeyer flask. 10 drop of 1% starch solution was added to each flask. A burette was rinsed with 10 ml iodine solution and then filled with the iodine solution which was used to titrate the vitamin C solution and the juice samples until the endpoint was reached (when the first sign of blue color that persisted after 20 sec of swirling the solution was seen).
Titration volume (consumed iodine solution) was recorded and each time titration was repeated three times until results that agreed within 0.1 ml were obtained. Vitamin C content of juice samples was calculated based on results from titration of standard vitamin C solution using the following formula:
CVitC(juice) = Vt(juice) X [CVitC(SS)/Vt(SS)]
Where, CVitC(juice) is the concentration of vitamin C in sample juices, Vt(juice) is the average titration volume of sample juices, CVitC(SS) is a concentration of vitamin C in the standard solution, and Vt(SS) is the average titration volume of standard solution.
RESULTS AND DISCUSSION:
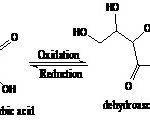
Determination of Vitamin C Content by Iodine Titration: The chemical principle of iodine titration employed in the present experiment can be described as follows: to improve the relatively water-insoluble nature of iodine, it is complexed with iodide to form triiodide (equation 1). During titration, triiodide oxidizes vitamin C to dehydro-ascorbic acid (equation 2) and Fig. 1. As long as vitamin C is present in the solution, the triiodide is converted to iodide ion very quickly. Once all the vitamin C has been oxidized, the excess iodine and triiodide are free to react with the starch indicator, forming blue-black starch-iodine complex, which is the endpoint of the titration.
I2 + I- ↔ I3- Eq. 1
C6H8O6 + I3- + H2O → C6H6O6 + 3I- + 2H+ Eq. 2
FIG. 1: SHOWS OXIDATION OF VITAMIN C TO DEHYDROASCORBIC ACID
Iodine Titration method is suitable for testing the amount of vitamin C in vitamin C tablets, juices, and fresh, frozen, or packaged fruits and vegetables. The titration can be performed using just iodine (not iodate) solution, but the iodate solution is more stable and gives a more accurate result, and hence the iodate solution was used in this experiment. Results of measurements done following this principle are summarized in Table 1.
TABLE 1: TITRATION VOLUMES AND CALCULATED AMOUNT OF VITAMIN C IN SAMPLE JUICES AND STANDARD VITAMIN C SOLUTION
| Samples | Titration volumes (ml) | Amount of Vit C | ||||
| 1 | 2 | 3 | Average | (mg/ml) | (mg/100 ml) | |
| Standard Solution | 18 | 18 | 18 | 18.00 | 1.00 | 100.00 |
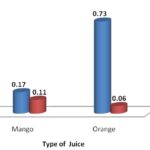
| Fresh Mango Juice | 3.1 | 3.2 | 3.1 | 3.13 | 0.17 | 17 |
| Packed Mango Juice | 2 | 1.9 | 2 | 1.97 | 0.11 | 11 |
| Fresh Orange Juice | 13 | 13.25 | 13.25 | 13.17 | 0.73 | 73 |
| Packed Orange Juice | 1.1 | 1.2 | 0.9 | 1.07 | 0.06 | 6 |
Titration of four juice samples (two fresh and two commercial) made from mango and orange fruits as well as standard vitamin C solution was carried out an average titration volume (consumed iodine solution) from triple measurements were recorded. The amount of vitamin C in each juice sample was calculated based on results from titration of standard vitamin C solution which contained a known concentration of 1 mg/ml.
FIG. 2: SHOWS VITAMIN C CONTENT IN MANGO AND ORANGE JUICES OF PACKED AND FRESH TYPES
Fresh and packed juices of mango and orange were compared by their vitamin C content; and as can be seen from Fig. 2, the calculated amount of vitamin C in fresh juice was higher than packed juice in both mango and orange. However, the difference between fresh and packed juice was more pronounced in orange. The observation that vitamin C content in fresh juices was higher than packed counterparts is expected phenomena since vitamin C is lost during processing and storage of packed juices. This loses through destruction, mainly by oxidation, is a serious problem in that a considerable quantity of vitamin C content of food is lost during processing, storage and preparation 10. Another similar report showed that content of vitamin C in industrial lemon juice was significantly lower than the mean value of vitamin C in fresh lemon juice, and the amount of vitamin C in the industrial lemon juice significantly reduced as storage time increased 11.
However, the observation that vitamin C content in packed juice of mango was higher than in packed juice of orange was not expected as citrus juices are claimed to be relatively good options for gaining vitamin C 12. Results of the present experiment compared mango and commercial orange juices of similar manufacturing date and indicated that commercial mango might provide better vitamin C than an orange. Similarly, Hossain et al., (2012) 12 reported that mango juice might provide a level in excess vitamin C than orange juices.
Being an important vitamin for human nutrition and health, vitamin C intake recommendations have been established that depend on age, sex, and health status. The recommended daily allowance (RDA) for vitamin C ranges from 40 mg for infants below 6 months to 120 mg for lactating women. For non-smoker adults, the RDA is 75 mg per day for women and 90 mg per day for men. Individuals who smoke require 35 mg/day more vitamin C than non-smokers 13.
In the present experiment, one orange fruit produced an average of 30 ml fresh juice; one mango fruit produced an average of 50 ml fresh juice, and the pack size of both commercial juices was 500 ml. Technically speaking, if for someone the juices studied in this experiment are the only sources of vitamin C, someone has to consume at least 55, 235, 364, and 650 ml of fresh orange juice, fresh mango juice, packed mango juice, and packed orange juice, respectively in order to cross the minimum amount in the RDA range of vitamin C. If it is considered that most individuals take liquid juices of up to 500 ml, it seems that the first three may provide the required vitamin C while the packed orange juice may not.
CONCLUSION: In conclusion, based on the results of this experiment, it could be suggested that in case of mango juices, both fresh and packed may be considered as vitamin C supplements whereas in case of orange juices, the fresh one is far more preferred to the packed one because the packed one may not be sufficient source of vitamin C, especially if supplied in liquid packed form.
ACKNOWLEDGEMENT: One of the authors (AY) is grateful to Pharmacognosy Course and Research Unit, Department of Pharmacy, College of Health Sciences, Mekelle University. He also thankful to Ato Abadi Aynalem and Teklebrhan Hailu for their laboratory helps while doing his experiment.
CONFLICT OF INTEREST: Nil
REFERENCES:
- NIH: Vitamin C Fact Sheet for Consumers. National Institute of Health, Office of Dietary Supplements-Strengthening Knowledge and Understanding of Dietary Supplements, 2011. (Accessed on Dec 12, 2014) at: http://ods.od.nih.gov/factsheets/VitaminC-Consumer/
- Pisoschi AM, Pop A, Negulescu GP and Pisoschi A: Determination of ascorbic acid content of some fruit juices and wine by voltammetry performed at Pt and carbon paste electrodes. Molecules 2011; 16: 1349-1365.
- Tadese A, Subramanian PA, Woldu A and Pal R: Electrochemical determination and comparison of ascorbic acid in freshly prepared and bottled fruit juices: A cyclic voltammetric study. Journal of Chemical and Pharmaceutical Research 2014; 6(5): 880-888.
- Ogunlesi M, Okiei W, Azeez L, Obakachi V, Osunsanmi M and Nkenchor G: Vitamin C contents of tropical vegetables and foods determined by voltammetric and titrimetric methods and their relevance to the medicinal uses of the plants. International Journal of Electrochemical Science 2010; 5: 105 -115.
- Liu RH: Health-promoting components of fruits and vegetables in the diet. Advances in Nutrition 2013; 4: 384S-392S.
- Hossu AM and Magearu V: Determination of vitamin C in pharmaceutical products with physicochemical and bioanalytical techniques. Roumanian Biotechnological Letters 2004; 9(1): 1497-1504.
- Mequanint T, Moges G, Tessma M and Mehretu S: All-solid-state Iodide selective electrode for iodimetry of iodized salts and vitamin C. Oriental Journal of Chemistry 2012; 28(4): 1547-1555.
- Njoku PC, Ayuk AA and Okoye CV: Temperature effects on vitamin C content in citrus fruits. Pakistan Journal of Nutrition 2011; 10 (12): 1168-1169.
- Helmenstine AM: Vitamin C determination by iodine titration. 2014. (Accessed on Sep 12, 2014) at: http:// chemistry.about.com/od/demonstrationsexperiments/ss/vitctitration.htm
- Oyetade OA, Oyeleke GO, Adegoke BM and Akintunde AO: Stability studies on ascorbic acid (Vitamin C) from different sources. IOSR Journal of Applied Chemistry (IOSR-JAC) 2012; 2 (4): 20-24.
- Babashahi-Kouhanestani M, Salehi M, Mazloomi SM and Almasi-Hashyani A: Quantitative evaluation of vitamin C in industrial lemon juice by titration method. Journal of Biology and Today’s World 2014; 3 (6): 139-141.
- Hossain MA, Rahman M and Shabuz MZR: Quality of industrially processed fruit juices: An assessment using multivariate framework. Dhaka University Journal of Science 2012; 60(2): 169-173.
- Dickinson A: Recommended intakes of vitamin C and essential Minerals. From the Benefits of Nutritional Supplements, Council for Responsible Nutrition, 2002. (Accessed on Dec 12, 2014) at: http://www.crnusa. org/benpdfs/CRN012benefits_recs.pdf
- NIH: Vitamin C Fact Sheet for Health Professionals. National Institutes of Health, Office of Dietary Supplements-Strengthening Knowledge and Under-standing of Dietary Supplements, 2013. (Accessed on Dec 18, 2014) at: http://ods.od.nih.gov/ factsheets/ Vitamin C-Health Professional/
How to cite this article:
Yebio A, Gebrelibanos M, Karim A, Gebremedhin G, Sintayehu B and Periasamy G: Comparison of vitamin C content in fresh and packed juices of orange and mango. Int J Pharmacognosy 2015; 2(2): 88-92. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(2).88-92.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
88-92
492
4471
English
IJP
A. Yebio, M. Gebrelibanos*, A. Karim, G. Gebremedhin, B. Sintayehu and G. Periasamy
Department of Pharmacy, Course and Research Unit of Pharmacognosy, College of Health Sciences, Mekelle University, Ethiopia.
mebrahtomgs@yahoo.com
31 December 2014
19 January 2015
27 January 2015
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(2).88-92
01 February 2015