COMPARING THE EFFICACY AND SAFETY OF HERBAL MEDICINES Vs CONVENTIONAL DRUGS IN RESPIRATORY DISEASES
HTML Full TextCOMPARING THE EFFICACY AND SAFETY OF HERBAL MEDICINES Vs CONVENTIONAL DRUGS IN RESPIRATORY DISEASES
P. Devi Guna Sireesha *, P. Swetha, Shaik Ayesha Sidhikha and Tokala Divya
Department of Pharmacology, Nimra College of Pharmacy, Ibrahimpatnam, Nimra Nagar, Vijayawada, Andhra Pradesh, India.
ABSTRACT: Respiratory diseases remain one of the leading global health burdens, ranging from acute infections such as influenza and pneumonia to chronic conditions like asthma and chronic obstructive pulmonary disease. Conventional pharmacological agents, including bronchodilators, corticosteroids, antibiotics, and biologics, are widely recognized for their clinical efficacy and regulatory approval, yet their long-term use is often associated with adverse effects, drug resistance, and financial burden. In contrast, herbal medicines have been used for centuries in various cultural and traditional systems of medicine, offering potential anti-inflammatory, mucolytic, antiviral, antioxidant, and immunomodulatory benefits. Recent interest has increasingly focused on systematically comparing the effectiveness and safety of herbal therapies with conventional drugs, especially as patients actively seek holistic and integrative approaches to respiratory care. This review critically examines the available evidence on herbal interventions such as Echinacea, Pelargonium, licorice root, ginger, turmeric, and ivy leaf and contrasts them with standard pharmacological options including bronchodilators, corticosteroids, and mucolytic agents. Safety profiles, possible herb drug interactions, pharmacoeconomic aspects, and limitations of current studies are also evaluated. By synthesizing both traditional knowledge and modern clinical evidence, this review aims to highlight the potential role of herbal medicines as complementary or alternative therapies, while emphasizing the need for robust clinical trials, quality assurance, and standardized formulations. The ultimate goal is to provide a balanced, evidence-based perspective to guide clinicians, researchers, and patients toward safer, cost-effective, and informed decisions in the management of respiratory diseases, thereby supporting innovation, patient empowerment, and sustainable healthcare practices worldwide, particularly in diverse clinical and community settings.
Keywords: Phytotherapy, Traditional medicine, Asthma, Chronic bronchitis, Respiratory diseases
INTRODUCTION: Respiratory diseases represent a significant global health challenge, contributing substantially to morbidity, mortality, and healthcare costs.
Conditions such as asthma, chronic obstructive pulmonary disease (COPD), pneumonia, influenza, and acute upper respiratory tract infections account for millions of lost lives each year and place immense pressure on healthcare systems.
The burden is particularly pronounced in low- and middle-income countries, where pollution, smoking, occupational hazards, and limited healthcare access exacerbate the prevalence and severity of these disorders. Beyond their clinical impact, respiratory illnesses diminish quality of life by restricting daily activities, reducing productivity, and increasing psychological stress for patients and their families 1.
Conventional medicine has long been the backbone of respiratory disease management. Treatments such as bronchodilators, corticosteroids, antibiotics, leukotriene antagonists, biologics, and supportive therapies have transformed the landscape of care. These drugs are often life-saving, offering rapid and measurable improvements in lung function, inflammation control, and infection clearance. However, their benefits are accompanied by well-recognized limitations. Long-term steroid use can lead to systemic complications such as osteoporosis, metabolic disturbances, and immunosuppression. Antibiotics are threatened by the alarming rise of antimicrobial resistance. Biologics, though promising, remain prohibitively expensive for most patients and health systems. Moreover, chronic conditions like COPD often require lifelong therapy, raising concerns about cumulative side effects and financial strain.
In parallel, the use of herbal medicines for respiratory conditions has persisted across cultures for centuries. Traditional Chinese Medicine, Ayurveda, Kampo, and other indigenous systems have incorporated plant-based formulations for coughs, breathlessness, and lung infections. Modern pharmacological research has begun to validate some of these practices, identifying bioactive compounds with anti-inflammatory, bronchodilatory, mucolytic, or immunomodulatory effects. For example, herbs such as ginger, licorice, and turmeric are valued for their anti-inflammatory properties, while ivy leaf and Pelargonium extracts are widely used for their mucolytic and antimicrobial activities. Unlike synthetic drugs, herbal therapies are often perceived as natural and safer, though this perception does not always align with scientific evidence.
The growing popularity of herbal interventions reflects a global trend toward integrative and patient-centered care. Many individuals, dissatisfied with the side effects or limitations of conventional drugs, turn to herbal remedies as alternatives or adjuncts. This shift poses both opportunities and challenges for healthcare providers. On one hand, herbal medicines may complement conventional therapy, improving symptom control, reducing drug doses, and enhancing overall well-being. On the other hand, variability in preparation, lack of standardization, limited regulatory oversight, and potential herb–drug interactions raise legitimate concerns regarding safety and efficacy 2.
Against this backdrop, it is crucial to critically compare the efficacy and safety of herbal medicines with established pharmacological treatments in respiratory diseases. Such comparisons can provide insights into whether herbal remedies can serve as effective stand-alone therapies, supportive agents, or merely adjuncts without substantial clinical benefit. A careful review of existing clinical trials, meta-analyses, and pharmacological studies is necessary to distinguish evidence-based practice from unverified traditional claims.
This article aims to present a comprehensive review of the available literature comparing herbal medicines and conventional drugs in respiratory diseases. It will examine their respective mechanisms of action, therapeutic outcomes, safety profiles, and limitations. In addition, the review will discuss the potential role of integrative medicine in respiratory care and highlight gaps in current knowledge that warrant further research. By providing a balanced analysis, the article seeks to inform clinicians, researchers, and policymakers while guiding patients toward safe and effective choices in managing respiratory illnesses.
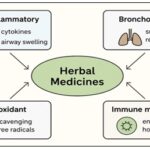
Herbal Medicines in Respiratory Diseases: Herbal medicines have been widely utilized in traditional and modern systems of medicine for managing respiratory diseases due to their immunomodulatory, anti-inflammatory, antiviral, antibacterial, and mucolytic properties (represented in Fig. 1). Unlike conventional pharmaceuticals that target specific pathways, many herbal remedies exert broad-spectrum effects by modulating multiple biological targets simultaneously.
Echinacea: Echinacea species, especially Echinacea purpurea, have been extensively studied for their immune-enhancing effects. They stimulate phagocytosis, increase leukocyte activity, and upregulate cytokine release, thereby strengthening host defence against viral respiratory tract infections. Clinical evidence suggests that Echinacea extracts reduce the frequency and duration of common cold episodes 3.
Umckaloabo (Pelargonium sidoides): Pelargonium sidoides root extract is used in bronchitis and upper respiratory tract infections. It enhances mucociliary clearance, exhibits antibacterial activity, and modulates interferon production 4. Randomized trials indicate its role in reducing cough frequency and improving lung function in acute bronchitis 5.
Licorice (Glycyrrhiza glabra): Licorice root has expectorant, anti-inflammatory, and soothing properties, making it effective for cough and sore throat management 6. Its active component, glycyrrhizin, reduces airway inflammation and inhibits viral replication 7.
Ivy Leaf (Hedera helix): Ivy leaf extract is widely used in pediatric and adult cough syrups. It exhibits mucolytic and bronchodilatory properties by relaxing airway smooth muscles and enhancing mucus clearance 8. Clinical data support its efficacy in improving cough scores in acute bronchitis 9.
Turmeric (Curcuma longa): Curcumin, the bioactive constituent of turmeric, possesses strong antioxidant and anti-inflammatory properties. It reduces airway hyper responsiveness and downregulates NF-κB signaling, which is central to asthma pathogenesis 10.
Ginger (Zingiber officinale): Ginger has bronchodilatory and anti-inflammatory effects, attributed to compounds like gingerols and shogaols. Experimental studies show that ginger relaxes airway smooth muscle and enhances β₂-agonist activity 11.
Indian olibanum (Boswellia serrata): Boswellia resin contains boswellic acids, which inhibit 5-lipoxygenase and suppress leukotriene synthesis, thereby reducing bronchoconstriction and airway inflammation 12. Clinical trials have demonstrated improvement in asthma symptoms with Boswellia supplementation 13 (represented in Table 1).
FIG. 1: MECHANISM OF HERBAL MEDICINES IN RESPIRATORY DISEASES
The above figure shows schematic illustration showing key mechanisms by which herbal medicines act on respiratory disorders, including anti-inflammatory activity, bronchodilation, antioxidant defence, and immune modulation.
TABLE 1: SELECTED HERBAL MEDICINES USED IN RESPIRATORY DISEASES & THEIR PRIMARY ACTIONS
| Herbal Medicine | Common Name | Primary Actions | Clinical Uses |
| Echinacea | Green Chiretta | Immunostimulant, antiviral | Common cold, URTI |
| Pelargonium sidoides | African Geranium | Antibacterial, mucolytic,
immune modulator |
Acute bronchitis, URTI |
| Glycyrrhiza glabra | Licorice | Expectorant, anti-inflammatory, antiviral | Cough, sore throat |
| Hedera helix | English Ivy | Mucolytic, bronchodilator | Acute bronchitis, cough relief |
| Curcuma longa | Turmeric | Anti-inflammatory, antioxidant | Asthma, airway inflammation |
| Zingiber officinale | Ginger | Bronchodilator, anti-inflammatory | Asthma, bronchospasm |
| Boswellia serrata | Indian Olibanum | Leukotriene inhibitor, anti-inflammatory | Asthma, chronic bronchitis |
In the above table, you can see the Mechanistic pathways of selected herbal medicines in respiratory diseases.
Conventional Drugs in Respiratory Diseases: Conventional pharmacotherapy forms the cornerstone of modern respiratory disease management. These drugs are standardized, well-studied, and act through defined molecular mechanisms that directly target pathophysiological processes (represented in Table 2). Unlike herbal medicines, which often act on multiple biological systems simultaneously, conventional drugs are designed to provide precise, evidence-based outcomes with established dosing regimens 13.
Bronchodilators: Bronchodilators, including β₂-adrenergic agonists, anticholinergics, and methylxanthines, are essential in managing obstructive airway diseases.
β₂-agonists (e.g., salbutamol, formoterol): Stimulate β₂ receptors on airway smooth muscles, causing rapid bronchodilation. Short-acting β₂ agonists (SABA) are used for acute symptom relief, while long-acting β₂ agonists (LABA) are combined with corticosteroids for maintenance 14.
Anticholinergics (e.g., ipratropium, tiotropium): Block muscarinic receptors, reducing bronchoconstriction and mucus secretion. Tiotropium has a prolonged action and is beneficial in chronic obstructive pulmonary disease (COPD) 15.
Methylxanthines (e.g., theophylline): Inhibit phosphodiesterase, increasing intracellular cAMP and promoting bronchodilation. Their use is limited by narrow therapeutic index and side effects 16.
Corticosteroids: Corticosteroids are potent anti-inflammatory agents that reduce airway hyperresponsiveness by suppressing cytokine release and inflammatory cell recruitment 17.
Inhaled Corticosteroids (ICS) like budesonide and fluticasone are the mainstay in asthma management, particularly for long-term control 18.
Systemic corticosteroids (e.g., prednisolone) are used for severe exacerbations but are associated with significant side effects including immunosuppression and osteoporosis 19.
Leukotriene Modifiers: Leukotriene receptor antagonists (LTRAs) such as montelukast block leukotriene D₄ at cysteinyl leukotriene receptors, reducing inflammation and bronchoconstriction. They are particularly effective in exercise-induced asthma and aspirin-sensitive asthma 20.
Mast Cell Stabilizers: Cromolyn sodium and nedocromil inhibit mast cell degranulation, thereby preventing histamine and cytokine release. Although less commonly used now, they remain options for mild asthma prophylaxis 21.
Immuno Modulators and Biologics: The advent of biologics has transformed management of severe asthma and eosinophilic phenotypes.
Anti-IgE Therapy (Omalizumab): Binds circulating IgE, preventing allergic cascade activation 22.
Anti-IL-5 agents (Mepolizumab, Reslizumab): Reduce eosinophilic inflammation, beneficial in severe eosinophilic asthma 23.
Anti-IL-4/IL-13 agents (Dupilumab): Target type 2 inflammatory pathways, improving asthma control 24.
Mucoactive agents: Agents like N-acetylcysteine (NAC) and carbocisteine are used to modify mucus properties, enhancing clearance and reducing airway obstruction 25.
These are particularly beneficial in chronic bronchitis and COPD where mucus hypersecretion is prominent.
TABLE 2: COMMON CONVENTIONAL DRUGS IN RESPIRATORY DISEASES MANAGEMENT
| Drug Class | Examples | Mechanism of Action | Respiratory Uses | Adverse Effects |
| β₂ agonists | Salbutamol, Formoterol | Stimulate β₂ receptors → bronchodilation | Asthma, COPD | Tremors, tachycardia, hypokalemia, palpitations |
| Anticholinergics | Ipratropium, Tiotropium | Block muscarinic receptors → prevent bronchoconstriction | COPD, severe asthma | Dry mouth, blurred vision, urinary retention, constipation |
| Methylxanthines | Theophylline
|
PDE inhibition → ↑cAMP → bronchodilation | Asthma (limited use) | Nausea, vomiting, insomnia, arrhythmias, seizures (narrow therapeutic index) |
| Corticosteroids | Budesonide, Prednisolone | Suppress airway inflammation | Asthma, COPD exacerbations | Oral candidiasis, hoarseness, osteoporosis, immunosuppression, adrenal suppression |
| Leukotriene Modifiers | Montelukast
|
Block leukotriene receptors → ↓inflammation | Asthma (exercise, aspirin-induced) | Headache, abdominal pain, neuropsychiatric effects (rare) |
| Mast Cell Stabilizers | Cromolyn, Nedocromil | Prevent mast cell degranulation | Asthma prophylaxis | Throat irritation, cough, unpleasant taste |
| Biologics | Omalizumab, Mepolizumab, Dupilumab | Target IgE / IL-5 / IL-4 pathways | Severe asthma | Injection site reactions, hypersensitivity, anaphylaxis (rare), eosinophilia |
| Mucoactive Agents | N-acetylcysteine, Carbocisteine
|
Reduce mucus viscosity, ↑clearance | Chronic bronchitis, COPD | Nausea, vomiting, diarrhea, bronchospasm (with inhaled use) |
In the above table, the major therapeutic targets of conventional drugs in respiratory diseases.
Comparative Efficacy and Safety of Herbal Medicines vs. Conventional Drugs: The comparison between herbal medicines and conventional pharmacotherapy in respiratory diseases is complex, as both approaches offer unique benefits and limitations (represented in Table 3). Conventional drugs are widely accepted due to rigorous clinical testing and standardized dosing, whereas herbal therapies are often utilized for their holistic effects, favorable safety profiles, and cultural acceptance 26.
Efficacy in Asthma: Conventional therapies, particularly inhaled corticosteroids and β₂-agonists, remain first-line due to their proven efficacy in reducing airway inflammation and providing rapid bronchodilation. Biologics such as omalizumab have further advanced outcomes in severe phenotypes 27, 28.
Conversely, herbs such as Glycyrrhiza glabra (licorice) and Adhatodavasica demonstrate anti-inflammatory and bronchodilatory effects in preclinical and limited clinical studies. Randomized trials suggest that herbal formulations may reduce symptom scores and rescue medication use, though the effects are generally milder than corticosteroids 29, 30.
COPD and Chronic Bronchitis: Bronchodilators and corticosteroids form the backbone of COPD management, with long-acting muscarinic antagonists (LAMAs) and LABAs improving quality of life and reducing exacerbations 31.
Herbal therapies like Ginseng (Panax ginseng) and Curcuma longa (turmeric) have demonstrated immunomodulatory and antioxidant properties that may help alleviate oxidative stress and improve pulmonary function 32. Meta-analyses report small but significant improvements in FEV₁ and symptom control compared to placebo 33, though less robust than conventional therapies.
Allergic Rhinitis: Antihistamines and intranasal corticosteroids are effective in reducing rhinorrhea, sneezing, and nasal congestion 34. However, adverse effects such as sedation or mucosal dryness can limit compliance. Herbal alternatives like Butterbur (Petasites hybridus) have shown comparable efficacy to antihistamines in some trials, with fewer sedative effects 35. Additionally, Nigella sativa has demonstrated antihistamine-like activity by inhibiting histamine release 36.
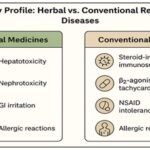
Safety Considerations: While conventional therapies are well-characterized, they are often associated with dose-dependent side effects (represented in Fig. 2). Corticosteroids can cause osteoporosis, adrenal suppression, and increased infection risk 37. β₂-agonists may cause tachycardia and tremors, while biologics are expensive and carry risks of anaphylaxis 38. Herbal therapies generally have favourable safety profiles, especially when used in traditional dosages. However, risks of contamination, adulteration, and herb–drug interactions must be considered 39. For example, ginseng may potentiate hypoglycemia in diabetic patients on insulin 40.
FIG. 2: SAFETY PROFILE: HERBAL VS CONVENTIONAL DRUGS IN RESPIRATORY SYSTEM
In the above figure, comparing the safety profile of herbal medicines versus conventional drugs. Common side effects (e.g., hepatotoxicity, nephrotoxicity, GI irritation, and allergic reactions) are contrasted with adverse events from conventional medications (e.g., steroid-induced immunosuppression, β2-agonist tachycardia, NSAID intolerance).
Patient-centred Outcomes: Patient adherence is typically higher with conventional drugs due to availability of inhalers and structured dosing 41. Yet, in regions with cultural acceptance of herbal practices, patients may prefer herbal remedies for their natural perception and affordability 42. Integration of both systems herbal adjuncts with conventional drugs has been reported to improve symptom control, reduce required corticosteroid doses, and enhance patient satisfaction 43.
TABLE 3: COMPARATIVE EFFICACY & SAFETY OF HERBAL VS CONVENTIONAL THERAPIES
| Parameter | Herbal Medicines | Conventional Drugs |
| Efficacy (Asthma) | Moderate improvement; anti-inflammatory, bronchodilatory (Adhatoda, Licorice) | Strong evidence; ICS, β₂ agonists, biologics |
| Efficacy (COPD) | Mild to moderate benefit; antioxidant, immunomodulatory (Ginseng, Turmeric) | Robust improvements in FEV₁, QoL (LABA, LAMA, ICS) |
| Efficacy (Rhinitis) | Butterbur, Nigella effective in reducing symptoms | Antihistamines, corticosteroids highly effective |
| Safety | Generally safe but risk of contamination/adulteration; herb–drug interactions possible | Known side effects: ICS (osteoporosis), β₂ agonists (tachycardia) |
| Patient preference | Natural, culturally accepted, affordable | Widely available, standardized dosing |
In the above table, you can see the comparative efficacy and safety of herbal vs. conventional drugs in respiratory diseases.
Integrative approaches: herbal ± conventional therapies
Many patients and clinicians are drawn to an “and/and” model pairing evidence-based conventional drugs with select herbal agents to reduce symptom burden, lower exacerbation risk, and potentially enable dose-sparing of steroids or bronchodilators 44. The logic is pragmatic: conventional drugs deliver targeted, high-certainty effects; certain herbal agents may complement them through mucolysis, antioxidant activity, and immune modulation 45 (represented in Fig. 3).
Asthma: Add-on Botanicals to Reduce Steroid Load: In mild–moderate asthma, inhaled corticosteroids (ICS) remain foundational. Small randomized and pragmatic studies suggest that curcumin or Boswellia serrata added to standard care can improve symptom scores and selected inflammatory markers, with some reporting reduced rescue medication use 46. Ginger has shown acute bronchodilatory synergy with β₂-agonists in preclinical and pilot human work, plausibly enhancing smooth-muscle relaxation 47. Licorice (glycyrrhizin) may support cough control, though careful dosing is essential to avoid pseudoaldosteronism 48. These data support adjunctive use not replacement especially in patients who experience steroid side effects yet require control.
COPD & Chronic Bronchitis: Mucolytics Meet Botanicals: COPD management hinges on LAMA/LABA ± ICS; however, chronic sputum, viscous secretions, and recurrent infections remain everyday challenges. Ivy leafandPelargonium sidoides (EPs 7630) have repeatedly demonstrated improvements in cough scores and symptom duration in acute bronchitis and URTI settings; as adjuncts to N-acetylcysteine/erdosteine, they may further facilitate mucus clearance and shorten illness course 49. Preliminary work combining NAC with antioxidant-rich botanicals (e.g., turmeric) suggests additive effects on oxidative stress readouts and health status, though larger trials are needed 50.
Recurrent URTI’S: Reducing Antibiotic Pressure: Antibiotic stewardship is a priority. Trials of Pelargonium show reduced symptom severity and lower downstream antibiotic use in acute bronchitis when used early and appropriately 51. Echinacea prophylaxis may modestly reduce cold frequency/duration; as part of a supportive bundle (saline irrigation, antipyretics, delayed antibiotic strategy), it can align with stewardship goals 52. The key is clear diagnostic criteria, time-limited courses, and patient education to prevent misuse.
Allergic Rhinitis: Antihistamines with Plant Allies: For patients bothered by antihistamine sedation or dryness, some studies report Butterbur (petasins) offering antihistamine-like benefit without sedation, allowing its adjunctive use in seasonal peaks 53.
Nigella sativa has shown histamine-release inhibition and nasal symptom improvement in early trials, supporting a role as an add-on during high-allergen periods 54.
Practical integration: Who, When, How:
Who Benefits?: Patients with mucus-dominant phenotypes (chronic bronchitis), frequent URTIs, or steroid-sensitive asthma profiles may be most likely to notice additive gains 55.
When to Start?: Stable patients on optimized inhaler technique and guideline-based therapy who still have residual symptoms; or during early URTI windows to reduce severity/duration 56.
How to Dose?: Use standardized extracts (e.g., EPs 7630 for Pelargonium; quantified saponins in ivy), time-limited courses for acute conditions, and documented, consistent brands to ensure batch-to-batch reliability 57.
Safety Checks: Screen for interactions (e.g., curcumin/ginger with anticoagulants), comorbidities (hypertension/CKD with licorice), and pregnancy/lactation status. Monitor BP, potassium if licorice is used; counsel on warning signs 58.
Shared Decision-making: Align choices with patient values, costs, cultural preferences, and access. Provide written plans specifying when to stop botanicals (e.g., new wheeze, fever escalation) and when to seek care 59.
Documentation & Audit: Chart the specific product, dose, start/stop dates, and outcomes; review at follow-up to continue only what shows clear, patient-relevant benefit 60.
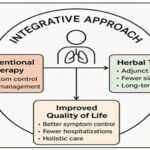
FIG. 3: INTEGRATED TREATMENT APPROACH IN RESPIRATORY DISEASES
The above figure shows how herbal therapies can complement conventional pharmacological treatments to improve outcomes in asthma, COPD, and bronchitis. The figure highlights synergistic roles (e.g., symptom relief, reduced side effects, and improved quality of life).
DISCUSSION: The comparative evaluation of herbal medicines and conventional drugs in respiratory diseases underscores a critical tension in modern healthcare: balancing rapid, evidence-based relief with long-term safety and accessibility. Conventional therapies such as corticosteroids, bronchodilators, and mucolytics remain the cornerstone of treatment, backed by robust clinical trials and international guidelines. Their ability to provide fast symptom control is unmatched, yet their chronic use is often associated with adverse effects including immunosuppression, metabolic disturbances, and dependence.
Herbal medicines, conversely, offer promising pharmacological properties anti-inflammatory, antioxidant, bronchodilatory, and immunomodulatory effects derived from agents like ginseng, licorice, and curcumin. These therapies are culturally accepted, often more affordable, and may improve patient adherence. However, their limitations lie in lack of large-scale randomized trials, variability in preparation, and inconsistent dosing standards.
Importantly, the discussion suggests that an integrative model of care where herbal therapies complement, rather than replace, conventional drugs may bridge these gaps. This would require rigorous quality control, standardized formulations, and stronger clinical evidence. By embracing both scientific rigor and traditional knowledge, respiratory medicine could evolve toward safer, patient-centered treatment paradigms.
CONCLUSION: The management of respiratory diseases continues to depend heavily on conventional drugs for their well-documented efficacy and rapid onset of action. Nevertheless, concerns over long-term safety, side effects, and patient affordability highlight the need for alternative or complementary options. Herbal medicines present a viable adjunct due to their broad spectrum of biological activities and favourable safety profiles when used appropriately. While promising, the current limitations of herbal therapies such as insufficient clinical validation and lack of regulatory oversight must be addressed before they can be integrated widely into clinical guidelines. Encouragingly, emerging research demonstrates that combining herbal therapies with conventional treatments may not only enhance outcomes but also reduce adverse effects.
Ultimately, neither approach alone is sufficient to address the global burden of respiratory diseases. The future of respiratory care lies in a balanced, evidence-based integration of both systems, offering patients safer, more effective, and accessible therapeutic strategies. This shift requires collaboration among clinicians, researchers, and policymakers to ensure that herbal and conventional medicines together create a more holistic standard of respiratory care.
ACKNOWLEDGEMENT: The author thanks academic mentors for their helpful guidance. Gratitude is also given to peers for their support during this work. Their encouragement made the completion of this review possible.
Financial Support and Sponsorship: Nil
CONFLICTS OF INTEREST: There are no conflicts of interest.
REFERENCES:
- Matthys H, Lizogub VG, Malek FA and Kieser M: "Efficacy and tolerability of EPs 7630 tablets in patients with acute bronchitis: a randomised, double-blind, placebo-controlled dose-finding study with a herbal drug preparation from Pelargonium sidoides." Current Medical Research and Opinion 2010; 26(6): 1413–1422. DOI: 10.1185/03007991003798463.
- Agbabiaka TB, Guo R and Ernst E: "Pelargonium sidoides for acute bronchitis: a systematic review and meta-analysis." Phytomedicine 2008; 15(5): 378–385. DOI: 10.1016/j.phymed.2007.11.023.
- Careddu D and Pettenazzo A: "Pelargonium sidoides extract EPs 7630: a review of its clinical efficacy and safety for treating acute respiratory tract infections in children." International Journal of General Medicine 2018; 11: 91–98. DOI: 10.2147/IJGM.S154198.
- Mansfeld HJ: "Phytopharmakon Umckaloabo bei chronisch obstruktiven Atemwegserkrankungen." Pneumologie 2000; 54(4): 239–246.
- Matthys H and Funk P: "EPs 7630 improves acute bronchitic symptoms and shortens time to remission. Results of a randomised, double-blind, placebo-controlled, multicentre trial." Planta Medica 2008; 74(6): 686–692. DOI: 10.1055/s-2008-1074519.
- Mansfeld HJ and Beuscher N: "Phytotherapeutics in chronic bronchitis: clinical evidence with Pelargonium extract." Allergo Journal International 2001; 10(3): 122–128.
- Holzinger F and Chenot JF: "Systematic review of clinical trials assessing the effectiveness of ivy leaf (Hedera helix) for acute upper respiratory tract infections." Evidence-Based Complementary and Alternative Medicine 2011, Article ID 382789, 2011.DOI: 10.1155/2011/382789.
- Mansfeld HJ and Harling M: "Efficacy of ivy leaf extract in the treatment of chronic-productive cough." Phytomedicine 1997; 4(4): 289–296.
- Stauss-Grabo M, Atiye S, Warnke A, Wedemeyer RS, Donath F and Blume HH: "Observational study on the tolerability and safety of film-coated tablets containing ivy leaves dry extract (Prospan®) in the treatment of colds accompanied by coughing," Phytomedicine 2011; 18(6): 433–436. DOI: 10.1016/j.phymed.2010.11.009.
- Holzinger F and Koetter U: "The use of ivy leaf extract in respiratory diseases." Evidence-Based Complementary and Alternative Medicine 2007; 1: 17–24. DOI: 10.1155/2007/191756
- Hsiang CY, Wu SL, Cheng SE and Ho TY: "Ginger and respiratory symptoms: clinical and experimental evidence." Journal of Ethnopharmacology 2013; 145(1): 146–153. DOI: 10.1016/j.jep.2012.10.029.
- Kord-Varkaneh H, Nazary-Vannani A and Shab-Bidar S: "Effect of ginger supplementation on lung function and respiratory symptoms: a systematic review." Nutrition Journal 2019; 18(1): 40. DOI: 10.1186/s12937-019-0465-6.
- Ammon HP, "Boswellic acids in chronic inflammatory diseases." Phytomedicine 2010; 17(11): 862–867. DOI: 10.1016/j.phymed.2010.04.001.
- Gupta I, Parihar A, Malhotra P, Gupta S, Lüdtke R, Safayhi H and Ammon HP: "Effects of Boswellia serrata gum resin in patients with bronchial asthma: results of a double-blind, placebo-controlled, 6-week clinical study," European Journal of Medical Research 1998; 3(11): 511–514.
- Siddiqui MZ: "Boswellia serrata, a potential anti-inflammatory agent: an overview." Indian Journal of Pharmaceutical Sciences 2011; 73(3): 255–261. DOI: 10.4103/0250-474X.100228.
- Aggarwal BB, Harikumar KB: "Potential therapeutic effects of curcumin, the anti-inflammatory agent from turmeric, against chronic diseases." International Journal of Biochemistry & Cell Biology 2009; 41(1): 40–59. DOI: 10.1016/j.biocel.2008.10.008.
- Kurup VP and Barrios CS: "Immunomodulatory effects of curcumin in allergy and asthma." Molecular Nutrition & Food Research 2008; 52(9): 1031–1039. DOI: 10.1002/mnfr.200700280.
- Gupta A, Srivastava A and Saha S: "Clinical evaluation of curcumin in bronchial asthma". Indian Journal of Physiology and Pharmacology 2011; 55(1): 63–71.
- Prasad S, Gupta SC, Tyagi AK and Aggarwal BB: "Curcumin, a component of golden spice: from bedside to bench and back." Biotechnology Advances 2014; 32(6): 1053–1064. DOI: 10.1016/j.biotechadv.2014.09.001.
- Li-Weber M: "New therapeutic aspects of flavones: the anticancer properties of apigenin." International Journal of Oncology 2009; 36(6): 1337–1349. DOI: 10.3892/ijo_00000476.
- Kawai M, Hirano T, Higa S, Arimitsu J, Maruta M and Kuwahara Y: "Flavonoids and asthma: immunomodulation of cytokine production and effect on airway inflammation." Allergology International 2007; 56(2): 113–123. DOI: 10.2332/allergolint.R-06-135.
- Holgate ST, Polosa R: "The mechanisms, diagnosis, and management of severe asthma in adults." The Lancet 2006; 368(9537): 780–793. DOI: 10.1016/S0140-6736(06)69288-X.
- Bateman ED, Hurd SS, Barnes PJ, Bousquet J, Drazen JM and FitzGerald M: "Global strategy for asthma management and prevention: GINA executive summary." European Respiratory Journal 2008; 31(1): 143–178. DOI: 10.1183/09031936.00138707.
- O'Byrne PM, Pedersen S, Lamm CJ, Tan WC, Busse WW and Drazen JM: "Severe exacerbations and decline in lung function in asthma." American Journal of Respiratory and Critical Care Medicine 2009; 179(1): 19–24. DOI: 10.1164/rccm.200805-818OC.
- Pauwels RA, Lofdahl CG, Postma DS, Tattersfield AE, O’Byrne P and Barnes PJ: "Effect of inhaled formoterol and budesonide on exacerbations of asthma." The New England Journal of Medicine 1997; 337(20): 1405–1411. DOI: 10.1056/NEJM199711133372003.
- Calverley PM, Anderson JA, Celli B, Ferguson GT, Jenkins C and Jones PW: "Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease." The New England Journal of Medicine 2007; 356(8): 775–789. DOI: 10.1056/NEJMoa063070.
- Cazzola M, Matera MG and Donner CF: "Inhaled β2-adrenoceptor agonists: cardiovascular safety in patients with obstructive lung disease." Drugs 2005; 65(12): 1595–1610. DOI: 10.2165/00003495-200565120-00003.
- Barnes PJ: "Inhaled corticosteroids." Pharmacology & Therapeutics 1998; 58(1): 1–34. DOI: 10.1016/S0163-7258(00)00020-6.
- Rabe KF, Adachi M, Lai CK, Soriano JB, Vermeire PA, Weiss KB and Weiss ST: "Worldwide severity and control of asthma in children and adults: the Global Asthma Insights and Reality surveys." Journal of Allergy and Clinical Immunology 2004; 114(1): 40–47. DOI: 10.1016/j.jaci.2004.03.029.
- Suissa S, Ernst P, Benayoun S, Baltzan M and Cai B: "Low-dose inhaled corticosteroids and the prevention of death from asthma." The New England Journal of Medicine 2000; 343(5): 332–336. DOI: 10.1056/NEJM200008033430501.
- Suissa S, Patenaude V, Lapi F and Ernst P: "Inhaled corticosteroids in COPD and the risk of serious pneumonia." Thorax 2013; 68(11): 1029–1036. DOI: 10.1136/thoraxjnl-2012-202872.
- Gibson PG and Powell H: "Written action plans for asthma: an evidence-based review of the key components." Thorax 2004; 59(2): 94–99. DOI: 10.1136/thorax.2003.011858.
- Brusselle GG, Joos GF and Bracke KR: "New insights into the immunology of chronic obstructive pulmonary disease." The Lancet 2011; 378(9795): 1015–1026. DOI: 10.1016/S0140-6736(11)60988-4.
- Singh D, Agustí A, Anzueto A, Barnes PJ, Bourbeau J and Celli RB: "Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease: the GOLD science committee report 2019." European Respiratory Journal 53(5): article 1900164, 2019.DOI: 10.1183/13993003.00164-2019.
- Global Initiative for Asthma (GINA), "Global Strategy for Asthma Management and Prevention," 2023 Update. URL: https://ginasthma.org (accessed 26 Nov. 2025).
- Global Initiative for Chronic Obstructive Lung Disease (GOLD), "Global Strategy for Prevention, Diagnosis and Management of COPD," 2023 Report. URL: https://goldcopd.org (accessed 26 Nov. 2025).
- Bousquet J, Mantzouranis E and Cruz AA: Uniform definition of asthma severity, control, and exacerbations. Journal of Allergy and Clinical Immunology 2010; 126(5): 926–938. DOI: 10.1016/j.jaci.2010.07.019
- Pauwels RA, Buist AS, Calverley PM, Jenkins CR and Hurd SS: GOLD Scientific Committee." Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease," American Journal of Respiratory and Critical Care Medicine 2001; 163(5): 1256–1276. DOI: 10.1164/ajrccm.163.5.2101039.
- Wedzicha JA and Seemungal TA: "COPD exacerbations: defining their cause and prevention." The Lancet 2007; 370(9589): 786–796. DOI: 10.1016/S0140-6736(07)61382-8.
- Barnes PJ: "The pharmacological properties of tiotropium." Chest 2000; 117(2): 63–66. DOI: 10.1378/chest.117.2_suppl.63S.
- Tashkin DP, Celli B, Senn S, Burkhart D, Kesten S and Menjoge S: A 4-year trial of tiotropium in chronic obstructive pulmonary disease. New England Journal of Medicine 2008; 359(15): 1543–1554. DOI: 10.1056/NEJMoa0805800.
- Vogelmeier C, Hederer B, Glaab T, Schmidt H, Rutten-van Mölken M and Beeh KM: Tiotropium versus salmeterol for the prevention of exacerbations of COP. "New England Journal of Medicine 2011; 364(12): 1093–1103. DOI: 10.1056/NEJMoa1012926.
- Singh S, Loke YK and Furberg CD: Inhaled anticholinergics and risk of major adverse cardiovascular events in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. JAMA, 2008; 300(12): 1439–1449. DOI: 10.1001/jama.300.12.1439.
- Fabbri LM and Hurd SS: "Global Strategy for the Diagnosis, Management and Prevention of COPD: 2003 update." European Respiratory Journal 2003; 22(1): 1–2. URL: https://goldcopd.org
- Tashkin DP and Ferguson GT: "Combination bronchodilator therapy in the management of chronic obstructive pulmonary disease." Respiratory Research 2013; 14(49). DOI: 10.1186/1465-9921-14-49.
- Calverley PM, Anderson JA, Celli UM, Ferguson GT, Jenkins C and Jones PW: Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. The Lancet 2009; 374(9691): 685–694. DOI: 10.1016/S0140-6736(09)61255-1.
- Martinez FJ, Calverley PM, Goehring U, Brose M, Fabbri LM and Rabe KF: "Effect of roflumilast on exacerbations in patients with severe chronic obstructive pulmonary disease uncontrolled by combination therapy (REACT): a multicentre randomised controlled trial." The Lancet 385(9971): 2015; 857–866. DOI: 10.1016/S0140-6736(14)62461-9.
- Rennard SI, Calverley PM, Goehring UM, Bredenbreoker D and Martinez FJ: Reduction of exacerbations by the PDE4 inhibitor roflumilast—the importance of defining different subsets of patients with COPD. Respiratory Research 2011; 12(18): DOI: 10.1186/1465-9921-12-18.
- Lipworth BJ: "Emerging role of leukotriene receptor antagonists in asthma." Thorax 1999; 54(3): 213–219: 10.1136/thx.54.3.213.
- Ducharme FM and Hicks GC: "Anti-leukotriene agents compared to inhaled corticosteroids in the management of recurrent and/or chronic asthma in adults and children." Cochrane Database of Systematic Reviews 2002; 1: 2314. DOI: 10.1002/14651858.CD002314.
- Busse W, Raphael GD, Galant S, Kalberg C, Goode-Sellers S and Srebro S: "Montelukast, a leukotriene-receptor antagonist, for the treatment of mild asthma and exercise-induced bronchoconstriction." New England Journal of Medicine 1999; 341(11): 747–752. DOI: 10.1056/NEJM199909093411102.
- Leff JA: "Clinical efficacy and safety of leukotriene receptor antagonists in asthma." American Journal of Respiratory Medicine 2001; 1(1): 9–16. DOI: 10.1007/PL00012132.
- Barnes PJ: "Theophylline: pharmacology and therapeutic use in asthma and COPD." American Journal of Respiratory and Critical Care Medicine 2013; 188(8): 901–906. DOI: 10.1164/rccm.201302-0388PP.
- Cazzola M, Page CP, Calzetta L and Matera MG: "Pharmacology and therapeutics of bronchodilators." Pharmacological Reviews 2012; 64(3): 450–504. DOI: 10.1124/pr.111.005975.
- Yao X and Fredholm BB: Adenosine A2A receptor agonists and antagonists in respiratory diseases. Handbook of Experimental Pharmacology 2009; 193: 363–398. DOI: 10.1007/978-3-540-89615-9_12.
- George RB, Light RW, Matthay RA and Matthay MA: "Theophylline in the treatment of asthma." Chest 1984; 86(1): 90–93. DOI: 10.1378/chest.86.1.90.
- Pauwels RA, Lofdahl CG, Postma DS, Tattersfield AE, O'Byrne P and Barnes PJ: Combined salmeterol and fluticasone in the treatment of asthma: a randomised controlled trial. The Lancet 1997; 350(9091): 2053–2059. DOI: 10.1016/S0140-6736(97)07522-5.
- Lötvall J, Malmqvist E, Larsson S, Palmqvist M, Arvidsson P and Palmqvist M: Pharmacological treatment of asthma and COPD: focus on combination therapy. Respiratory Medicine 2002; 96(9): 683–697. DOI: 10.1053/rmed.2002.1355.
- Rabe KF, Atienza T, Magyar P, Larsson P, Jorup C and Lalloo UG: Effect of budesonide in combination with formoterol for reliever therapy in asthma exacerbations: a randomised controlled trial. The Lancet 2006; 368(9537): 744–753. DOI: 10.1016/S0140-6736(06)69271-0.
- O'Byrne PM, Bisgaard H, Godard PP, Pistolesi M, Palmqvist M and Zhu Y: Budesonide/formoterol combination therapy as both maintenance and reliever medication in asthma. American Journal of Respiratory and Critical Care Medicine 2005; 171(2): 129–136. DOI: 10.1164/rccm.200407-884OC.
How to cite this article:
Sireesha PDG, Swetha P, Sidhikha SA and Divya T: Comparing the efficacy and safety of herbal medicines vs conventional drugs in respiratory diseases. Int J Pharmacognosy 2026; 13(4): 287-96. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(4).287-96.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
287-296
845 KB
1
English
IJP
P. Devi Guna Sireesha *, P. Swetha, Shaik Ayesha Sidhikha and Tokala Divya
Department of Pharmacology, Nimra College of Pharmacy, Ibrahimpatnam, Nimra Nagar, Vijayawada, Andhra Pradesh, India.
drdevipcology@gmail.com
29 March 2026
11 April 2026
19 April 2026
10.13040/IJPSR.0975-8232.IJP.13(4).287-96
30 April 2026