COMPARATIVE STUDY OF THE EFFECTS OF DILTIAZEM VERSUS ALPHA TOCOPHEROL ON RETINAL DEGENATION IN RABBITS
HTML Full TextCOMPARATIVE STUDY OF THE EFFECTS OF DILTIAZEM VERSUS ALPHA TOCOPHEROL ON RETINAL DEGENERATION IN RABBITS
Bassant M. M. Ibrahim *, Abeer A. A. Salama, Heba M. I. Abdallah and Gehad A. Abdel Jaleel
Department of Pharmacology, National Research Centre, Dokki, Giza, Egypt.
ABSTRACT: Disturbance in retinal blood supply can have an effect on the supply of oxygen and the substrates which leads to retinal ischemia, the result is irreversible, ischemic and apoptotic cascades resulting in cell death. The present study was conducted to study the protective effects of diltiazem and vitamin E "alpha-tocopherol," against degenerative ischaemic retinal changes induced by Middle Cerebral Artery Occlusion (MCAO) in rabbits. Animals were classified into normal control group which received tap water throughout the experiment, positive control where induction of retinal ischemia was done by MCAO and three treated groups: a group received diltiazem (15 mg/kg), another received vitamin E (200 mg/kg body), and a third received both diltiazem and vitamin E for three successive weeks followed by induction of retinal ischemia by MCAO for all treated groups. When assessment of the protective effects of diltiazem and vitamin E and their combination, was done by clinical examination of the eyes, measurement of reduced glutathione, malondialdehyde and nitric oxide levels, electroretinography and histopathologically, it showed that both diltiazem and vitamin E and their combination had protective effects against degenerative ischaemic retinal changes and retinitis pigmentosa in rabbits.
| Keywords: |
Retina, Diltiazem, Tocopherol, Degeneration, Middle cerebral artery
INTRODUCTION: Retinal ischemia is a common cause of irreversible visual impairment and even loss of vision. Retinal ischemia takes place when the blood supply to the retina is insufficient or doesn't meet the metabolic needs of the retina. Since the retina has a high metabolic rate, therefore, any disturbance in its blood supply can have an effect on the supply of oxygen and the substrates which leads to retinal ischemia, that's why if no treatment is given to repair this imbalance, the result is irreversible, ischemic and apoptotic cascades resulting in cell death.
Moreover retinal disorders characterized by a progressive loss of rod and cone photoreceptors and eventual atrophy of the entire retinal lead to defective light to dark and dark to light adaptation which is known as night blindness (nyctalopia), a symptom of retinitis pigmentosa 1. The retina is widely used for studying the pathophysiology of ischemia and mechanism of damage triggered by ischemia and excitotoxicity. Occlusion of the middle cerebral artery (MCA) leads to retinal ischemia. Middle cerebral artery occlusion (MCAO) is a vascular model of retinal ischemia that mimics human vision loss. This model occludes arterial blood flow intraluminally. The ophthalmic artery that mainly supplies the inner retina originates from the internal carotid artery proximal to the origin of the middle cerebral artery. That's why it is expected that MCAO subsequently obstructs blood flow in the ipsilateral retina.
This model is non-invasive regarding the eye and does not cause blood-retinal barrier disruption or any mechanical damage to the retina. Therefore, MCAO is a more relevant model for studying any changes in the retina and testing the efficacy of therapeutic strategies in cases of retinal ischemia 2.
Calcium (Ca2+) channel blockers play an important role in the management of cardiovascular diseases. They block membrane-bound calcium channels and inhibit the calcium influx, causing relaxation of smooth muscle cells in blood vessels' walls, so lead to a decrease in vascular tone, and an improvement in blood flow 3. Both verapamil and diltiazem are a calcium channel blockers which showed relaxation of cat opthalmocilliary artery ring segments in-vitro 4.
There is considerable evidence that oxidative stress causes an increase in the incidence of disease; that's why consumption of foods rich in antioxidants can potentially scavenge excess radicals and plays an important role in the prevention of many diseases 5. Vitamin E is the major lipid-soluble antioxidant in the cell antioxidant defense system and is exclusively present in diet 6.
Rabbits represent a useful animal model for the study of the pathophysiology and new treatments of retinal degeneration as a result of ischemia because they have large eyes 7. That's why they were chosen to be the animal model in the present study.
The objective of the Study: This study was conducted to study the protective effects of either a selected vasodilator drug from calcium channel blocker class which is diltiazem and Vitamin E "alpha tocopherol" which is an antioxidant present in the diet, and their combination against degenerative ischaemic retinal changes and retinitis pigmentosa induced by MCAO in rabbits.
MATERIALS AND METHODS: The experiment was done in 2016, in the Pharmacology department of National research center and the Institute of Ophthalmology.
Materials:
Animals Used: Adult Giza white rabbits (weighing 2500 - 2750 gm) were used. They were obtained from the animal house of Institute of Ophthalmology, and were individually housed in metallic cages in laminar flow cages maintained at 22 ± 2 °C, 50 -60% relative humidity, under a 12 h light: 12 h dark cycle and in well-ventilated rooms, under hygienic conditions. Animals were given water ad libitum and fed with green leafy vegetable available from the local market. Animals were cared for by "Principles of Laboratory Animal Care" 8 and were reviewed and approved by the ethics committee of National Research Centre, Dokki, Egypt, before starting the experiment, it had certificate registration number: 15153.
Chemicals and Drugs Used: Ketamine/xylazine solution, formaldehyde, tropicamide, phenylephrine hydrochloride, and atropine sulfate were purchased from Sigma Aldrich Chemical Co., USA. Diltiazem was purchased from EPICO, Egypt (Egyptian International Pharmaceutical Industries Company). Vitamin E "alpha tocopherol" was purchased from SD. Fine Chem. Limited, Mumbai. Kits for the assessment of sera level of malondialdehyde (MDA), Nitric oxide (NO) metabolites, and reduced glutathione (R-GSH) were purchased from Biodiagnostic Company, Egypt.
Apparatus Used: Electroretinogram apparatus, Keeler Fison binocular indirect ophthalmoscope, Cooling centrifuge, Spectrophometer, Enzyme-linked immunosorbent assay (ELISA) apparatus and Light microscope.
Methods:
Experimental Design: Animals were classified into five groups consisting of six rabbits each as follows: The first group was normal rabbits which received distilled water throughout the experiment, the second group was positive control which received distilled water for three successive weeks followed by induction of retinal ischemia by middle cerebral artery occlusion (MCAO),the third group received diltiazem solutions which were prepared by adding diltiazem in a dose of 15 mg/kg body weight to distilled water (10 ml) and stirring for 1 h 9, and then administered orally through a catheter for three successive weeks followed by induction of retinal ischaemia by MCAO , the fourth group received vitamin E alpha-tocopherol orally in a dose of 200 mg/kg body weight 10, for three successive weeks followed by induction of retinal ischemia by MCAO and the fifth group received orally a combination of both diltiazem in a dose of 15 mg/kg body weight and alpha-tocopherol in a dose of 200 mg/kg body weight for three successive weeks followed by induction of retinal ischaemia by MCAO.
Method of Middle Cerebral Artery Occlusion (MCAO): After three successive weeks of administration of each drug, all animals in second to fifth groups were subjected to MCAO according to 11. The animals were kept fasting for 12 hr then anesthetized with ketamine/xylazine solution 35/5 mg/kg intramuscularly 12, then intubated orotracheally and mechanically ventilated with 0.8% halothane in a mixture of 70% N2O and 30% O2 to maintain normal arterial blood gases. MCAO was done on the right side, by insertion of 1-0 surgical nylon monofilament (Ethicon) with its tip rounded by heat through the external carotid artery (ECA) 13. Briefly, the right cervical carotid bifurcation was exposed, and the internal carotid artery (ICA) was distally dissected free from the adjacent tissue under the operating microscope.
The ECA was then ligated and cut distal to the superior thyroid artery after a micro clip was temporarily placed on the common carotid artery (CCA) and ICA. The filament was gently advanced through the ECA until there was a perceptible resistance. The filament was secured at the ECA by a tight ligature and a micro chip. There was a modification done for the previously mentioned method referred to 11, where thinner nylon monofilaments (3-0 and 2-0) were used in two positive control rabbit groups, in a trial to induce retinal changes as a result of MCAO, but their effects were negligible so thicker filament (1-0) was used in a third positive control rabbit group, and it produced the desired effect on the retina in the present experiment, and this was the selected thickness for induction of retinal changes by MCAO in all the three treated groups. Then each animal was kept in an isolated, sterile cage under aseptic conditions to avoid infection.
Ophthalmic Examination: Before starting the experiment and before doing MCAO, the external ocular examination was done and also fundus examination was performed using Keeler Fison binocular indirect ophthalmoscope for each animal in all four groups.
After doing MCAO, the external ocular examination was done every other day, and fundus examination was performed using Keeler Fison binocular indirect ophthalmoscope weekly until ischaemic retinal changes were observed in the right eye of the positive control group, four weeks after MCAO. Ischaemic retinal changes severity was given the following scores according to the degree of pallor: 0 = no pallor, 1 = mild pallor, 2 = moderate pallor, 3 = severe pallor.
Electroretinography Recording Methods: When ischaemic retinal changes of the right eye were observed in the positive control group, Four weeks after MCAO, electr-retinogram (ERG) was done for both eyes of all animals in all four groups according to 14. Pupils were dilated with 0.8% tropicamide, 5.0% phenylephrine hydrochloride, and 1% atropine sulfate. Each trial consisted of 100 consecutive, computer-averaged stimulations. Two trials (i.e., a total of 200 stimulations) were made for each test and each period.
The average amplitude of the two trials is the reported value. Noise recordings were obtained by identical methods, except that the light or electrical stimulus did not reach the animal. Ambient illumination was 480 lumen / ft2 (i.e. photopic condition). For the ERG, recordings were made under photopic conditions because we wanted the same recording conditions before and after surgery, and surgery required bright light. The corneal recording electrode was a modified Machemer magnifying vitrectomy lens (Storz, Ophthalmic Co.) 15. The common reference was on an ear. The retina was stimulated with a Grass PS22 photo-stimulator (Mini Ganzfeld, model PSD22D) positioned 2 cm above the cornea.
The photo-stimulator flash intensity setting was 2 (equal to 2.75 lumen s/ft2, for a 10 µs flash), and flash frequency was 2 Hz. The b-wave amplitude was measured from the negative peak of the a-wave to the positive peak of the b-wave. Percent (%) change of the difference between b-wave amplitude in left assumed normal eye and right assumed diseased eye with degenerative ischaemic retinal changes was calculated for all rabbits in all groups. Statistical analyses for significant % change difference was done.
Biochemical Parameter Estimation: Four weeks after MCAO, at the end of the in vivo experiment after electroretinogram was done all animals in all five groups were kept fasting for 18 h, then they were anesthetized with ketamine/xylazine solution 35/5 mg/kg intramuscularly 12, and then 5 ml of blood was drawn from the auricular marginal vein, using a 25-gauge needle. Blood samples were allowed to stand for 10 min at room temperature then centrifuged at 4 ºC using cooling centrifuge (Laborezentrifugen, 2k15, Sigma, Germany) at 3000 r. p. m for 10 min and sera were separated for the assessment of sera level of malondialdehyde (MDA), Nitric oxide (NO) metabolites, and reduced glutathione (R-GSH) according to 16-19, using commercially available kits (Biodiagnostic, Egypt).
Histopathologic Examination: After the withdrawal of blood samples all animals from different groups, the animals were sacrificed under general anesthesia then their globes were dissected. Autopsy samples were taken from the eye of rabbits in different groups and fixed in 10% formol saline for twenty four. Washing was done in tap water then serial dilutions of alcohol (methyl, ethyl and absolute ethyl) were used for dehydration. Specimens were cleared in xylene and embedded in paraffin at 56 degrees in a hot air oven for twenty-four h. Paraffin bees wax tissue blocks were prepared for sectioning at 4 microns thickness by slidgemicrotome. The obtained tissue sections were collected on glass slides, deparaffinized, stained by hematoxylin and eosin stain for examination through the light electric microscope 20.
Statistical Analysis: Comparison between means was carried out using one-way analysis of variance (ANOVA) followed by Tukey Kramer multiple comparison test. P ˂ 0.05 was considered as being significant in all types of statistical tests. Graph pad prism software (version 6) was used to carry out all statistical tests.
RESULTS AND DISCUSSION:
Results of Ophthalmic Examination and Electro-Retinogram (ERG): Retinal ischemia is a common cause for visual loss. It takes place when the blood supply of the retina is not sufficient to meet its demands. Retinal degeneration (rd) in rodents is a good model to evaluate treatment of retinitis pigmentosa; because the pattern of photoreceptor loss is similar to that in affected humans where rod cell death is followed by cone cell degeneration 21. That's why rabbits were used in the present study.
In the present study ophthalmic examination was done once for both rabbit's eyes of each rabbit in all groups, before starting the experiment at zero time, then after administration of medication for three successive weeks and it revealed: Normal adnexa, anterior segments, fundi, normal pupillary reflexes, and normal ocular movements. When the ophthalmic examination was done after induction of MCAO, it revealed: Normal adnexa, anterior segments, fundi, normal pupillary reflexes and normal ocular movements of both eyes of all rabbits in the normal control group.
On the other hand, it revealed: Normal adnexa, anterior segments, but there was severe pallor of the retina of the right eyes only of the positive control group which was significantly more than the normal control and the three treated groups, this pallor started to be significant four weeks after MCAO. The pallor was moderate to mild in the right eyes only of the groups treated with diltiazem (15 mg/kg) alone, vitamin E α tocopherol (200 mg/kg) alone, and group treated with combination of both diltiazem and vitamin E α tocopherol, but it was significantly more than the normal control group at the same onset. There was also loss of light reflexes of the right eyes associated with limited ocular movements of the four groups (positive control and the three treated groups) compared to left normal eyes of all rabbits.
The effect of MCAO may be due to pro-apoptotic and anti-apoptotic genes that include p53, which is a DNA-binding transcription factor and is involved in DNA damage and repair, as it was reported that ischemia leads to increase in expression of p53, which causes selective vulnerability of inner retina to transient ischemia 22.
The effect of diltiazem on pallor which is a sign of ischemia is expected due to its action as a Ca2+ channel blocker. However, the effect of Vitamin E is may be due to what has been suggested in previous studies, that vitamin E supplementation in doses of (200 - 400 mg/d) to humans may be appropriate to reduce tissue injury arising from ischemia and reperfusion during surgery and delay cataract development 23. Results of the degree of pallor scored on fundus examination are expressed in Table 1.
TABLE 1: EFFECT OF PRETREATMENT WITH DILTIAZEM (15 mg/kg) AND VITAMIN E α TOCOPHEROL (200 mg/kg) AND THEIR COMBINATION FOLLOWED BY MCAO, ON FUNDUS EXAMINATION AND ELECTROPHYSIOLOGICAL CHANGES IN THE RETINAE OF RABBITS
| Group | Severity of retinal
pallor (a sign of degenerative ischaemic retinal changes) |
Electro retinograghy | ||
| Left eye | Right eye | % change of
b wave amplitude |
||
| Normal control | ------------ | 3.25 ± 0.03 | 3.28 ± 0.03 | 0.67 ± 0.2 |
| Positive control | 2.66 ± 0.21a | 3.23 ± 0.04 | 1.85 ± 0.03 | 42.59 ± 1.48a |
| Diltiazem (15 mg/kg) | 1.16 ± 0.16ab | 2.92 ± 0.04 | 2.62 ± 0.02 | 2.62 ± 0.02bcd |
| Vitamin E α tocopherol (200 mg/kg) | 1.66 ± 0.21ab | 3.92 ± 0.04 | 2.5 ± 0.03 | 36.06 ± 1.49a |
| Diltiazem (15mg/kg) + Vitamin E α tocopherol (200mg/kg) | 1.83 ± 0.16ab | 2.63 ± 0.01 | 1.67 ± 0.01 | 36.56 ± 0.62a |
Results are expressed as means of Severity of retinal pallor ± SE and % change of b wave amplitude between left normal and diseased eyes ± SE. n = 6, significance at p˂0.05.(a) significantly different from normal control (b) significantly different from positive control, (c) significantly different from group treated with Vitamin E α tocopherol (200 mg/kg), (d) significantly different from group treated with combination of diltiazem (15 mg/kg) and Vitamin E α tocopherol (200 mg/kg).
Four weeks after MCAO, electroretinogram (ERG) was done, and the difference in amplitude of b-wave in left normal eye and right assumed eye to have degenerative ischaemic retinal changes was calculated, and the percentage of this change was calculated. There was a significant decrease in % change in all groups when compared to the positive control group. As a result of ischemia, the voltage-gated Ca2+ channels are opened, leading to a rise in intracellular levels of Ca2+ ions, which inhibit the mitochondrial metabolism and lead to excitotoxicity 24 which affects most cells. This is consistent with the effect of MCAO in our study, on the electrophysiologic changes manifested by a marked reduction of b-wave amplitude in the diseased side due to effect of ischemia on photoreceptors.
Calcium channels in the retina, are mainly from the L-type that do not inactivate with time When animals were subjected to retinal degeneration (rd) by increasing the intraocular pressure, it reduced the cell count, these animals received L-type Ca2+ channel blocker, diltiazem injections which induced a less severe reduction in retinal cells; and was significant in the ganglion cell layer 25. It was also reported that diltiazem treatment rescued the functional photoreceptors in the rd mice 26. The effect of diltiazem on the b-wave ERG also indicated that it could enter the retina and permeate through the hematopoietic barrier, so it was concluded that diltiazem rescued photoreceptors and interfered with photoreceptor degeneration by blocking L-type calcium channels rather than by increasing blood flow 27.
This may contribute to an explanation for the results in our study attributed to the effect of diltiazem, as it's worth noting that there was significant reduction in % change difference between right diseased and left normal eyes in ERG done for rabbits when compared to positive control group, moreover there was significant decrease in % change in the group treated with diltiazem alone when compared to the group treated with vitamin E α tocopherolalone and the group treated with a combination of diltiazem and vitamin E α tocopherol. However, our electrophysiology results obtained from the ERG are in contradiction with the results of another study which didn't show any therapeutic effect of d-cis-diltiazem in a canine model of retinal degeneration 28. On the contrary, it was revealed that the diltiazem treated mice had markedly subnormal dark-adapted ERG responses, and the light-adapted ERG responses were similar in the waveform to the dark-adapted responses but smaller so it was rejected trying diltiazem for treatment of retinitis pigmentosa 29.
On the other hand vitamin E α tocopherol group and combination group showed a significant increase in % change when compared to the normal control group and diltiazem group. The effect of vitamin E α tocopherol as well as its combination with diltiazem in the present study is a significant protective effect as it showed a reduction in % difference between right diseased and left normal eyes when compared to the positive control group. This may be attributed to its protection against ischemia. Results of ERG for all groups are expressed in Table 1 and Fig. 1.
FIG. 1: 1(A) LEFT EYE OF NORMAL CONTROL, AMPLITUDE OF B WAVE IS 3.231mv. 1(B) RIGHT EYE OF NORMAL CONTROL, AMPLITUDE OF B WAVE IS 3.201mv. 1(C) LEFT NORMAL EYE OF POSITIVE CONTROL, AMPLITUDE OF B WAVE IS 3.069 mv. 1(D) DEGENERATIVE ISCHAEMIC RETINAL CHANGES OF RIGHT EYE OF POSITIVE CONTROL, AMPLITUDE OF B WAVE IS 1.85 mv. 1(E) LEFT NORMAL EYE OF RABBIT PRETREATED WITH DILTIAZEM (15 mg/kg), AMPLITUDE OF B WAVE IS 2.977 mv. 1(F) RIGHT EYE OF RABBIT PRETREATED WITH DILTIAZEM (15 mg/kg), AMPLITUDE OF B WAVE IS 2.649 mv. 1(G) LEFT NORMAL EYE OF RABBIT PRETREATED WITH VITAMIN E "ALPHA TOCOPHEROL" (200 mg/kg), AMPLITUDE OF B WAVE IS 3.93 mv. 1(H) RIGHT EYE OF RABBIT PRETREATED WITH VITAMIN E "ALPHA TOCOPHEROL" (200 mg/kg), AMPLITUDE OF B WAVE IS 2.507 mv. 1(I) LEFT EYE OF RABBIT PRETREATED WITH COMBINATION OF DILTIAZEM (15 mg/kg) AND VITAMIN E "ALPHA TOCOPHEROL" (200 mg/kg), AMPLITUDE OF B WAVE IS 2.644 mv. 1(J) RIGHT EYE OF RABBIT PRETREATED WITH COMBINATION OF DILTIAZEM (15 mg/kg) AND VITAMIN E "ALPHA TOCOPHEROL" (200 mg/kg), AMPLITUDE OF B WAVE IS 1.681 mv
Results of Biochemical Parameters: Since, the retina has a high metabolic rate, therefore deprivation of glucose and oxygen can harm the whole retina and causes irregular cell loss, as well as oxidative stress due to ischemia, the most affected cells, are the photoreceptor cells as they have the maximum oxidative metabolic rates 30. This explains our biochemical results of oxidative stress parameters as R-GSH, MDA and NO in rabbits subjected to MCAO to induce retinal changes mimicking retinal degeneration in humans revealed significant reduction in R-GSH, an increase in MDA levels in sera of rabbits subjected to MCAO in the positive control group without prior administration of Diltiazem, Vitamin E α tocopherol or combination of both therapies, when compared to normal control, and the three treated groups. The level of NO in the serum of positive control group significantly higher than that of the normal control and groups treated with vitamin E α tocopherol and combination of Diltiazem + vitamin E α tocopherol but not the group treated with diltiazem alone.
Moreover the level of R-GSH was significantly higher in the group treated with Diltiazem than groups treated with vitamin E α tocopherol and combination of Diltiazem + vitamin E α tocopherol, while MDA level was less and NO level was higher than the group treated with a combination of Diltiazem + vitamin E α tocopherol. The level of MDA in the group treated with vitamin E α tocopherol alone was significantly less, and the level of NO was significantly higher than the group treated with a combination of Diltiazem + vitamin E α tocopherol. Also, the antioxidant effect of diltiazem in the present study was in agreement with Anjaneyulu and Chopra (2005) 31, who reported in their study that administration of diltiazem to diabetic rats for eight weeks caused attenuation in oxidative stress along with increased NO levels.
Vitamin Eα tocopherol led to an increase in R-GSH and reduction in MDA levels, but the non-significant effect on NO levels, when compared to the positive control group. Since, vascular smooth muscle cells have tocopherol-binding abilities 32, therefore they may act as vitamin E receptors which adapt the molecule within the membrane for optimum antioxidant function, which in turn reduced the ischaemic effect of MCAO, this is explained by the fact that vitamin E α tocopherol is a phenolic antioxidant which is primarily located in cell and organelle membranes and exerts its maximum protective effect by protecting polyunsaturated fatty acids (PUFAs) and other components of cell membranes and low-density lipoprotein (LDL) from oxidation by free radicals 6.
In a study done in 2001, vitamin E α tocopherol significantly improved the impaired endothelium-dependent relaxations, additionally it decreased the contraction induced by phenylephrine and 5-hydroxytryptamine in diabetic rat rings, these results were explained by the effect of Vitamin E as it decreased membrane lipid peroxidation, these findings provide an explanation for the protective effect of vitamin E in our study 33. Results of biochemical parameters are expressed in Table 2.
TABLE 2: EFFECT OF PRETREATMENT WITH DILTIAZEM (15 mg/kg) AND VITAMIN E α TOCOPHEROL (200 mg/kg) AND THEIR COMBINATION, FOLLOWED BY MCAO ON OXIDATIVE STRESS BIOMARKERS: R-GSH, MDA AND NO LEVELS IN SERA OF RABBITS
| Group | R-GSH (µmol/ml) | MDA (nmol/ml) | NO (µmol/ml) |
| Normal control | 0.822 ± 0.01 | 21.75 ± 1.37 | 3.07 ± 0.12 |
| Positive control | 0.35 ± 0.01a | 45.02 ± 0.73a | 5.9 ± 0.04a |
| Diltiazem (15 mg/kg) | 0.707 ± 0.03abcd | 29.15 ± 1.24ad | 5.52 ± 0.01ad |
| Vitamin E α tocopherol
(200 mg/kg) |
0.49 ± 0.007ab | 28.53 ± 0.75ad | 5.35 ± 0.03abd |
| Diltiazem (15mg/kg) + Vitamin E α tocopherol (200mg/kg) | 0.485 ± 0.006ab | 36.7 ± 1.36a | 3.78 ± 0.17ab |
Results are expressed as means of R-GSH, MDA and NO levels in sera of rabbits ± SE.N = 6, significance at p˂0.05. (a) significantly different from normal control (b) significantly different from positive control, (c) significantly different from the group treated with Vitamin E α tocopherol (200 mg/kg), (d) significantly different from the group treated with a combination of diltiazem and vitamin E α tocopherol.
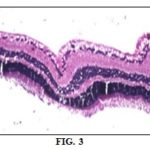
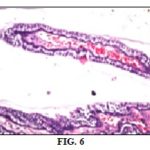
Results of Histopathologic Examination: Regarding histopathologic examination of rabbits' globes it was consistent with the results of the ophthalmic examination, electroretinogram and biochemical parameters as it revealed the following: normal control group globes revealed no histopathological alteration in the retina, choroid, and sclera as recorded in Fig. 2, while The retina of the positive control group had cellular degeneration Fig. 3 associated with congestion in the blood capillaries in the choroid and sclera Fig. 4. Histopathologic examination of globes of rabbits treated orally with diltiazem (15 mg/kg) before MCAO showed normal histological structure of the retina and choroid while the sclera showed congestion in the blood vessels Fig. 5, while examination of globes of rabbits treated orally with vitamin E α tocopherol (200 mg/kg) followed by MCAO showed that the retina, as well as the choroid, were intact histologically associated with edema of the sclera Fig. 6, on the other hand, there was no histopathological alteration in the retina, choroid, and sclera of rabbits treated with a combination of diltiazem (15 mg/kg) and Vitamin E α tocopherol (200 mg/kg) followed by MCAO Fig. 7.
Photomicrographs of retinae of Normal control group Fig. 5 showing no histopathological alteration in the retina choroid and sclera, positive control group Fig. 6 showing cellular degeneration Fig. 6 and congestion in the blood capillaries of the choroid and sclera Fig. 7 (H & EX200).
CONCLUSION: The results of the present study show that application of diltiazem as well as vitamin E has potential retinal neuroprotection in case of retinal ischemia and might offer the possibility to postpone the occurrence of blindness in people suffering retinitis pigmentosa. However, further clinical studies are recommended before it can be applied for human protection against retinal degeneration or retinitis pigmentosa.
FUNDING: The study was Funded by the internal house project unit in National Research Centre, Dokki, Giza, Egypt under the project grant no: P100513, entitled: Comparative Study of the effects of Vasodilators versus Antioxidants on Retinal Degeneration in Rabbits.
ACKNOWLEDGEMENT: The authors would like to thank Professor Dr. Adel Bakeer, Professor of Pathology for his effort in performing the histopathologic work in this study, and Dr. Kareem Abd El Hameed Risk, Assistant Professor of Ophthalmology, at Institute of Ophthalmology for performing the ophthalmology examination.
DECLARATION OF INTEREST: The authors declare that there is no conflict of interest regarding the present study.
REFERENCES:
- Heckenlively JR: RP syndromes. In: Heckenlively JR, ed. Retinitis Pigmentosa. Philadelphia. JB Lippincott 1988; 221-52.
- Gillipsie M and Akshay A: Animal Models of Retinal Ischemia. In: Brain Injury -Pathogenesis, Monitoring, Recovery and Management. Edited by Amit Agrawal 2012; 534.
- Harino S, Riva CE and Petrig BL: Intravenous nicardipine in cats increases optic nerve head but not retinal blood flow. Invest - Opthalmol Vis Sci 1992; 33: 2885-90.
- Winterkorn JM and Beckman RL: Recovery from ocular ischemic syndrome after treatment with verapamil. J Neuro-ophthalmol 1995; 15: 209-11.
- Duthie GG: Lipid peroxidation. Eur J Clin Nutr 1993; 47: 759-64.
- Scott G: Antioxidants in science, technology, medicine and nutrition. Chichester Albion Publishing 1997.
- Mineo K, Takao S and Keiichi K: Generation of a Transgenic Rabbit Model of Retina Degeneration. Investigative Ophthalmology & Visual Science 2009; 50(3): 1371-7.
- NIH Guide for the Care and Use of Laboratory Animals. a) DHEW Publication Number (NIH) 78-23, Revised 1978 and b) NIH Publication Number. 85-23, Revised 1985, U.S. Department of Health, Education and Welfare. Research Triangle Park, North Carolina.
- Muneerbabu C and Pandey VP: Effect of piperine on oral bioavailability of diltiazem HCl in rabbits. International Journal of Pharmaceutical Applications 2012; 3(4): 406-13.
- Castellini C, Lattaioli PDA and Minelli A: Oxidative status and semen characteristics of rabbit buck as affected by dietary vitamin E, C and n-3 fatty acids Reprod Nutr Dev 2003; 43: 91-03.
- Schmid-Elsaesser R, Zausinger S and Hungerhuber E: Critical reevaluation of the intraluminal thread model of focal cerebral ischemia evidence of inadvertent premature reperfusion and subarachnoid hemorrhage in rats by laser-doppler flowmetry. Stroke 1998; 29(10): 2162-70.
- Flecknell PA: Laboratory Animal Anesthesia (Third Edition). Academic Press, Harcourt Brace Jovanovich, London 2009.
- Longa EZ, Weinstein PR and Carlson S: Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989; 20: 84-91.
- Montezuma SR, Rizzo JF and Ziv OR: Differential recovery of the ERG, VECP and EECP following vitrectomy: implications for acute testing of an implanted retinal prosthesis. J Rehabil Res Devel 2004; 41: 113-20.
- Montezuma SR, Rizzo JF and Ziv OR: Combined vitrectomy lens and contact electrode for ERG recording during surgery. Retina 2002; 22(6): 828-29.
- Ruiz-Larrea MB, Leal AM and Liza M: Antioxidant effects of estradiol and 2-hydroxyestradiol on iron-induced lipid peroxidation of rat liver microsomes. Steroids 1994; 59: 383-8.
- Miranda KM, Espey MG and Wink DA: A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 2001; 5: 62-71.
- Ellman GL: Tissue sulfhydryl groups. Arch Biochem Biophys 1959; 82:70-7.
- Bulaj G, Kortemme T and Goldenberg DP: Ionization-reactivity relationships for cysteine thiols in polypeptides. Biochemistry 1998; 37: 8965-72.
- Bancroft J, Stevens A and Turner D: Theory and practice of histological techniques: Churchill Livingstone New York. The text 1996; 766.
- Farber DB, Flannery JG and Bowes-Rickman C: In Progress in Retinal and Eye Research (eds. Osborne, NN and Chader, GJ) Pergamon, Oxford, 1994; 13: 31-62
- Zhang SX, Ma J and Sima J: Genetic difference in susceptibility to the blood-retina barrier breakdown in diabetes and oxygen-induced retinopathy. American Journal of Pathology 2005; 166(1): 313-21.
- Packer L: Vitamin E: Biological activity and Health Benefits: Overview. In: Vitamin E in health and disease. New York, Packer, L, Fuchs, J., eds. Marcel Dekker, Inc 1993; 977-82.
- Romano C, Price M and Bai HY: Neuroprotectants in Honghua: glucose attenuates retinal ischemic damage. Investigative Ophthalmology and Visual Science 1993; 34(1): 72-80.
- Géraldine VD, Céline F and David C: Diltiazem-induced neuroprotection in glutamate excitotoxicity and ischemic insult of retinal neurons. Documenta Ophthalmologica 2005; 110(1): 25-35.
- Frasson M, Sahel JA and Fabre M: Retinitis pigmentosa: rod photoreceptor rescue by a calcium-channel blocker in the rd mouse. Nat Med 1999; 5(10): 1183-7.
- Buckley MMT, Grant SM and Goa KL: Diltiazem A reappraisal of its pharmacological properties and therapeutic use. Drugs 1990; 39: 757-806.
- Pearce-Kelling SE, Aleman TS and Nickle A: Calcium channel blocker D-cis-diltiazem does not slow retinal degeneration in the PDE6B mutant rcd1 canine model of retinitis pigmentosa. Mol Vis 2001; 7: 42-47
- Basil SP, Tiansen L and Michael SS: Absence of Photoreceptor Rescue with d-cis-Diltiazem in the rd Mouse. Investigative Ophthalmology and Visual Science 2002; 43: 1912-15.
- Osborne NN, Safa R and Nash MS: Photoreceptors are preferentially affected in the rat retina following permanent occlusion of the carotid arteries. Vision Research 1999; 39: 3995-02.
- Anjaneyulu M and Chopra K: Diltiazem attenuates oxi-dative stress in diabetic rats. Ren Fail 2005; 27(3): 335-44.
- Block F, Buckley MMT and Grant SM: Diltiazem A reappraisal of its pharmacological properties and therapeutic use. Drugs 1990; 39: 757-06.
- Çınar MG, Ülker S and Alper G: Effect of dietary Vitamin E supplementation on vascular reactivity of thoracic aorta in streptozotocin-diabetic rats. Pharmacology 2001; 62: 56-64.
How to cite this article:
Ibrahim BMM, Salama AAA, Abdallah HMI and Jaleel GAA: Comparative study of the effects of diltiazem versus alpha tocopherol on retinal degenation in rabbits. Int J Pharmacognosy 2018; 5(3): 158-66. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(3).158-66.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
158-166
707
1495
English
IJP
B. M. M. Ibrahim *, A. A. A. Salama, H. M. I. Abdallah and G. A. A. Jaleel
Department of Pharmacology, National Research Centre, Dokki, Giza, Egypt.
bmmih1974@gmail.com
21 October 2017
12 November 2017
18 November 2017
10.13040/IJPSR.0975-8232.IJP.5(3).158-66
01 March 2018