COMPARATIVE MACROSCOPIC AND MICROSCOPIC STUDY ON ACACIA ARABICA & PROSOPIS JULIFLORA
HTML Full TextCOMPARATIVE MACROSCOPIC AND MICROSCOPIC STUDY ON ACACIA ARABICA & PROSOPIS JULIFLORA
Sarika Nigam * 1, Vikram Singh 2, Archana Dongray 1 and Dilip Kumar Chanchal 3
College of Pharmacy 1, SRGI Ambabai, Jhansi - 284002, Uttar Pradesh, India.
Department of Pharmacognosy 2, SRI, Datia - 475661, Madhya Pradesh, India.
Department of Pharmacognosy 3, Insitute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Aim of Study: Compare the macroscopic and microscopic study of Acacia arabica and Prosopis juliflora. Material and Methods: The ethanolic extract of on Acacia arabica and Prosopis juliflora were using physio-chemical parameters and preliminary phytochemical investigation. Results and Discussion: In this study. I have done the comparative pharmacognostic study on Acacia arabia and Prosopis juliflora and conclude that the ethanolic extract of Acacia arabica plays a more significant role and has more significant value than the extract of Prosopis juliflora. Conclusion: The present study was aimed at the pharmacognostical study. Plants Acacia arabica and Prosopis juliflora were studies for pharmacognostical characteristic, namely, morphology, microscopy, which can be of utilized in identification and authentication of plants.
| Keywords: |
Acacia Arabica, Prosopis juliflora, Phyto-Pharmacognostic
INTRODUCTION: India has a rich heritage of traditional medicine constituting with its different components like Ayurveda, Siddha, and Unani, and traditional health care has been flourishing in this country for many centuries. Botanicals constitute of major part of these traditional medicines. With the emerging worldwide interest, in adopting traditional practices, in the health care systems by exploiting their potential, the evaluation of the botanicals in these systems of medicine in India is utmost essential 1. Herbal medicines have a vital role in the prevention and treatment of cancer, and medicinal herbs are commonly available and comparatively economical 2.
Herbalism (herbal medicine) as an alternative medical therapy is defined as the use of plants or substances derived from them, in treating disease, usually by medical herbalists without an orthodox medical qualification. Before the relatively recent application of scientific method into diagnosis and therapeutics, traditional medicines were mostly herbal 3. According to the WHO, 80% of the world population continues to rely mainly on traditional medicine for their health care 4.
Ayurvedic system understanding the knowledge of plants used for Ayurvedic preparations in relation to their use as therapeutic agents, pharmacological properties, medicinal plants being imported; medicinal plant parts being exported, endangered medicinal plants and availability of medicinal plants in different bio-geographical zones of India can be utilized in drawing strategies for rational and more scientific use of medicinal plants in a way that can be extended for future scientific investigation in different aspects 5.
There were thought to be roughly 1300 species of Acacia worldwide, about 960 of them native to Australia, with the remainder spread around the tropical to warm-temperate regions of both hemispheres, including Europe, Africa, southern Asia, and the Americas.
Natural Products in Medicine: Natural products are products from various natural sources, plants, microbes, and animals. They can be an entire organism (e.g. a plant, an animal or a micro-organism), a part of an organism (e.g. leaves or flowers of a plant, an isolated animal organ), an extract of an organism or part of an organism and an exudate, or pure compound (e.g. alkaloids, coumarins, flavonoids, lignans, steroids and terpenoids) isolated from plants, animals or micro-organisms. The use of natural products, especially plants, for healing is an ancient and universal as medicine itself.
Natural products have been an integral part of the ancient traditional medicine systems, e.g., Chinese, Ayurvedic, and Egyptian. Even now, continuous traditions of natural product therapy exist throughout the third world, especially in the orient, where numerous minerals, animal substances, and plants are still in common use. This recent resurgence of interest in plant remedies has been spurred on by several factors 6:
- The effectiveness of plant medicines.
- The preference of consumers for natural therapies, a greater interest in alternative medicines, and a commonly held erroneous belief that herbal products are superior to manufactured products.
- Dissatisfaction with the results from synthetic drugs and the belief that herbal medicines might be effective in the treatment of certain diseases where conventional therapies and medicines have proven to be inadequate.
- The high cost and side effects of most modern drugs.
- Improvements in the quality, efficacy, and safety of herbal medicines with the development of science and technology.
- Patients’ belief that their physicians have not properly identified the problem; hence, they feel that herbal remedies are another option.
- A movement towards self-medication.
Medicinal plants are generally known as “Chemical Goldmines” as they contain natural chemicals, which are acceptable to human and animal systems. Of the 2,50,000 higher plant species on earth, more than 80,000 are medicinal. The Red Data Book of India has 427 entries of endangered species of which 28 are considered extinct, 124 threatened, 81 vulnerable, 100 rare and 34 insufficiently known species 7.
The Origin, Scope and Practice of Pharmacognosy: The history of herbal medicines is as old as human civilization. The documents, many of which are of great antiquity, revealed that plants were used medicinally in China, India, Egypt and Greece long before the beginning of the Christian era. One of the most famous surviving remnants is Papyrus Ebers, a scroll some 60 feet long and a foot wide, dating back to the sixteenth century before Christ 6. Indians also, worked meticulously to examine and classify the herbs which they came across, into groups called Gunas. Charaka made fifty groups of ten herbs, each of which, according to him, would suffice an ordinary physician’s need. Similarly, Sushrutha arranged 760 herbs in 7 distinct sets based on some of their common properties. A large portion of the Indian population even today depends on the Indian System of Medicine - Ayurveda, ‘An ancient science of life.’ The well-known treaties in Ayurveda are Charaka Samhita and Sushruta Samhita. The first pharmacist, Galen, was known to have had several pain-relieving materials, including opium in his apothecary 9.
Quality Control of Herbal Drugs: Herbal drug technology is used for converting botanical materials into medicines, where standardization and quality control with proper integration of modern scientific techniques and traditional knowledge is important 10.
One of the impediments in the acceptance of herbal products worldwide is the lack of standard quality control profiles. Quality control of traditional medicines involves the quality of both raw materials, mostly plants but also animals, metals and minerals, as well as of finished products. The starting point is establishing the identity of the raw material. It is very important to be aware that the primary identity of the material can only be decided by a reliable traditional source. After this identification, morphological, microscopical, chemical and biochemical methods are available to undertake standardization of traditionally identified material, and they should be appropriately applied.
A practical problem that sometimes occurs in this regard is that a traditionally named plant material, for instance, may have more than a single botanical source. In the context of Ayurveda, the plant name brahmi, for instance, refers to at least two botanical species, viz, Bacopa monnieri and Centella asiatica, both of which possess similar properties and can be substituted for each other. Most of the herbal formulations, especially the classical formulations of traditional medicine, are poly-herbal. Each formulation contains 10-20 or more ingredients; a few have even 50-75 ingredients.
Many preparations are either liquid or semisolid. For such formulations, it is very difficult to establish parameters for quality control. Even official standards are not available. The unique processing methods followed for the manufacture of these drugs turn the single drugs into the very complex mixture, from which separation, identification, and analysis of the components are a very difficult 11.
Many times two or more different plants have the same name in Ayurveda. Ayurveda use plants by Sanskrit names and there are instances where the same name stands for two or three different plants. So, in spite of botanical identification, there is still confusion concerning some Ayurvedic drugs. Boerhaavia diffusa widely used as ‘quality of life enhancer’ and the plant Trianthema portulacastrum both, for instance, are known as “Punarva,” and both plants may be used at the same time.
Another well-known example is of Sankhapushpi, an important medhya drug used for the improvement of memory power, and intellect. Shankhapushpi is equated with one or other of the following plants depending on the region in India: Canscora decussata, Evolvulus alsinoides, and Clitoria ternatea and sometimes Convulvularis pluricalis. There seems to be a lot of confusion in correlating the terms Vishnukranti, Shankhapuspi, Aparajita, Girikarni, etc. to the respective botanical sources. All local trade occurs by these vernacular names add to the confusion.
Another problem for Ayurvedic drugs is that there are 56 standard books and different manufacturers use different reference book, which brings about manufacture to manufacturing variation in the same product.
There are various factors affecting the identification of plant material;
- Collection of wildly growing plants from forests and wastelands.
- Traders or suppliers generally have limited knowledge of medicinal plants.
- Folk populace and laborers who are not fully aware of the identity of the drugs, always do collections.
Non-homogeneity of plant material due to collection from wild sources and different geographical locations 12.
Plant Profile:
Botanical name : Acacia arabica
Hindi name : Babul, Pankikar
Family : Fabaceae
Prosopis juliflora (Sw.) DC (Mimosaceae) commonly known as mesquite is a shrub or small tree native to Mexico, South America, and the Caribbean. P. juliflora probably originates from Peru; it occurs naturally in dry areas of northern South America and Central America, Mexico and the southern USA. It has been introduced into many tropical areas, including northeastern Brazil, Africa, Australia, Southeast Asia, and the Indian subcontinent.
P. juliflora is xerophytic and is adapted to many soil types under a wide range of moisture conditions. The value of the tree lies in its exceptional tolerance of drought and marginal soils. It tolerates strongly saline soils and seasonal waterlogging. P. juliflora has been planted successfully on soils with acid to an alkaline reaction. It is sometimes said to dry out the soil and compete with grasses, particularly in dry areas 10.
FIG. 4: FLOWER OF PROSOPIS JULIFLORA
Chemical Constituents: Steroids, tannins, leucoanthocyanidin, and ellagic acid glycosides. A new monocyclic diketone, prosopidione, and two alkaloids, namely, juliprosinene and juliflorinine, have been isolated from the leaves 11.
Parts Used: Leaves, gum, bark, pods, flowers.
Botanical name : Prosopis juliflora
Hindi : Kabuli kikar, angarajii
babul, vilayati babul
Family : Fabaceae
MATERIALS AND METHODS:
Materials, Instruments, and Chemicals: Plant materials, glass slide, grinding mixer, hot air oven, silica crucible, ashless filter paper (Whatman no. 44), Petri dish, stoppered conical flask, rotary flask shaker alcohol (95%), chloroform water, chloral hydrate solution, water.
Collection of Plant: The plant materials were collected from the Jhansi and Lucknow.
Authentication of Plant: The materials were authenticated at Indian Grassland and Fodder Research Institute, Jhansi, India. Sample specimens have been identified as Prosopis juliflora (SW.) DC. of the family Fabaceae.
Processing of Plant Material for Study: The materials for the final study were prepared by the following procedure:
Washing: Foreign material was identified and discarded through washing.
Drying: Plant material was dried in the shed to prevent decomposition of the chemical constituents.
Grinding: Material ground till homogeneous powder was formed.
Study of Entire Material:
Macroscopical Study: It included determination of size, shape, surface characteristics, texture, and fracture characteristics.
Microscopical Study:
Stem Bark: Materials of bark were broken into pieces of about 1-2 cm long and 0.5-1 cm wide and boiled in a test tube for 1-3 min, to make it soft. Soft pieces were then cut into T. S. forms. Cut sections were dehydrated with a successive series of ethanol (i.e., 30, 50, 70 and 80 percent v/v) before staining with the saffranine solution (1% solution of saffranine in 50% alcohol w/v). The sections were mounted on glass slides in 50% (v/v) glycerine and covered with coverslip. All samples were examined under the microscope, and photographs were taken.
Leaf: The leaf was cut into small squares of 1-2 cm and treated with concentrated aqueous chloral hydrate to make the leaf colorless. As the leaves were thick and were taking time to be cleared. The section cutting of leaf was done by cutting the leaf into small pieces and keeping it in between the potatoes to get the fine section and staining the section with the saffranine solution (1% solution of saffranine in 50% alcohol w/v). The sections were mounted on glass slides in 50% (v/v) glycerine and covered with coverslip. All samples were examined under the microscope, and photographs were taken.
Twig: The fine section of the twig was directly cut with the help of sharp blade, and the sections were stained with the saffranine solution (1% solution of saffranine in 50% alcohol w/v). The sections were mounted on glass slides in 50% (v/v) glycerine and covered with coverslip. All samples were examined under the microscope, and photographs were taken.
Histochemical Analysis: It deals with localization of chemical compounds within the cells using specific colors of the compounds. The sections of stem bark, leaf, twig were treated with various stains such as ferric chloride solution (10%), Sudan-III, conc. HCl, conc. H2SO4, a pinch of phloroglucinol + conc. HCl and saturated solution of Sudan IV in 70% alcohol and the compounds present in the cells were identified with the help of microscope through the colors, which are specific to the compounds when stained with specific dyes.
Powder Study:
Microscopy: The powders of stem bark, leaf, and twig were examined for its microscopic characters. The powders were passed through sieve no. 60 and treated with chloral hydrate to remove coloring matter and viewed under a microscope at the 10X eyepiece and 40X objective for stone cells, calcium oxalate crystals and other characters.
Fluorescence Analysis: The powder was subjected to fluorescence analysis for the detection of the presence of compounds, which are fluorescent. Many substances when suitably illuminated, emit light of different wavelengths or color from that what falls on them. Fluorescence of powders of stem bark, leaf, flower, and twig were observed in daylight and UV light (254 nm & 366 nm). The powdered drugs were treated with different solvents in the glass slides. The solvents used were 1N HCl (aqueous), 1N HNO3 (aqueous), 1N H2SO4 (aqueous), CH3COOH, 1N NaOH (aqueous), Aq. NaOH, Meth. NaOH, I2, 1N KOH, Aq. KOH, Meth. KOH, alcohol as such, acidic alcohol and basic alcohol 13.
Organoleptic Study: It included determination of color, odor, and taste.
RESULTS: Study of Entire Material:
TABLE 1: STUDY OF ENTIRE MATERIAL
| Parameter | A. Arabica (Bark) | P. juliflora (Bark) |
| Shape | Curved pieces | Curved pieces |
| Surface
|
Irregular longitudinal ridges and sometimes transverse cracks. Inner surface is longitudinally striated. | Irregular longitudinal ridges and sometimes transverse cracks. Inner surface longitudinally striated |
| Fracture | Irregular and coarsely fibrous | Rough |
| Texture | shallowly fissured | Medium to coarse |
TABLE 2: ORGANOLEPTIC STUDY
| Parameter | Acacia arabica
(Leaves) |
Acacia arabica
(Bark) |
Acacia arabica
(Twig) |
Prosopis juliflora
(Leaves) |
Prosopis juliflora
(Bark) |
Prosopis juliflora
(Twig) |
| Colour | Green | Brown | Light Green | Green | Redish brown | Light green |
| Odor | Odorless | Odorless | Odorless | Odorless | Odorless | Odorless |
| Taste | Bitter | Astringent | Astringent | Palatable taste | Palatable taste | Palatable taste |
TABLE 3: MACROSCOPICAL STUDY
| Parameters | Acacia arabica (Leaf) | Prosopis juliflora (Leaf) |
| Size | 1.2-2.5 cm length and 0.25 cm in breadth | 1.8-2.5 cm length and 0.3 cm in breadth |
| Shape | Oblong | Oblong |
| Apex | Blunt | Blunt |
| Surface | Glabrous | Glabrous |
| Leaflet | 10-12 pairs, subsessile | 15-18 pairs, petiolate |
| Type | Bipinnately compound | Bipinnately compound |
| Venation | Reticulate | Reticulate |
| Stipule | Stipular spines are variable | Stipular spines are variable |
| Margin | Entire | Entire |
| Base | Round | Round |
| Arrangement | Alternate | Alternate |
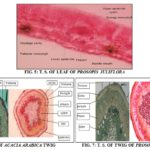
Microscopy: Thick and straight walled epidermal cells, large mucilage cavities in the mesophyll tissue and paracytic type stomata, prismatic type of calcium oxalate crystals in the mesophyll tissue, dense deposition of tannin content. The mesophyll has a palisade cell, and spongy mesophyll tissue has three or four layers of loosely arranged parenchyma cells. The vascular bundle is collateral with a conical mass of thick-walled, angular xylem elements, and a thin arc of phloem elements. A thick arc of gelatinous sclerenchymatous cells occurs on both upper and lower sides of the bundle.
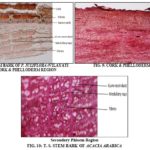
TS of the twig is almost circular in outline. The outermost region consists of a multilayered cork followed by multilayered secondary cortex embedded with mucilage canals, stone cells, and crystals. The phloem region beneath the secondary cortex is very broad traversed by medullary rays; phloem is made up of phloem parenchyma, fibers, sieve tubes, and companion cells. Fibers are arranged in small groups and radiating rows towards the cortical region. Xylem consists of xylem vessels, fibres, trachied and xylem parenchyma. Medullary rays in this region are also radiating 1-2 cells broadly, vessels are arranged mostly in radial rows of 2 to 7. Some vessels are solitary, the central portion is occupied by parenchymatous cell.
TABLE 8: MICROSCOPY OF BARK
| Acacia arabica (Bark) | Prosopis juliflora (Bark) | |
| Cork
|
15-25 layered, thin-walled, slightly flattened mostly rectangular, brown colored cork cells, | 10-15 layered, thin-walled, rectangular brown cork cells. |
| Cortex
|
A few lenticels formed by rupturing of cork cells, secondary cortical cells ovate to elongated, many tanniniferous stone cells, variable in shape and size present in large groups | A few lenticels formed by rupturing of cork cells, tannin cell present and fibers also present in the cortex. |
| Phloem
|
Phloem consists of sieve tubes, companion cells, fibers, crystal fibers and phloem parenchyma, phloem tissues filled with reddish or brown contents present, crystal fibers thick-walled, elongated, divided by transverse septa into segments, each contain a prismatic crystal of calcium oxalate, medullary rays uni to-multi- seriate, crystals of calcium oxalate found scattered amongst the stone cell" cells of secondary cortex and phloem parenchyma | Phloem consists of sieve tubes, companion cells, fibers, and phloem parenchyma, medullary rays uni to multiseriate. Gum resin duct present in phloem |
FIG. 11: PROSOPIS JULIFLORA BARK A) CORK CELL, B) MEDULLARY RAYS AND PHLOEM, C) FIBER
FIG. 12: TWIG OF ACACIA ARABICA A) PRISMATIC CALCIUM OXALATE CRYSTAL, B) EPIDERMAL CELL WITH FIBER, C) TRICHOME
FIG. 13: PROSOPIS TWIG
Fluorescence Analysis:
TABLE 4: FLUORESCENCE ANALYSIS IN DAYLIGHT
| S. no. | Reagent
used |
A. arabica
(leaf) |
A. arabica (bark) | A. arabica (twig) | P. juliflora (leaf) | P. juliflora (bark) | P. juliflora (twig) |
| 1 | Powder as such | Dark green | Brown | Yellow | Green | Light yellow | Light orange |
| 2 | Powder + 1N NaOH in H2O | Reddish brown | Black | Yellow | Green | Yellow | Light green |
| 3 | Powder + 1N HCI | green | Brown | Yellow | Green | Brownish yellow | Light green |
| 4 | Powder + 1N H2SO4 | green | Blackish brown | Yellow | Green | Yellow | Light green |
| 5 | Powder + HNO3 | Brown | orange | Light orange | Yellowish green | Bright yellow | Orange |
| 6 | Powder + 1N NaOH in MeOH | Black | Black | Dark yellow | Dark green | Dark brown | Light orange |
| 7 | Powder + KOH | brown | Reddish brown | Yellow | Light green | Bright yellow | Orange |
| 8 | Powder + H2SO4 | Dark brown | Reddish brown | Black | Blackish green | Black | Black |
| 9 | Powder + GAA | Black green | Brown | Yellow | Green | Yellow | Green |
| 10 | Powder + Methanol | Dark green | Brown | Yellow | Light green | Yellow | Green |
| 11 | Powder + Acetone | Dark Green | Dark brown | Yellow | Light green | Yellow | Yellow |
| 12 | Powder + EtOH | Brown | Black | Yellow | Light green | Yellow | Yellow |
| 13 | Powder + Alc FeCI3 | green | Dark green | Yellow | Yellowish green | Black | Green |
TABLE 5: FLUORESCENCE ANALYSIS IN 254 nm
| S. no. | Reagent
used |
A. arabica
(leaf) |
A. arabica
(bark) |
A. arabica
(Twig) |
P. juliflora (leaf) | P. juliflora (bark) | P. juliflora (twig) |
| 1 | Powder as such | Greenish brown | green | Green | Green | Green | Green |
| 2 | Powder + 1N NaOH in H2O | Dark green | Dark
green |
Dark
green |
Green | Green | Green |
| 3 | Powder + 1N HCI | green | Light Green | Green | Yellow green | Green | Green |
| 4 | Powder + 1N H2SO4 | Dark green | Black | Green | Yellow-green | Green | Green |
| 5 | Powder + HNO3 | Dark green | Light green | Green | Green | Green | Green |
| 6 | Powder + 1N NaOH in MeOH | Dark green | Black | Green | Green | Green | Dark
green |
| 7 | Powder + KOH | Black | green | Green | Yellow green | Green | Green |
| 8 | Powder + H2SO4 | Dark green | Black | Dark green | Dark green | Blackish green | Blackish green |
| 9 | Powder + GAA | Dark green | Green | Green | Green | Green | Green |
| 10 | Powder + Methanol | Brown | Grey | Green | Green | Green | Green |
| 11 | Powder + Acetone | Dark Green | Light Green | Green | Green | Green | Green |
| 12 | Powder + EtOH | Green | Black | Green | Green | Green | Green |
| 13 | Powder + Alc FeCI3 | Dark green | Dark green | Green | Green | Dark green | Bright green |
TABLE 6: FLUORESCENCE ANALYSIS IN 365 nm
| S. no. | Reagent
used |
A. arabica
(leaf) |
A. arabica
(bark) |
A. arabica
(Twig) |
P. juliflora (leaf) | P. juliflora (bark) | P. juliflora (twig) |
| 1 | Powder as such | Black | Dark black | Black | Blackish green | Black | Black |
| 2 | Powder + 1N NaOH in H2O | Black | black | Black | Blackish green | Black | Black |
| 3 | Powder + 1N HCI | Black | Dark black | Black | Blackish green | Black | Black |
| 4 | Powder + 1N H2SO4 | Black | Black | Black | Blackish green | Black | Black |
| 5 | Powder + HNO3 | Dark black | Dark black | Black | Blackish green | Black | Black |
| 6 | Powder + 1N NaOH in MeOH | Dark black | Dark black | Black | Blackish green | Black | Black |
| 7 | Powder + KOH | Black | Dark black | Black | Blackish green | Black | Black |
| 8 | Powder + H2SO4 | Black | Black | Black | Blackish green | Black | Black |
| 9 | Powder + GAA | Black | Dark black | Black | Blackish green | Black | Black |
| 10 | Powder + Methanol | Black | Black | Black | Blackish green | Black | Black |
| 11 | Powder + Acetone | Dark black | Black | Black | Black green | Black | Black |
| 12 | Powder + EtOH | Black | Black | Black | Blackish green | Black | Black |
| 13 | Powder + Alc FeCI3 | Black | Black | Dark black | Blackish green | Black | Dark black |
DISCUSSION:
Pharmacognostic Study: Botanical study is of prime importance in establishing quality control (identification) of herbal drugs. It may also provide suitable criteria to differentiate the different parts used of Acacia Arabic and Prosopis juliflora (Sw). DC. A detailed study of macroscopical, micro-scopical and powder microscopy and organoleptic study of powdered drug was done of stem bark, leaf, and twig microscopy.
Histochemical Analysis of the Sections of Different Parts of Acacia arabica and Prosopis juliflora: The results of the histochemical analysis showed that tannins were present in all the parts of the Acacia arabica and Prosopis juliflora (Sw.) DC. The lignins were present in the walls of the vessels, tracheids, fibers, and sclereids, the lignin is impregnated in between the cellular framework of the secondary, calcium oxalate crystals were present in stem bark, petiole, and twig, cellulose was present in leaf, and twig and gum-resin duct present in stem bark. Calcium oxalate crystals are more common among diversified plant group. They exhibited the unique properties of pleomorphism and birefringence and were present in stem bark, twigs. Absence of cutin and suberin shows the absence of phellem cells in all the sections. All parts of all the sections stained black when treated with ferric chloride 10%, which shows the presence of tannins. Stone cell present in a twig of both species.
CONCLUSION: It is concluded that given plant is Acacia arabica and Prosopis juliflora, I have done the comparative pharmacognostical study between Acacia arabica and Prosopis juliflora and conclude that Acacia arabica plays a more significant role and has a more scientific value. The present study was aimed at pharmacognostical study. Plants Acacia Arabica and Prosopis juliflora were studies for pharmacognostical characteristic, namely, morphology, microscopy, physicochemical, parameters which can be of utilized in identification and authentication of plants.
ACKNOWLEDGEMENT: The authors thankful with our deepest core of heart to Mr. Vikram Singh (Assistant Professor), for his valuable guidance.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Mukherjee PK: Exploring Botanicals in Indian System of Medicine-Regulatory Perspectives, Vol. 20, 2003: 249-264.
- Bijauliya RK, Alok S, Singh M and Mishra SB: A comprehensive review on cancer and anticancer herbal drugs. Int J Pharm Sci Res 2017; 8(7): 2740-61.doi: 10.13040/IJPSR.0975-8232.8(7).2740-61.
- Smith G: Herbs in medicine Triple Helix Autumn 04, 12.
- Chanchal DK, Niranjan P, Alok S, Kulshreshtha S, Dongray A and Dwivedi S: A brief review on medicinal plant and screening method of antilithiatic activity. Int J Pharmacognosy 2016; 3(1): 1-9. doi link: http://dx.doi.org/ 10.13040/ IJPSR.0975-8232.IJP.3(1).1-9.
- Raghavendra1 MP: Alkaloid extracts of Prosopis juliflora (Sw.) DC. (Mimosaceae) Against Alternaria alternate. Journal of Biopesticides 2009; 2(1): 56-59.
- Chanchal DK, Niranjan P, Alok S, Singh S and Saurabh: An Update on Ayurvedic Herb Kachhnar (Bauhinia purpurea )- A Review. Int J Pharmacognosy 2015; 2(8): 381-90.
- Thomas J: Medicinal and aromatic plants research in India. In UNDP. 1997. Proc. Training course on Industrial Exploitation of Indigenous Medicinal and Aromatic Plants. Beijing, China, 1997.
- Kokate CK: Pharmacognosy’ Nirali Prakashan, Edition 37th, 2008: 105-120, 1-3.
- Agrawal SS and Paridhavi M: Herbal drug technology; Universities Press (India) Private Limited, 2007: 1-5.
- Chanchal DK, Niranjan PS, Alok S and Rashi S: Evaluation of Macroscopical and Microscopical Study, Phytochemical Analysis, TLC and HPTLC Fingerprinting of Bauhinia Purpurea Leaves. Int J Pharm Sci Res 2016; 7(8): 3539-44.
- Wickramasinghe and Bandaranayake M: Quality control, screening, toxicity, and regulation of Herbal drugs, modern phytomedicine. Turning medicinal plants into drugs, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006.
- Shinde V: Exploration of molecular markers in quality control of herbal medicines. Master thesis. Bharati Vidyapeeth University, Department of Pharmacognosy 2004.
- Khandelwal KR: Practical Pharmacognosy, Nirali Prakashan, Pune 2001.
How to cite this article:
Nigam S, Singh V, Dongray A and Chanchal DK: Comparative macroscopic and microscopic study on Acacia arabica & Prosopis juliflora. Int J Pharmacognosy 2018; 5(11): 746-55. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(11).746-55.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
746-755
958
1514
English
IJP
S. Nigam *, V. Singh, A. Dongray and D. K. Chanchal
College of Pharmacy, SRGI Ambabai, Jhansi, Uttar Pradesh, India.
sarikabbd@gmail.com
12 September 2018
25 October 2018
27 October 2018
10.13040/IJPSR.0975-8232.IJP.5(11).746-55
01 November, 2018