COMPARATION BETWEEN TRADEMARK AND ANALYTICAL STANDARD CHLORPYRIFOS ADMINISTRATION ON BUTYRYLCHOLINESTERASE AND GLUCOSE LEVELS IN RATS
HTML Full TextCOMPARATION BETWEEN TRADEMARK AND ANALYTICAL STANDARD CHLORPYRIFOS ADMINISTRATION ON BUTYRYLCHOLINESTERASE AND GLUCOSE LEVELS IN RATS
Galang Cahyo Pamungkas, Elly Nurus Sakinah *, Cholis Abrori, Supangat, Cicih Komariah, Desie Dwi Wisudanti, Pipiet Wulandari, Jauhar Firdaus and Yuanita Nirmala Putri
Department of Pharmacology, Faculty of Medicine, University of Jember, Jember, Indonesia.
ABSTRACT: Indonesia is one of the largest agricultural countries in the world. In 2022, approximately 88.89% of the workforce was employed in the agricultural sector. One of the most important agricultural practices is the use of pesticides, which are essential for pest control. Organophosphates are widely used pesticides, and chlorpyrifos is one of the most commonly applied organophosphate insecticides. However, chlorpyrifos exposure may cause adverse health effects, including poisoning, organ damage, mortality, and insulin resistance. Butyrylcholinesterase (BChE) activity and serum glucose levels are important biomarkers for evaluating chlorpyrifos toxicity, as exposure to chlorpyrifos can decrease BChE activity and increase serum glucose levels. Chlorpyrifos is available in two forms, analytical standard chlorpyrifos (Pestanal) and trademark formulations such as Dursban 200 EC. However, studies comparing the effects of trademark and analytical standard chlorpyrifos on BChE and serum glucose levels are still limited. This study aimed to compare BChE activity and serum glucose levels between control and treatment groups, as well as between Pestanal and Dursban treatments. Rats were orally administered Pestanal or Dursban at a dose of 5 mg/kg body weight for 28 days. Serum BChE activity and glucose levels were measured and analyzed using an independent t-test with SPSS software. The results showed that serum BChE activity differed significantly compared to the control group and between the Pestanal and Dursban groups. Serum glucose levels were significantly higher in the treatment groups compared to the control group. However, no significant difference was observed between the Pestanal and Dursban groups.
Keywords: Chlorpyrifos, Trademark, Analytical standard, Butyrylcholinesterase, serum glucose
INTRODUCTION: The Indonesia is one of the largest agricultural country in the world. There are 8 million hectare agricultural land in 2015 in Indonesia and 88,89% workers work on agricultural sector 1, 2.
Large amount of agricultural activity cause pesticide in Indonesia are widely used. There are 1,6 tons of pesticide used between 2011 and 2020 3.
Pesticides are beneficial to eliminate the pests in agricultural land. Beside that beneficial function, pesticides can cause poisoning and make residue 4. Organophosphate as one of the pesticide group is widely used around the world. 50% of all pesticide in the world used organophosphate 5. Chlorpyrifos as one of the organophosphate pesticide is being used as insecticide in agricultural land 6.
There are 133 tons of chlorpyrifos used between 2020-2022 around the world (European Chemicals Agency, 2022). One of the trademark of chlorpyrifos used is Dursban 7.
Exposure to chlorpyrifos for the long period can cause negative impact because of its residue. Chlorpyrifos can cause disturbance such as neurotoxicity and alteration in enzymatic activity. In acute period such as nausea, vomiting, dizziness, seizure, loss off consciousness. In chronic period can also cause alteration of liver, testis, kidney functions, and even death 8. Enzymatic activity is also altered. One of them is the activity of cholinesterase 9. Chlorpyrifos can inhibits activity of cholinesterase. The inhibition of cholinesterase can cause sign and symptoms of neurotoxicity and disturbance of enzymatic activity 10. Butyrylcholinesterase (BChE) as one of the cholinesterases can be used to identify pesticides poisoning for asymptomatic, sub clinical, and also low dose exposure. This enzyme can also be used to determine of liver function 11. Low dose exposure of chlorpyrifos can also alter metabolism and cause hyperglycemia 12.
Previous study showed that chlorpyrifos exposure inhibit activity. Given dose 2,84 and 4,26 mg/KgBW of rats for 28 days significantly decrease butyrylcholinesterase serum level compare to control 13. Other study also showed that 10 mg/KgBW also decrease level of butyrylcholinesterase serum level on rats orally for 28 days 14. Study about glucose serum level for sub chronic time period showed that 16 weeks exposure of chlorpyrifos 4,75 orally to rats can increase glucose level 15. In 90 days exposure of chlorpyrifos 3,26 and 8,15 mg/KgBW orally could increase glucose serum levels in rats 16.
Pestanal as analytical standard chlorpyrifos is commonly use in laboratory because of its 99% purity of chlorpyrifos 17. Dursban as one of the trademark of chlorpyrifos in the data showed that it has 12,6% of chlorpyrifos and 87,4% inert solvent of aromatic and aliphatic hydrocarbon 18. Carbon tetrachloride (CCl4) as one of the aliphatic hydrocarbon can cause fibrosis, vacuolization, and necrosis of hepatocyte that givel orally 0,5 mL/KgBW for 8 weeks 19. CCl4 can also lead to lipid peroxidation and elevate glucose level 3 mL for 4 weeks 20. Ethanol 4 mg/KgBW orally for 12 weeks as one of the aromatic hydrocarbon also increased Kupffer and stellate cells in rats hepatocytes 21. Many study variations about the effect of chlorpyrifos for rats was already implemented. Until now none of those study compare the differences between analytical standard and trademark chlorpyrifos.
MATERIALS AND METHODS:
Chemicals: Analytical standard chlorpyrifos (99% purity)/Pestanal was bought from sigma-aldrich company in Indonesia. Dursban 200 EC was also bought from DOW agro Indonesia. All other chemicals were generous gift from Medical Faculty of Jember University.
Animal Treatments: Adult male wistar (2-3 months) rats were obtain from malang, Indonesia. The rats were housed at Medical Faculty experimental animal house. Acclimatization firstly conducted to rats for 7 days. The temperature controlled in 25o C, given turbo 30 mg/ day as food, and tap water ad libitum for the drink. Previously the study was approved by Medical Faculty Ethical Committee of Jember University. The body weight of the rats maintained at 150-200 mg. Rats divided into 4 groups that assigned with simple random sampling. Each group contains of 5 rats. The first Group (K1) was given tween 20 and normal saline by oral gavage for 28 days. The second group (K2) was given distilled water by oral gavage for 28 days. The third group (P1) was given Pestanal 5 mg/KgBW by oral gavage for 28 days. The last group (P2) was given Dursban 5 mg/KgBW by oral gavage for 28 days. The Pestanal was dissolved used 5% tween 20 and normal saline to. Dursban was dissolved using distilled water. Upon sacrifice by given 200 mL/KgBW of pentobarbital to the rats, blood were soon collected from intracardiac. The blood sample waited for 1 hour and then turn them into serum and ready to to be analized for glucose and BChE serum level.
Butyrylcholinesterase Analysis: Butyryl-cholinesterase was analyse with kinetic photometry method. Measurement conducted with reagen kit from DiaSys. Reagent 1 contain pyrophosphate with 7,6 PH 95 mmol/L and 2,5 mmol/L hexacyanoferrate kalium. Reagent 2 contain 75 mmol/L butyrylcholine. Kinetic photometry method were based on cholinesterase reaction that based on hydrolysis of butyrylcholine resulting thiocholine and butyrate. Thiocholine after that decreasing yellow Kalium hexacyanoferrate (II) to colorless.
Glucose Analysis: Butyrylcholinesterase was analyse with kinetic photometry method. Measurement conducted with reagen kit from DiaSys. Reagent 1 contain pyrophosphate with 7,6 PH 95 mmol/L and 2,5 mmol/L hexacyanoferrate kalium. Reagent 2 contain 75 mmol/L butyrylcholine. Kinetic photometry method were based on cholinesterase reaction that based on hydrolysis of butyrylcholine resulting thiocholine and butyrate. Thiocholine after that decreasing yellow Kalium hexacyanoferrate (II) to colorless.
Statistical Analysis: The statistical analysis wereuse independent t-test with help of SPSS software. At first, the K1 and P1 also K2 and P2. The significant results for the groups continue to compare P1 and P2.
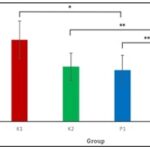
RESULTS AND DISCUSSION: Based on mean of the butyrylcholinesterase IBChE) level the highest level is in the K1 group (746.43 ± 170.53). P1 group has lower BChE level than K1 (498.74 ± 125.00) and the lowest in the P2 group (322.00 ± 26.17) compare to the K2 group (498.74 ± 125.00). The BChE level is shown on the Table 1.
TABLE 1: SERUM BChE LEVELS
| Group | BChE Serum Levels (U/L) (Mean ± SD) |
| K1 | 746.43 ± 170.53 |
| K2 | 498.74 ± 125.00 |
| P1 | 468.33 ± 134.34 |
| P2 | 322.00 ± 26.17 |
Independent t-test Sig. 2 tailed for the BChE serum level for the all the group compare show significant result (P <0.05). K1 and P1 t-test result is (0.031), K2 and P2 (0.002), and P1 and P2 (0.004). The independent t-test results are shown in Table 2, and the differences in serum BChE levels are presented in Fig. 1.
TABLE 2: INDEPENDENT T-TEST FOR BChE SERUM LEVEL
| Group | Sig. 2 tailed | Mean difference |
| K1 and P1 | 0.031 | 247.69 |
| K2 and P2 | 0.002 | 129.03 |
| P1 and P2 | 0.004 | 219.71 |
FIG. 1: DIFFERENCES IN SERUM BChE LEVELS AMONG CONTROL AND TREATMENT GROUPS. Values are expressed as mean ± SD. p< 0.05 (*); p< 0.01 (**).
BChE serum levels show significant different compare to control and between Pestanal and Dursban group. Oral administration of Chlorpyrifos for 28 days could reduce BChE level that is corresponding to previous study. 5 mg/KgBW for 28 days could reduce BChE serum level compare to control 22, 23. Other study that use trademark chlorpyrifos for 28 days also show same result compare to control. 2.84 and 4.26 mg/KgBW chlorpyrifos given by oral gavage for 28 days could reduce BChE serum level 13, 24. This condition occurred because chlorpyrifos that metabolized by live transform into chlorpyrifos-oxon and inhibits cholinesterase and binding to it irreversibly 25. Sub-chronic and chronic exposure could also change liver function because of inflammation caused by chlorpyrifos-oxon and created vacuoliazion, congestion, and fibrosis to hepatocytes. Liver as the place to synthesize BChE if there are change of structure could lead to reducing of BChe synthesis lower than normal 26, 27, 28.
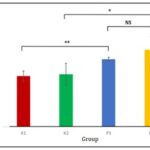
The significant reduction in BChE activity observed in chlorpyrifos-treated groups indicates enzymatic inhibition and possible hepatic involvement following sub-chronic exposure. As BChE is synthesized in the liver, its decreased activity may reflect hepatocellular dysfunction, which plays an important role in metabolic regulation. Hepatic impairment and cholinergic disruption induced by organophosphate exposure have been reported to interfere with insulin signaling, glucose utilization, and gluconeogenesis 12. Therefore, alterations in BChE activity may be associated with subsequent changes in glucose homeostasis, providing a biological basis for evaluating serum glucose levels as a metabolic outcome of chlorpyrifos exposure 15. Glucose serum level shown that K2 is lower than P2 (75.53 ± 15.64) and (110.58 ± 18.83). Meanwhile K1 is also lower than P1 (72.80 ± 7.40) and (96.97 ± 2.95). The glucose level shown on Table 3.
TABLE 3: SERUM GLUCOSE LEVELS
| Group | Glucose Serum Levels (mg/dL) (Mean ± SD) |
| K1 | 72.80 ± 7.40 |
| K2 | 75.53 ± 15.64 |
| P1 | 96.97 ± 2.95 |
| P2 | 110.58± 18.83 |
Independent t-test Sig. 2 tailed for the glucose serum level show significant level compare to control, but not P1 and P2 group. K1 and P1 t-test result is (0.001), K2 and P2 (0.029). There is no significant different from Mann-Whitney analysis for P1 and P2 result is (0.386). The independent t-test results are shown in Table 4, and the differences in serum glucose levels are presented in Fig. 2.
TABLE 4: INDEPENDENT T-TEST FOR GLUCOSE SERUM LEVEL
| Group | Sig. 2 tailed | Mean difference |
| K1 and P1 | 0.001 | -24.17 |
| K2 and P2 | 0.029 | -35.04 |
FIG. 2: DIFFERENCES IN SERUM GLUCOSE LEVELS AMONG CONTROL AND TREATMENT GROUPS. Values are expressed as mean ± SD. p< 0.05 (*); p< 0.01 (**); Not significant (NS).
Glucose serum also shown that compare to control the groups have significant differences. Organophosphate in previous study could elevate glucose serum level by oral gavage administration for 28 days. Diazinon ¼ LD50 administration for 28 days made insulin resistance and elevated glucose serum level 29. Other study gave malathion for 28 days by oral gavage 200 mg/KgBW also elevated glucose serum level 30.
Study of chlorpyrifos administration 5 mg/KgBW for 28 days by oral gavage also made elevation of glucose serum level 31. Sub-chronic and chronic exposure of chlorpyrifos could induce β pancreatic cell death. Hyperglycemia occurred when there are β pancreatic cell death 32. Elevated glucose level could also caused by liver gluconeogenesis and glycogenolysis from other tissues 33. There were no significant different between pestanal and dursban group according to glucose serum level. It was indicate that between trademark and analytical standard had no different refer to glucose serum level for 28 days administration of chlorpyrifos.
CONCLUSION: Oral administration of Dursban and Pestanal for 28 days have significant diffirences compare to control group for BChE and glucose serum level. Between Dursban and Pestanal group there is significant different of BChE serum level. There is no significant different glucose serum level elevation in Dursban and pestanal group.
ACKNOWLEDGMENTS: We wish to acknowledge the contribution of all members of this article.
CONFLICT OF INTEREST: There are no conflict of interest in this study.
REFERENCES:
- Badan Pusat Statistik: Data Penggunaan Pestisida 2018.
- Badan Pusat Statistik: Data Jumlah Pekerja Bidang Pertanian 2022.
- STATISTA: Volume of pesticides used in Indonesia from 2011 to 2020. STATISTA, 2022.
- Darmiati, D: Faktor-faktor yang berhubungan dengan risiko keracunan pestisida pada petani. Jurnal SAGO Gizi Dan Kesehatan 2021; 2(1): 81.
- Javeres MNL, Habib R, Laure NJ, Shah STA, Valis M, Kuca K and Nurulain S: Chronic exposure to organophosphates pesticides and risk of metabolic disorder in cohort from pakistan and cameroon. International Journal of Environmental Research and Public Health 2021; 1–13.
- Kurnia: Analisis Residu Klorpirifos pada Tanah dan Validasinya. Jurnal Agrikultura 2018; 61–65.
- Chairul, S: Pengaruh Iradiasi Sinar Gamma Terhadap Residu Insektisida Klorpirifos Pada Buah Anggur (Vitis vinifera L.). Ganendra Majalah Iptek Nuklir 2012.
- Akbar F: Analisis Risiko Kesehatan Pajanan Residu Klorpirifos Dalam Bayam (Amaranthus Sp.) Pada Masyarakat Di Kabupaten Gowa. [Thesis] 2013.
- Thetkathuek A, Yenjai P, Jaidee W, Jaidee, P & Sriprapat P: Pesticide Exposure and Cholinesterase Levels in Migrant Farm Workers in Thailand. Journal of Agromedicine 2017; 118–130.
- Masson P & Lockridge O: Butyrylcholinesterase for protection from organophosphorus poisons: Catalytic complexities and hysteretic behavior. In Archives of Biochemistry and Biophysics 2010; 107–120.
- Suemizu H, Kawai K, Murayama N, Nakamura M & Yamazaki H: Chimeric mice with humanized liver as a model for testing organophosphate and carbamate pesticide exposure. Pest Management Science 2018; 1424–1430.
- Li J, Li X, Zhang Z, Cheng W, Liu G & Zhao G: High-Fat Diet Aggravates the Disorder of Glucose Metabolism Caused by Chlorpyrifos Exposure in Experimental Rats Foods 2023; 816.
- Afify MM, Noaishi MA, Abd Allah AA & Afify MM: Oral and dermal exposure of chlorpyrifos and cypermethrin mixture induced cytogenetic, histopathological damage and oxidative stress in rats. Journal of American Science 2013; 6–7.
- Wielgomas B & Krechniak J: Effect of α-Cypermethrin and Chlorpyrifos in a 28-Day Study on Free Radical Parameters and Cholinesterase Activity in Wistar Rats. Polish J of Environ 2005; 1–7.
- Uchendu C, Ambali SF, Ayo JO & Esievo KA: The protective role of alpha-lipoic acid on long-term exposure of rats to the combination of chlorpyrifos and deltamethrin pesticides. Toxicology and Industrial Health 2017; 159–170.
- Wang HP, Liang YJ, Long DX, Chen JX, Hou WY & Wu YJ: Metabolic Profiles of Serum from Rats after Subchronic Exposure to Chlorpyrifos and Carbaryl. Chemical Research in Toxicology 2009; 1026–1033.
- Sigma Aldric: Chlorpyrifos. Sigma Aldrich 2023.
- M. James: Dursban Insecticide. EPA Encyclopedia of Pesticide, First Edition 1999.
- Kang J, Hong J and Lee S: Melatonin enhances mitophagy and mitochondrial biogenesis in rats with carbon tetrachloride‐induced liver fibrosis. Journal of Pineal Research 2016; 383–393.
- Khan RA, Khan MR, Sahreen S, Ahmed M and Shah NA: Carbon tetrachloride-induced lipid peroxidation and hyperglycemia in rat. Toxicology and Industrial Health 2015; 546–553.
- Sun F, Zhuang Z, Zhang D, Chen Y, Liu S, Gao N, Shi J and Wang B: Chronic moderate alcohol consumption relieves high‐fat high‐cholesterol diet‐induced liver fibrosis in a rat model. Clinical and Experimental Pharmacology and Physiology 2018; 1046–1055.
- Barski D and Spodniewska A: Effect of chlorpyrifos and enrofloxacin on selected enzymes in rats. Polish Journal of Veterinary Sciences 2018; 2(7): 234–236.
- Imam A, Sulaiman NA, Oyewole AL, Chengetanai S, Williams V, Ajibola MI, Folarin RO, Muhammad AS, Shittu STT & Ajao MS: Chlorpyrifos- and Dichlorvos-induced oxidative and neurogenic damage elicits neuro-cognitive deficits and increases anxiety-like behavior in wild-type rats. Toxics 2018; 6(4).
- Newairy AA, Abdou and H. M: Effect of propolis consumption on hepatotoxicity and brain damage in male rats exposed to chlorpyrifos. African Journal of Biotechnology 2013; 12(33): 5232–5243.
- Koshlukova SE and Reed NR: Encyclopedia of Toxicology. Elsevier Third Edition 2014.
- Cakici O and Akat E: Effects of oral exposure to diazinon on mice liver and kidney tissues: biometric analyses of histopathologic changes. Analytical and Quantitative Cytopathology and Histopathology 2013; 35(1): 7–16.
- Pohanka M and Zakova J: A smartphone camera colorimetric assay of acetylcholinesterase and butyrylcholinesterase activity. Sensors 2021; 21(5): 1–12.
- Tanvir EM, Afroz R, Chowdhury MAZ, Gan SH, Karim, N, Islam MN and Khalil MI: A model of chlorpyrifos distribution and its biochemical effects on the liver and kidneys of rats. Human and Experimental Toxicology 2016; 35(9): 991–1004.
- Alahyary P, Ilkhani Po M, Fathy Azar F and Nejati V: The Potential toxicity of diazinon on physiological factors in male rat. Pakistan Journal of Biological Sciences 2007; 11(1): 127–130.
- Lasram MM, Bouzid K, Douib IB, Annabi A, El Elj N, El Fazaa S, Abdelmoula J and Gharbi N: Lipid metabolism disturbances contribute to insulin resistance and decrease insulin sensitivity by malathion exposure in Wistar rat. Drug and Chemical Toxicology 2015; 227–234.
- Łukaszewicz-Hussain A: Serum Glucose Concentration in Subacute Intoxication with Chlorpyrifos - Organophosphate Insecticide. Medycyna Pracy 2013.
- Anuradha R, Saraswati M, Kumar KG and Rani SH: Apoptosis of Beta Cells in Diabetes Mellitus. DNA and Cell Biology 2014; 743–748.
- Farkhondeh T, Amirabadizadeh A, Samarghandian S and Mehrpour O: Impact of chlorpyrifos on blood glucose concentration in an animal model: a systematic review and meta-analysis. Environmental Science and Pollution Research 2010; 2474–2481.
How to cite this article:
Pamungkas GC, Sakinah EN, Abrori C, Supangat, Komariah C, Wisudanti DD, Wulandari P, Firdaus J and Putri YN: Comparation between trademark and analytical standard chlorpyrifos administration on butyrylcholinesterase and glucose levels in rats. Int J Pharmacognosy 2026; 13(1): 35-39. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(1).35-39.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
35-39
481 KB
203
English
IJP
Galang Cahyo Pamungkas, Elly Nurus Sakinah *, Cholis Abrori, Supangat, Cicih Komariah, Desie Dwi Wisudanti, Pipiet Wulandari, Jauhar Firdaus and Yuanita Nirmala Putri
Department of Pharmacology, Faculty of Medicine, University of Jember, Jember, Indonesia.
ellyns.fk@unej.ac.id
28 December 2025
28 January 2026
29 January 2026
10.13040/IJPSR.0975-8232.IJP.13(1).35-39
31 January 2026