CHEMICAL STANDARDIZATION OF MUCAMPARAP PARRU
HTML Full TextCHEMICAL STANDARDIZATION OF MUCAMPARAP PARRU
R. Shakila * 1, R. Jayachandran 1, Shyamala Rajkumar 1, A. Kanagarajan2 and P. Sathiyarajeswaran 1
Siddha Central Research Institute (Central Council for Research in Siddha) 1, Arignar Anna Hospital Campus, Arumbakkam, Chennai - 600106, Tamil Nadu, India.
Siddha Clinical Research Unit 2, Palayamkottai, Tirunelveli - 627002, Tamil Nadu, India.
ABSTRACT: Aim of the present study is to analyze Mūcāmparap paṛṛu, a poly herbomineral Siddha formulation for physicochemical parameters, TLC photo documentation, and HPTLC fingerprint profile studies. The physicochemical parameters such as ash contents, solubility in water and ethanol, pH, loss on drying and successive extraction with hexane, chloroform, and ethanol were carried out. Hexane, chloroform, and ethanol extracts were subjected to TLC photo documentation, HPTLC analysis. HPTLC fingerprint profile under UV 254 nm, hexane extract showed 13 peaks, chloroform showed 9 peaks and ethanol showed 10 peaks; under UV 366 nm, hexane showed 11 peaks, chloroform showed 15 peaks and ethanol showed 12 peaks; after derivatization with vanillin-sulphuric acid all the extracts showed 11 peaks. The results obtained in the study are specific and could be used as a reference in the quality control of the drug.
| Keywords: |
Mūcāmparap Paṛṛu, Kuṇrip Paṛṛu, Karuvangam, Mercury, Inflammation, Edema
INTRODUCTION: Analytical chemists have broad scope in the field of herbal drug analysis due to the recent increasing popularity of herbal drugs treatment. Siddha system of medicine is an indigenous system of India practiced in Southern states. Siddha Formulary of India is the collection of Siddha formulations gathered from the Siddha literature and published by Government of India. Mūcāmparap paṛṛu is enlisted in the Siddha Formulary of India, the official publication for Siddha medicines. It is also known as Kuṇrip paṛṛu. It is one of the external medicines used for the treatment of inflammation, sprain, edema during pregnancy 1.
It is a combination of six herbal drugs, two metallic drugs, egg, and sesame oil. As there is no report on the standardization on this drug in literature, the authors aim to analyze the drug for physicochemical parameters, TLC photo documentation and HPTLC fingerprint profile studies.
MATERIALS AND METHODS:
Procurement of Drug: The drug was procured from M/s. SKM Siddha and Ayurveda Company (India) Limited, Erode, Tamil Nadu, India. The drug consists of ten raw materials including mercury and lead. The list of all raw materials and the composition are presented in Table 1.
Chemicals and Solvents: Toluene, ethyl acetate, formic acid, hexane, chloroform, ethyl alcohol (Merck), sulphuric acid, hydrochloric acid and vanillin (SDFCL) of analar grade were used in the study. For visualization of spots, vanillin-sulphuric acid reagent (1% vanillin in 5 ml of sulphuric acid mixed with 95 ml ethyl alcohol) was prepared and used as visualizing reagent.
Physicochemical Analysis: The loss on drying at 105 ºC, total ash, water soluble ash, the alkalinity of the water-soluble ash, insoluble acid ash, water-soluble extractives, alcohol-soluble extractive and pH of 10% aqueous solution was carried out as per the methods described in Pharmacopoeial texts 2, 3.
TABLE 1: LIST OF RAW MATERIALS OF MUCAMPARAP PARRU
| S. no. | Regional Name
of the Drug |
Botanical/Chemical
Name of the Drug |
Quantity |
| 1 | Iracam | Mercury | 10 parts |
| 2 | Karuvaṅkam | Lead | 10 parts |
| 3 | Mūcāmparam | Aloe vera (L.) Burm. f. dried juice | 10 parts |
| 4 | Kantupāraṅki | Pygmaeopremna herbacea (Roxb.) Mold. root | 10 parts |
| 5 | Kuṇṛi vittup paruppu | Abrus precatorius L. seed | 10 parts |
| 6 | Āli vittu | Lepidium sativum L. seed | 10 parts |
| 7 | Eṭṭi vittu | Strychnous nux-vomica L. seed | 10 parts |
| 8 | Peruṅkāyam | Ferula foetida Regel exudate | 5 parts |
| 9 | Kōḷimuṭṭai veṇkaru | Egg white | Sufficient quantity |
| 10 | Nalleṇṇey | Sesame oil | Sufficient quantity |
TLC Photo Documentation / HPTLC Fingerprint Analysis:
Sample Preparation: Four gram of the drug was successively extracted with hexane, chloroform, and ethanol in a soxhlet extractor. The extracts were separately filtered, concentrated over a water bath and made up to 10 ml in standard flasks.
TLC Plate: Aluminium sheets precoated with 0.2 mm thick silica gel 60F254 (Merck) was used as a TLC plate for the TLC photo documentation and HPTLC fingerprint analysis.
Solvent System: Various solvent systems with different solvent ratio were tried to attain improved separation of spots. Toluene: Ethyl acetate (6:1.5, v/v) was selected as a suitable solvent system. Chloroform: Methanol: Glacial acetic acid (10:0.5:0.1, v/v) was finalized for ethanol extract.
Instruments: Twin trough chamber (CAMAG) of 10 × 10 size was used for the development of the plate. Automatic TLC Sampler 4 (ATS4) applicator, visualizer (CAMAG), scanner (CAMAG) equipped with WINCATS software were the instruments used for TLC photo documentation and HPTLC fingerprinting.
Procedure: Extracts (15 µl) were applied as 10 mm band and developed up to 8 cm in the above-mentioned solvent system. The developed plate was air dried, visualized under UV 254, 366 nm for documenting the TLC chromatograms; Then scanned in both wavelengths for generating the finger print profiles. The photo documentation and finger printing were also done at 575 nm after dipping the plate in the vanillin-sulphuric acid reagent, followed by heating in an oven till the appearance of the color of the spots.
RESULTS AND DISCUSSION:
Physico-Chemical Analysis: All the results of physicochemical parameters are presented in Table 2. The loss on drying was found to be 7.65% which is indicating the safe storage of the drug from microbial contaminations. The total ash remained 21.73% which shows the entire inorganic matter present in the drug; 1.64% water soluble ash indicates the soluble salts present; insoluble acid ash was determined as 15.71% which may be due to the lead added in the drug. Water and ethanol soluble extracts are nearly the same (17%) indicating the presence of polar phytoconstituents. The pH value shows the slightly acidic nature of the drug. The successive extract value also indicates the total extractable phytoconstituents.
TLC Photo Documentation / HPTLC Fingerprints: The Rf and color of spots of TLC of hexane extract of the drug before and after derivatization are presented in Table 3 and the corresponding photos in Fig. 1. Similarly, the results of chloroform extract and ethanol extract are detailed in Table 4, 5, Fig. 2 and Fig. 3. All these results showed the presence of numerous phytoconstituents.
TABLE 2: PHYSICO-CHEMICAL RESULTS OF MUCAMPARAP PARRU
| S. no. | Physico-chemical parameters | Mean (n=2) |
| 1 | Loss on drying at 105 ºC (%, w/w) | 7.65 |
| 2 | Total ash (%, w/w) | 21.73 |
| 3 | Water soluble ash (%, w/w) | 1.64 |
| 4 | Acid insoluble ash (%, w/w) | 15.71 |
| 5 | Water soluble extractives (%, w/w) | 16.70 |
| 6 | Ethanol soluble extractives (%, w/w) | 17.70 |
| 7 | Alkalinity (ml of 0.1 N HCl/ g) | 0.15 |
| 8 | pH (10% solution, w/v) | |
| 9 | Extractive value | |
| Hexane (%, w/w) | 8.80 | |
| Chloroform (%, w/w) | 4.70 | |
| Ethanol (%, w/w) | 6.60 | |
TABLE 3: Rf AND COLOUR OF SPOTS OF TLC OF HEXANE EXTRACT OF MUCAMPARAP PARRU
| S. no. | Under UV 254 nm | Under UV 366 nm | After Dipping in vanillin-sulphuric acid | ||
| Rf value | Rf value | Colour of the spot | Rf value | Colour of the spot | |
| 1 | 0.06* | - | - | 0.06 | Purple |
| 2 | - | 0.10 | Pink | 0.10 | Purple |
| 3 | 0.17* | 0.16 | Blue | 0.17 | Purple |
| 4 | - | - | - | 0.20 | Purple |
| 5 | 0.23* | 0.23 | Blue | 0.23 | Purple |
| 6 | 0.57* | 0.36 | Blue | - | - |
| 7 | - | 0.46 | Blue | - | - |
| 8 | - | - | - | 0.51 | Purple |
| 9 | - | 0.57 | Blue | 0.57 | Violet |
| 10 | - | 0.62 | Pale blue | 0.62 | Purple |
| 11 | - | 0.67 | Pale blue | 0.67 | Purple |
| 12 | - | 0.77 | Blue | - | - |
| 13 | - | 0.86 | Blue | - | - |
| *Green color | |||||
TABLE 4: Rf AND COLOUR OF SPOTS OF TLC OF CHLOROFORM EXTRACT OF MUCAMPARAP PARRU
| S. no. | Under UV 254 nm | Under UV 366 nm | After Dipping in vanillin-sulphuric acid | ||
| Rf value | Rf value | Colour of the spot | Rf value | Colour of the spot | |
| 1 | 0.12* | 0.12 | Pink | - | - |
| 2 | - | 0.19 | Pale blue | - | - |
| 3 | 0.22* | 0.22 | Pale blue | 0.23 | Purple |
| 4 | 0.25* | 0.25 | Pale blue | 0.25 | Purple |
| 5 | - | 0.31 | Bluish white | - | - |
| 6 | 0.37* | 0.37 | Blue | - | - |
| 7 | 0.44* | 0.43 | Pale blue | - | - |
| 8 | - | 0.52 | Pale blue | 0.55 | Purple |
| 9 | 0.57* | 0.59 | Bluish white | - | - |
| 10 | 0.62* | 0.62 | Pale blue | 0.62 | Purple |
| 11 | - | 0.66 | Blue | - | Grey |
| 12 | - | 0.75 | Blue | - | Blue |
| 13 | - | 0.87 | Blue | - | Pink |
| *Green color | |||||
TABLE 5: RF AND COLOUR OF SPOTS OF TLC OF ETHANOL EXTRACT OF MUCAMPARAP PARRU
| S. no. | Under UV 254 nm | Under UV 366 nm | After Dipping in vanillin-sulphuric acid | ||
| Rf value | Rf value | Colour of the spot | Rf value | Colour of the spot | |
| 1 | - | 0.03 | Pale blue | 0.06 | Purple |
| 2 | - | 0.14 | Blue | - | - |
| 3 | 0.17* | 0.17 | Creamy white | - | - |
| 4 | - | - | - | - | - |
| 5 | 0.29* | - | - | 0.29 | Purple |
| 6 | - | 0.34 | Blue | 0.32 | Purple |
| 7 | 0.38* | 0.38 | Blue | - | - |
| 8 | 0.44* | 0.42 | Pale Blue | 0.42 | Purple |
| 9 | - | 0.50 | Blue | - | - |
| 10 | - | 0.59 | Blue | - | - |
| 11 | - | 0.72 | Blue | - | - |
| 12 | 0.92* | 0.92 | Blue | - | - |
| 13 | - | - | - | ||
| *Green color | |||||
1. Under UV 254 nm; 2. Under UV 366 nm; 3. White light after derivatization with vanillin-sulphuric acid
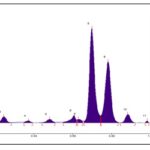
HPTLC fingerprint profile of hexane extract under UV 254 nm Fig. 4 showed 13 peaks in which peak at Rf 0.60 contributes to a maximum of 43.43% and a peak at Rf 0.24 remains as the second largest peak, and all other peaks are small; under UV 366 nm Fig. 5 showed 11 peaks in which peak at Rf 0.70 contributes to a maximum of 47.33% and a peak at Rf 0.78 remains as the second the largest peak with an area of 34.10% and all other peaks are small; under white light after derivatization at 575 nm Fig. 6 also exhibited 11 peaks in which peak at Rf 0.53 contributes to a maximum of 51.47%, a peak at Rf 0.59 remains as the second largest peak with an area % of 18.41 and all other peaks are small.
FIG. 4: HPTLC FINGER PRINT PROFILE OF HEXANE EXTRACT UNDER UV 254 nm
FIG. 5: HPTLC FINGER PRINT PROFILE OF HEXANE EXTRACT UNDER UV 366 nm
FIG. 6: HPTLC FINGER PRINT PROFILE OF HEXANE EXTRACT UNDER WHITE LIGHT AFTER DERIVATIZATION
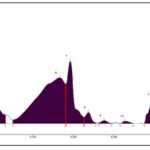
HPTLC fingerprint profile of chloroform extract under UV 254 nm Fig. 7 showed 9 peaks in which peak at Rf 0.96 was the major with 36.74% area, a peak at Rf 0.61 remains as the second largest peak, a peak at Rf 0.22 remains as the third major peak and all other peaks are small; under UV 366 nm Fig. 8 showed 15 peaks in which peak at Rf 0.66 contributes to 20.52%, a peak at Rf 0.96 remains as the second largest peak with an area of 18.12%, a peak at Rf 0.92 was the third major peak with an area of 12.27% and all other peaks are small; under white light after derivatization at 575 nm Fig. 9 showed 11 peaks in which peak at Rf 0.56 contributes to a maximum of 32.16%, a peak at Rf 0.25 remains as the second largest peak with an area % of 16.95, a peak at Rf 0.22 was the third largest peak with an area of 14.38% and all other peaks are small.
FIG. 7: HPTLC FINGER PRINT PROFILE OF CHLOROFORM EXTRACT UNDER UV 254 nm
FIG. 8: HPTLC FINGER PRINT PROFILE OF CHLOROFORM EXTRACT UNDER UV 366 nm
FIG. 9: HPTLC FINGER PRINT PROFILE OF CHLOROFORM EXTRACT UNDER WHITE LIGHT AFTER DERIVATIZATION
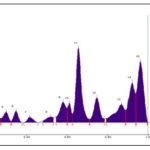
HPTLC fingerprint profile of ethanol extract, under UV 254 nm Fig. 10 showed 10 peaks in which peak at Rf 0.16 was the major with 40.06 % area, a peak at Rf 0.44 remains as the second largest peak with the peak are of 11.65 % and all other peaks are minor peaks; under UV 366 nm Fig. 11 showed 12 peaks in which peak at Rf 0.35 and 0.45 contributes to 14.78% and 13.19% respectively. A peak at Rf 0.02 with an area of 36.69 % need not be considered as major as it is very close to the application position; under white light after derivatization at 575 nm Fig. 12 showed 11 peaks in which peak at Rf 0.44 contributes to 33% and a peak at Rf 0.96 remains as the second largest peak with an area of 14.63%.
FIG. 10: HPTLC FINGER PRINT PROFILE OF ETHANOL EXTRACT UNDER UV 254 nm
FIG. 11: HPTLC FINGER PRINT PROFILE OF ETHANOL EXTRACT UNDER UV 366 nm
FIG. 12: HPTLC FINGER PRINT PROFILE OF ETHANOL EXTRACT UNDER WHITE LIGHT AFTER DERIVATIZATION
CONCLUSION: From the results of the study, it is evident that all the physicochemical parameters, TLC photo documentation of hexane, chloroform, and ethanol extracts under different conditions and the HPTLC fingerprint profiles of these extracts under UV 254 nm, 366 nm and white light at 575 nm can be considered as a reference data for the quality assessment of the drug in future. Though the drug is an external medicine, needs to be standardized for confirming its efficacy.
ACKNOWLEDGEMENT: Authors are thankful to the Director General, CCRS for facilities and support.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Anonymous: The Siddha Formulary of India, Part 1, Ministry of Health & Family Welfare, Government of India, New Delhi 1992: 165.
- Anonymous: The Siddha Formulary of India, Part-I, Department of ISM & H, Ministry of Health & Family Welfare, Government of India, New Delhi 1992.
- Anonymous: Quality Control Methods for Medicinal Plant Materials. World Health Organization, Geneva, Switzerland 1998.
How to cite this article:
Shakila R, Jayachandran R, Rajkumar S, Kanagarajan A and Sathiyarajeswaran P: Chemical standardization of Mucamparap Parru. Int J Pharmacognosy 2016; 3(1): 35-42. doi: 10.13040/IJPSR.0975-8232.3(1).35-42.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
35-42
841
1940
English
IJP
R. Shakila *, R Jayachandran, S. Rajkumar, A. Kanagarajan and P. Sathiyarajeswaran
Siddha Central Research Institute (Central Council for Research in Siddha), Arignar Anna Hospital Campus, Arumbakkam, Chennai,Tamil Nadu, India
shakilasiva@gmail.com
21 October 2015
26 November 2015
06 December 2015
10.13040/IJPSR.0975-8232.IJP.3(1).35-42
31 January 2016