CHEMICAL COMPOSITION AND ANTIFUNGAL ACTIVITY OF ESSENTIAL OILS FROM AERIAL PARTS OF MENTHA PIPERITA AND MENTHA ARVENSIS
HTML Full TextCHEMICAL COMPOSITION AND ANTIFUNGAL ACTIVITY OF ESSENTIAL OILS FROM AERIAL PARTS OF MENTHA PIPERITA AND MENTHA ARVENSIS
Harpreet Kaur 1, Ritu Tandon * 1, Anu Kalia 2 and Charu Maini 1
Department of Chemistry 1, Department of Soil Science 2, Punjab Agricultural University, Ludhiana - 141004, Punjab, India.
ABSTRACT: The present study deals with the chemistry and antifungal activity of essential oil (EO) of two species of Mentha viz. Mentha piperita and Mentha arvensis which was extracted using Clevenger-type apparatus. The percent yield of EO of M. arvensis (2.8%) was found higher than M. piperita (2.1%). Compositional analysis of M. piperita and M. arvensis EO oil by GC-MS revealed the presence of 51 and 35 compounds, respectively. Vibration spectroscopy performed in the 500-4000 cm-1 wave number region depicted variations in the presence of functional groups among the two EOs. The M. piperita EO exhibited better antifungal potential against Penicillium digitatumin comparison to M. arvensis EO. The results were found at par with the standard Carbendazim 50 WP at 250 µg/mL. Optical research microscopy study depicted profound morphological changes in hyphae, and spore-bearing organs of EO treated fungus, thereby supporting the better antifungal activity of M. piperita EO.
| Keywords: |
Essential oil, Mentha piperita, Mentha arvensis, GC-MS, FT-IR, Antifungal activity
INTRODUCTION: Postharvest damages in fruits have been predicted to range between 10 to 40% 1. Kinnow has very diminutive storage life at ambient conditions and economically stored at low temperature by sellers for marketing in the off period. Among various pathogens, attack by fungal pathogens is the major cause for the postharvest losses. The main fungal pathogens of kinnow during storage period are P. digitatum, Penicillium italicum, and Aspergillus niger. Out of these, P. digitatum, the green mold, causes the significant loss and is the most dominant mesophilic fungus of division Ascomycota. It is found in soils of citrus producing areas and is responsible for prevalent post-harvest diseases in citrus fruit causing huge losses due to decay caused by P. digitatum 2.
Essential oils are emerging as possible alternatives for preservatives in the food industry due to their diverse antimicrobial properties. Essential oils are natural compounds produced as secondary metabolites in the plant system. These are aromatic oily volatile colorless liquids characterized by distinct odor and usually are less denser than water 3. These are rich in secondary compounds such as flavonoids, polyphenols, and terpenoids and are generally synthesized in a specific cell or tissue types in leaves and plant stems. The main volatile components of EOs include monoterpenes, sesquiterpenes and their oxygenated derivatives such as aldehyde, ketones, alcohols, esters, and acids 4.
Many EOs and their primary compounds exhibit diverse biological activities such as antibacterial, antifungal, antioxidant, antimutagenic, and anti-microbial activities. Being natural and rich in antioxidants, these are considered safe for human consumption 5, 6. There are 17,500 medicinal plant species among higher plants, and approximately 3,000 EOs exist among which 300 are commercially important having applications in perfume, pharmaceuticals and cosmetics industries 7, 8. Among various plants, the genus Mentha (mint), an imperative member of the Lamiaceae family, is very diverse 9. It is signified by approximately 19 species and 13 natural hybrids, mainly persistent herbs growing enthusiastically in damp or wet places. Mints can grow under an extensive range of agro-climatic conditions and are invasive and fast growing 10.
Many species and hybrids of Mentha also have considerable economic importance such as Mentha spicata, Mentha aquatica, Mentha arvensis, Mentha canadensis, Mentha pulegium and Mentha piperita 11. Different species of Mentha are utilized across the globe for their medicinal and culinary properties. Mints are extensively cultivated for their oils and terpenoid components such as menthol, carvone, linalyl acetate, and linalool, for use in cosmetic, food, pharmaceutical, flavor, beverage and allied industries 12. Mentha species essential oils have high antibacterial and antifungal activity and hence can be used as natural fungicides 13. Commercially, the most sustainable species is peppermint (M. piperita), which is a hybrid of corn-mint (M. canadensis) and spearmint (M. spicata) 5. Major bioactive compounds of Mentha are menthol, menthone and pulegone 14.
A lot of studies related to antimicrobial activities of EOs have been performed but the anti-mycotic potential of Mentha EOs against the postharvest pathogen, P. digitatum has not been explored as yet. The present investigation has been carried out on the compositional analysis of M. arvensis, and M. piperita leaves EOs focusing on the morphological variations of P. digitatum caused by the essential oils.
MATERIALS AND METHODS:
Plant Material: At the full flowering stage during May 2017, the mature fresh leaves of two Mentha species namely M. arvensis and M. piperita were collected from Herbal Garden, Department of Agronomy, Punjab Agricultural University, Ludhiana, India. The species were identified and authenticated by a plant agronomist, Dr. Amanpreet Singh from Department of Agronomy, Punjab Agricultural University, Ludhiana.
Extraction of Essential Oil: The fresh leaves were washed twice with double distilled water to remove dust, at room temperature. The fresh leaves of M. arvensis and M. piperita were coarsely grounded prior to the operation and after that subjected to hydro-distillation in Clevenger-type apparatus for 5h at 60 °C (until no more oil was obtained). The extracted essential oil was refluxed with diethyl ether and dried over anhydrous sodium sulfate, filtered and stored at 4 °C in dark amber colored vials till further analysis. The yield of essential oils was determined by using the formula:
Yield (%) = The volume of essential oil extracted (ml)/ Weight of sample taken (g) × 100
Chemical Characterization of Essential Oils: The essential oils were analyzed using GC-MS (QP2010 Plus, Shimadzu, Japan), equipped with Rtx-5 MS capillary column (30.0 m × 0.25 mm i.d., 0.25 μm film thickness) for the separation of the components of essential oil. The injector was maintained at 260 °C and operated in split injection mode with split ratio 120.0 the split valve closed for 1 min. Helium gas was used as the carrier gas at a constant pressure of 69 kPa. The column oven was initially maintained at 50 °C for 2 min, raised to 210 °C at 3°C per min for 2 min and then to 250 °C at 6 °C per min for 6 min. The interface temperature was 270 °C and the sample was run in EI mode. Data acquisition was started 4 min after injection. The MS operating parameters used were; ionization voltage (EI) 70 eV, peak width 2 s, scan speed 1250 m/z; scan range 40-650 m/z and detector voltage 1.5 V.
Essential Oils Identification: The compounds of essential oils were identified based on the comparison of their relative retention times and mass spectra with literature data including Flavors and Fragrances of Natural and Synthetic Compounds 2 (FFNSC2), WILEY 8 and National Institute of Standards and Technology 14 (NIST14) libraries.
FT-IR Analysis: The vibrational spectroscopy of both Mentha species essential oil was performed on a Thermo Nicolet model 6700 FT-IR spectrometer at room temperature using ATR assembly at a resolution of 3.86 cm-1 in the 500-4000 cm-1 domains.
Determination of Antifungal Activity:
Procurement of Test Fungus: The culture of the test fungus P. digitatum isolated from kinnow fruit surface was procured from IARI (Indian Agriculture Research Institute), New Delhi with ITCC No. 7190.
Poisoned Food Technique: The antifungal activity of the Mentha essential oils against P. digitatum culture was evaluated by poisoned food technique 15. All tests were performed in triplicates with different working concentrations (µg/mL) of the oil, i.e. 1000, 500, 400, 300, 200 and 100 µg/mL of M. piperita and M. arvensis essential oils along with standard Carbendazim 50 WP (250 µg/mL). For this purpose, each essential oil was serially diluted using 0.5% tween 20 to prepare test solutions of 1000, 500, 400, 300, 200, and 100 µg/mL. One ml of each concentration was incorporated into 15 ml molten potato dextrose agar medium and mixed well in Petri dishes. After solidification of the medium, 100 µl of prepared spore suspension containing 104 colony forming units (CFU ml-1) of fungal spores were spread over medium. The Petri dishes were tightly sealed with parafilm and incubated at 25 °C in the incubator. The colony count was recorded until the 7th day after inoculation (DAI). The percentage inhibition was calculated from the mean value of colony numbers of treated and untreated fungus.
Optical Research Microscopy of Fungal Growth: The morphological changes in fungus after treatment were studied by optical microscopy. The fungal samples were stained with lactophenol cotton-blue dye and observed under Optical research microscope (make Leica DM 5000B) at 200 and 400 X magnifications to examine any morphological changes. Untreated samples were also stained and examined. Optical micrographs were taken with the help of CCD camera attachment (Leica DFC 420C camera).
Statistical Analysis: The experimental results were expressed as the mean of three replicates. The data were analyzed using the SAS software 9.0. Differences between means were tested using the least significant difference, and treatment means were compared with Duncan’s multiple range test (P<0.05) and t-grouping and P-value of P<0.05 was considered as significant.
RESULTS:
Yield and Chemical Composition of Essential Oils: Essential oils isolated from fresh leaves of both Mentha species were pale yellow. The percentage yield of M. arvensis essential oil (2.8%) was found to be higher than M. piperita essential oil (2.1%). The major compounds identified by GC-MS analysis in M. piperita and M. arvensis essential oils are shown in Table 1. A total of 51 and 30 components were identified in M. piperita and M. arvensis essential oil, respectively.
TABLE 1: MAJOR CONSTITUENTS (AREA %) IN MENTHA PIPERITA AND MENTHA ARVENSIS
| Name | Area (%) | |
| M. piperita | M. arvensis | |
| Menthol | 58.80 | 84.63 |
| Pulegone | 6.62 | - |
| Isomenthne | 6.42 | - |
| L-menthol | - | 4.58 |
| Menthyl acetate | 3.94 | 1.11 |
| Menthofuran | 3.11 | - |
| Cyclohexanone | - | 2.97 |
| Neomenthone | 2.64 | - |
| (E)-Caryophyllene | 1.95 | - |
| Limonene | 1.62 | 1.54 |
| Neoisomenthol | - | 1.46 |
| Germacrene D | 1.43 | - |
| 2-Cyclohexen-1-one | 1.41 | - |
| 2-Isopropyl-5-methyl cyclohexanol | 1.35 | - |
| Eucalyptol | 0.90 | 0.07 |
| α-Pinene | 0.71 | 0.10 |
| (E)- β-Famesene | 0.58 | - |
| 2-Isopropenyl-5-methylcyclohexanol | 0.57 | - |
| Viridiflorol | 0.55 | - |
Table 2 showed the comparative existence of some compounds in M. piperita and M. arvensis. Menthol, the major component of both essential oils was found in higher concentration in M. arvensis (84.63%) than in M. piperita (58.80%). However, other compounds such as pulegone (6.62%), isomenthone (6.42%), menthofuran (3.11%), neomenthone (2.64%), caryophyllene (1.94%), germacrene (1.43%), 2-cyclohexen-1-one (1.41%), 2 -isopropyl -5 -methyl cyclohexanol (1.35%) etc. were found to be absent in essential oil of M. arvensis.
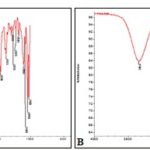
FT-IR Analysis: The FT-IR spectra of essential oil of both Mentha species revealed distinct variation for occurrence of several peaks indicating variation in compounds possessing different functional groups. The FT-IR spectrum of M. piperita showed a broad band at 3367.1 cm-1 due to bonded O-H stretching, a band at 1706 cm-1 due to C=O stretching and a sharp band at 2888.7 cm-1 due to C−H stretching whereas in M. arvensis the similar stretching bands were observed at 3346 cm-1, 1656 cm-1, and 2884.8 cm-1 respectively Fig. 1A, and Fig. 1B. However, a sharp bonded O−H stretch band and small overtone due to C=O band was observed in the FT-IR spectrum of M. arvensis in comparison to M. piperita essential oil.
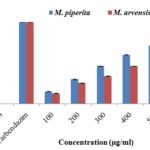
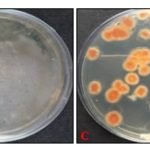
Antifungal Activity Assay: The antifungal activities of Mentha species essential oils were assessed against P. digitatum w.r.t. absolute control and standard Carbendazim 50 WP. Results of the antifungal activity on the seventh day after inoculation (DAI) showed 100% inhibition at 1000 µg/mL in essential oils obtained from both species whereas at 100-500 µg/mL M. piperita essential oil showed better percent inhibition compared to M. arvensis essential oil Fig. 2. The maximum inhibition was observed at 500 µg/mL for both essential oils, i.e. 71% and 55% M. piperita and M. arvensis essential oils, respectively. Standard Carbendazim 50 WP showed 99.7% inhibition, which was found higher than both the essential oils Fig. 3B, 3C, and 3D. All the interactions, i.e. between different concentrations of both essential oils concerning control (i.e., without treatment) Fig. 3A was significant with a coefficient of variance value of 0.765.
TABLE 2: COMPARISON OF MINOR CONSTITUENTS OF MENTHA PIPERITA AND MENTHA ARVENSIS
| S. no. | Name | M. piperita | M. arvensis |
| 1 | Ethyl hexanol | + | - |
| 2 | Bicyclogermacrene | + | - |
| 3 | Caryophyllene oxide | + | - |
| 4 | Phytol acetate | + | - |
| 5 | Lavandulyl acetate | + | - |
| 6 | Neomenthol | + | - |
| 7 | α-Cadinol | + | - |
| 8 | α-Bourbonene | + | - |
| 9 | Neomenthyl acetate | + | - |
| 10 | δ-Cadinene | + | - |
| 11 | Bicyclo[3.1.0]hexane-6-methanol | + | - |
| 12 | 1,6-Octadien-3-ol | + | - |
| 13 | Spathulenol | + | - |
| 14 | Trans-Nerolidol | + | - |
| 15 | 2,5-diethyl-Tetrahydrofuran | + | - |
| 16 | Isomenthyl acetate | + | - |
| 17 | Eugenol | + | - |
| 18 | α-Gurjunene | + | - |
| 19 | (+)-4-Carene | + | - |
| 20 | 3-Cyclohexene-1-methanol | + | - |
| 21 | BHT | + | - |
| 22 | 1-Octen-3-ol | + | - |
| 23 | 4,8-β-epoxy-Caryophyllane | + | - |
| 24 | P-Menthane-1,2,3-triol | + | - |
| 25 | Ispulegyl acetate | + | - |
FIG. 1: FT-IR SPECTRA OF ESSENTIAL OILS EXTRACTED FROM MENTHA PIPERATA (1A) AND MENTHA ARVENSIS (1B) USING ATTENUATED TOTAL REFLECTANCE MODE
FIG. 2: ANTIFUNGAL ACTIVITY OF THE ESSENTIAL OIL OF M. PIPERITA AND M. ARVENSIS AGAINST P. DIGITATUM
FIG. 3: ANTIFUNGAL ACTIVITY OF M. PIPERITA AND M. ARVENSIS ESSENTIAL OIL AT 500 µg/mL 7th DAY AFTER INOCULATION (3A) CONTROL (3B) CARBENDAZIM 50 WP (3C) M. PIPERITA AND (3D) M. ARVENSIS
Optical Research Microscopy of Essential Oil - P. Digitatum Studies: Optical research micro-graphs of control P. digitatum showed the presence of spores in groups and normal morphology of hyphae possessing dense cytoplasm Fig. 4A and 4B. Whereas, on treatment with M. piperita Fig. 4C, 4D and M. arvensis 4E and 4F essential oil, scattered spores in the form of smaller groups and as short spore chains respectively.
FIG. 4: OPTICAL RESEARCH MICROSCOPY SHOWING HYPHAE AND SPORE MORPHOLOGY OF CONTROL 4(A) & 4(B) FUNGUS TREATED WITH 500 µg/mL M. ARVENSIS EO 4(C) & 4(D) FUNGUS TREATED WITH M. PIPERITA EO 4(E) & 4(F)
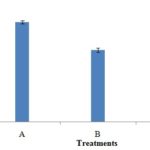
FIG. 5: HYPHAL DIAMETER (µM) OF CONTROL (A) AND FUNGAL MYCELIA TREATED WITH M. PIPERITA (B) AND M. ARVENSIS(C)
The optical micrographs also exhibited modified hyphal morphology indicated by a decrease in diameter and number of hyphal branches than control. Moreover, the cytoplasm was less dense exhibiting shrinkage which led to the fragmentation of fungal hyphae, loss of linearity including irregular hyphae and formation of warty surfaces (cell wall pitting phenomena) in fungus treated with M. piperita essential oil. The hyphal diameter was observed to be greater in untreated fungus followed by fungus treated with M. arvensis and M. piperita essential oil Fig. 5.
DISCUSSION: Percent yield of essential oils of both the species are well in correlation with the available literature with minute variations 13, 16. The GC-MS of essential oil of both the Mentha species showed a broad spectrum of compounds with distinct variations. These variations are well explained due to different climatic conditions in which the crop was reared. Further, time, the season of sample collection and soil composition of the sampling area also contributed to the composition of essential oil.
Almost all chemical compounds reported in essential oil from fresh leaves of M. piperita exhibited one or the other biological activity 17, 18. Differences in the compositional analysis by GC-MS correlate very well with the FT-IR spectra of both species essential oils since more predominant bands were observed for M. piperita essential oil than M. arvensis essential oil. The FT-IR spectra of M. piperita depicted more interactions between various compounds may be because of greater number of compounds in it as shown by its GC-MS. The position of the absorption band due to valence vibration of O-H bonds can be used to quantify hydrogen bonding. When hydrogen bonding is stronger, the O-H bond length increases and bond force constant decreases, so the valence vibration is identified at lower frequency values compared to the values determined in the absence of association with hydrogen bonds. The association through hydrogen bonds led to wide band at 3367.1 cm-1 in M. piperita essential oil and sharp band at 3346 cm-1 in M. arvensis essential oil and a red shift occurred due to O-H stretching by 11.1 cm-1 of M. arvensis essential oil in comparison to M. piperita essential oil.
Samfira et al., 19 reported O-H stretch at 2950 cm-1 and C=O peak at 1700 cm-1 for M. piperita essential oil extracted from leaves collected from the western part of Romania. Prakash and Yunus 20 found O-H stretch at 3355.6 cm-1 and C=O peak at 1702.5 cm-1 for M. arvensis essential oil extracted from leaves collected from Lucknow city. The shift in frequency bands of functional groups was observed due to climatic changes. The results from poisoned food technique followed by percentage inhibition calculation showed more percent inhibition with less number of colonies in M. piperita at all tested concentrations than in M. arvensis at similar concentrations. At the concentration of 1000µg/mL, both species essential oils showed antifungal activity at par with standard Carbendazim 50 WP. Results observed in our studies confirmed enhanced efficacy of M. piperita essential oil (71%) compared to M. arvensis essential oil (55%) at 500µg/ml in controlling the dreaded post-harvest fungus P. digitatum. The essential oil of M. piperita leaves possess significant antifungal activity may be due to the presence of pulegone, isomenthone, menthofuran, and neomenthone, which were identified absent in M. arvensis essential oil. Optical research microscopy of fungus treated with essential oils revealed fragmentation of fungal hyphae, loss of linearity including irregular hyphae and formation of warty surfaces. In fungus treated with M. piperita essential oil malformed conidiophore structures were recorded which culminated to the formation of impaired conidia.
All these findings indicate that the antifungal mode of action of essential oil on P. digitatum is a result of damage to the cell plasma membrane integrity. Owing to our results, the similar phenomenon of the lipophilicity of essential oils causing membrane expansion, augmented membrane permeability and fluidity, inhibition of respiration and variation in ion transport processes of fungi which results into induced the outflow of other cellular components was observed in earlier studies 21, 22, 23.
CONCLUSION: The percent yields of M. piperita and M. arvensis essential oil obtained from leaves was calculated. The comparison showed that M. arvensis leaf essential oil yield (2.8%) was higher as compared to Mentha piperita (2.1%). Gas chromatography-Mass Spectrometry (GC-MS) of M. piperita and M. arvensis essential oil revealed the presence of 51 and 30 compounds respectively, menthol being the major compound in both essential oils. The FT-IR spectra of essential oil of both Mentha species showed prominent variation in compounds indicated by different shape and position of IR bands. Screening of antifungal activity revealed that M. piperita essential oil (71%) was found to be more effective than M. arvensis (55%) against P. digitatum. Optical research microscopy further revealed that the essential oils showed their efficacy by disrupting the cell plasma membrane, thereby causing the fragmentation of fungal hyphae.
ACKNOWLEDGEMENT: The authors wish to thank the Director, Punjab Horticultural Post-Harvest Technology Centre, Punjab Agricultural University, Ludhiana, Punjab and the Director, EMNL, for providing the necessary infrastructural facilities and Head, Department of Chemistry, College of Basic Sciences and Humanities, Punjab Agricultural University, Ludhiana, Punjab, India for providing the contingency for purchase of consumables for this study.
CONFLICT OF INTEREST: The author declares no conflict of interest.
REFERENCES:
- Kaur N, Bala A and Kumar A: Prevent kinnow produce from post harvest disorders and diseases. Rashtriya Krishi 2014; 9(1): 45-46.
- Mandal G: Effect of lac-wax, citrashine and individual shrink wrapping of fruits on storage life of late harvested kinnow under ambient condition. IJBEAS 2015; 1(3): 84-89.
- Miguel MG: Antioxidant and anti-inflammatory activities of essential oil: A short review. Molecules 2010; 15: 9252-9287.
- Suntar I, Akkol EK, Tosun A and Keles H: Comparative pharmacological and phytochemical investigation on the wound-healing effects of the frequently used essential oils. J Essent Oil Res 2014; 26(1): 41-49.
- Bozovic M, Pirolli A and Ragno R: Mentha suaveolensehrh (Lamiaceae) essential oil and its main constituent piperitenone oxide: Biological activities and chemistry. Molecules 2015; 20: 8605-8633.
- Dadalioglu I and Evrendilek GA: Chemical compositions and antibacterial effects of essential oils of turkish oregano (Origanum minutiflorum), bay laurel (Laurus nobilis), spanish lavender (Lavandula stoechas) and fennel (Foeniculum vulgare) on common food borne pathogens. J Agric Food Chem 2004; 52: 8255-8260.
- Bruneton J: Pharmacognosy, Phytochemistry, Medicinal plants: Essential oils. Lavoisier Publishing, Paris. Edition 2nd, 1999: 461-780.
- Bakkali F, Averbeck S, Averbeck D and Idaomar M: Biological effects of essential oils-A review. Food Chem Toxicol 2008; 46: 446-475.
- Elansary HO and Ashmawy NA: Essential oils of mint between benefits and hazards. J Essent Oil Bear Plant 2013; 16(4): 429-438.
- Kumar P, Mishra S, Malik A and Satya S: Insecticidal properties of Mentha species. Ind Crops Prod 2011; 34: 802-817.
- Mirzaie-Nodoushan H, Rezale MB and Jaimand K: Path analysis of the essential oil-related characters in Mentha sp. Flavour Frag J 2001; 16(6): 340-343.
- Chaddha KL and Gupta R: Japanese mint. Adv Horticult 1995; 11: 687-725.
- Moghtader M: In-vitro antifungal effects of the essential oil of Mentha piperita and its comparison with synthetic menthol on A. niger. Afr J Plant Sci 2013; 7(11): 521-27.
- Luca A and Ciucanu I: Solid-phase microextraction of menthol from peppermint. Ser Chem 2011; 20(1): 71-80.
- Balouiri M, Sadiki M and Ibnsouda SK: Methods for in-vitro evaluating antimicrobial activity: A review. J Pharma Anal 2016; 6: 71-79.
- Gende LB, Mendiara S, Fernandez NJ, Baren CV, Lira AD, Bandoni A, Fritz R, Floris I and Eguaras M: Essentials oils of some Mentha sp. and their relation with antimicrobial activity against larvae, the causative agent of American foulbrood in honey bees, by using the bio-autography technique. Bul of Insectol 2014; 67(1): 13-20.
- Satoyanova A and Georgiev E: Chemical composition and antimicrobial activity of Bulgarian peppermint oils. Bul Ess Oil Perfum Cosm (Bulgaria) 2003; 43-47.
- Georgiev S and Topalova V: Chemical composition and antimicrobial activity of Bulgarian peppermint oils. Plant Sci 1989; 26: 33-37.
- Samfira I, Rodino S, Petrache P, Cristina RT, Butu M and Butnariu M” Characterization and identity confirmation of essential oils by mid infrared absorption spectro-photometry. Digest Journal of Nanomaterials and Bio-structures 2015; 10(2): 557-566.
- Prakash N and Yunus M: Fourier transforms infrared spectroscopy analysis of oil of Mentha arvensis grown at sites varying with vehicular traffic loads in Lucknow city, India. Int J Environ 2013; 2(1): 16-25.
- Wu Y, Yang O and Tao N: Plasma membrane damage contributes to the antifungal activity of citronellal against digitatum. J Food Sci Technol 2016; 53(10): 3853-58.
- Tyagi AK and Malik A: Liquid and vapor-phase antifungal activities of selected essential oils against albicans: microscopic observations and chemical characterization of C. citratus. BMC Complement Altern Med 2010; 10: 1-65.
- Zhou HE, Tao NG and Jia L: Antifungal activity of citraloctanal and α-terpineol against Geotrichumcitri aurantii. Food Control 2014; 37: 277-283.
How to cite this article:
Kaur H, Tandon R, Kalia A and Maini C: Chemical composition and antifungal activity of essential oils from aerial parts of Mentha piperita and Mentha arvensis. Int J Pharmacognosy 2018; 5(12): 767-73. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(12).767-73.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
767-773
799
1539
English
IJP
H. Kaur, R. Tandon *, A. Kalia and C. Maini
Department of Chemistry, Punjab Agricultural University, Ludhiana, Punjab, India.
ritutandon@pau.edu
31 October 2018
22 November 2018
27 November 2018
10.13040/IJPSR.0975-8232.IJP.5(12).767-73
01 December 2018