CHARACTERIZATION OF PHYTOCHEMICAL AND ANTIOXIDANT PROPERTIES OF CITRUS AURANTIFOLIA (KAGOJI LEBU) AVAILABLE IN BANGLADESH AND ITS PROTECTIVE EFFECTS AGAINST EXPERIMENTALLY INDUCED MYOCARDIAL INFARCTION IN RATS
HTML Full TextCHARACTERIZATION OF PHYTOCHEMICAL AND ANTIOXIDANT PROPERTIES OF CITRUS AURANTIFOLIA (KAGOJI LEBU) AVAILABLE IN BANGLADESH AND ITS PROTECTIVE EFFECTS AGAINST EXPERIMENTALLY INDUCED MYOCARDIAL INFARCTION IN RATS
Alamgir Kabir, Rezaul Karim, Md. Yousuf Ali, Md. Ibrahim Khalil and M. Ibrahim Hossain *
Laboratory of Preventive and Integrative Biomedicine, Department of Biochemistry and Molecular Biology, Jahangir Nagar University, Savar, Dhaka - 1342, Bangladesh.
ABSTRACT: Plants and fruits have medicinal purposes, and they are being used long before the prehistoric period. Among the different varieties of citrus fruits, Citrus aurantifolia (Kagoji labu) is widespread in tropical and subtropical regions around the World. It is one of the major citrus fruits and widely consumed, but there is limited evidence about its health-promoting properties. In the present study, we evaluated the phytochemical and antioxidant properties of this citrus fruit since phytochemicals through anti-oxidant properties have played a significant role in human health protection and treatment of many diseases. Polyphenol, flavonoid, tannin and reducing sugar contents were found 4.68 ± 0.36 g GAE / 100 g extract, 615.38 ± 17.75 mg CE / 100 g extract, 12.5 ± 6.25 g TAE / 100 g extract and 51.69 ± 7.50 g GE / 100 g extract, respectively. Significant antioxidant activities of C. aurantifolia via 2, 2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging and ferric reducing antioxidant power (FRAP) were detected. Earlier studies have reported that almost all species of citrus fruits have significant antioxidant properties and are effective against stress-induced ulcers, cancer, and other chronic diseases. Therefore, next, we asked about the protective effects of C. aurantifolia extract on isoproterenol (ISO) induced myocardial infarction (MI). Subcutaneously injected ISO (a well-known chemical to produce MI in experimental animals) causes a significantly (p<0.01) increase of serum concentrations of lactate dehydrogenase (LDH) and creatine kinase-MB (CK-MB), two myocardial cell-specific enzymes which are increased in blood after MI, indicating ISO injection produced MI. Hematoxyline and eosin (H and E) staining of the heart showed relatively thin and abnormal myocardial cell fibers in ISO injected rats when compared with normal control. Treatment of rats with C. aurantifolia (1000 mg/kg body weight) followed by ISO injection significantly (p<0.01) decrease the serum concentrations of LDH and CK-MB, indicating the protective effects of this fruit against ISO induced MI. H and E staining also showed treatment with C. aurantifolia followed by ISO injection causes the myocardial cell fibers to remain as thick as normal control rats. Together all data suggests that C. aurantifolia are rich in phytochemical properties, and it has protective effects against ISO induced MI.
| Keywords: |
Phytochemical, Antioxidant, Myocardial infraction, Isoproterenol
INTRODUCTION: Despite improved clinical care, the availability of modern medicines and
greater health awareness, the world health organization (WHO) has predicted that cardio-vascular disease (primarily myocardial infarction) will be a major cause of death worldwide by the year 2020 1, 2.
Myocardial infarction (MI), a common presentation of ischemic heart disease (IHD) occurs when cardiac ischemia surpasses a clinical threshold, resulting in irreversible myocardial damage.
IHD is an acute condition leading to necrosis of the myocardium as a result of an imbalance between myocardial metabolic demands and the coronary supply of oxygen and nutrients 3.
MI leads to free radical generation in the heart, which contributes to further toxic reactions and eventually, cardiac cell death 4. Reactive oxygen species (ROS) such as free radicals, oxygen ions, and peroxides are generated during aerobic metabolism as by-products and are tightly controlled by antioxidants 5. However, excess production of ROS or depletion of antioxidants can lead to a state of oxidative stress that can inflict damage to lipids, proteins, and DNA 6. Following MI, ROS production is usually increased, which can lead to further damage to the myocardium. The first line of cellular defense against oxidative injury in the heart, as well as most tissues, includes antioxidant enzymes 7.
Dietary antioxidants can prevent the deleterious effects of ROS by restoring the balance between production and clearance of ROS by mechanisms such as scavenging ROS or enhancing endogenous antioxidant enzyme activity 8. Natural products have high global demands because of their purported superiority in terms of both safety and efficacy against oxidative stress-induced cardiovascular disease, including MI 9.
Citrus fruits have been a natural boon to mankind for years. Earlier studies have reported that almost all species of citrus fruits have significant antioxidant properties and are effective against stress-induced ulcers, cancer, and other chronic diseases 10. Citrus aurantifolia (locally known as Kagoji labu) is widespread in tropical and subtropical regions around the world, such as North America (Florida, Texas, California, Mexico, etc.), India, Bangladesh, Egypt and Central America 11.
It is one of the major citrus fruits and widely consumed, but there is limited evidence about its health-promoting properties. Lime essential oils are not only used as flavoring agents in beverages, manufactured foods, and pharmaceutical firms but also as ingredients in perfumes 11. Additionally, C. aurantifolia is used in traditional medicine as an antiseptic, anthelmintic, mosquito bite repellent, for stomach ailments, tonic, antiscorbutic, astringent, diuretic, headache and arthritis and digestive and appetite stimulant and for colds, coughs and sore throats 12.
Previous investigations have shown the presence of different phytochemical compounds such as flavonoids, coumarins, and terpenoids in C. aurantifolia 13. Lime peel oil has shown antimicrobial 14, radical scavengings, anti-cholinesterase 15, anthelmintic 16, and anticancer activities 17. Furthermore, leaves of lime showed a protective effect against osteoporosis 18 and induced platelet aggregation 19. Isoproterenol (ISO) is a synthetic catecholamine and β-adrenergic agonist that causes severe stress to the myocardium, resulting in infarct-like necrosis of the heart muscle if administered in high doses. It has been reported that path physiological and morphological changes of ISO-induced cardiac dysfunctions in laboratory animals are comparable to those in humans suffering from MI. Studies have shown that hypoxia is the major cause of ISO-induced cardiac damage because of myocardial hyperactivity, coronary hypotension, and excessive generation of highly cytotoxic free radicals resulting from the auto-oxidation of catecholamine’s 20.
Following oxidation, catecholamine’s form quinoid compounds, which stimulate the production of superoxide anions and subsequently, hydrogen peroxide. Hydrogen peroxide becomes a highly reactive hydroxyl radical in the presence of iron, causing oxidative damage to preserved lipids, proteins, and DNA, ultimately affecting the infracted myocardium 21. In the present study, we first characterized the phytochemical and antioxidant properties of Citrus aurantifolia and asked that Citrus aurantifolia has protective effects against ISO-induced MI in rats. For this, we made MI in rats by subcutaneous injection of ISO. The serum concentration of creatine kinase-MB (CK-MB) and lactate dehydrogenase (LDH) were significantly increased following ISO injection, and thin heart muscle of ISO injected rats observed by H and E staining indicating ISO causes MI in rats. Treatment of ISO injected rats with ethanolic extract of Citrus aurantifolia reverse all the serum parameters used to monitor MI in rats indicating protective effects these citrus fruits against MI in rats.
MATERIALS AND METHODS:
Chemicals and Reagents: All chemicals were newly purchased for experiments. Gallic acid (3, 4, 5-trihydroxybenzoic acid), tannic acid, catechin, folin-ciocalteu reagent, rutin, quercetin, sodium acetate (CH3COONa), sodium hydroxide (NaOH), vanilic acid, acetic acid (CH3COOH), 2, 4, 6-Tri (2-pyridyl)-s-triazine (TPTZ), green vitriol (FeSO4-7H2O), aluminum chloride (AlCl3), ferric chloride (FeCl3) 2, 2-diphenyl-1-picrylhydrazyl (DPPH), ascorbic acid (vitamin C), sulfuric acid (H2SO4), 2, 4-dinitrophenyl hydrazine-thiourea-copper (DTC), potassium ferricyanide [K3Fe(CN)6], trichloroacetic acid (TCA), blue vitriol (CuSO4-5H2O), sodium potassium tartrate (KNaC4H4O6-4H2O) and calcium chloride (CaCl2) were used for the determination of phytochemical and antioxidant activities.
Sample Collection and Extract Preparation: Mature and fresh Citrus aurantifolia fruits were purchased from the markets of Tangail district, Bangladesh, in September 2017. The fruits were authenticated as Citrus aurantifolia by a specialist, and selected fruits were washed with clean, sterile water and air-dried. Fruits were then cut into small pieces and dried with sunlight. The ethanolic extract of Citrus aurantifolia was prepared according to the previous method 22.
In brief, using a blender (Jaipan Commando, Mumbai, India) fine powder of Citrus aurantifolia was made, and then, powder samples were dissolved with sufficient amount (20% w/v) of pure ethanol (100%) and were put into a shaker (IKA 400 i.e., Germany) at 150 rpm at 30 °C for 72 h. Whatman No. 1 was used to filter the extract of fruits, and the crude extract was evaporated using a rotatory evaporator (R-215 BUCHI, Switzerland) under reduced pressure (100 Psi) and controlled temperature at 40 °C. It was finally concentrated and stored at -20 °C until further use.
Phytochemical Analysis: The following tests were conducted to estimate different phytochemical properties of C. aurantifolia.
- The total polyphenol content of methanolic extract of whole aurantifolia was estimated by spectrometric determination following modified Folin-Ciocalteu’s method 23. Gallic acid was used as the standard for this method.
- The total flavonoid content of whole aurantifolia was estimated by using aluminum chloride colorimetric assay 24. Catechin was used as the standard.
- The total tannin content in methanol extracts of whole aurantifolia were estimated by using Folin-Ciocalteu’s method with slight modifications 25. Tannic acid was used as the standard.
- The total protein content of whole aurantifolia was estimated using Lowry’s method 26 of protein estimation. Bovine serum albumin (BSA) was used as the standard.
- Reducing sugar content was determined using nelson-somogyi method 27, and glucose was used as the standard in this process.
In vitro Antioxidant Activity Analysis:
In-vitro Anti-oxidant Properties of C. aurantifolia were Examined by the Following Two Methods:
DPPH Free Radical-Scavenging Activity: The percentage of antioxidant activity (AA %) of methanol extracts of whole C. aurantifolia was assessed by DPPH (1, 1-diphenyl-2-picryl-hydrazyl) free radical assay. The measurement of the DPPH radical scavenging activity was performed according to the methodology established by Manzocco et al. 28. Ascorbic acid was used as the standard. DPPH free radical scavenging activity of ethanolic extract of C. aurantifolia was investigated to determine their antioxidant properties.
Ferric Reducing Antioxidant Power (FRAP) Assay: The measurement of FRAP assay was performed according to the methodology established by Benzie and Strain 29 in which green vitriol was used as the standard.
Experimental Animals and Induction of MI: 60 adult male Wister rats with a bodyweight range of 125 to 200 g at between 15-18 weeks of age. All the animals were reared in the animal house facility of the Department of Biochemistry and Molecular Biology, Jahangir Nagar University, at the standard condition of temperature and humidity. A standard laboratory pellet diet and water ad libitum were provided to the rats on a regular basis. The experimental protocol was approved by the biosafety, biosecurity, and ethical. Committee of Jahangirnagar University, Savar, Dhaka.
MI was induced by subcutaneous injection of isoproterenol (ISO) (85 mg/kg body weight dissolved in physiological saline) at an interval of 24 h for two consecutive days. The ISO dose was based on a pilot study for ISO dose fixation and the results of previous studies 30. Animals were sacrificed 48 h after the first ISO dose. After acclimatization, the experimental rats were randomly divided into the three groups consisting of 20 rats each-
Group 1 (Control): Animals received standard laboratory diet and drinking water ad libitum and serve as a normal control group.
Group 2 (ISO-control): Animals were injected with isoproterenol (85 mg/kg bw) subcutaneously on 27th and 28th day (at an interval of 24 h) in saline and serve as a negative control group.
Group 3 (ISO-kagoji): Animals were orally treated with C. aurantifolia (1000 mg/kg body weight) for a period of 28 days and were injected with isoproterenol (85 mg/kg BW) subcutaneously on 27th and 28th day (at an interval of 24 h) in saline and serve as a preventive group. After the experimental periods, all the rats were perfused with 4% paraformaldehyde (PFA) transcardially. Before perfusion, a sufficient amount of blood samples were collected. Blood samples (4 mL) were placed in dry test tubes and were allowed to coagulate at ambient temperature for 30 min.
Serums were separated by centrifugation at 2000 rpm for 10 min. Serums were stored at -80 °C until use. After perfusion, brains were quickly removed and proceed to paraffin embedding for histological staining.
Estimation of Biochemical Parameters: Serum levels of cardiac function parameters (CK-MB and LDH) were measured by standard protocols provided by the manufacturer.
Statistical analysis: Results are represented as Mean ± SEM. P< 0.05 was considered statistically significant. Statistical analysis was conducted using internet-based one-way or two-way ANOVA with Tukey’s test when applicable.
RESULTS AND DISCUSSION: Phytochemicals or bioactive compounds such as polyphenols, flavonoids, and tannin can donate electrons and are therefore regarded as very powerful antioxidants. Besides acting as antioxidants against singlet oxygen molecules, hydrogen peroxide, hydroxyl radicals, superoxide radicals and other pro-oxidants, they play a major role as colouring and flavouring agents of plants which also contribute to their functional features.
Polyphones are effective in histone modification, miRNA regulation and DNA methylation and thus can alter the epigenetic processes against carcinogenesis and cancer development 32. Accumulating data indicates that Citrus aurantifolia has different health beneficial roles such as an antiseptic, anthelmintic, mosquito bite repellent, for stomach ailments, tonic, ant scorbutic and astringent, diuretic, headache, arthritis and digestive and appetite stimulant and for colds, coughs and sore throats 12. In the present study, first we determined the photochemical (such as polyphenols, flavonoids, tannins and reducing sugar content) and antioxidant properties of the ethanolic extract of C. aurantifolia and second we examined the protective effects of ethanolic extract of C. aurantifolia against MI.
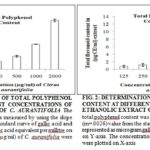
In order to determine the total polyphenol content, we used gallic acid as standard Fig. 1 and therefore, the results are represented as µg GAE/ml of extract of C. aurantifolia. The maximum total polyphenol content was obtained in ethanolic extract of C. aurantifolia (4.68 ± 0.36 g GAE/100 g extract) at a concentration of 125 µg/ml with a very marginal difference to other concentrations like 4.60 ± 0.34 g GAE / 100 g extract at 250 µg/ml and 3.98 ± 0.13 g GAE / 100 g extract at a concentration of 500 µg/ml of C. aurantifolia. Therefore, it suggests that at different concentrations of the ethanolic extract of C. aurantifolia there is an almost maximum and same total polyphenolic compound. The health benefits of phenolics are primarily derived from their antioxidant potentials because the radicals produced after hydrogen or electron donation is resonance stabilized and thus relatively stable 33.
Use of phenolics is also reported for effective secretion of dopamine, lowering and preventing obesity and prevention of oxidative stress 34. These phenolic compounds from natural sources are recommendable as natural food additives and they are considered more suitable for application in food products than butylated hydroxytoluene and butylated hydroxyanisole which are artificial compounds with antioxidant properties 35.
Total flavonoid content (TFC) was measured by suing catechin as the standard Fig. 2, and the results are represented as µg CE/ml of an extract of C. aurantifolia. The maximum total flavonoid content was obtained in the ethanolic extract of C. aurantifolia (615.38 ± 17.75 mg CE / 100 g extract) at a concentration of 1000 µg/ml.
Flavonoids are the plant pigments responsible for plant colors and exert their health-promoting activities through their high pharmacological potentials as radical scavengers 36. Flavonoids are the antioxidants that can prevent or delay the oxidation of substrates even when it is present in low concentrations, so as to prevent oxidation by the prooxidants (ROS and RNS).
These non-enzymatic antioxidants (phenolics and flavonoids) react with the pro-oxidants leading to inactivation. In the redox reaction, the antioxidants act as reluctant and serve as the first-line defense to suppress the formation of free radicals 37.
The flavonoids have a strong inherent ability to modify the body’s reaction to allergens, viruses, and carcinogens. Flavonoids are a class of secondary plant metabolites with significant antioxidant and chelating properties. They have anti-allergic, anti-inflammatory, anti-microbial, and anti-cancer activity 38.
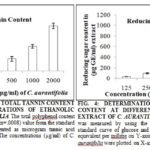
Total tannin content (TTC) was measured by using tannic acid as the standard Fig. 3 Results are represented as µg TAE/ml of an extract of C. aurantifolia. The maximum TTC was obtained in ethanolic extract of C. aurantifolia (12.5 ± 6.25 g TAE / 100 g extract) at a concentration of 125 µg/ml.
A number of uses of different parts of Citrus fruits in medicine, especially in traditional Chinese medicine, have also been reported, and this might be attributed to the presence of tannin in them. Many developing countries have been using plant materials in primary health care for the treatment of various types of diseases. These parts, namely peel and pulp of this Citrus species, could be therefore used in ethnomedicine as drugs. Their extracts could also be used in cosmetic industries also as antimicrobial agents. Therefore, they could be used in the treatment of animal diseases. It is noted that tannin-containing plants could possibly be used to prevent diarrhea in pigs 39.
This is probably because of the antidiarrheal property of tannin. Tannins serve as a natural defense mechanism against microbial infections. Tannins have also been reported to exert other physiological effects, such as to accelerate blood clotting, reduce blood pressure, decrease the serum lipid level, produce liver necrosis, and modulate immune responses.
Reducing sugar content was determined using Nelson-Somogyi method and glucose was used as the standard Fig. 4. Results are represented as µg GE/ml of extract of C. aurantifolia. Reducing sugar is an important component in Kagoji lemon. Ethanolic extract of kagoji lemon showed a high amount of reducing sugar (51.69 ± 7.50 g GE / 100 g extract). Carbohydrate-containing compounds are promising way to synthesize drugs that do not only save pharmacological properties of an initial agent but also acquire a number of advantageous features namely increased bioavailability, water solubility, and protection from quick metabolism in the body 40.
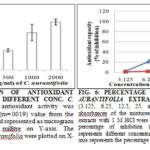
Next, we tried to evaluate the antioxidant properties of extract of C. aurantifolia. Antioxidant potential of ethanolic extract of kagoji lemon was investigated though ferric reducing antioxidant power (FRAP) assay and DPPH free radical scavenging assay. In FRAP assay, green vitriol was used as standard and results are represented as µmol GVE/1000 ml extract of kagoji lemon Fig. 5. The highest FRAP value of ethanolic extract of kagoji lemon was found to be 6315.78 µmol GVE / 100 g extract at the concentration of 500 µg/ml. FRAP assay treats the antioxidants in the sample as reductants in a redox reaction and measures the reducing potential of test sample.
The antioxidant exerts its activities by donating electron or hydrogen atoms to the ferric complex before being further converted to ferrous complex (Fe3+ to Fe2+ -TPTZ complex) thus, breaking the radical chain reaction. In other methods, the ability to scavenge 2, 2-diphenyl-1-picrylhydrazyl (DPPH) free radicals was determined where ascorbic acid was used as the standard. DPPH free radical scavenging activity of ethanolic extract of kagoji lemon was investigated to determine their antioxidant properties.
The findings are expressed as a percentage (%) of inhibition against the concentrations. In comparison with the standard, kagoji lemon showed very mild anti-oxidant activities, Fig. 6.
These phytochemicals serve many important functions in our body, such as polyphenols are effective in reducing the risk of cardiovascular diseases (CVD). It also plays an active role in endothelial dysfunction, inflammation and inflammatory diseases. Therefore, our next aim was to examine the protective effects of ethanolic extracts of C. aurantifolia extracts on ISO induced MI in rats. We found that extracts of C. aurantifolia significantly improves cardiac functions as measured by heart functions specific enzymes (LDH and CK-MB) after ISO induced MI in rats. We also reported that ISO-induced MI causes a change in lipid profile with a significant increase in TG and TC. C. aurantifolia extracts significantly lowers the serum TG levels in rats after MI induced by ISO but serum TC levels were not significantly decreased by this citrus fruit extracts after MI. Our data indicates that C. aurantifolia has protective effects against cardiovascular diseases like MI.
ISO is a well-known chemical to produce abnormal myocardial functions by different mechanisms like oxidative stress, coronary insufficiency, altered metabolism, ionic imbalance, necrosis and apoptosis.31 Due to necrosis and apoptosis, different cellular enzymes come into the blood circulation. Therefore, at first, we checked ISO could potentially produce MI in our experimental rats or not by measuring the serum concentration of LDH and CK-MB, two heart-specific cellular enzymes.
Lactate dehydrogenase (LDH) has five isozymes, which are found in different tissues such as heart, liver, lungs, RBC, brain etc. Among these five isozymes, LDH-1 (more commonly known as only LDH) is predominantly found in the heart.41 When there is any necrosis or apoptosis in myocardial cells due to heart attack or MI, LDH is released from apoptotic cells into the bloodstream and subsequently its concentration increases in the blood serum. In the blood, the concentration of LDH increases more quickly than other heart marker troponin-I (TnI) after a heart attack or MI 42. Therefore, we also determined the serum concentrations of LDH after ISO injection.
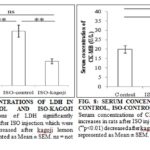
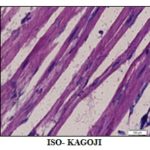
Serum concentrations of LDH are significantly (p<0.01) higher in the ISO-control rats (rats received only ISO injection) than that of normal control rats (rats not received ISO injection) Fig. 7. Therefore, ISO injection significantly increases serum concentrations of LDH, indicating the necrosis or apoptosis of myocardial cells. This result suggests that ISO injection cause MI. In order to verify the result showed by LDH that ISO induce MI, we determined the serum concentrations of another myocardial cell-specific enzyme, namely CK-MB. Creatine kinase (CK) is an enzyme which is also found in different tissues such as heart, brain, and muscle. CK-MB is the isozymes of CK, which is specifically found in the heart. Therefore, we verified the results of LDH by determining the serum concentrations of CK-MB and compared between normal control and ISO-control. Serum concentrations of CK-MB also significantly (p<0.01) increased after ISO injection Fig. 8. Results of both LDH and CK-MB indicate that ISO injection induces MI in rats. Hematoxyline and Eosin staining of the heart showed that the fibers of the heart of ISO injected rats were thinner than that of normal control. After C. aurantifolia treatment, myofibers looked very much similar to that of the normal control indicating protecting functions of C. aurantifolia on heart muscle cells after ISO injection Fig. 9.
FIG. 9: HEMATOXYLENE AND EOSIN (H AND E) STAINING OF HEART MUSCLE OF NORMAL CONTROL, ISO-KAGOJI AND ISO-CONTROL ISO-control showed thin my fibers than normal control and ISO-KAGOJI.
Next, we checked C. aurantifolia treatment might have some beneficial effects on the ISO induced MI. Treatment with the extract of C. aurantifolia significantly (p<0.01) decreased the serum concentrations of both LDH and CK-MB Fig. 7 and Fig. 8 indicating the protective effects of C. aurantifolia on ISO induced MI. ISO is a well-known chemical compound that is used to produce experimental MI in animals. This statement is also consistent with our experimental rats as we found increased serum concentrations of LDH and CK-MB in ISO injected rats, indicating ISO produces MI in our experimental rats. Serum concentrations of both of the myocardial specific enzymes (LDH and CK-MB) decreased to almost normal levels when these rats are treated with C. aurantifolia extract after ISO injection indicating protective effects of this fruit extract.
CONCLUSION: It has already found that C. aurantifolia possesses various beneficial effects such as antimicrobial, radical scavenging, anti-cholinesterase, anthelmintic, and anticancer activities. In addition to these, the current study demonstrated that C. aurantifolia extract also showed protective roles against MI. Here we show that C. aurantifolia extract possesses phytochemical properties like significant amounts of polyphenols, flavonoids, tannins, and reducing sugar properties as well as antioxidant properties. Next examined the protective effects of C. aurantifolia extract against ISO induced MI. We found that serum concentration of LDH and CK-MB were significantly decreased after ISO injection when the rats treated with C. aurantifolia extract. Therefore, current study uncovers another beneficial role of C. aurantifolia in protecting heart.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Aronow WS: Epidemiology, path physiology, prognosis, and treatment of systolic and diastolic heart failure. Cardiology in Review 2006; 14: 108-24.
- Murray CJ and Lopez AD: Alternative projections of mortality and disability by cause 1990-2020: Global burden of disease study. The Lancet 1997; 349: 1498-04.
- Anversa P and Sonnenblick EH: Ischemic cardiomyopathy: Path physiologic mechanisms. Progress in Cardiovascular Diseases 1990; 33: 49-70.
- Cai Q, Rahn RO and Zhang RR: Dietary flavonoids, quercetin, luteolin and genistein, reduce oxidative DNA damage and lipid per oxidation and quench free radicals. Cancer Letters 1997; 119: 99-07.
- Droge W: Free radicals in the physiological control of cell function. Physiological Reviews 2002; 82: 47-95.
- Papaharalambus CA and Griendling KK: Basic mechanisms of oxidative stress and reactive oxygen species in cardiovascular injury. Trends in Cardiovascular Medicine 2007; 17: 48-54.
- Rodrigo R, Prieto JC and Castillo R: Cardio protection against ischemia/reperfusion by vitamins C and E plus n− 3 fatty acids: molecular mechanisms and potential clinical applications. Clinical Science 2013; 124: 1-15.
- Han X, Shen T and Lou H: Dietary polyphenols and their biological significance. International Journal of Molecular Sciences 2007; 8: 950-88.
- Patel V, Upaganlawar A, Zalawadia R and Balaraman R: Cardio protective effect of melatonin against isoproterenol induced myocardial infarction in rats: A biochemical, electrocardiographic and histoarchitectural evaluation. European Journal of Pharmacology 2010; 644: 160-68.
- Sood S, Muthuraman A, Arora B, Bansal S, Bali M and Sharma PD: Potential effect of Citrus decumana extract on stress induced peptic ulcer in rat. Lat Am J Pharm 2010; 29: 52-56.
- Morton JF: A textbook of Fruits of warm climates. Echo Point Books & Media. 1987.
- Apraj V, Thakur DN, Bhagwat A, Mallya R, Sawant L and Pandita N: Pharmacognostic and phytochemical evaluation of Citrus aurantifolia (christm) swingle peel. Pharmaco Journal 2011; 3: 70-76.
- Piccinelli AL, Garcia Mesa M, Armenteros DM, Alfonso MA, Arevalo AC, Campone L and Rastrelli L: HPLC-PDA-MS and NMR characterization of c-glycosyl flavones in a hydro alcoholic extract of Citrus aurantifolia leaves with ant platelet activity. Journal of Agricultural and Food Chemistry 2008; 56: 1574-81.
- Chisholm MG, Wilson MA and Gaskey GM: Characterization of aroma volatiles in key lime essential oils aurantifolia swingle. Flav and Frag Jl 2003; 18: 106-15.
- Tundis R, Loizzo MR, Bonesi M, Menichini F, Mastellone V, Colica C and Menichini F: Comparative study on the antioxidant capacity and cholinesterase inhibitory activity of Citrus aurantifolia Swingle, aurantium L. and C. bergamia risso and poit. Peel essential oils. Journal of Food Science 2012; 77: 40-46.
- Taur DJ: Anthelmintic activity of Ocimum sanctum and Citrus aurantifolia Pharmacology Online 2009; 3: 495-99.
- Gharagozloo M, Doroudchi M and Ghaderi A: Effects of Citrus aurantifolia concentrated extract on the spontaneous proliferation of MDA-MB-453 and RPMI-8866 tumor cell lines. Phyto Medicine 2002; 9: 475-77.
- Kawaii S, Tomono Y, Katase E, Ogawa K and Yano M: Isolation of furocoumarins from bergamot fruits as HL-60 differentiation-inducing compounds. Journal of Agricultural and Food Chemistry 1999; 47: 4073-78.
- Shalaby NMM, Abd-Alla HI, Ahmed HH and Basoudan N: Protective effect of Citrus sinensis and Citrus aurantifolia against osteoporosis and their phytochemical constituents. J of Medicinal Plants Res 2011; 5: 579-88.
- Rona G: Catecholamine cardio toxicity. Journal of Molecular and Cellular Cardiology 1985; 17: 291-06.
- Dhalla NS, Temsah RM and Netticadan T: Role of oxidative stress in cardiovascular diseases. Journal of Hypertension 2000; 18: 655-73.
- Paul S, Islam MA, Tanvir EM, Ahmed R, Das S, Rumpa NE, Hossen MS, Parvez M, Gan SH and Khalil MI: Satkara (Citrus macroptera) fruit protects against acetaminophen-induced hepatorenal toxicity in rats. Evid Based Complement Alternat Med 2016: 9470954.
- Singla P and Kaur G: A textbook on Citrus fruits and cruciferous vegetables: boon for immunity 1983.
- Chang CC, Yang MH, Wen HM and Chern JC: Estimation of total flavonoid content in propolis by two complementary colorimetric methods. Journal of Food and Drug Analysis 2002; 10: 178-82.
- Afroz R, Tanvir EM, Islam MA, Alam F, Gan SH and Khalil MI: Potential antioxidant and antibacterial properties of a popular jujube fruit: apple kul (Zizyphus mauritiana). J of Food Biochemistry 2014; 38: 592-01.
- Lowry OH, Rosebrough NJ, Farr AL and Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 1951; 193: 265-75.
- Nelson N: A photometric adaptation of the somogyi method for the determination of glucose. J Biol Chem 1944; 153: 375-80.
- Manzocco L, Anese M and Nicoli M: Antioxidant properties of tea extracts as affected by processing. LWT-Food Science and Technology 1998; 31: 694-98.
- Benzie IF and Strain JJ: The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical Biochemistry 1996; 239: 70-76.
- Khalil MI, Tanvir EM, Afroz R, Sulaiman SA and Gan SH: Cardio protective effects of Tualang honey: Amelioration of cholesterol and cardiac enzymes levels. Bio Med Research International 2015; 1-8.
- Wong ZW, Thanikachalam PV and Ramamurthy S: Molecular understanding of the protective role of natural products on isoproterenol-induced myocardial infarction: A review. Biomed Pharmacother 2017; 94: 1145-66.
- Henning SM, Wang P, Carpenter CL and Heber D: Epigenetic effects of green tea polyphenols in cancer. Epigenomics 2013; 5: 729-41.
- Denre M: The determination of vitamin C, total phenol and antioxidant activity of some commonly cooking spices crops used in west bengal. Int J of Plant Physiolo and Biochem 2014; 6: 66-70.
- Dalar A, Turker M, Zabaras D and Konczak I: Phenolic composition, antioxidant and enzyme inhibitory activities of Eryngium bornmuelleri Plant Food for Hum Nutri 2014; 69: 30-36.
- Ghafoor K, Al-Juhaimi F and Choi YH: Effects of grape (Vitis labrusca) peel and seed extracts on phenolics, antioxidants and anthocyanins in grape juice. Pak J Bot 2011; 43: 1581-86.
- Cook N and Samman S: Flavonoids: Chemistry, metabolism, cardio protective effects and dietary sources. Nutritional Biochemistry1996; 7:66-76.
- Tilak JC, Banerjee M, Mohan H and Devasagayam TP: Antioxidant availability of turmeric in relation to its medicinal and culinary uses. Phytothe R 2004; 18: 798-04.
- Okwu D: Phytochemicals, vitamins and mineral contents of two Nigerian medicinal plants. Int J Mol Med Adv Sci 2005; 1: 375-81.
- Krisper P, Tisler V, Skubic V, Rupnik I and Kobal S: The use of tannin from chestnut (Castanea vesca), in plant polyphenols. Springer 1992; 1013-19.
- Tolstikova T, Khvostov MB and Ryzgalov A: The complexes of drugs with carbohydrate-containing plant metabolites as pharmacologically promising agents. Mini Reviews in Medicinal Chemistry 2009; 9: 1317-28.
- Hammond GL, Nadal-Ginard B, Talner NS and Markert CL: Myocardial LDH isozyme distribution in the ischemic and hypoxic heart. Circulation 1976; 53: 637-43.
- Li L, Hessel M, van der Valk L, Bax M, Linden IVD and Laarse AVD: Partial and delayed release of troponin-I compared with the release of lactate dehydrogenase from necrotic cardiomyocytes. Pflu Arch 2004; 448: 146-52.
How to cite this article:
Kabir A, Karim R, Ali MY, Khalil MI and Hossain MI: Characterization of phytochemical and antioxidant properties of Citrus aurantifolia (Kagoji lebu) available in Bangladesh and its protective effects against experimentally induced myocardial infarction in rats. Int J Pharmacognosy 2019; 6(12): 397-06. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(12).397-06.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
397-406
487
1120
English
IJP
A. Kabir, R. Karim, M. Y. Ali, M. I. Khalil and M. I. Hossain *
Laboratory of Preventive and Integrative Biomedicine, Department of Biochemistry and Molecular Biology, Jahangir Nagar University, Savar, Dhaka, Bangladesh.
Ibrahim_bmb@juniv.edu
28 November 2019
23 December 2019
26 December 2019
10.13040/IJPSR.0975-8232.IJP.6(12).397-06
31 December 2019