BIOSURFACTANT PRODUCED BY NOVEL HALO-THERMOPHILIC BACTERIAL STRAIN FROM UNCONVENTIONAL SUBSTRATES
HTML Full TextBIOSURFACTANT PRODUCED BY NOVEL HALO-THERMOPHILIC BACTERIAL STRAIN FROM UNCONVENTIONAL SUBSTRATES
Chinyere Jennifer Igbonekwu 1, Uduma Eke Osonwa 2, Chukwuebuka Emmanuel Umeyor * 2, Moses Nkechukwu Ikegbunam 1, Emmanuel Maduabuchi Uronnachi 2 and Charles Okechukwu Esimone 1
Department of Pharmaceutical Microbiology and Biotechnology 1, Faculty of Pharmaceutical Sciences, Nnamdi Azikiwe University, Awka 422001, Anambra State, Nigeria.
Department of Pharmaceutics and Pharmaceutical Technology 2, Faculty of Pharmaceutical Sciences, Nnamdi Azikiwe University, Awka 422001, Anambra State, Nigeria.
ABSTRACT: Our ecosystem is laden with hazardous and non-hazardous wastes that pose a serious threat to bio-existence. Most pharmaceutical formulations contain chemically-synthesized surfactants. The purpose of this work is to suggest an alternative source of surfactant which would also aid in ridding our environment of harmful wastes. Bacillus licheniformis B-4 strain isolated from a hydrocarbon dump site produces biosurfactant with excellent interfacial properties utilizing unconventional substrates (kerosene and diesel) as carbon sources, and glucose as an energy source. Acid precipitation and foam separation methods were used for biosurfactant recovery and its physicochemical properties evaluated which include the effect of the biosurfactant on the surface tension of water, critical micelle concentration (CMC), emulsification stability (ES) of kerosene and diesel, emulsification index, hemolytic activity, and antimicrobial activity. The results showed that the biosurfactant reduced the surface tension of water from 70.2 to 14.7 Nm-1, CMC was estimated to be 50 mg/l, emulsification stability of kerosene and diesel were obtained as 0.06 and 0.02 min-1, while the emulsification indices were 72.2 and 50 % respectively for kerosene and diesel. Hemolytic activity of the biosurfactant showed concentration-dependent lyses of blood cells while antimicrobial studies also showed concentration-dependent inhibition of bacterial cells. The results obtained from the analysis showed that the biosurfactant could serve as a good alternative to chemically-synthesized surfactants.
| Keywords: |
Biosurfactant, Bacillus licheniformis, Surface tension, Emulsification index, Critical Micelle Concentration, Halo-thermophilic, Hemolytic, Physicochemical, Isolate and the substrate
INTRODUCTION: Many microorganisms (fungi and bacteria) can produce a wide range of amphipathic compounds, with both hydrophilic and hydrophobic moieties present within the same molecule, which allow them to exhibit surface activities at interfaces and are generally called Biosurfactant.
These Surface Active Compounds (SAC) are mainly classified according to their modes of action, molecular weight and general physicochemical properties 1. Biosurfactants or microbial surface-active compounds are a range of structurally diverse molecules produced by different microorganisms and are mostly therefore classified by their structural features, the producing organism, and their molecular mass. Their hydrophilic moiety is mainly comprised of an acid, peptide cations, or anions, mono-, di- or polysaccharides while their hydrophobic moiety can be unsaturated or saturated hydrocarbon chains of fatty acids.
The structural orientation on the surfaces and interfaces confer the range of properties, such as the ability to lower surface and interfacial tension of liquids and the formation of micelles and microemulsions between these different phases 2.
Although the functions of biosurfactants in microorganisms are not fully understood, it is believed that these secondary metabolites can enhance nutrient transport across membranes, act in various host-microbe interactions, and provide bactericidal and fungicidal functions to the producing organism 3. Several types of research have been carried out on biosurfactants with great success, and the products recommended for use because they are environment-friendly. They are bio-degradable and non-toxic to the ecosystem, unlike the chemically-synthesized surfactants. In recent years, biosurfactants have found quite significant applications in the pharmaceutical industries, agrochemical industries, petrochemical industries, and paper and pulp industries 4.
Some of the prominent advantages of biosurfactants include biodegradation, specific activity at extreme temperatures, environmental protection and management, bioremediation and crude oil recovery, and antimicrobial agents in healthcare and food processing industries 5. Over the years, biosurfactants have been produced using various substrates, mainly organic, which are mostly renewable. They include distillery wastes, vegetable oils and materials, and dairy wastes. Productions have also been made using tropical agro-materials such as cassava, soybean, potato, sugarcane, corn, and rice and tea waste materials.
This initiative is laudable because it rids the environment of hazardous and non-hazardous wastes through reducing, reusing and recycling of these organic materials. However, millions and tons of crude oil and refined petroleum products spillage have been reported and witnessed throughout the world, and cleaning of this spill poses a great and outrageous financial burden on industries and governments especially in the face of the current economic recesses worldwide. Unfortunately, the synthesis of biosurfactant from these petroleum products has received little attention, especially in areas prone to oil spillage. The production of biosurfactant utilizing these products could help to ease the financial burden required for the mechanical cleaning of the oil spill.
In this study, we report the production of biosurfactant using unconventional substrates (diesel and kerosene) by halo-thermophilic bacteria isolated from a petrochemical wastes dump site, and evaluation of the physicochemical properties as well as some essential pharmaceutical applications of the synthesized biosurfactant.
MATERIALS AND METHODS: Safranin red, D-glucose (Fluka chemical, Poole, England), Malachite green (Kem-light laboratory, UK), Kerosene, diesel, Shaker (Barloworld Scientific Limited Stone, Staffordshire ST 15 OSA UK), Incubator, Oven (Gen Lab, Cheshire, UK), Autoclave (Equitron Medica Instrument Mfg. Co. Mumbai, India). All other reagents used were of analytical grade and were used as received without any treatment.
Test Microorganisms: Some clinical isolates of Salmonella typhi, Klebsiella pneumonia, Escherichia coli, Staphylococcus aureus, Streptococcus pneumonia, Pseudomonas aeruginosa, and Proteus vulgaris were collected from Nnamdi Azikiwe Teaching Hospital Laboratory, Nnewi, Anambra State while the fungi (Aspergillus niger and Candida albicans) were collected from our laboratory.
Collection of Samples and Cultivation of Bacteria: Samples of oil-contaminated water and soil were collected at different hydrocarbon sites (at a mesophilic temperature < 40 ºC, pH 6.8 - 7.8; and thermophilic temperature 45 - 50 ºC, pH 8 - 10) in Nigeria in mid-November 2011. A total of 8 sites were sampled and labeled A-1, A-2, A-3, A-4 & B-1, B-2, B-3, B-4 with four bacterial strains (B-1, B-2, B-3, B-4) isolated. 50 ml each was added to an Erlenmeyer flask containing 50 ml of the Mineral Salt Medium (MSM). 1% (v/v) each of (kerosene and diesel) and 2% (v/v) of glucose respectively, were added as a sole carbon and energy sources and incubated at 50 ºC in a shaker at 160 rpm for 72 h. The degree of emulsification was observed, and only emulsified samples were serially diluted in 0.85% NaCl and streaked onto the solid medium. Plates were examined, and preliminary identification of isolates made by colony morphologies, microscopy, and biochemical tests. Isolates were purified by repeated single colony isolation and purity of cultures checked periodically by streaking liquid cultures onto the MSM.
Preparation of Inoculums: The isolated organism was stored in a refrigerator at 37 ºC and was streaked on freshly prepared nutrient agar, and incubated at 50 ºC for 24 h. A distinct colony of the isolate was grown in a nutrient broth medium at 50 ºC for 72 h. The seed culture was used for inoculation of the fermentation medium.
Morphological and Biochemical Determination: The morphological and biochemical tests were done to determine the microorganism and the genus of the unknown bacterial isolate. The micro-biochemical tests performed include gram staining, oxidase, catalase, starch hydrolysis, and spore staining. Results from the analysis were used to find the closest match with known bacterial genus and to assign the bacterial signature.
Extraction of Biosurfactant: After 72 h of incubation of bacterial cells in the fermentation medium, the hydrocarbon oil layer in the conical flask was removed firstly, followed by the bacterial cells which were removed by centrifugation at 8,000 rpm at 4 ºC for 10 min. The supernatant was subjected to acid precipitation by adding conc. HCl to adjust the pH to 2, and precipitate the lipids and proteins 6. The grey-white pellets formed by precipitation were collected by centrifugation at 8,000 rpm at 4 °C for 20 min. The precipitate was re-suspended with little-distilled water (pH 7) to dissolve most of them, and they were finally lyophilized. The crude biosurfactant was lyophilized and weighed for quantification.
For further purification of biosurfactant, chloroform: methanol (2 : 1 v/v) was added to the dry product and incubated in a rotary shaker at 250 rpm, 30 ± 0.5 ºC for 15 min 7. The extract was evaporated to dryness, weighed and used for analysis.
Characterization of the Biosurfactant Producing Isolate:
Blood Hemolysis Test: Fresh cultures of the isolates were prepared by streaking on nutrient agar followed by incubation at 50 ºC for 24 h. A single fresh colony of the isolates respectively was then re-streaked on blood agar and incubated at 37 ºC for 24 h. The bacterial colonies were then observed for the presence of clear zones of hemolysis around the colonies on blood agar.
Oil Displacement Test: 20 ml distilled water (pH 7) was added to a Petri dish (90 mm diameter), and then 10 ml each of kerosene and diesel, was dropped on the surface of the distilled water. A loopful of the culture solutions were gently put on the center of the oil film, and the diameter and area of displacement as visualized under visible light were measured after 30 s 8. The tendency of the biosurfactant- producing isolate to displace both the diesel and kerosene after 2 min was recorded.
Evaluation of Some Physicochemical Properties of the Biosurfactant:
Surface Tension Measurement: Several different concentrations of the biosurfactant solution (10, 30, 50, 80, 100, 120, 150 and 200 mg/l) were prepared, and their surface tension was measured using a tensiometer. The surface tension of distilled water served as the control. This was repeated in triplicates.
Determination of Critical Micelle Concentration (CMC): The CMC was calculated as the concentration where the micelle formation was observed during the measurement of the effect of the biosurfactant on surface tension.
Estimation of Emulsification Index, E24: A solution (biosurfactant and water, as solvent) was mixed with kerosene and diesel in the ratio 1:1, and then vigorously shaken for 2 min using a vortex. After equilibration for 24 h, the solution divided into three layers: a water layer, an emulsion layer, and an oil layer. Water mixed with kerosene and diesel was used as a control. The emulsification index was calculated using the equation below: 9.
Emulsification index (E24, %) = Height of emulsion layer × 100% / The total height of the solution
Determination of Emulsion Stability: 4 ml of biosurfactant solution was diluted with 1 ml substrate (kerosene and diesel) respectively. The mixture was vigorously shaken in a vortex mixer for 2 min. The mixture was allowed to stand for 10 min, and the turbidity was measured at 540 nm using a colorimeter with distilled water as the blank. The turbidity was measured every 10 min for 1 h. The log of the absorbance was plotted against time, and the emulsification stability was calculated as the slope of the graph. For the control test, the 4 ml biosurfactant in the above protocol was replaced with distilled water.
Assay of Hemolytic Activity: The hemolytic activity of the biosurfactant was determined using agar diffusion method on a blood agar plate. Using a cork borer, a 6 mm-wide hole was bored on the blood agar plate, and different concentrations (0, 1.6, 3.2, 6.4, and 25.6 mg/l) of the biosurfactant was added into each hole. The plates were incubated at room temperature for 24 h. Their zones of inhibition were measured.
Study of Anti-microbial Activity: Nutrient agar and Sabouraud Dextrose Agar were used for the test. The media were seeded with each test organism (Salmonella typhi, Klebsiella pneumonia, Escherichia coli, Staphylococcus aureus, Streptococcus pneumonia, Proteus vulgaris, Bacillus Licheniformis strain B-4, Aspergillus niger, and Candida albicans) and adjusted using 0.5 McFarland Standard 10. The agar wells were dug using a sterile cork borer of (6 mm) and various concentrations of the biosurfactant (1.6, 3.2, 6.4, 12.8 and 15 μg/l) were introduced into the wells, and incubated at 37 ºC for 24 h (bacteria) and at 30 ºC for 48 h (for fungi). Zones of inhibition were measured after the incubation. The experiments were repeated in triplicates for good statistical analysis.
Statistical Analysis: The results obtained were expressed as mean ± SD. ANOVA and student’s t-test were performed on the data generated using SPSS. Differences were considered significant for p<0.05.
RESULTS AND DISCUSSIONS:
Morphological and Biochemical Tests: The morphological tests showed that all the isolates are Gram-positive rod-like microorganisms, and probably of the same strain. This could be confirmed from the biochemical tests where they also produced the same results. Since the tests are used to identify Bacillus licheniformis (BL), and the tests results are positive for all the isolates, it is an indication that they are BL strains. The isolate, B. licheniformis B-4 selected for the study produced gliding and swimming features on nutrient agar which could be essential as biofilms on medical devices. The results of the morphological and biochemical screening of the isolates are shown in Table 1 below.
TABLE 1: RESULTS OF MORPHOLOGICAL AND BIOCHEMICAL TESTS OF ISOLATES
| ISOLATE / TEST | B1 | B2 | B3 | B4 |
| Gram staining | +ve rods | +ve rods | +ve rods | +ve rods |
| Oxidase | -ve | -ve | -ve | -ve |
| Catalase | +ve | +ve | +ve | +ve |
| Starch Hydrolysis | +ve | +ve | +ve | +ve |
| Spore staining | +ve | +ve | +ve | +ve |
| Incubating in 6.5 % NaCl at 55 ºC | +ve | +ve | +ve | +ve |
Blood Hemolysis and Oil Displacement Screenings: It is worthy to note that the MSM used for the isolation of the test organisms supports the growth of halo-thermophilic bacteria. From Table 2, it could be seen that the test isolates were positive for blood hemolysis and oil displacement tests respectively, while the control organisms were negative. This is an indication that the test organisms isolated from the hydrocarbon waste could be good synthesizers of biosurfactant. The test organism could be cultured and used in the degradation of oil during the spill, and reduce the contaminating effects of crude oil. Though the strains of the organisms could not be determined from these studies, the ability of the organisms to proliferate in MSM is a clear proof that they are resistant to extreme effects of heat and salt. At temperature conditions above normal for instance, the biosurfactants produced would still lower surface tension of a particular medium, and micelles could also be formed to protect the lipophilic regions of molecules, while the hydrophilic ends go into solution with the aqueous environment. The results are shown below.
TABLE 2: SCREENING OF BIOSURFACTANT PRODUCING BACTERIAL ISOLATES (B-1, B-2, B-3, B-4)
| Isolates | Blood Haemolysis | Oil Spreading/Displacement | |
| Diesel | Kerosene | ||
| B-1 | β | + | + |
| B-2 | β | + | + |
| B-3 | β | + | + |
| B-4 | β | + | + |
| Control | |||
| a | α | - | - |
| b | Β | - | - |
Key: + = Oil displaced; β = Beta haemolysis; α = Alpha haemolysis, a = Control (Bacillus subtilis); b = Standard for Beta haemolysis
Surface Tension Study: The evaluation of the pharmaceutical applications of a biosurfactant by measurement of the surface tension activity has been used by many researchers to measure the surface properties of biosurfactants 11, 12. From the results of the surface tension study shown in Table 3 below, increase in the concentrations of the biosurfactant from the BL strain B-4 produced a corresponding decrease in surface tension values measured from 70.2 - 14.7 Nm-1. This is an indication that the biosurfactant from this test isolate is viable. This reduction could be due to the adsorption of the surfactant at the interface of its solutions with the lipophilic and hydrophilic regions orientating themselves with the appropriate interface. It, therefore, means that the dual nature of the biosurfactant, which is energetically favorable in a solution, is important for this surface activity. The results of the study are shown below.
TABLE 3: RESULT OF THE EFFECT OF BIOSURFACTANT ON SURFACE TENSION
| Biosurfactant Concentration (mg/l) | Surface tension (Nm-1) |
| 0.0 | 70.2 ± 1.1 |
| 10.0 | 55.3 ± 1.0 |
| 30.0 | 43.5 ± 1.5 |
| 50.0 | 17.3 ± 2.0 |
| 80.0 | 15.6 ± 1.8 |
| 100.0 | 14.7 ± 2.1 |
| 150.0 | 14.8 ± 2.2 |
| 200.0 | 14.7 ± 2.1 |
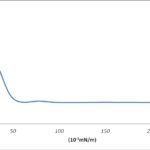
Determination of Critical Micelle Concentration (CMC): From the data obtained, the CMC of the biosurfactant was estimated at 50 mg/l. This is the critical concentration at which the surface energy of the solution has been reduced to allow the formation of micelles. From Fig. 1 below, as the surface layer of the biosurfactant solution became saturated, further increases in its concentration did not cause further change in the surface energy of the solution, resulting in the formation of micelles at that optimum concentration. This is shown below.
FIG. 1: CRITICAL MICELLE CONCENTRATION (CMC) OF BIOSURFACTANT FROM BACILLUS LICHENIFORMIS B-4 STRAIN (THE ARROW ON THE GRAPH INDICATES THE POINT OF MICELLE FORMATION)
Estimation of Emulsification Index, E24: After Equilibration, the calculated indices of emulsification for the substrates (kerosene and diesel) and the biosurfactant were 72.2 and 50% respectively. From these results shown in Table 4 below, it could be seen that the biosurfactant has good emulsification index. Hence, it is a confirmation that the biosurfactant is a good emulsifier, and could be used in accelerated emulsification of the hydrophobic phase of an emulsion in the hydrophilic phase. However, with kerosene substrate, a better emulsification activity was recorded while diesel substrate gave the least emulsifying activity with the biosurfactant. The test solutions gave positive values for the test as shown in the table and figure below.
TABLE 4: EMULSIFICATION INDICES OF BIOSURFACTANT WITH SOME HYDROCARBONS
| Substrate | Emulsification Index, E24 (%) |
| Kerosene | 72.2 |
| Diesel | 50.0 |
FIG. 2: DETERMINATION OF EMULSIFICATION INDEX OF THE BIOSURFACTANT WITH THE SUBSTRATES (KEROSENE AND DIESEL)
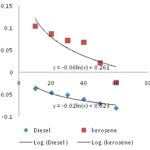
Determination of Emulsion Stability: The emulsification stability values were obtained as - 0.06 and - 0.02 min-1 for the substrates (kerosene and diesel) respectively. Generally, emulsions are thermodynamically unstable, and will eventually separate into oil and water phases. Therefore, emulsion stability is a measure of the rate of separation of an emulsion into oil and water phases 13, and this depends on the amount of the emulgent adsorbed at the oil/water interface. This is because the ability of the surfactant to reduce the interfacial tension will facilitate the formation of a stable disperse system. Stability of emulsions is expressed as the decay constant (Kd) obtained from the slope of the graphical representation of log. absorbance against time. An emulsion formulated with a biosurfactant is stable when the value of the decay constant (Kd) is < -2.00 min-1 14. Therefore, the emulsions formed by the biosurfactant with the oils were stable because Kd values for kerosene and diesel are -0.061 and -0.024 min-1 respectively. Thus, the more negative the slope or the lesser the Kd, the more stable the bioemulsifier/biosurfactant. The result is shown in the figure below.
FIG. 3: GRAPH OF EMULSION STABILITY SHOWING DECAY CONSTANT (KD) VALUES FOR THE SUBSTRATES
Assay of Hemolytic Activity: The zones of lyses obtained from the study of hemolytic activity showed that the lysis of the biosurfactant are concentration dependent as zones of lyses increased with an increase in surfactant concentration. This activity could be due to the adsorption of the surfactant at the surface of the erythrocytes, and distortion of the permeability of the cell membrane. The erythrocytes loose essential substances are causing their death. Thus, they disorganize the membrane’s lipidic bilayers and solubilize proteins. This technique was first discovered by Bernheimer and Avigad (1970) 15 who reported that the production of Biosurfactant (surfactin) by Bacillus subtilis might cause red blood cells (RBCs) to lose. It has also been used for the isolation and quantification of surfactin and rhamnolipids. This result is presented in the table below.
TABLE 5: RESULT OF HAEMOLYTIC ACTIVITY OF BIOSURFACTANT
| Concentration (mg/l) | Zone of lysis (mm) |
| 0.0 | 0.0 ± 0.0 |
| 0.8 | 0.0 ± 0.0 |
| 1.6 | 1.0 ± 0.1 |
| 3.2 | 2.0 ± 1.1 |
| 6.4 | 3.0 ± 1.1 |
| 25.6 | 7.0 ± 1.5 |
The Result of Antimicrobial Study: From the result of the antimicrobial study shown in Table 6 below, it could be seen that the biosurfactant gave a concentration-dependent inhibition of microbial growth. Essentially, only bacterial cells were inhibited while fungal cells were not. At surfactant concentration ≤ 5 μg/ml, there was no inhibition of microbial viability; microbial growth was minimally inhibited (1-2 mm) at 6 μg/ml while at maximum concentration of biosurfactant (Cmax) of 15 μg/ml, the viability of Salmonella typhi was maximally inhibited (13 mm) followed by the extended-spectrum beta-lactamase (ESBL) organisms (Klebsiella pneumonia and Escherichia coli) and methicillin-resistant Staphylococcus aureus (MRSA). This expressed antibacterial activity could be due to the damaging effect of the biosurfactant on the cell membranes of the bacterial cells due to its adsorptive effect on them, destroying their integrity, preventing the uptake of essential growth and survival elements as well as leakage of intracellular substrates ultimately leading to cell death. This explains why preparations of a certain class of surfactants are used as disinfectants for instruments and skins, antibacterial creams and throat lozenges. The observed low zones of inhibition from this study could be due to the multi-resistant nature of the clinical isolates, low concentration of the biosurfactant used and the inoculum size of the test microorganisms. The results are as shown below.
TABLE 6: RESULTS OF INHIBITION OF GROWTH OF MICROORGANISMS BY BIOSURFACTANT
| Mean IZD ± SD (mm) | |||||||
| Biosurfactant Concentration (μg/ml) | |||||||
| Test Organism | 0.0 | 0.8 | 0.16 | 3.2 | 6.4 | 12.8 | 15.0 |
| Pseudomonas aeruginosa | - | - | - | - | - | - | 7.0 ± 1.5 |
| Salmonella typhi | - | - | - | - | - | - | 13.0 ± 2.7 |
| Staphylococcus aureus | - | - | - | - | - | - | 7.0 ± 1.5 |
| Streptococcus pneumonia | - | - | - | - | - | - | - |
| Bacillus licheniformis (β-4) | - | - | - | - | 1.0 ± 0.4 | 2.0 ± 1.1 | 5.0 ± 1.8 |
| Proteus vulgaris | - | - | - | - | - | - | - |
| Klebsiella pneumonia | - | - | - | - | 2.0 ± 1.1 | 4.0 ± 2.0 | 9.0 ± 3.2 |
| Escherichia coli | - | - | - | - | - | - | 9.0 ± 3.2 |
| Aspergillus niger | - | - | - | - | - | - | - |
| Candida albicans | - | - | - | - | - | - | - |
IZD = Inhibition Zone Diameter
SD = Standard Deviation
CONCLUSION: The Bacillus licheniformis B-4 strain (BL B-4) is a halo-thermophilic bacteria isolated from a hydrocarbon source capable of producing biosurfactant. The result obtained from the evaluations of its pharmaceutical applications inferred that the biosurfactant possesses excellent interfacial properties because it could reduce the surface energy of suspensions, stabilizes emulsions and has good emulsification index. Its hemolytic and anti-bacterial activities are concentration-dependent and could be veritable alternatives in the preparation of detergents and disinfectants for domestic, clinical and industrial use.
Cultures of BL B-4 could also be used to produce biosurfactants to lower the damaging effects of hydrocarbon - contaminated sites through accelerated bioremediation. The discovery of the existence of this novel bacterial strain (Bacillus licheniformis B-4) has given a new direction and improved already existing data of bacteria capable of synthesizing biosurfactants. Its use of kerosene and diesel as substrates for biosurfactant production makes it even more interesting.
However, further researches should be conducted to design better extraction methods to improve the yield of biosurfactants from petroleum products, as well as carrying out extensive and thorough isolation of more microorganisms with the potential of producing biosurfactants using uncommon substrates. The toxicity profile of these bio-surfactants should be extensively studied to ensure the safety of products which could be formulated using them. Finally, compatibility and interaction studies should be carried out before combining these biosurfactants with chemically-synthesized surfactants so as not to destroy their important properties or degrade any formulation in which they are incorporated.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Fracchia L, Cavallo M, Martinotti MG and Banat IM: Biosurfactants and bioemulsifiers biomedical and related applications – present status and future potentials. In: Ghista DN, editor. Biomedical Science, Engineering and Technology 2012; 14: 326-335.
- Chen ML, Penfold J, Thomas RK, Smyth TJP, Perfumo A, Marchant R and Banat IM: Solution self-assembly and adsorption at the air-water interface of the mono and di-rhamnose rhamnolipids and their mixtures. Langmuir. 2010(a); 26: 18281-18292.
- Chen ML, Penfold J, Thomas RK, Smyth TJP, Perfumo A, Marchant R and Banat IM: Mixing behaviour of the biosurfactant, rhamnolipid, with a conventional anionic surfactant, sodium dodecyl benzene sulfonate. Langmuir 2010(b); 26: 17958-17968.
- Jennings EM and Tanner RS: Biosurfactant-producing bacteria found in contaminated and uncontaminated soils. Paper presented at: Conference on hazardous waste research 2000; 299-306.
- Abouseoud M, Maachi R and Amrane A: Biosurfactant production from olive oil by Pseudomonas fluorescens. In: Mendez-Vilas A, editor. Communicating current research and educational topics and trends in applied microbiology. Formatex 2007; 340-347.
- Cao XH, Cai P, Li F, Wang CL and Lu MF: Medium optimization for lipopeptide produced by Bacillus natto TK-1 using response surface methodology. China Biotechnol 2007; 27: 59-65.
- Dhouha G and Semia E: Enhancement of Bacillus subtilis lipopeptide biosurfactants production through optimization of medium composition and adequate control of aeration. Biotechnology Research International 2011; 1-6.
- Maneerat S and Phetrong K: Isolation of Biosurfactant-producing marine bacteria and characteristics of selected biosurfactant. Songklanakarin J Sci Technol 2007; 29: 781-791.
- Lee SC, Lee SJ, Kim SH, Park IH, Lee YS, Chung SY and Choi YL: Characterization of new biosurfactant produced by Klebsiella sp. Y6-1 isolated from waste soybean oil. Bioresource Technology 2008; 99(7): 2288-2292.
- Okore VC: Principles of Pharmaceutical Microbiology: Evaluation of chemical antimicrobial agents. 2nd ed, Enugu, Nigeria: El Demark Publishers 2009.
- Youssef NH, Duncan KE, Nagle DP, Savage KN, Knapp RM and McInerney MJ: Comparison of methods to detect biosurfactant production by diverse microorganisms. J. Microbiol. Methods 2004; 56: 339-347.
- Neu TR and Poralla K: Emulsifying agent from bacteria isolated during screening for cells with hydrophobic surfaces. Microbiol Biotechnol 1990; 32: 521-525.
- Pearce KN and Kinsella JE: Emulsifying properties of proteins: Evaluation of a turbidimetric technique. J. Agric. Food Chem 1978; 26(3): 716-723.
- Kim SH, Lim EJ, Lee SO, Lee JD and Lee TH: Purification and characterization of biosurfactants from Nocardia sp. L-417. Biotechnol. Appl. Biochem 2000; 31: 249-253.
- Bernheimer AW and Avigad LS: Nature and properties of a cytolytic agent produced by Bacillus subtilis. J Gen Microbiol 1970; 6: 361-366.
How to cite this article:
Igbonekwu CJ, Osonwa UE, Umeyor CE, Ikegbunam MN, Uronnachi EM and Esimone CO: Biosurfactant produced by novel halo-thermophilic bacterial strain from unconventional substrates. Int J Pharmcognosy 2014; 1(1): 73-81. doi: 10.13040/IJPSR.0975-8232. 1(1).73-81.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
9
73-81
641
2331
English
IJP
C. J. Igbonekwu, U. E. Osonwa, C. E. Umeyor*, M. N. Ikegbunam, E. M. Uronnachi and C. O. Esimone
Department of Pharmaceutical Microbiology and Biotechnology, Faculty of Pharmaceutical Sciences, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria.
chuks4live@gmail.com
10 November 2013
14 December 2013
29 December 2013
http://dx.doi.org/10.13040/IJPSR.0975-8232.1(1).73-81
01 January 2014