BI-LAYER TABLET: A CONTROLLED RELEASE DOSAGE FORM
HTML Full TextBI-LAYER TABLET: A CONTROLLED RELEASE DOSAGE FORM
Vivek Mahavir Satpute
Naldurg Tal Tuljapur, Osmanabad - 413601, Maharashtra, India.
ABSTRACT: Bi-layer tablets are developed to get immediate and sustained delivery of various drugs that have pre-defined release. In the past few decades, development in a combination of active pharmaceutical ingredients (API) in a single fixed dosage form has increased in the pharmaceutical industry, also promotes patient convenience and compliance. Bi-layer tablet plays a crucial role in the development of controlled release in order to give a successful drug delivery. Bi-layer tablets can be said to be a better option to reduce chemical incompatibilities taking place between Active Pharmaceutical Ingredients and to enable the development of various drug release profiles. Bi-layer tablet suits for sequential release of two drugs in combination or to incorporate two incompatible substances in same tablet. The bi-layer tablets are preparing by using different techniques such as OROS® push-pulls Technology, L-OROSTM Technology, EN SO TROL Techno-logy. This review article explains various techniques of bi-layer tablets and why the development and production of quality bi-layer tablets need to be carried out and basic challenges faced during the production of Bi-layer tablets. This review also describes the rationale for this combination therapy and the clinical trials that have demonstrated that these two agents can be combined without the loss of efficacy for either agent or an increase in the incidence of adverse events.
| Keywords: |
Bi-layer tablets, OROS®, API
INTRODUCTION: Solid oral dosage forms are mostly preferred over other routes for many drugs and are still the most widely used formulations. The controlled-drug delivery systems typically require more demanding mechanical testing, characterization, and monitoring techniques with faster response times than those possible with traditional measurement approaches 1. Over 90% of the formulations manufactured today are ingested orally. It shows that this class of the formulation is the most popular worldwide, and the major attention of the researcher is in this direction.
The major aim of controlled drug delivery is to reduce the frequency of dosing 2. The objective of sustained release is to ensure safety and also to improve the efficacy of drugs and patient compliance. A bi-layer tablet is a fixed-dose combination (FDC) intended for oral application. It consists of two layers the first layer have immediate release part of single; the next layer is controlled release part of single or multiple actives.
They are called “Bi-layer tablets.” For the identification of two drugs, various colors were used. A bi-layer tablet is a very improved technique to overcome the single-layered tablet. Bi-layer tablets contain immediate, sustained release layers, and the immediate release layer delivers the initial dose. It includes super disintegrates, that increases the release rate of the drug and also attains the onset of action quickly.
Whereas sustained-release (maintenance dose) layer releases the drug in a sustained manner for a prolonged period of time by using various polymers as release in retardants. Diabetes, anti-hypertensive, antihistamines, analgesics, antipyretics, and antiallergenic agents are mainly suitable for this type of drug delivery 3. Bi-layer tablets have an advantage as compared to conventional monolayer tablets. These tablets are commonly used, so as there is less use of chemical incompatibilities of formulation components by physical separation. Moreover, bi-layer tablets have also enabled the development of controlled delivery of an API by combining slow-release with immediate-release layers. However, such drug delivery devices are complicated mechanically to manufacture and also difficult to judge their long term properties as they have poor mechanical and compression of the materials in the adjacent layers which is compacted, insufficient hardness, inaccurate individual mass control, reduced yield, cross-contamination between the layers and their potency to delaminate at the interface while on various stages of the compaction process. The major problem that has to be overcome is to find out in detail the sources of these problems at lower scales and to find out remedies to solve them. A major challenge is the lack of appropriate bonding at the interface between the adjacent compacted layers. When the compacted layers are beyond a certain limit, soft or hard, they might not bond appropriately with each other, and that can lead to compromised mechanical integrity. Challenges during development also include layer weight ratio, establishing the order of layer sequence, first layer tamping force, elastic mismatch of the adjacent layers, and cross-contamination between layers 4, 5.
TABLE 1: VARIOUS ADVANCEMENTS IN THE FIELD OF BILAYER TABLETS 7
| Drug(s) | Dosage Form | Rationale |
| Atorvastatin,
Atenolol |
Bi-layer gastro retentive
matrix Table |
Treatment of hypertension and hypercholesterolemia |
| Nifedipine | Gastroretentive floating
Bi-layer tablets |
Treatment of hypertension and angina pectoris |
| Aspirin, Isosorbide
5-mono-nitrate |
Sustained tablets | Treatment of pain, fever and other inflammatory
Conditions |
| Pioglitazone HCl, Gliclazide | Bi-layer Tablets | Treatment of Type II Diabetes |
| Losartan potassium | Bi-layer tablet | Treatment of hypertension |
| Trimetazidine HCl, Clopidogrelbisulphate | Bi-layer tablets | Cytoprotective anti-ischemic |
| Diclofenac, Cyclobenza-prine | Bi-layer tablets | Synergistic effect in pain |
| Granisetron HCl | Bi-layer buccal tablets | To overcome the bioavailability problem |
| Metformin HC1, Glimepiride | Bi-layer tablets | Synergistic effect in diabetes |
| Indomethacin | Bi-layer floating tablets | Biphasic drug release |
| Metformin HC1, Atorvastatin
Calcium |
Bi-layer tablets | To develop polytherapy for the treatment of NIDDS & hyperlipidemia |
| CefiximeTrihydrate,
Dicloxacillin Sodium |
Bi-layer tablets | Synergistic effect in bacterial infections |
| Piracetam, Vinpocetin | Bi-layer tablets | Synergistic effect in Alzheimer disease |
| Metformin HCl, Pioglitazone | Bi-layer tablets | Synergistic effect in diabetes mellitus |
| Atenolol | Bi-layer buccal tablets | To overcome the bioavailability problem, reducing side
effects and frequency of administration |
| Cefuroxime Axetil Potassium
Clavulanate |
Bi-layer tablets | Synergistic effect against microbial infections and to minimize dose-dependent side effects |
| Amlodipine BesylateMetoprolol
Succinate |
Bi-layer tablets | Synergistic effect in hypertension |
| Diclofenac Sodium, Paracetamol | Bi-layer tablets | Synergistic effect in pain |
| Ibuprofen, Methocarbamol | Bi-layer tablets | Synergistic effect of drugs in back pain |
| Atorvastatin Calcium | Bi-layer buccal tablets | To overcome the bioavailability problem, reducing side effects and frequency of administration |
Cardiovascular disease (CVD) has a multi-factorial nature, and their risk factors accompany various different problems. The combination of risk factors like hypertension and dyslipidemia act synergistically, which in turn increases the risk of CVD events. This synergistic action is recognized by the major clinical guidelines currently to help in the management of patients with CVD or at risk of CVD, as they recommend a strategy of treating CVD risk factors simultaneously rather than in isolation. There is an increase in evidence describing the advantages of a combined / multi-factorial approach to reducing CV risk vs. the older sequential approach of treating risk factors individually. The driving forces for the development of such a bi-layer tablet were the poor level of control of CV risk factors, despite the widespread availability of efficacious anti-hypertensive and lipid-lowering mediations. A single tablet combination of antihypertensive and lipid-lowering medication may address some of the issues thought to hinder the management of CVD, such as poor adherence to multiple treatments due to high pill burden and the reluctance of physicians to manage more than one CV risk factor simultaneously.
The anti-hypertensive component(s) must also be free from drug-drug interactions with other BP-lowering medications due to the frequent need for multiple antihypertensive to achieve BP goals in certain difficult-to-treat populations, such as patients with diabetes. The antihypertensive amlodipine Besylate satisfies these criteria in that it has been demonstrated to reduce CV events in various patient populations and is effective when combined with other classes of antihypertensive. There are a number of important requirements for therapies used in combination medication, regardless of the condition being treated. Firstly, the medications must have the same dosing regimen. There should be no negative pharmacokinetic or pharmacodynamic interactions between the proposed components. Thirdly, from a patient’s perspective, the tablet should be of a reasonable size, and the formulations should allow flexible dosing 5, 6.
Need of Bi-layer Tablet: 8
- For purpose of administration of dual release fixed dose combinations of different APIs.
- For the purpose of developing novel drug delivery systems such as buccal / Mucoadhesive delivery system and floating tablets for gastro retentive drug delivery system. It helps in controlling the drug delivery rate of single or two APIs.
- To modify bi-layer tablets in such a way that surface area available for active ingredient layer by placing between one or two in active layers for achieving swellable/erodible barrier for modified release.
- To incorporate two incompatible API in one dosage, this helps in the controlled release of API from one layer by utilizing the property of another layer.
General Properties of Bi-layer Tablet Dosage Forms: 9
- A bi-layer tablet should possess an elegant product identity and should be free of defects like cracks, chips, contamination, and discoloration.
- Must have sufficient strength, which will handle mechanical shock during its production.
- It must have the chemical and physical stability to maintain its physical attributes over time.
- Must have a chemical stability shelf-life.
The Advantages of the Bi-layer Tablet Dosage Form are: 10
- They are unit dosage forms and offer the greatest capabilities of all oral dosage form for the greatest dose precision and the least content variability.
- Cost is lower compared to all other oral dosage form.
- Lighter and compact.
- Easiest and cheapest to package and strip.
- Easy to swallowing with least tendency for hang-up.
- Objectionable odour and bitter taste can be masked by coating technique.
- Suitable for large scale production.
- Greatest chemical and microbial stability overall oral dosage forms.
- Product identification is easy and rapid, requiring no additional steps when employing an embossed and/or monogrammed punch face.
Disadvantages of Bi-layer Tablet Dosage Form are: 10
- Difficult to swallow in case of children and unconscious patients.
- Some drugs resist compression into dense compacts, owing to amorphous nature, low-density character.
- Drugs with poor wetting, slow dissolution properties, optimum absorption high in GIT may be difficult to formulate or manufacture as a tablet that will still provide adequate or full drug bioavailability.
- Bitter testing drugs, drugs with an objectionable odor or drugs that are sensitive to oxygen may require encapsulation or coating.
Preparation of Bi-layer Tablets: 10, 11, 12
Quality and Good manufacturing practice (GMP) requirements of bi-layer tablets: 12
- To produce a quality bi-layer tablet, in a validated and GMP-way, it is important that the selected press is capable of:
- Preventing capping and separation of the two individual layers that constitute the bi-layer tablet.
- Providing sufficient tablet hardness.
- Preventing cross-contamination between the two layers.
- Producing a clear visual separation between the two layers.
- High yield.
- Accurate and individual weight control of the two layers.
These requirements seem very obvious but are not very easy to observe in manufacturing a bi-layer tablet dosage form as there are several critical factors that are to be taken into consideration while manufacturing bi-layer tablets. Bi-layer tablets require fewer materials than compression coated tablets weigh less and maybe thinner. These tablets are manufactured where one layer of the drug is for immediate release while the second layer is for release of drug afterward, it can be as a second dose of an extended-release form 8.
The bi-layer tablets can also be produced, taking into account two incompatible drugs, which is done by compressing individual layers of each drug to reduce the area of contact between two layers. There are certain requirements to develop a proper tablet formulation, like sufficient mechanical strength and desired drug release profile must be met. This might be a tough task for a formulator to achieve these conditions, especially in bi-layer tablet formulation in which a double compression technique is involved, because of poor flow and compatibility characteristics of the drug. The compaction of the material involves compressibility and consolidation.
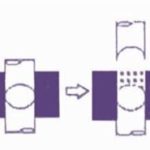
- Filling of the first layer
- Compression of the first layer
- Ejection of the upper punch
- Filling of the second layer
- Compression of the second layer
- Ejected bi-layer tablet fully
FIG. 1: STEPS IN BI-LAYER TABLET FORMULATION
Various Techniques for Bi-layer Tablet: 13, 14
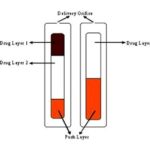
OROS® Push Pulls Technology: It includes two or three layers amongst which the one layer is essential of the drug, and the other layer is the push layer. The drug layer consists of drugs, along with two or more various agents.
So, this drug layer comprises of drug which is in poorly soluble form. Suspending agents and osmotic agents can also be added. A semi-permeable membrane surrounds the tablet core Fig. 2.
FIG. 2: BI-LAYER AND TRI-LAYER OROS PUSH PULL TECHNOLOGY
L-OROSTM Technology: This system used for the solubility issue Alza developed the L-OROS system where a lipid soft gel product containing drug in a dissolved state is initially manufactured and then coated with a barrier membrane, then osmotic push layer and then a semi-permeable membrane, drilled with an exit orifice Fig. 3.
FIG. 3: L–OROSTM TECHNOLOGY
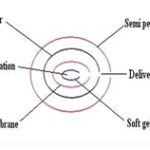
EN SO TROL Technology: Enhancement of solubility or to create an optimized dosage form uses an approach to drug delivery focusing on identification and incorporation of the identified enhancer into controlled release technologies.
DUROS Technology: This system is also called as “Miniature drug dispensing technology”. Its functioning resembles a miniature syringe that releases drugs in a continues and consistent manner in small concentrated form for a long period of time. Drug molecules are protected from enzyme with the help of cylindrical titanium alloy reservoir present outside having high impact strength.
FIG. 4: EN SO TROL TECHNOLOGY
DUREDAS™ Technology: This technology gives a combination release pattern of the drug, i.e., immediate or sustained release. This system provides one drug with a different release pattern or two drugs of the combination release pattern. In this system, different release patterns achieved by using a combination of the hydrophilic polymer. This technology provides a number of advantages, i.e., combination release in one tablet or another advantage is two drugs incorporated in a single dosage form. During the process of manufacturing a bi-layer tablet by using DUREDAS™ Technology, immediate-release granulate compressed first followed by a sustained release layer. DUREDAS™ Technology first used for the development of OTC controlled release analgesics
Geminex Technology: With the help of this, the therapeutic efficacy of drugs can be increased greatly, also useful in minimizing side effects. This technology delivers one or more drugs with different release rates in a single dosage form. It is very useful both for the industry as well as patients. Geminex Technology actively applied by pen west in following areas – diabetes, cardiovascular diseases, cancer, and CNS disorders
Various Approaches used in the Bi-layer Tablet: 15, 16
- Floating Drug Delivery System: These are designed to have a low density and thus float on gastric contents after administration until the system either disintegrates or the device absorbs fluid until its density is such that it can pass easily from the stomach responsible for gastric emptying. The design of a bi-layer tablet is such that one layer gives the immediate dosing of the drug, and it gives faster onset of action. The other layer is designed as a floating layer that floats in the stomach (GI-fluid).
Disadvantages:
- It might not control the loss of density required to exit from the stomach.
- These tablets cannot be applicable to high doses of highly water-soluble drugs.
- The performance of floating formulation may also be posture dependant. Hence, floating dosage forms might be expected to only have limited applications.
- Polymeric Bio-adhesive System: These are made to imbibe fluid after administration in such a way that the outer layer is seen to be viscous, which adheres to the gastric mucus layer. It is required that gastric retention increases until the adhesive forces are weakened. These are designed, such as one layer has immediate dosing, and the next layer has Bioadhesive property.
Disadvantages:
- The success seen in animal models is not as that of human subjects due to differences in mucous amounts, consistency between animals and humans.
- The system adheres to mucous, not mucosa.
- The mucous layer in humans can slough off easily and can carry drugs along with it.
- Swelling System: These systems are small on administration, so ingestion of the dosage form is made easy. Upon ingestion, they swell rapidly or unfold to a size that precludes passage through the pylorus until after drug release has progressed to a required degree.
Slow erosion or its breakdown into smaller particles allows it to leave the stomach. The bi-layer tablet might contain an immediate-release layer with the other layer as a conventional release.
Challenges in Bi-layer Manufacturing: Conceptually, bi-layer tablets can be seen as two single-layer tablets compressed into one. In Practice, there are some manufacturing challenges 17, 18.
Delamination: Tablet falls apart when the two halves of the tablet do not bond completely.
Cross-contamination: When the granulation of the first layer intermingles with the granulation of the second layer or vice versa, cross-contamination occurs. Proper dust collection can prevent cross-contamination.
Production Yields: In order to prevent cross-contamination, dust collection is required, which leads to losses. Thus, bi-layer tablets have lower yields than single-layer tablets.
Cost: Bi-layer tableting is expensive than single layer tableting for several reasons. The tablet press costs more. The press generally runs slowly in bi-layer mode.
Third, the development of two compatible granulations is mandatory, which means more time spent on formulation development, analysis, and validation.
FIG. 5: DRUG RELEASE FROM BILAYER TABLET 18
Evaluation of Bi-layer Tablets:
- General Appearance: It includes its visual identity; “elegance” is required for consumer acceptance. Other parameters are tablet’s size, color, shape, presence or absence of an odor, taste, surface texture, physical flaws and consistency, and legibility of any identifying marking 19.
- Size and Shape: The size and shape of the tablet can be dimensionally described, monitored, and controlled.
- Tablet Thickness: Tablet thickness is an important characteristic when it comes to reproducing appearance and also in counting by using filling equipment. Some filling equipment utilizes the uniform thickness of the tablets as a counting mechanism.
- Weight Variation: Standard procedures are followed as described in the official books 20.
- Friability: The friability test relates to the hardness of the tablet and is planned to test the ability of the tablet to withstand abrasion in all the processes like the packaging. It is measured by the Roche friabilator. Tablets are placed in the apparatus after they are weighed where they are exposed to rolling and repeated shocks as they fall 6 inches in each turn within the apparatus. It continues for four minutes or 100 revolutions. The loss occurring due to abrasion is measured by tablet friability. The value is expressed as a percentage.
A maximum weight loss of not more than 1% of the weight of the tablets being tested during the friability test is considered generally acceptable, and any broken or smashed tablets are not picked up. Normally, when capping occurs, friability values are not calculated.
A thick tablet may have less tendency to cap whereas thin tablets of large diameter often show extensive capping, thus indicating that tablets with greater thickness have reduced internal stress the loss in the weight of the tablet is the measure of friability and is expressed in percentage as 20:
% Friability = 1‐ (loss in weight / Initial weight) × 100
- Hardness (Crushing strength): The hardness of the tablet will be carved out using Monsanto type hardness tester. The hardness of the tablet is measured in Kg/Cm2. The hardness is considered as an important parameter which helps to overcome resist the tablets to shipping or breakage under conditions of storage 21.
- Stability Study (Temperature dependent): The bi-layer tablets are packed in suitable packaging and stored under the following conditions for a period as prescribed by ICH guidelines for accelerated studies.
TABLE 2: RECOMMENDED LONG TERM AND ACCELERATED STORAGE CONDITION
| Study | Storage
condition |
Minimum time period |
| Long term | 25 °C±2 °C / 60% RH ± 5% RH
30 °C±2 °C / 65% RH ± 5% RH |
12 months |
| Intermediate | 30 °C±2 °C / 65% RH ± 5% RH | 6 months |
| Accelerated | 40 °C±2 °C / 75% RH ± 5% RH | 6 months |
Applications:
- Bi-layer tablets deliver two different drugs having a different release profile.
- Bi-layer tablets deliver the loading dose and maintenance dose
- Bi-layer tablets are mainly used in combination with the modified release.
- Bi-layer tablets are used for bi-layer floating tablets in which one layer is a floating layer; another one is an immediate release layer of the drug.
CONCLUSION: Bi-layer tablet is selected for sequential release of two drugs in combination or separate two substances which are incompatible, for sustained release tablet where one layer is immediate release and the second layer is the maintenance dose. Bi-layer tablets give an opportunity for producers to improve their products’ efficacy, and protect against impersonator products. Bi-layer tablet GMP requirements can differ widely. When a bi-layer tablet requires to be developed along with accurate weight control of the two layers, compression force-controlled presses are limited as to their insufficient sensitivity and thus lack of accuracy at low compression forces required to secure interlayer bonding.
Accurate individual layer weight monitoring/control at high speed and in combination with reduced layer separation risk can be achieved with the displacement weight control system based presses. A bi-layer tablet has been done with various or various combinations, which is useful for various ailments. Thus, bi-layer formulation is a convenient dosage form, safe and posses greater advantages to both patient and clinician that it may be administered as a single tablet once a day. Bi-layer tablet is quality, and GMP requirements can vary widely.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Abebe A, Akseli I, Sprockel O, Kottala N and Cuitiño AM: Review of bilayer tablet technology. International Journal of Pharmaceutics 2014; 461(1-2): 549-58.
- Kiran B, Rao PS, Babu GR and Kumari MV: Bilayer tablets-a review. International Journal of Pharmaceutical, Chemical & Biological Sciences. 2015; 5(3): 510-16.
- Reddy P, Rao D and Kumar RK: Bi-layer technology-an emerging trend: a review. Int J Res Dev Pharm L Sci 2013; 2(3): 404-11.
- Swain RP and Pendela S: Formulation and evaluation of gastro-bilayer floating tablets of simvastatin as immediate release layer and atenolol as sustained release layer. Indian Journal of Pharmaceutical Sciences 2016; 78(4): 458-68.
- Breech JA, LucisanoLJ and Franz RM: Investigation into substrate cracking of a film‐coated Bilayered tablet. Journal of Pharmacy and Pharma 1988; 40(4): 282-83.
- Vishwakarma AG, Mogal RT and Pawar AY: Bi-layer Tablet-A new ways in oral drug delivery system. Int Journal of Pharm Tech Research 2014; 6(5): 1416-28.
- Lende LK, Banerjee SK, Gadhave MV, Gaikwad DD and Gaykar AJ: Review on: bilayer floating tablet. Asian Journal of Pharma Res and Develop 2013; 1(1): 31-39.
- Devtalu SV, Patil AE, Bari MM and Barhate DS: A Review on Novel Approach–Bi-layer Tablet Technology. Int J Pharm Sci Rev Res 2013; 21(1): 46-52
- Patel M and Shah N: A Sequential Review on Bilayered Tablets. Journal of Pharmaceutical Science and Bio Scientific Research 2013; 3(5): 163-69.
- Rudnic EM and Kottke MK: Tablet dosage form. Modern Pharmaceutics 72: 369.
- Deshpande RD, Gowda DV, Mahammed N and Maramwar DN: Bi-layer tablets-An emerging trend: a review. International Journal of Pharmaceutical Sciences and Research 2011; 2(10): 2534-44.
- Li SP, Karth MG, Feld KM, Di Paolo LC, Pendharkar CM and Williams RO: Evaluation of bilayer tablet machines a case study. Drug Development and Industrial Pharmacy 1995; 21(5): 571-90.
- Kalam MA, Humayun M, Parvez N, Yadav S, Garg A, Amin S, Sultana Y and Ali A: Release kinetics of modified pharmaceutical dosage forms: a review. Cont J Pharm Sci 2007; 1: 30-35.
- Zamorano J and Edwards J: Combining antihypertensive and antihyperlipidemic agents–optimizing cardiovascular risk factor management. Integrated Blood Pressure Control 2011; 4: 55-71.
- Pujara ND, Gokani RK and Paun JS: Bilayer tablet–an emerging trend. International Journal of Pharmaceutical Research and Development 2012; 4(04): 102-11.
- Shirwaikar AA, Kumar SM, Jacob S, Rashi W and Ravi K: Recent developments in floating drug delivery systems for gastric retention of drugs, an overview. Indian Drugs 2006; 43(9): 697-04.
- Gohel MC, Parikh RK, Nagori SA and Jethwa BA: Fabrication and evaluation of bi-layer tablet containing conventional paracetamol and modified release diclofenac sodium. Indian Journal of Pharmaceutical Sciences 2010; 72(2): 191-96.
- Hiremath D, Goudanavar P, Azharuddin M, Udupi R H and Sarfaraz M: Design and characterization of bilayer controlled release matrix tablets of losartan potassium. Int J Pharm Res 2010; 2(4): 34-39.
- Ramesh A: Formulation and evaluation of bilayer sustained release matrix tablets of Metformin HCl and Pioglitazone. Amer-Euras J Sci Res 2010; 5(3): 176-82.
- Udayakumar T and Suresh AG: Formulation and Evaluation of Immediate and Sustain Release bilayered tablet with Glibenclamide and Metformin Hydrochloride. International Journal of Research and Development in Pharmacy and Life Sciences 2013; 2(2): 337-43.
- Dhumal RS, Rajmane ST, Dhumal ST and Pawar AP: Design and evaluation of bilayer floating tablets of cefuroxime axetil for bimodal release. Journal of Scientific and Industrial Research 2006; 65: 812-16.
How to cite this article:
Satpute VM: Bi-layer tablet: a controlled release dosage form. Int J Pharmacognosy 2020; 7(7): 175-82. doi link: http://dx.doi.org/ 10.13040/IJPSR.0975-8232.IJP.7(7).175-82.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
175-182
772
1201
English
IJP
V. M. Satpute
Naldurg Tal Tuljapur, Osmanabad, Maharashtra, India.
Correspondence to Author: Vivek Mahavir Satpute Naldurg Tal Tuljapur, Osmanabad - 413601, Maharashtra, India. E-mail: satputevivek9@gmail.com
11 April 2020
18 June 2020
29 June 2020
10.13040/IJPSR.0975-8232.IJP.7(7).175-82
01 July 2020