AUTHENTICATION OF POLYHERBAL FORMULATIONS USING PCR TECHNIQUE
HTML Full TextAUTHENTICATION OF POLYHERBAL FORMULATIONS USING PCR TECHNIQUE
Juwairiya Butt * 1, Saiqa Ishtiaq 1, Bushra Ijaz 2, Zulfiqar Ali Mir 3, Shumaila Arshad 4 and Sophia Awais 4
Department of Pharmacognosy 1, Punjab University College of Pharmacy, Lahore, Pakistan.
Functional Genomics Lab 2, Department of Bioinformatics 3, Center of Excellence in Molecular Biology, Lahore, Pakistan.
Faculty of Pharmacy 4, The University of Lahore, Lahore, Pakistan.
ABSTRACT: Unprecedented growth in popularity of complementary medicines raised concerns about their quality and safety. So, there is a need to develop methods for their sensitive, specific and accurate analysis. Authenticity and similarity of medicinal nutrients and strict procedures of material handling are significant features to maintain the quality of herbal preparations. Genome-based methods to authenticate these plants revolutionized the authentication process. Developing DNA molecular markers by sequencing a standard zone of the DNA is the best technique to identify the adulterants as well as to authenticate the required species of medicinal plants. Application of molecular biological technique serves as one of the very consistent systems for authentication of natural herbal materials. The progress of authentic analytical methods is a major challenge to scientists as natural products are optimized as drug-like molecules. Current work is based on the use of PCR technique for authentication of Glycyrrhiza glabra L. standard crude root and in polyherbal formulations i.e., Hamdard’s Joshanda, Marhaba’s Joshanda, GNC Herbal supplement and Joshaba Sadar (Chest Tea) available in the market, provided isolated DNA from dried roots of the sample was used as templates in PCR. All the products gave the desired results except one. It proved to be a complementary tool to control the quality of herbal materials alone and in a different marketable herbal product having Glycyrrhiza sp. as their active ingredient.
| Keywords: |
Glycyrrhiza, Authentication, cGMP, Chloroplast DNA, Herbal medicine, Polymerase chain reaction, Species-specific primers
INTRODUCTION: Commercialization of formulations based on natural herbal materials requires reproducible standards for effective quality control. So, the standardization and pharmaceutical quality of all the phytopharmaceuticals must be determined. The quality of herbal drugs must be as high as that of other medicinal preparations. In the finished herbal medicinal products quality is difficult to achieve and is more complex than for other pharmaceuticals.
Increased cases of poisoning are demanding to identify the risks and set standards for the safety of drugs of natural origin. Authentication means to prove that material is true in itself; it involves many parameters like morphology, microscopy, chemical analysis and DNA fingerprinting.
Previously, regulatory principles most of the times emphasized upon just quantifiable analysis of formulations 1. Currently, there is a marked an increase in natural products investigation that leads to, search for new leads for drug development, biotechnological applications for pharmaceuticals, nutraceuticals, authentication of old-fashioned prescriptions. Many people consider products stamped "natural" are always safe which is not essentially true 2.
Passionate work of Pharmacognosist played a vital role in the introduction of new technologies like microscopy, TLC, GC HPLC, MS, NMR to check the quality of pharmaceutical preparations. Herbal materials cause intrinsic reactions (predictable toxicity or idiosyncratic reaction i.e. type A or type B) due to active medicine and extrinsic reactions (contaminants e.g. microbes, pesticides or heavy metals) due to inappropriate handling or failure of GMP 3.
Previously, the amount of herbal extract in preparation conforms to claim on label but the suppliers these days provide standardized extracts, to achieve target strength. If the selective marker is present in high concentration or a non-standardized pharmacologically active component is present it will not contribute appreciably to the herb activity. The plants are variable raw materials as many factors contribute to this variation 4. Such variants contribute considerably in many batches of a product so modify its efficacy. Defining the authenticity and quality of drugs of natural origin remain as much a frontier as it is a vital science in guaranteeing clinical use of herbal drugs 2.
It is essential to investigate the current market of drugs of natural origin and find reasons for various confusions not solved by conventional methods of authentication. Real and consistent identification of herbal materials can only be attained by exercising advance molecular genetics tools 2, 5, 6. The advance practice of genomic authentication has extended the scope of pharmacognostical science 7 to the molecular level so it should be added as a parameter to control the quality of drugs of natural origin 8.
A genetic marker is a measurable character that can detect the variation. Molecular markers are based on biochemical macromolecule deoxyribonucleic acid (DNA). These have revolutionized research activities in biological science. DNA based molecular markers act as an efficient and versatile tool and complement classical strategies for the genetic analysis. Since the development of DNA based molecular markers, they are constantly modified to increase their use and the automation of the process of genome analysis. The genetic markers present in plants are playing their role in genome-based procedures, e.g. SNP 9, 10, 11, 12, 13 and micro-satellites (SSR) 14, 15, 16, 17, 18. The application of sequence-independent array technology to determine genomic polymorphism, PCR for the amplification of a minute locus of genomic DNA and RT-PCR to quantify amplified markers is exclusive progress 20. As current DNA based techniques only show the presence of a target DNA, Simply verifying the presence of a species does not give any indication of its quantity in the mixture. It’s a limitation of PCR based DNA identification technique.
In this research, PCR technique is applied to the species identification of Glycyrrhiza glabra targeting one gene loci. One standard sample was obtained from Botanical garden of GC University, Lahore and was compared with licorice, sweet wood present in Pakistan’s most famous herbal remedy “Johar Joshanda”, Joshaba Sadar (Chest Tea) and capsules of Licorice root used for digestive problems. It is necessary to discern the false herb from the genuine herb in terms of their origins, distribution areas. Only in this way quality can be guaranteed 21.
Pakistan is the world’s sixth largest country with an area of 796095 Km2 and population in millions. However, more than 70 million live in urban areas and around 110 million in rural areas 22. Due to this large number of Population in rural areas with low daily income complementary medicine is taken as the first line of treatment. As a direct result of this, the Unani and homeopathic system is practiced quite heavily, so there is a need to develop a relationship between conventional allopathic physicians and complementary alternative medicine 23.
Glycyrrhiza glabra Linn. sweet wood commonly known as ‘liquorice’ is a potent and unique medicinal herb 24, in Pakistan commonly known as “mulethi” Glycyrrhiza glabra is a perennial herb. Liquorice root consists of the dried unpeeled or peeled, whole or cut root of Glycyrrhiza glabra Linn. and / or Glycyrrhiza inflata Bat. and / or Glycyrrhiza uralensis Fisch (dried drug) 25.
Liquorice presents demulcent and expectorant properties for dissolving and facilitating the discharge of mucus in catarrhs and for upper respiratory tract diseases and is currently employed in cough preparations. Ulcer-healing properties, anti-inflammatory, and mild laxative activities have been documented 26 and in the treatment of atopic dermatitis 27. According to the British Herbal Pharmacopoeia, Glycyrrhiza glabra has anti-inflammatory and expectorant activities 28. The anti-tussive and expectorant properties of the drug have also been attributed to glycyrrhizin, which accelerates tracheal mucus secretion 2.
MATERIALS AND METHODS: Dried root samples of Glycyrrhiza glabra (fresh sample, intact form, powdered form, crushed form, and capsule) for this study were acquired from the botanical garden of GC University, local Dawakhana and Pharmacy store or local market of Lahore, Pakistan. To shelter the producer’s identity, sample formulations are named as fresh sample, intact form, powdered form, crushed form, and capsule.
TABLE 1: DOSAGE FORMS OF THE HERBAL FORMULATIONS
| Formulation | Targeted specie | Part Used | Quantity | Dosage Form | Therapeutic Uses |
| Standard Material / Botanical garden of GC University | Glycyrrhiza glabra Linn. (Mulethi) | Dried root and rhizome | 3.00g | Raw
form |
Anti-tussive, expectorant |
| Hamdard’s
Joshanda |
Glycyrrhiza glabra Linn. (Mulethi) | Dried
rhizome |
3.00 g | Intact
form |
Natural cure for cough and cold relieve |
| Marhaba
Joshanda |
Glycyrrhiza glabra Linn. (Mulethi) | Dried
rhizome |
3.00 g | Intact
form |
Natural cure for cough and cold relieve |
| Joshaba Sadar
(Chest Tea) |
Glycyrrhiza glabra Linn. (Mulethi) | Dried
rhizome |
1.80 g / 12.00 g | Crushed
from |
Chest
tea |
| GNC Herbal Plus
Whole Herb |
Glycyrrhiza glabra Linn. (Mulethi) | Licorice root herbal supplement | 450 mg / capsule | Powdered | Traditional digestive herb |
Extraction of DNA: CTAB / Chloroform-isoamyl alcohol DNA extraction protocol was used as mentioned by 29, 30. The CTAB buffer was prepared by mixing CTAB, PVP, and β-mercaptoethanol with the ratios as given below.
TABLE 2: COMPOSITION OF DIFFERENT VOLUMES OF CTAB BUFFER
| S. no. | CTAB | PVP-40 | β-merc | Tris-Cl | EDTA | NaCl | H2O |
| 1 | 0.5 ml | 0.02 gm | 2.5 μl | 0.5 ml | 0.2 5 ml | 2 ml | 1.15 ml |
| 2 | 5 ml | 0.2 gm | 25 μl | 5 ml | 2.5 ml | 20 ml | 11.5 ml |
| 3 | 20 ml | 0.8 gm | 100 μl | 20 ml | 10 ml | 80 ml | 46 ml |
CTAB (Cetyl trimethylammonium bromide), PVP (Polyvinylpyrrolidone), β-merc (β-Mercaptoethanol), Tris (tris (hydroxyl-methyl) aminomethane) Cl, EDTA (Ethylenediaminetetra acetic acid), NaCl (Sodium chloride)
TABLE 3: PRIMER SEQUENCE FOR GLYCYRRHIZA GLABRA
| Gene | Forward Primer | Reverse Primer | Product Size (bp) | Annealing Temp (Tm) °C |
| trnH-trnK1 intergenic spacer region cpDNA | ACGGGAATTGAACCCGCGCA | CATATGACTTCACAATGTAAAATC | Around ± 220 | 57.3 |
+base pairs (bp)
Applying the modified CTAB method, fine powder sample (40-50mg) was mixed in extraction buffer (as mentioned in Table 2) and incubated in a water bath at 65 °C for 20 minutes. 700 μl of chloroform-isoamyl alcohol (24:1) was mixed with the sample and vortexed well to form an emulsion. Eppendorf tubes containing emulsion were centrifuged at 15000 rpm for 30 min.
Pre-chilled (900 μl) isopropanol was added to the supernatant and incubated for 1 h at RT followed by centrifugation at 14000 rpm for 30 min so DNA can be precipitated. Treatment with RNase A (10 mg/ml) was done to eliminate RNA from crude DNA. The quality and quantity of DNA can be confirmed by using NanoDrop™ by noticing the absorbance ratio at A260/280.
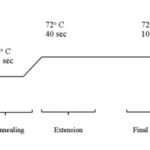
PCR reaction mixture containing 10X reaction buffer 1.5 μl, dNTPs 0.3 μl, 50 mM MgCl2 0.45 μl, forward primer (ACGGGAATTGAACCCGCG CA) 0.5 μl, reverse primer (CATATGACTTCACA ATGTAAAATC) 0.5 μl, Taq Polymerase 0.14 μl, water, and template was used with following PCR cycling parameters: initial denaturation step of 30 sec at 98 °C; denaturation of 20 sec at 98°C, annealing for 30 sec at 57 °C and extension for 30 seconds at 72 °C; and final extension period of 10 min at 72 °C and with 35 times repeated cycles. The entire process was optimized after several runs of PCR and Gradient PCR for the right Tm of the primer range from 55 ºC - 60 ºC and that is found to be 57 ºC, and reactions without template DNA were utilized as controls. Typically, PCR is a process of runs of 35 times repeated cycles involving 2 - 3 different stages of temperature.
FIG. 1: POLYMERASE CHAIN REACTION THERMAL CYCLE STAGES
The PCR products were electrophoresed on 1.8% (w/v) agarose gel, 0.5X TAE buffer and ethidium bromide stain at 80 V for ~30 min 31. An electronic system called gel documentation system (Dolphin gel doc system) was used to visualize amplified DNA bands under UV light, and the results were recorded in system for further analysis.
TABLE 4: VOLTAGE AND TIME FOR THE GEL ELECTROPHORESIS
| Volts | Recommended Time in min | |
|
80 |
Minimum | Maximum |
| 30 | 45 min | |
RESULTS: This study determined the presence of Glycyrrhiza glabra in herbal drugs by using PCR based molecular technique. Agarose separates DNA molecules of the same size which appear in the form of a clear band, and the size can be determined by comparing with a DNA ladder and analyzed using Illuminator with a Gel documentation system and GrabIt™ software.
Sample A: Standard Sample of Botanical Garden of GCU: Electrophoresis separated DNA fragments by size, and it was found to be around ± 220 bp, and the band is shown in Fig. 2 and was compared with other samples.
Sample B: Licorice Root Herbal supplement (GNC): A full band of product size around ± 220 bp on agarose gel electrophoresis was observed under UV light shown in Fig. 3.
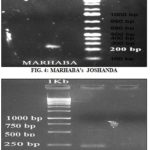
Sample C: Marhaba Joshanda (Marhaba Laboratories): Product size of around ± 220 bp against DNA ladder of known fragments when visualized under UV light as shown in Fig. 4.
Sample D: Hamdard’s Joshanda (Hamdard Laboratories): Under UV light appearance of band is observed against DNA ladder and product size is measured that is found to be around ± 220 bp as shown in Fig. 5.
Sample E: Joshaba Sadar (Chest Tea): The DNA extracted from this sample could not be identified with this primer; the sample is labelled as a mixture of plant materials. There are various reasons for this, the amplified DNA might have come either from another species or it might have been degraded (due to DNA shearing), and more than one herb increases the complexity and difficulty in analysis.
Chemical analysis of such a mixed preparation containing many compounds from different species produces a highly complex profile. However, other methods, e.g. the multiplex Plant ID system could potentially identify all different species present in the preparation and also test for species that are known to be used as adulterants and sequencing can be used to identify DNA of concerned species from highly degraded DNA.
TABLE 5: DNA QUANTIFICATION OF GLYCYRRHIZA GLABRA BY SPECTROPHOTOMETER
| Samples | Manufacturers | Label Claim
(Glycyrrhiza glabra) |
Product
Description |
Concentration
ng/μl |
| A) Standard crude sample | Botanical Garden of GCU Lahore | N/A | Crude
state |
58.6 |
| B) Licorice root Herbal supplement | GNC Herbal Plus whole herb | 450 mg whole herb per capsule | Filled capsules with dried ground material | 119.9 |
| C) Marhaba
Joshanda |
Marhaba Laboratories Pvt. Ltd., | 5 gm of pure herb per packet | Washed in
crude form |
67.6 |
| D) Hamdard’s Joshanda | Hamdard Laboratories
(Pvt.) Ltd., |
5 gm of standardized herb | Washed in a
crude state |
107.5 |
| E) Joshaba Sadar Chest Tea | Hamdard laboratories Pvt. Ltd., | 1.8 gm / 12 gm ground state mixed with other herbs | Ground mixed materials of all the herbs present in the formulation | 100 Highly degraded and fragmented DNA was detected |
DISCUSSION: With the population growth in the developing world and increasing interest in the industrialized nations has greatly expanded the demand for medicinal plants themselves and the products derived from them. Even the growth of the pharmaceutical industry and the unceasing development of new and more effective synthetic and biological medicinal products have not diminished the importance of medicinal plants in many societies. Herbs are no doubt precious natural source and economic crops due to their role in health care, i.e. traditional and modern medicine.
In Pakistan, the total turnover of crude drugs is worth around in millions although its cultivation is intermittent and grown as minor crops. Pakistan does not meet its total requirements and has to import major part of herbal materials from Nepal, Sri Lanka, India, China, Kenya, and Uganda. Pakistan annually spends millions on the import of raw herbal materials and medicinal/pharmaceutical products. So, it is required by higher authorities to monitor how much plant material is collected/ cultivated and actual quantity reaches to the market after passing through the process of refinements. The herbs are sold either dried or fresh to the local traders who sell them to wholesalers and ultimately to Pharmaceutical concerns or exporters. Pakistan’s major exporters of medicinal herbs and herbal medicines are Hamdard Laboratories (Pvt.) Ltd.; Herbasian (Pvt.) Ltd.; Hashmi Surma; Qarshi Industries (Pvt.) Ltd.; Tayyebi Dawakana; Marhaba and Medics Laboratories out of them Hamdard Laboratories is one of the leading stake holders.
Efforts should be made to explore herbal product development. As safety is imperative, though herbal product’s use has been embraced as complementary and alternative medicines, safety is conceded due to lack of suitable quality procedures, inappropriate labeling, adulteration, substitution and lack of patient data which has resulted in the decline of faith in crude drug promotion 44. Safety of natural origin medicines has become a major concern to both national health authorities and the general public 32. Therefore, regulatory policies on herbal medicines need to be standardized and strengthened on a global scale and with this adequate training regarding the use of herbal products with prescription or non-prescription medicine to conform to standards of safety, quality, and efficacy are to be considered for public health. Regulations and WHO support is helping in the preparation of model guidelines in this field and the herbal medicines and phytonutrients, or nutraceuticals continues to grow worldwide.
DNA based molecular markers, however, are important tool in quality assurance of medicinal plant species in the plant kingdom. This serves as qualitative/quantitative diagnostic tool for identification of medicinal herbs from harvest to finished product. The recent era is the use of molecular biological techniques that helps in identification of species 33. Although, species-specific constituents are helpful in the identification of species, e.g., three kinds glycoumarin, glabridin and licochalcone A (G. uralensis, G. glabra, G. inflata) but with the help of this technology, we can distinguish their kinds. In this study species identification of Licorice was done present in polyherbal formulation, because when licorice is used for medicinal purposes, the licorice species should be selected with recognition of even its constituent properties, e.g. G. uralensis mostly used as tonic, an antipyretic, treat acne, pimples, antidot while G. glabra to cure influenza, uterine complaints, etc. 34 and it provides vital and absolute mean of authentication.
These days herbal medicines are present in all forms i.e. simple as well as in highly processed form mostly marketed as supplements for health augmentation, but there are still concerns to be raised about the authenticity and efficacy of herbal medicines. Different techniques of molecular biotechnology can be utilized for the identification/ authentication of herbal materials, in well-refined dosage forms such as tablets, capsules, troches, injections and oral liquids. Usually, a PCR based method is applied to audit highly processed medicines and also assist in monitoring their quality and legality. A lot of work has been completed by researchers on Chinese highly processed patent drugs consisting of different kinds of plants or animals 35. In this research Glycyrrhiza glabra Linn. is chosen to test the reliability of the quality control using PCR technique which is a widely grown herb in Pakistan due to its high medicinal value such as antioxidant, antibacterial, digestive, anti-tussive, cough and cold treatment, anti-inflammatory activities due to various constituents e.g. flavonoids, coumarins, chalcons, glycyrrhetinic acid. Even synthetic antibiotic cannot withstand heat treatment at 100 ºC but Glycyrrhiza glabra after boiling at 100 ºC shows antibiotic activity against Staphylococcus aureus. Its roots, rhizomes and whole herb are used in different herbal medicines. There are different dosage forms available in the market containing Glycyrrhiza glabra e.g. capsules (root powder by GNC Herbal plus), sachet (entire roots by Hamdard’s Joshanda and Marhaba’s Joshanda) and cough syrups e.g. Inflo syrup, Myocid syrup, Ivy syrup (Herbasian Laboratories Pakistan Pvt. Ltd.,).
G. glabra have different varieties depending on the habitat i.e. G. glabra var. typica (Spanish liquorice) having purplish blue colored papilionaceous flowers and large number of stolons, G. glabra var. glandulifera (Russian liquorice) which is a big root stock along with several elongated roots without stolons. G. glabra var. violacea (Persian liquorice) has violet flowers, various hybrids and a major adulterant Abrus precatorious. The sweetish and mucilaginous roots of Abrus precatorious are similar in morphology with roots of G. glabra. Based on morphology and histology, it is very difficult to distinguish them. Herb authentication has presented a great challenge for people using them for medical purposes. Wrong herb usage can worsen the condition and even cause 36, 37. So, intentional and unintentional adulteration is needed to be avoided.
The objective of this research was to authenticate the presence of Glycyrrhiza glabra in different formulations available in market, i.e. capsules (GNC Herbal Plus), sachet (Marhaba Joshanda, Hamdard’s Joshanda) and (Joshaba Sadar Chest-Tea) and Crude G. glabra (as standard by Herbasian Laboratories Pvt. Ltd., Each capsule of Licorice root herbal supplement by GNC Herbal Plus contains 450 mg root powder per capsule Marhaba’s Johar Joshanda contains 5 gm of root per packet, Hamdard’s Johar Joshanda contains 5gm of root herb per sachet and Joshaba Sadar Chest - Tea (OTC) contains 1.8gm/12gm of Glycyrrhiza glabra.
With the help of PCR, it becomes possible to detect DNA of specific herb in processed drugs which has undergone industrial extraction, filtration, and sterilization. There is no doubt about the discriminatory power of DNA among different species 38. To carry out this work G. glabra pure sample was authenticated from botany department GC University to compare with the commercial samples. DNA extraction protocol depends on the types of tissues, e.g., roots, seeds, leaves, etc. and concentration of DNA required. Using a modified CTAB method by Salim et al., (2007) resulted in moderately degraded genomic DNA in concentrations detectable by agarose gel electrophoresis but was without purification and viscous which can’t be amplified by PCR. There is a possibility the presence of pigments prohibits amplification by PCR. Later to obtain the DNA of high quality and purity independent of secondary metabolites was extracted using Cullings (1992), 29, 30 methods and resulted in successful PCR amplification using specific primers.
This work helped to attain a protocol for detecting all commercial licorice root/rhizome samples to species. Herbal samples used in this analysis were containing licorice root whole (Hamdard Joshanda, Marhaba Joshanda), chopped-up / comminuted (Joshaba Sadar, Chest tea) or powdered form (Herbal supplement by GNC Herbal Plus). Polymerized agarose acts as a sieve and separates the DNA by size. Around ± 220 bp size was determined with specific primer trnH-trnK1 in the crude sample by crude authenticated standard sample of GCU, Marhaba Joshanda, Hamdard Joshanda and Herbal supplement by GNC Herbal Plus confirmed the presence of Glycyrrhiza glabra in the marketed formulations. Amusingly, among five investigated samples, Joshaba Sadar (Chest Tea) was found to contradict the species labeled by the manufacturer, signs of admixture were located. This is the basis of failure in crude drug marketing that results in altered efficacy 44.
This technique proved to have specifically authenticated medicinal herb and have extensive applications in quality control of raw materials. The PCR based method can be carried out in any laboratory using unknown genomic DNA from any developmental stage and any part of herb 39. It was found that repeated thaw cycles didn't affect the quality of DNA. These results obtained are highly reproducible and DNA extracted was also very stable even after keeping DNA at room temperature for one week produced the desired results 19, 40.
Genome sizes in plants are remarkably diverse, with 2350 fold range from 63-149,000 Mb. Each plant species possess a characteristic number of base pairs in its nuclei known as it’s genome size / nuclear DNA content which is constant within a species. Bennetlt & Leithch (2011) states the average genome size of angiosperm 5800 Mb. The advantage of this approach is it’s rapidity, simplicity, reproducibility, discriminatory power and user-friendliness. Major drawbacks of this approach are a risk of contamination, the requirement of sequence information for the design of primers and it does not provide information about the presence of species which is not the target of primer.
An additional disadvantage is a need for performing electrophoresis to confirm amplification success of target sequences. However, by using Nested PCR the specificity and sensitivity of PCR can be enhanced. Real-time PCR is also known as quantitative PCR that can quantify the starting amount of specific DNA sequence in the sample utilizing fluorescent technology. Benefiting from PCR technique DNA markers have become a powerful tool for authentication of plant, animal, fungal and botanical sp. 41. Contrary to chemical fingerprinting which are strongly influenced by the age of the sample, physiological conditions environmental factors and can be recovered from any state (fresh, dried, processed) beside marker molecule is not tissue specific.
There is a need to increase consumer confidence in herbal medicine. Along with traditional this new innovative automated assays and specific tools DNA analysis are emerging and contribute to the next generation of technologies, e.g. mini-sequencing, nano-scale DNA sequence, Next Generation sequencing NGS 42. Further, extremely promising development is nanopore technology for identification of DNA bases and APEX (enzymatic genotyping to analyze variations of the genome in single multiplexed reaction), OLA to detect highly polymorphic gene. These will provide complete genome analysis, high multiplexing capacity, and future taxon identification. Recently, massively parallel sequencing technology produces millions of DNA sequence reads, i.e. Giga base pairs in a single run. These revolutionized research in medicine. However, there is no perfect DNA-typing method, and the choice of a particular technique is often a compromise that depends on several factors, including resources of the laboratory, financial constraints, available expertise, time limitations and more importantly, the research question pursued. All factors should be scrutinized to avoid an inappropriate choice.
CONCLUSION: Molecular techniques can be utilized to maintain the quality of herbal materials to ensure safety and efficacy. The developed method is one of the efforts to give the guidelines for the identification and authentication of Glycyrrhiza glabra as raw material and as an ingredient in different dosage forms.
FUTURE PROSPECTIVE: By using PCR technique we can develop the methods for the authentication of other herbal materials.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Li S, Han Q, Qiao C, Song J, Lung Cheng C and Xu H: Chemical markers for the quality control of herbal medicines: an overview. Chinese Medicine 2008; 3: 7.
- Zhao Z, Yuen J, Wu J, Yu T and Huang W: World Health Organization 1999. Glycyrrhiza glabra A systematic study on confused species of Chinese Materia Medica in the Hong Kong market. Annals Academy of Medicine, Singapore 2006; 35(11): 764-769.
- Drew AK and Myers SP: Safety issues in herbal medicine: implications for the health professions. Medical Journal of Australia 1997; 166(10): 538-541.
- Tilton R, Paiva AA, Guan JQ, Marathe R, Jiang Z, Van Eyndhove W, Bjoraker J, Prusoff Z, Wang H, Liu SH Dubey NK, Kumar R and Tripathi P: Global promotion of herbal medicine: India’s opportunity. Current Science 2004; 86(1): 37-41.
- Cheng YC: A comprehensive platform for quality control of botanical drugs (Phytomics QC): a case study of Huangqin Tang (HQT) and PHY906. Chinese Medicine 2010; 5(1): 30.
- Ali MA, Gyulai G, Hidvégi N, Kerti B, Al Hemaid FM, Pandey AK and Lee J: The changing epitome of species identification-DNA barcoding. Saudi Journal of Biological Sciences 2014; 21(3): 204-231.
- Ganie SH, Upadhyay P, Das S and Sharma MP: Authentication of medicinal plants by DNA markers. Plant Gene 2015; 4: 83-99.
- Heubl G: New aspects of DNA-based authentication of Chinese medicinal plants by molecular biological techniques. Planta Medica 2010; 76(17): 1963-1974.
- Huang LQ: Molecular Pharmacognosy; Beijing: Press of Beijing Medical University 2012: 2: 217.
- Hyten D, Song Q, Fickus E, Quigley C, Lim JS, Choi IY, Hwang EY, Pastor CM and Cregan P: High-throughput SNP discovery and assay development in common bean. BMC Genomics 2010; 11: 475.
- Myles S, Chia JM, Hurwitz B, Simon C, Zhong GY, Buckler E and Ware D: Rapid genomic characterization of the genus Vitis. Plos One 2010; 5: 8219.
- Arai-Kichise Y, Shiwa Y, Nagasaki H, Ebana K, Yoshikawa H, Yano M and Wakasa K: Discovery of genome-wide DNA polymorphisms in a Landrace cultivar of Japonica rice by whole genome sequencing. Plant and Cell Physiology 2011; 52: 274-282.
- Barbazuk WB and Schnable PS: SNP discovery by transcriptome pyrosequencing. cDNA Libraries: Methods and Applications 2011; 225-246.
- Marroni F, Pinosio S, Di CE, Jurman I, Boerjan W, Felice N, Cattonaro F and Morgante M: Large scale detection of rare variants via pooled multiplexed next-generation sequencing: towards next generation Ecotilling. Plant Journal 2011; 67: 736-745.
- Csencsics D, Brodbeck S and Holderegger R: Cost-effective, species-specific microsatellite development for the endangered dwarf Bulrush (Typha minima) using next-generation sequencing technology. Journal of Heredity 2010; 101: 789-793.
- Buehler D, Graf R, Holderegger R and Gugerli F: Using the 454 pyrosequencing - based techniques in the development of nuclear microsatellite loci in the alpine plant Arabis alpine (Brassicaceae). American Journal of Botany 2011; 98: 103-105.
- Delmas CEL, Lhuillier E, Pornon A and Escaravage N: Isolation and characterization of microsatellite loci in ferrugineum (Ericaceae) using pyrosequencing technology. American Journal of Botany 2011; 98: 120-122.
- Gardner MG, Fitch AJ, Bertozzi T and Lowe AJ: Rise of the machines-recommendations for ecologists when using next - generation sequencing for microsatellite develop-ment. Molecular Ecology Resources 2011; 11: 1093-1101.
- Michalczyk IM, Schumacher C, Mengel C, Leyer I and Liepelt S: Identification and characterization of 12 microsatellite loci in Cnidium dubium (Apiaceae) using next-generation sequencing. American Journal of Botany 2011; 98: 127-129.
- Appleby N, Edwards D and Batley J: New technologies for ultra- high throughput genotyping in plants. Plant Genomics 2009; 513: 19-39.
- Kumar A: Developments in Molecular Markers for Genetic Analysis of Plants. Plant Genetic Transformation and Molecular Markers, Pointer publisher, Jaipur, India Edition 2nd, 2010; 9: 173-203.
- Cao H, Cai JN, Liu YP, Wang ZT and Xu LS: Correlative analysis between geographical distribution and nucleotide sequence of chloroplast matK gene of Cnidium monnieri fruit in China. Chinese Pharmaceutical Journal 2001; 36: 373-376.
- NIPS: National Institute of Population Studies 2016. http://www.nips.org.pk/
- Flink S: International efforts spotlight traditional, complementary and alternative medicine. American Journal of Public Health 2002; 92(11): 1734-1739.
- Wang X, Zhang H, Chen L, Fan G and Gao X: Liquorice, a unique “guide drug” of traditional Chinese medicine: A review of it’s role in drug interactions. Journal of Ethnopharmacology 2013; 150(3): 781-790.
- European Medicines Agency: Assessment report on Glycyrrhiza glabra and / Glycyrrhiza inflata Bat. And / Glycyrrhiza uralensis Fisch., radix. European Medicines Agency 2010.
- Armanini D, Fiore C, Mattarello MJ, Bielenberg J and Palermo M: History of the endocrine effects of licorice. Experimental and Clinical Endocrinology and Diabetes 2002; 110: 257-261.
- Saeedi M, Morteza-Semnani K and Ghoreishi MR: The treatment of atopic dermatitis with licorice gel. Journal of Dermatological Treatment 2003; 14: 153-157.
- British Herbal Pharmacopoeia: Bournemouth: British Herbal Association 1990; 1.
- Doyle JJ: A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bulletin 1987; 19: 11-15.
- Doyle JJ and Dickson EE: Preservation of plant samples for DNA restriction endonuclease analysis. Taxon 1987; 36(4): 722.
- Joseph S and Russel DW: In-vitro Amplification of DNA by the Polymerase Chain Reaction, Edition 3rd, 2001; 8.
- Ekor M: The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Frontiers in Pharmacology 2013; 4: 177.
- Kondo Y, Ito T, Ma X, Watanabe S, Kreiswirth NB, Etienne J and Hiramatsu K: Combination of Multiplex PCRs for staphylococcal cassette chromosome mec type assignment: rapid identification system for mer, ccr, and major differences in Junkyard Regions. Antimicrobial Agents and Chemotherapy 2007; 51(1): 264-274.
- Asha R, Verma KN, Kumar SC, Chandra V, Singh PD and Panday KM: Phytochemical constituents, pharmacological activities and medicinal uses through the millenia of glabra Linn.: A review. International Research Journal of Pharmacy 2012; 3(8): 51.
- Rong C, Juan D, Xin C, Wei W, Afshan Y, Yun D, Xiaomao Z and Zhou T: DNA based identification of medicinal materials in Chinese patent medicines. Scientific Reports 2012; 2: 958.
- Bijoya C, Jigisha P, Jignesh K, Vasavdutta K, Preeti P and Bhat U: Significance of Molecular Markers and DNA Based Technology in Research and Standardization of Medicinal Plants: A review. International Journal of Research and Medicine 2015; 4(2): 5-16.
- Preethi JP, Padmini K, Lohita M, Swetha K, Priyanka B and Rao P: Adulterants and Substitutes of Foods and Herbs: A Review. International Journal of Medicinal Chemistry and Analysis 2014; 4(4): 213-217.
- Serrone DP, Attorri L and Palazzino G: Easy DNA extraction for rapid detection of Panax ginseng A. Meyer in commercial ginseng products. Natural Product Research 2007; 21(12): 1099-1103.
- Kethidi DR, Roden DB, Ladd TR, Krell PJ, Retnakaran A and Feng Q: Development of SCAR markers for the DNA-based detection of the Asian long-horned beetle; Anoplophora ghbripennis (Motschulsky). Archives of Insect Biochemistry and Physiology 2003; 52: 193-204.
- Schuster D and Appleby D: Does freezing and thawing of DNA solutions insert nicks in the double helix. Focus 1983; 5(2): 10.
- Kaplan SR, Ward DM, Shiflett SL and Kaplan J: Genome-wide analysis of iron-dependent growth reveals a novel yeast gene required for vacuolar acidification. The Journal of Biological Chemistry 2004; 279(6): 4322-4329.
- Lerner HR and Fleischer RC: Prospects for the use of next-generation sequencing methods in ornithology. Bio One 2010; 27(1): 4-15.
- Dubey NK, Kumar R and Tripathi P: Global promotion of herbal medicine: India’s opportunity. Current Science 2004; 86(1): 37-41.
How to cite this article:
Butt J, Ishtiaq S, Ijaz B, Mir ZA, Arshad S and Awais S: Authentication of polyherbal formulations using PCR technique. Int J Pharmacognosy 2018; 5(6): 382-90. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(6).382-90.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
11
382-390
704
1457
English
IJP
J. Butt *, S Ishtiaq, B. Ijaz, Z. A. Mir, S. Arshad and S. Awais
Department of Pharmacognosy, Punjab University College of Pharmacy, Lahore, Pakistan.
juwairiya.zulfiqar@pharm.uol.edu.pk
11 February 2018
26 March 2018
30 March 2018
10.13040/IJPSR.0975-8232.IJP.5(6).382-90
01 June 2018