ASSESSMENT OF ANTIOXIDANT, ANTIBACTERIAL AND ANTHELMINTIC ACTIVITIES OF ETHANOL EXTRACT OF LEAVES OF CROTALARIA PALLIDA (AITON)
HTML Full TextASSESSMENT OF ANTIOXIDANT, ANTIBACTERIAL AND ANTHELMINTIC ACTIVITIES OF ETHANOL EXTRACT OF LEAVES OF CROTALARIA PALLIDA (AITON)
M. H. Alam 1, M. M. Rahman 1, S. I. Sathi 1, S. F. Bellah 3, M. A. Mazid 3 and G. M. M. Murshid * 1
Pharmacy Discipline, Khulna University 1, Khulna - 9208, Bangladesh.
Department of Pharmaceutical Chemistry, Faculty of Pharmacy 2, Dhaka University, Dhaka - 1000, Bangladesh.
Department of Pharmacy, Manarat International University 3, Dhaka - 1216, Bangladesh.
ABSTRACT: The ethanol leaves extract of Crotalaria pallida (Aiton) was evaluated to investigate the Phytochemical nature and some selected pharmacological activities. The plant extract indicated the presence of combined reducing sugar, tannins, flavonoids, glycosides, alkaloids and steroids. The extract showed free radical scavenging activity with an IC50 value of 37.60 µg/ml, and standard ascorbic acid showed IC50 value of 16.95µg/ml. The total phenolics content (TPC) was found to be 14 ± 0.015 mg GAE/g of dried plant extract. Total flavonoids content was found to be 20.42 ± 0.001 mg QE / 100 g of dry extract. The extract showed mild antibacterial activity against the bacterial strains Vibrio cholera, Shigella flexneri, Shigella dysenteriae, in comparison with standard drug kanamycin (30 µg/disc). In the anthelmintic test, the extract showed a dose-dependent decrease in paralysis time and death time of worm Paramphistomum cervi (trematoda) when compared with standard albendazole. These results espouse the traditional use of the leaves of this plant.
| Keywords: |
Dysentery, Urinary tract infections, Vermifuge, Flavonoids
INTRODUCTION: For many years, plants are regarded as a puissance source of natural products especially as a source of viands and therapeutic agents for a vast range of ailments 1-2. Medicinal plants, containing versatile medicinal constituents, used in healthcare sectors and different parts of the world for a different therapeutic purpose, are a common application 3. In traditional uses, the leaves of C. pallida (Aiton) are used as the curative agent of urinary tract infections (UTI) and vermifuge 4. Moreover, the recent study has deciphered that it works as a potential HIV-1 protease inhibitor 5.
Oxidation is an imperative process for living organisms for different metabolic process. Usually, a tantamount of free radicals and antiradicals are produced in a normal physiologic system but when reactive oxygen species (ROS) are increased irrationally, incur oxidative damages, provoke biochemical changes and cause diseases. For instance, diabetes mellitus, aging, arthritis, inflammation and neurodegeneration, cancer and atherosclerosis 5-6.
Though many synthetic antioxidants have been established they are getting prohibited due to their toxic and carcinogenic comportment, and inquisition of novel antioxidant from plant parts has been started as it contains polyphenolics compounds, known properties of scavenging free radicals and can inhibit hydrolytic and oxidative enzymes 7. Anthelmintics may also be called vermifuges or vermicides 8.
Helminthiasis is one of the most important animal diseases, infringed upon the economy. The disease is highly rife especially in third world countries 9. Because of the easy availability and physical resemblance of this parasite (Paramphistomum cervi) with the parasite of human beings, the initial evaluation of anthelmintic compounds in vitro was performed.
Plants possess many antimicrobial properties as secondary metabolites for instances, alkaloids, phenolics compounds, etc. 10 The increasing case of drug-resistant of bacteria and reduced susceptibility of some strains to antibiotics raised the bacterial infections incurable and exhorted to the search for new antibacterial compounds in different plants 11-12.
A survey of literature revealed that no research on Anti-bacterial, Anti-oxidant and Anthelmintic activity of C. pallida (Aiton) leaves extract was performed. For this reason, it was thought worthwhile to investigate the Anti-bacterial, Anti-oxidant and Anthelmintic activity of leaves of this plant.
MATERIALS AND METHODS:
Plant Material Collection: The leaves of C. pallida was collected from the Boyra, Khulna, Bangladesh in 23rd August 2012 at evening and identified by the experts of Bangladesh National Herbarium (Accession number 45236), Mirpur, Dhaka, Bangladesh. A voucher specimen has been deposited in Pharmacy Discipline, Khulna University, Khulna, Bangladesh.
Extraction: The collected leaves were separated from undesirable materials and then were washed with water. They were shade-dried for five weeks. The leaves were ground into fine powder by a suitable grinder. About 150 g of powdered material was taken in a clean glass container and soaked in 600 ml of 95% ethanol. The container was then sealed and kept for 14 days accompanying occasional stirring by a clean glass rode. The mixture then underwent a filtration process by a piece of clean, white cotton material. Then it was filtered through filter paper. The filtrate (Ethanol extract) obtained was then evaporated.
Phytochemical Screening: A small portion of the extract was taken for phytochemical screening 13-14.
Test Microorganisms: Six species of both gram positive and gram negative bacteria were used for the antibacterial test. The bacterial strains were collected from the microbiology lab of Khulna University, Khulna, Bangladesh. The bacterial strains were used for the investigation are Gram-negative (Escherichia coli, Shigella dysenteriae, Vibrio cholera, Shigella flexneri) and Gram-positive (Staphylococcus aureus, Streptococcus pyrogens).
Antioxidant Assay: The free radical-scavenging activity of C. pallida leaves extract was evaluated by assessing its discoloration of 2, 2-diphenyl-1picryl-hydroxyl radical (DPPH) in ethanol by a slightly modified method 15. The radical scavenging activity was calculated using the following formula:
Inhibition (%) = [{(A-B)/A} ×100]
Where, A = The absorbance of blank sample and B = The absorbance of the extract
Determination of Total Phenolics Content: The total phenolics content of C.pallida leaves was determined by Folin-ciocalteu assay. Briefly, a standard solution of Gallic acid (20, 40, 60, 80 µg/ml), and 0.0025 gm of the extract was dissolved in 25 ml of 80% ethanol and sonicated for 20 minutes. Then each concentration of both standard and extract was added 9 ml of distilled deionized water in each test tube. Then 1 ml of Folin-Ciocalteu was added and shaken properly. After 5 min later 10 ml of 7% of Na2CO3 was added. The volume was adjusted to 25ml by adding distilled water and kept in the dark for 30 min. After 30 min the UV absorbance was measured at 750 nm 16.
Determination of Total Flavonoids Content: The total flavonoids content was measured with an aluminum chloride colorimetry assay. Briefly 300 µg/ml, 250 µg/ml, 200 µg/ml, 150 µg/ml, 100 µg/ml, 50 µg/ml concentration of solution of standard [Quercetin] was prepared in six test tubes. 1 ml of a solution of each test tube was taken in another six separate test tubes. 300 µg/ml concentration of the sample was prepared in another test tube. Then 4ml of distilled water was added to each test tube, and 0.3 ml of 5% NaNO2 was added to each test tube. After 5 minutes later 0.3ml of 10% AlCl3 was added to each test tube. 2 ml of 1M NaOH was added to each test tube. After 30 min the absorbance was taken at 510 nm wavelength 16.
Anthelmintic Activity Test: The Anthelmintic activity was done on adult worm’ Paramphistoma cervi’ (trematoda) due to its Physiological resemblance with the intestine parasites of human beings. Seven groups of approximately equal size of six worms in each group were used for the present study. Three groups were prepared as control (i.e. 0.2% tween-80 in water), reference i.e standard albendazole (10, 15 mg/ml) and extracts (25, 50, 100, 200 mg/ml). This test was carried out with the slight modification of the original one 17. Observations were made for the time required for paralysis was noted when no movement could be observed. Death was concluded when the worms lost their motility followed with fading away of their body color.
Antibacterial Activity Test: Antibacterial activity was performed by disc diffusion method 18-19. This method, test sample was dissolved in solvents to give the concentration of the solution to 250 µg/disc, and 500 µg/disc and kanamycin (30 µg/disc) was used as a standard. The sterile Matricel (BBL, Cockeysville, USA) filter paper discs were placed with a known amount of test samples using micropipette and dried. The disk of sample, positive control and negative control are then kept in sterile Petri dishes (120 mm in diameter) containing agar medium dipped with the test organisms using sterile transfer loop. The plates are then kept at 40 ºC for facilitating maximum diffusion. The plates were then kept in an incubator for 12-18 h to allow the growth of the microorganisms. If the test material has any antibacterial activity, it will inhibit the growth of microorganism giving a clear, distinct zone called “zone of inhibition.” The antibacterial activity of the test agent is determined by measuring the diameter of the zone of inhibition in term of a millimeter and compared with the standard antibiotic.
RESULTS:
Phytochemical Screening: The phytochemical analysis of C. pallida leaves extract revealed the presence of combined reducing sugar, steroids, tannins, alkaloids, flavonoids, and glycosides but gums and saponins were absent.
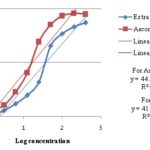
DPPH Scavenging Activity: The leaves extract showed DPPH radical scavenging activity in a concentration-dependent manner. The IC50 value for the standard and extract was measured by the following equation chronologically, y = 44.51x - 4.718 and y = 41.79x - 15.83 respectively. Where y is 50 and x is the log concentration of IC50 value Fig. 1. The extract showed IC50 value of 37.60 µg/ml which is comparable to that value of standard ascorbic acid that showed IC50 value of 16.95 µg/ml. The test was done triplicates.
FIG. 1: DPPH SCAVENGING ASSAY (% INHIBITION vs. LOG CONC.)
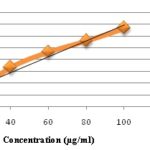
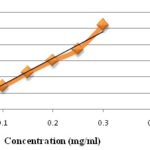
Determination of Total Phenolics Content and Total Flavonoids Content: A standard curve was prepared for the determination of total phenolics content and total flavonoids content using different concentrations of standard Gallic acid and quercetin respectively. The total phenolics content of leaves extracts of C. pallida was expressed in term of Gallic acid equivalent The standard curve equation: y = 0.003x + 0.001, R² = 0.962, Fig. 2 where y is absorbance at 750 nm and x is total phenolics content of the extracts of C. pallida in mg per g 20. And the Total Phenolics content was found to be 14.0 ± 0.015 mg GAE/g of dried plant extract. The total flavonoids content of leaves extracts was also expressed regarding Quercetin equivalent. The standard curve equation: y = 0.328x - 0.002, R² = 0.970, Fig. 3 where y is the absorbance at 510 nm, and X is the flavonoids content in mg per g 21. The Total flavonoids content was found to be 20.42 ± 0.001 mg QE/100 g of dried plant extract.
FIG. 2: LINEAR CURVE OF GALLIC ACID CONCENTRATION (µg/ml) vs. ABSORBANCE FOR DETERMINATION OF TOTAL PHENOLICS CONTENT (Abs = Absorbance)
FIG. 3: LINEAR CURVE OF QUERCETIN CONCENTRATION (mg/ml) vs. ABSORBANCE FOR DETERMINATION OF TOTAL FLAVONOIDS CONTENT (Abs = Absorbance)
Antibacterial Activity: The crude extract of the leaves of C. pallida showed activity against the bacterial strains Vibrio cholera, Shigella flexneri, Shigella dysenteriae, but no activity against, Staphylococcus aureus Streptococcus pyrogens, and Escherichia coli. Standard antibiotic discs of Kanamycin were used for comparison purpose.
From the results Table 1, the Activity Index (AI) Table 2 were calculated using the following formula:
Activity index (AI) = (Inhibition zone of sample / Inhibition zone of standard
TABLE 1: ANTIBACTERIAL ACTIVITY OF LEAVES EXTRACT OF C. PALLIDA
| S. no. | Bacterial strain | Blank | Zone of inhibition | ||
| Kanamycin (30 µg/disc) | Extract (250 µg/disc) | Extract (500 µg/disc) | |||
| 1 | E. coli | 0 | 19.8 | 0 | 0 |
| 2 | S. dysenteriae | 0 | 20.0 | 7.5 | 9.5 |
| 3 | S. pyrogens | 0 | 23.8 | 0 | |
| 4 | S. aureus | 0 | 32.0 | 0 | 0 |
| 5 | S. flexneri | 0 | 20.0 | 10.5 | 14 |
| 6 | V. cholera | 0 | 24.7 | 9 | 10.5 |
TABLE 2: ACTIVITY INDEX OF LEAVES EXTRACT OF C. PALLIDA
| S. no. | Bacterial strain | Extract (250 µg/disc) | Extract (500 µg/disc) |
| 1
2 3 4 5 6 |
E. coli
S. dysenteria S. pyrogens S .aureus S. flexneri V. cholera |
0
0.38 0 0 0.53 0.36 |
0
0.48 0 0 0.7 0.43 |
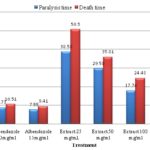
FIG. 4: ANTHELMINTIC ACTIVITY OF LEAVE EXTRACTS OF C. PALLIDA
TABLE 3: ANTHELMINTIC POTENCY OF ETHANOL LEAVES EXTRACT OF C. PALLIDA
| Group | Concentration (mg/ml) | Paralysis time (min) | Death time (min) |
| Control | -- | -- | -- |
| Standard
(Albendazole) |
10 | 8.73 ± 0.37 | 10.51 ± 0.39 |
| 15 | 7.55 ± 0.28 | 9.41 ± 0.34 | |
| 25 | 38.58 ± 0.53 | 50.5 ± 0.28 | |
| Extract
|
50 | 29.58 ± 0.53 | 35.81 ± 0.39 |
| 100 | 17.74 ± 0.46 | 24.48 ± 0.38 | |
| 200 | 10.56 ± 0.32 | 13.85 ± 0.49 |
All Values represent Mean ± SD; n=6 in each group. Comparisons made between standard versus treated groups, P<0.001 was considered significant
DISCUSSION: The presence of the bioactive plant metabolites in phytochemical screening possesses antibacterial activity. For instances, tannins are known to be helpful in treating diarrhea and dysentery.
In DPPH activity, the antioxidants were able to reduce DPPH to yellow colored diphenyl picrylhydrazone 22. The method based on the reduction DPPH in alcoholic solution in the presence of a hydrogen donating antioxidant due to the formation of the nonradical form DPPH-H in the reaction. DPPH is usually used as a reagent to assess free radical and accepts an electron or hydrogen radical to become a stable diamagnetic molecule 23. The extract showed significant antioxidant activity compared to standard.
The antioxidative activities observed can be due to the different polyphenolics compounds that are, tocopherols, flavonoids and other organic acids. Many studies have shown polyphenols contribute significantly to the antioxidant activity 24. They act as highly effective free radical scavengers which are mainly due to their redox properties, which can play an important role in absorbing and neutralizing free radicals, quenching singlet and triplet oxygen or decomposing peroxides 25. The extract showed phenolics and flavonoids content that possess a wide range of activity including antibacterial, antioxidant and anti-allergic.
The antibacterial activity of C. pallida probably due to the presence of flavonoids that revealed in phytochemical studies. The zone of inhibition varies within the ranges of 7.5-10.5 mm and 9.5-14 mm at the dose of 250 µg/disc and 500 µg/disc respectively. The highest zone of inhibition was found against Shigella flexeneri (14 mm) at 500µg/disc. As it showed moderate activity against S. dysenteriae, S. flexneri, V. cholera, the results buttress the traditional use of this plant as a remedy of urinary disorder 3, while the ethanol root extract showed moderate activity against Staphylococcus aureus, Escherichia coli, Salmonella typhi, Pseudomonas aeruginosa 26.
The leaves extract showed significant anthelmintic activity at 200 mg/ml concentration Table 3.
Statistical Analysis: All measurements were repeated three times. The results are expressed as the mean values ± standard deviation. The results were statistically analyzed by ANOVA and Duncan’s multiple range tests. Statistical significance was set at p<0.001.
CONCLUSION: The present study showed that some significant phytochemical components present in the leaves extract of C. pallida that helps to exhibit a potent antioxidant activity and moderate antibacterial and anthelmintic activity which support the traditional uses. Further advanced inquisitions are suggested to elucidate the underlying mechanism as well as to asunder the bioactive compounds responsible for each pharmacological activity. This study has contributed to the validation of the medicinal potential of extracts of leaves of C. pallida.
ACKNOWLEDGEMENT: The authors are grateful to the head of the pharmacy discipline, Prof. Dr. Md. Golam Hoassain for permitting the lab facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Etuk EU and Mohammed BJ: Informant consensus selection method: A reliability assessment of medicinal plants used in north western Nigeria for the treatment of diabetes mellitus. African Journal of Pharmacy and Pharmacology 2009; 3(10): 496-500.
- Okeniyi SO, Adedoyin BJ and Garba S: Phytochemical screening, cytotoxicity, antioxidant and antimicrobial activities of stem and leave extracts of Euphorbia heterophylla. Journal of Biology and Life Science 2013; 4(1): 24-31.
- Deshpande HA and Bhalsing SR: Recent advances in the phytochemistry of some medicinally important Cassia species: A review. International Journal of Pharma Medicine and Biological Science 2013; 3(2): 60-78.
- Govindappa M, Bharath N, Shruthi HB, Sadananda TS and Sharanappa P: Antimicrobial, antioxidant and anti-inflammatory activity and phytochemical screening of Crotalaria pallida African Journal of Pharmacy and Pharmacology 2011; 5 (21): 2359-2371.
- Khan RA: Evaluation of flavonoids and diverse antioxidant activities of sonchus arvensis. Chemistry Central Journal 2012; 6: 126.
- Soni H, Pandey H, Phatak AK, Nayak G, Singhai AK, Parihar A, Singh V and Rathur AS: Evaluation of antioxidant potential of hydroalcoholic extract of leaves of Coleus aromaticus. Adv Pharmacol Toxicol 2009; 10(1): 75-82.
- Barku VYA, Opoku-Boahen Y, Owusu-Ansah E and Mensah EF: Antioxidant activity and the estimation of total phenolics and flavonoids contents of the root extract of Amaranthus spinosus. Asian Journal of Plant Science and Research 2013; 3(1): 69-74.
- Lakshmanan B, Mazumder PM, Sasmal D, Ganguly S and Jena SS: In-vitro anthelmintic activity of some 1-substituted imidazole derivatives. Acta Parasitologica Globalis 2011; 2(1): 01-05.
- Lone BA, Chishti MZ, Ahmad F and Tak H: A survey of gastrointestinal helminth parasites of slaughtered sheep and goats in Ganderbal, Kashmir. Global Veterinaria 2012, 8(4): 338-341.
- Najafi S: Phytochemical screening and antibacterial activity of leaf extract of Ziziphus mauritiana International Research Journal of Applied and Basic Sciences 2013; 4(11): 3274-3276.
- Ogbodo SO, Okeke AC, Ugwuoru CDC and Chukwurah EF: Possible alternatives to reduce antibiotic resistance. Life Sciences and Medicine Research 2011; 1-9.
- Rojas JJ, Ochoa VJ, Ocampo SA and Munoz JF: Screening for antimicrobial activity of ten medicinal plants used in Colombian folkloric medicine: A Possible alternative in the treatment of non-nosocomial infections. BMC Complement Altern. Med 2006; 6: 2.
- Andrew O, Yusuf S, Jangabe LM, Lawal BS and Adamu AA: The α-Glucosidase inhibitory potential of selected antidiabetic plants used in North-Western Nigeria. Journal of Medicinal Plants Research 2013; 7(12): 2010-201
- Nisar M, Ali S and Qaisar M: Preliminary phytochemical screening of flowers, leaves, bark, stem and roots of Rhododendron arboretum. Middle-East Journal of Scientific Research 2011; 10(4): 472-476.
- Brand-williams W, Cuvelier ME and Berset C: Free radical method to evaluate antioxidant activity. Lebensmittel Wissenschaft Technol 1995; 28: 25-30.
- Atanassova M, Georgieva S and Ivancheva K: Total phenolic and total flavonoid contents, antioxidant capacity and biological contaminants in medicinal herbs. Journal of the University of Chemical Technology and Metallurgy 2011; 46(1): 81-88.
- Saha A, Shilpi JA, Mondal H, Gofur R, Billah M, Nahar L and Sarker SD: Bioactivity studies on Musa seminifera Pharmacognosy magazine 2013; 9(36): 315-322.
- Bauer AW, Kirby WMM, Sherris JC and Turck M: Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol 1966; 45: 493-496.
- Ahmed F, Das PK, Islam MA, Rahman KM, Rahman MM and Selim MST: Antibacterial activity of Cordyline terminalis leaves. J Med Sci 2003; 3: 418-422.
- Sahu R and Saxena J: Journal of Pharmacognosy and phytochemistry screening of total phenolics and flavonoids content in conventional and non-conventional species of Curcuma 2013; 2(1): 176-179.
- Bukhari SB, Bhanger MI and Memon S: Antioxidative activity of extracts from fenugreek seeds (Trigonella foenum-graecum). Pak J Anal Environ Chem 2008; 9(2): 78-83.
- Frankel E and Meyer A: The problems of using one-dimensional methods to evaluate multifunctional food and biological antioxidants. J Sci Food Agric 2000; 80: 1925-1941.
- Oyaizu M: Studies on products of browning reaction: Anti-oxidative activity of the product of browning reaction prepared from glucosamine. Jap J Nutrition 1986; 44: 307-315.
- Khan RA, Khan MR, Sahreen S and Ahmed M: Assessment of flavonoids contents and in-vitro antioxidant activity of Launaea procumbens. Chemistry Central Journal 2012; 6: 43.
- Sahu RK, Kar M and Routray R: DPPH free radical scavenging activity of some leafy vegetables used by tribals of Odisha, India. Journal of Medicinal Plants Studies 2013; 1(4): 21-27.
- Kwaji A, Atiko R and Friday E: Phytochemical screening and antibacterial activity of crude ethanol root extract of Crotalaria pallida (Aiton) plant. Top class Journal of Herbal Medicine 2013; 2(10): 235-238, 26.
How to cite this article:
Alam MH, Rahman MM, Sathi SI, Bellah SF, Mazid MA and Murshid GMM: Assessment of antioxidant, antibacterial and anthelmintic activities of ethanol extract of leaves of Crotalaria pallida (Aiton). Int J Pharmacognosy 2014; 1(7): 438-44. doi: 10.13040/IJPSR.0975-8232.1(7).438-44.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
438-444
553
2697
English
IJP
M. H. Alam *, G. M. M. Murshid, B. Bokshi, S. A. Suma, S. I. Sathi and A. Rahman
Pharmacy Discipline, Life Science School, Khulna University, Khulna, Bangladesh.
anik.ku.pharmacy@gmail.com
10 February 2014
18 May 2014
28 June 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(7).438-44
01 July 2014