ANTIOXIDANT EFFECT OF CRUDE WATER EXTRACT OF VETIVERIA ZIZANIOIDES (GRAMINEAE) IN MICE WITH ACETAMINOPHEN INDUCED HEPATOTOXICITY
HTML Full TextANTIOXIDANT EFFECT OF CRUDE WATER EXTRACT OF VETIVERIA ZIZANIOIDES (GRAMINEAE) IN MICE WITH ACETAMINOPHEN INDUCED HEPATOTOXICITY
R. P. Hewawasam * and K. A. P. W. Jayatilaka
Department of Biochemistry, Faculty of Medicine, University of Ruhuna, Ruhuna, Sri Lanka.
ABSTRACT: Acetaminophen (APAP) is widely used as an analgesic and antipyretic drug. The large dose of APAP results in severe hepatic necrosis due to the oxidative stress. This work evaluates the antioxidant activity of crude water extract of Vetiveria zizanioides roots against a single dose of APAP induced acute hepatotoxicity. Mice were treated with 300 mg/kg of APAP dissolved in saline and fasted for 16 h. N-Acetylcysteine (NAC, 500 mg/kg) was used as a positive control. The activities of serum alanine transaminase (ALT), aspartate transaminase (AST) and alkaline phosphatase (ALP) increased significantly 4 h after APAP administration. Oral administration of crude water extract of Vetiveria root at a dose of 0.9 g/kg before or after the oral administration of APAP significantly reduced (P<0.05) the elevated values of serum ALT, AST, ALP and hepatic morphologic changes induced in mice. Antioxidant status, reduced glutathione (GSH), glutathione reductase (GR) and glutathione S-transferase (GST) were reduced significantly (P<0.05) and the lipid peroxidation and the activity of GPx were enhanced significantly in the APAP treated group. Vetiveria root extract markedly inhibited lipid peroxidation, oxidation of glutathione and enhanced the activities of GR and GST in mouse liver homogenate (P<0.05). Although, the effects of NAC and Vetiveria root extracts were comparable in replenishing GSH in APAP toxicity, pre or post-treatment with Vetiveria showed better effects on GR and GST levels in liver homogenates. Hepatoprotective effect of the crude water extract of Vetiveria root against APAP induced acute toxicity may be mediated by preventing the decline of hepatic antioxidant status.
| Keywords: |
Acetaminophen Toxicity, antioxidant enzymes, liver, Vetiveria zizanioides
INTRODUCTION: Acetaminophen [N-acetyl-p-aminophenol (APAP)] is a well-known, long trusted, and an exceedingly available over-the-counter analgesic and antipyretic. Its use has become widespread, because of its efficacy, high toxic-to-therapeutic ratio, and the availability in liquid, tablet and suppository preparations.
Acetaminophen toxicity has replaced viral hepatitis as the most common cause of acute liver failure and the second most common cause of liver failure which requires transplantation in the United States 1. Therefore, it is the most reliable and widely studied animal model of drug-induced liver injury 2. APAP is metabolized by a cytochrome P450 system to N-acetyl-p-benzoquinoneimine (NAPQI).
The overdose of APAP causes the formation of a highly reactive metabolite NAPQI that depletes the intracellular pool of glutathione (GSH). The tripeptide glutathione (GSH) is the major antioxidant and redox regulator in cells that are important in combating oxidation of cellular constituents. Cells spend a great deal of energy to maintain high levels of reduced GSH, which in turn helps to keep proteins in a reduced state.
An alteration of the normal redox balance can alter cell signaling pathways in hepatocytes and may thus be an important mechanism in mediating the pathogenesis of many liver diseases. Much evidence has pointed to the importance of GSH defense mechanism against the hepatic damage induced by free radicals in many pathophysiological situations involving lipid peroxidation reactions 3. Oxidative stress plays an important role in APAP-induced hepatotoxicity.
The protective action of antioxidants points out the possible central role of reactive oxygen species in APAP toxicity 4, 5. Herbal medicines derived from plant extracts are being increasingly utilized to treat a wide variety of clinical diseases 6. More attention has been paid to the protective effects of natural antioxidants against drug-induced toxicities 7. However, the vast majority of information concerning their use is anecdotal. Well-performed animal studies would lend credence to the concept that some medicinal herbs may prevent or, at least ameliorate, hepatic dysfunction arising from drug-induced toxicity.
In the practice of traditional ayurvedic medicine in Sri Lanka, some herbs have been recognized for their potential benefits in the treatment of liver disorders. Vetiveria zizanioides (Gramineae) is one such plant. It is a perennial herb with a branched densely tufted rootstock and long spongy, aromatic, brown fibers. This plant occurs in India, Sri Lanka, Burma, tropical Africa and in the Philippines.
In Sri Lanka, it has been documented in the literature that the root extract acts as a bitter stomachic, carminative and cholagogues and as such is useful in anorexia, chronic dyspepsia, flatulence, acute and chronic congestion of the liver and jaundice 8. Vetiveria zizanioides root is one of the most important raw materials in perfumery, and its root is steam distilled to obtain the Vetiver oil 9, 10. Vetiver oil has been studied for termiticidal 11, antibacterial 12 and in-vitro antioxidant activities 13.
The study carried out by Kim et al., on crude Vetiver oil has demonstrated that three compounds β-vetivenene, β-vetivone, and α-vetivone have strong reducing power through the DPPH radical scavenging assay. Scientific studies on Vetiver roots as a hepatoprotective agent have not been conducted so far.
In Sri Lanka Vetiveria root extract is used by traditional medical practitioners as rice gruel to treat jaundice. Therefore based on the literature, its use by traditional medical practitioners and on the recent evidence on in-vitro antioxidant activity, it was decided to carry out a study to evaluate the antioxidant effect of Vetiveria zizanioides crude water extract on APAP induced acute hepato-toxicity in mice with comparison to N-acetylcysteine (NAC). N-Acetylcysteine is the current clinical treatment for APAP overdose.
MATERIALS AND METHODS:
Chemicals: Diagnostic kits for serum alanine aminotransferase (ALT, EC 2.6.1.2), aspartate aminotransferase (AST, EC2.6.1.1) and alkaline phosphatase (ALP, EC 3.1.3.3) were purchased from Randox (UK). APAP was a gift from the Sri Lanka Pharmaceutical Manufacturing Corporation. 5, 5’- Dithiobis (2-nitrobenzoic acid), thiobarbituric acid, glutathione reductase, cumine hydroperoxide, NADPH and 1-chloro 2, 4 dinitrobenzenes were purchased from Sigma (St. Louis, Missouri). N-Acetylcysteine (NAC) was obtained from the Teaching Hospital, Karapitiya, Galle, Sri Lanka. All other reagents were commercially available and of reagent grade.
Preparation of Plant Extract: The roots of the Vetiveria zizanioides were collected from Hirimbura in the Southern province of Sri Lanka. The sample was identified and authenticated by Prof. M.D. Dassanayaka in comparison with the herbarium specimen preserved at the National Herbarium in the Botanical Gardens, Peradeniya, Sri Lanka. A voucher specimen was deposited at the Department of Biochemistry, University of Ruhuna, Sri Lanka.
Normal therapeutic dose for humans (60 g for a 70 kg man) was extrapolated to be dosed in mice according to the calculation given based on the surface area to body weight ratio in Dhawan and Srimal 14. Each mouse was administered a dose of 0.9 g/kg orally by gavage. The extract given was prepared daily from the dried plant material. Only a single oral dosage of 0.9 g/kg each was administered daily over certain periods to each mouse observed.
Experimental Animals: Healthy male ICR (Institute of Cancer Research) mice 6-8 weeks old and weighing 30-35 g, were used. The animals were allowed free access to water and pelleted food ad libitum. All animals fasted for 16 h before administration of the hepatotoxin. The protocols used in the study were approved by the ethics committee of the University of Ruhuna, Sri Lanka, guided by the CIOMS (Councils for International Organizations of Medical Sciences) international guiding principles of biomedical research involving animals.
Treatment of Animals:
Control Groups: Mice were divided into two groups of 10 animals in each. The first group served as the normal control group and received distilled water orally. The second group was treated with the Vetiveria extract for 7 days. Animals were killed 7days after the administration of plant extract
APAP Treated Groups: Mice were divided into four groups (groups 3-6) of 10 animals each. 300 mg/kg of APAP (dissolved in saline and heated at 60 ºC) was administered orally after a 16 h fast. Group 3 was given APAP alone and killed 4h later. Group 4 received the same dose of APAP, and half an hour later 500 mg/kg of NAC was given orally. The mice were killed 4h later. In group 5, Vetiveria extract was administered instead of NAC. Vetiveria was administered for 7 days in group 6, and on the seventh day, APAP was administered half an hour after the administration of the plant extract. Animals were killed 4 h later.
Assessment of Liver Function: Blood was drawn by cardiac puncture under ether anesthesia to determine serum ALT, AST, and ALP. Serum ALT, AST and ALP activities were measured using assay kits from Randox, UK 15.
Histopathologic Analysis: Liver tissues were excised, weighed and a section of the liver was fixed in 10% phosphate buffered formalin for histopathological assessment. Haematoxylin-eosin staining was performed according to standard histological procedures on 4 µm sections. The remaining liver tissues were used for the measurement of reduced glutathione, and malondialdehyde concentrations and, glutathione reductase (GR) (EC 1.6.4.2), glutathione peroxidase (GPx) (EC 1.11.1.9) and glutathione S-transferase (GST) (EC 2.5.1.18) activities.
Reduced Glutathione: The liver GSH content was estimated by the method of Jollow et al., 16 using DTNB (Ellman reagent, [5, 5’-dithiobis (2-nitrobenzoic acid)) as the substrate. The yellow color developed was read immediately at 412 nm and expressed as µg of reduced glutathione per g of liver tissue.
Estimation of Lipid Peroxidation: The extent of lipid peroxidation in liver homogenates was estimated by the malondialdehyde formation (MDA) using the method of Muriel et al. 17 as described by Okawa et al. 18 The absorbance of the supernatant was measured at 532 nm. The MDA concentration was calculated using the extinction coefficient of 1.56 × 105 M-1cm-1. Results were expressed in nanomoles per milligram of protein.
Preparation of the Cytosolic Fraction: The liver sample was homogenized in three volumes of ice-cold 0.1 M phosphate buffer (pH 7.4) at 2500 rpm for 20 min. The clear supernatant was used for GR, GPx and GST assays 19.
Total Protein Content: The method of Lowry et al., 20 was used.
Antioxidant Enzyme Assays:
Glutathione Reductase Activity (GR, E.C 1.6.4.2): The activity of glutathione reductase was assayed in the cytosolic fraction according to the method of Mohandas et al., 21 as modified by Jodynis et al. 19 Absorbance was measured for 5 min at 1 min intervals at 340 nm, and the activity was expressed as nmol NADPH per min per mg protein using the extinction coefficient of 6.22 × 103 M-1cm-1
Glutathione Peroxidase Activity (GPx, E.C. 1.11.1.9): The activity of glutathione peroxidase in the cytosolic fraction was determined according to the method of Mohandas et al., 21 as modified by Jodynis et al. 19 The reaction was initiated by the addition of cumin hydroperoxide. Absorbance was measured for 5 min at 1 min intervals at 340 nm. The enzyme activity was determined using the extinction coefficient of 6.22 × 103 M-1cm-1.
Glutathione S-transferase Activity (GST, E.C. 2.5.1.18): The method of Habig & Jakoby 22 as described by Athar et al., 23 and Ansar et al., 24 was used in the estimation of glutathione S-transferase activity. The changes in absorbance were recorded for 5min at one-minute intervals at 340 nm, and the enzyme activity was calculated as nmol CDNB conjugate formed per minute per mg protein using molar extinction coefficient of 9.6 × 103 M-1cm-1.
Statistical Analysis: The results were evaluated by one-way analysis of variance and Tukey's multiple comparison tests. A probability (p) value less than 0.05 was considered significant.
RESULTS: The effects of APAP and the extract of Vetiveria zizanioides on the activities of serum enzyme levels of ALT, AST, ALP and liver MDA concentration in mice are summarized in Table 1.
TABLE 1: EFFECTS OF CRUDE WATER EXTRACT OF VETIVERIA ON SERUM ENZYMES AND HEPATIC LEVELS OF MALONDIALDEHYDE
| Group | Enzyme activity | MDA | ||
| ALT(U/L) | AST(U/L) | ALP(U/L) | (nmol/mg protein) | |
| 1 | 5.1 ± 0.7 | 12.4 ± 0.8 | 14.2 ± 2.1 | 0.107 ± 0.02 |
| 2 | 4.6 ± 0.9 | 9.7 ± 1.9 | 10.4 ± 1.9 | 0.103 ± 0.043 |
| 3 | 588.1± 38.1 | 609.4 ± 0.1 | 89.6 ± 9.1 | 0.350 ± 0.036 |
| 4 | 13.0 ± 1.7 | 30.6 ± 5.6 | 47.5 ± 4.9 | 0.125 ± 0.137* |
| 5 | 333.3 ± 22.9* | 335.3 ± 60.0* | 63.0 ± 9.0* | 0.172 ± 0.019* |
| 6 | 273.9 ± 41.9* | 240.3 ± 36.6* | 55.5 ± 6.8* | 0.210 ± 0.014* |
Mice were treated as follows: group 1, untreated: group 2, Vetiveria (0.9 g/kg p.o.) for 7 days: group 3, a single oral dose of acetaminophen (300 mg/kg in saline after a 16 h fast) and killed 4 h later; group 4, a single oral dose of acetaminophen (300 mg/kg in saline after a 16h fast) + a single dose of N-Acetylcysteine (500 mg/kg) and killed 4 h later: group 5, a single oral dose of acetaminophen (300 mg/kg in saline after a 16 h fast) + one dose of plant extract: group 6, 7 doses of plant extract over 7 consecutive days + a single dose of acetaminophen and killed 4 h later.
Results are given as mean ± SEM n=20. *P< 0.05 compared to acetaminophen control group. ALT (Alanine aminotransferase), AST (Aspartate aminotransferase) ALP (Alkaline phosphatase), MDA (Malondialdehyde).
APAP administration resulted in the highest increase in ALT, AST and ALP levels. Pre- or post-treatment with crude water extract of Vetiveria root protected from APAP toxicity, as shown by significantly reduced concentrations of ALT, AST and ALP (P<0.05 vs. control group: Table 1) relative to the APAP treated group. Serum enzyme concentrations of the pre-treated group were lower than the post-treated group.
Histopathological examination provided supportive evidence for the results obtained from the enzyme analysis. Microscopically, liver slices from control animals stained with hematoxylin and eosin showed normal parenchymal architecture with cords of hepatocytes, portal tracts and terminal veins without noticeable alterations.
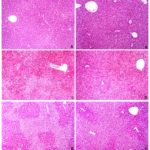
FIG. 1: ASSESSMENT OF ACETAMINOPHEN INDUCED HEPATOTOXICITY BY HISTOPATHOLOGY
Haematoxylin and eosin stained liver sections.
A: untreated;
B: Vetiveria (0.9 g/kg, p.o) for 7 days.
C: acetaminophen control group shows severe confluent necrosis 4 h after the administration of acetaminophen
D: acetaminophen + N acetylcysteine 4 h after the administration of acetaminophen does not show necrosis, but vacuolar degeneration is visible.
E: Post-treatment with Vetiveria zizanioides and sacrificed 4 h later show necrosis, congestion, ballooning degeneration and vacuolation
F: Pre-treatment with Vetiveria zizanioides for 7 days and sacrificed 4 h after the administration of acetaminophen shows no necrosis, but diffused vacuolar degeneration is visible 100 ´ magnification.
Macroscopically, the liver appeared dark and congested in APAP-intoxicated mice. Liver sections of mice challenged with APAP alone showed confluent necrosis with vacuolation and ballooning degeneration in the surviving hepatocytes. An improvement in the histo-pathological changes was observed in animals treated with Vetiveria. Pretreatment showed better results compared to the post-treated group. However, small areas of necrosis were visible in the pretreated group.
The formation of products of lipid peroxidation indicates cellular injury mediated by reactive oxygen intermediates with resultant destruction of membrane lipids and production of lipid peroxides 25.
This study showed that APAP significantly enhanced hepatic lipid peroxidation in the mouse (P<0.05 vs. control group: Table 1), whereas NAC treatment and pre and post-Vetivaria administration, significantly inhibited hepatic lipid peroxidation (P<0.05 vs. APAP only group). The influence of APAP toxicity on the liver GSH concentration is shown in Table 2.
TABLE 2: EFFECTS OF CRUDE WATER EXTRACTS OF VETIVERIA ON HEPATIC LEVELS OF REDUCED GLUTATHIONE AND THE ACTIVITY OF HEPATIC CYTOSOLIC ENZYMES
| Group | GSH | GPx | GR | GST |
| (µg/g liver) | (nmol NADPH/min/mg protein) | (nmol NADPH/min/mg protein) | (nmol CDNB/min/mg protein) | |
| 1 | 2916 ± 22.4 | 23.08 ± 4.91 | 12.89 ± 1.23 | 36.38 ± 3.65 |
| 2 | 3463.0 ± 101.9 | 23.43 ± 3.81 | 10.76 ± 1.34 | 36.37 ± 5.19 |
| 3 | 346.1 ± 64.6 | 90.06 ± 2.91 | 5.38 ± 0.25 | 8.38 ± 0.83 |
| 4 | 3363.3 ± 157.5 | 63.70 ± 3.58* | 7.64 ± 1.27 | 15.38 ± 2.95* |
| 5 | 1880.2 ± 120.1* | 79.78 ± 0.79* | 10.41 ±1.00* | 25.84 ± 3.04* |
| 6 | 2029.8 ± 51.9* | 78.98 ± 0.72* | 11.37 ± 0.71* | 24.17 ± 3.12* |
Mice were treated as follows: group 1, untreated: group 2, Vetiveria (0.9 g/kg p.o.) for 7 days: group 3, a single oral dose of acetaminophen (300 mg/kg in saline after a 16 h fast) and killed 4 h later: group 4, a single oral dose of acetaminophen (300 mg/kg in saline after a 16 h fast) + a single dose of N acetylcysteine (500 mg/kg) and killed 4 h later: group 5, a single oral dose of acetaminophen (300 mg/kg in saline after a 16 h fast) + one dose of plant extract: group 6, 7 doses of plant extract over 7 consecutive days + a single dose of acetaminophen and killed 4 h later. Results are given as mean ± SEM n=20. *P<0.05 compared to acetaminophen control group. Glutathione peroxidase (GPx), Glutathione reductase (GR) and Glutathione S-transferase (GST), Reduced glutathione (GSH).
It resulted in a marked decline in liver GSH concentration 4 h after the administration of APAP compared to the normal controls. Pre and post-treatment with Vetiveria crude extract significantly increased (P< 0.05) the liver GSH concentration. Pretreatment seems to be more effective than post-treatment. Although, the difference was not statistically significant, treatment with Vetiveria extract for seven days produced a higher level of GSH than the normal control group. Vetiveria thus possesses an action similar to NAC in the synthesis of glutathione.
The effects of crude water extracts of Vetiveria zizanioides roots on the activity of hepatic cytosolic enzymes in mice induced with a toxic dose of APAP is shown in Table 2. Within 4 h of APAP treatment, a significant reduction in hepatic GR and GST activities were observed in mice Table 2. Pre- or post-treatment with Vetiveria crude extract markedly increased the activities of these enzymes (P<0.05 vs. APAP control group). The activities of hepatic cytosolic GR and GST in the Vetiveria pre and post-treated groups were almost similar to the activities of these enzymes in the normal control group. The corresponding NAC treated groups had much lower enzyme activities. The activity of hepatic GPx was increased 4 h after the administration of APAP. The pre or post administration of the crude drug extract reduced the GPx activities (P<0.05 vs. APAP control group).
DISCUSSION: The primary objective of the present study was to evaluate the antioxidant effect of the crude water extract of Vetiveria zizanioides against APAP induced hepatotoxicity in mice. APAP toxicity is one of many human disease processes that involve oxidative stress. Antioxidant supplementation has been studied extensively as a method to counter disease associated with oxidative stress. Although, the commonly used antioxidants, which include vitamin C, vitamin K and lipoic acid can directly neutralize free radicals, they cannot replenish the cysteine required for GSH synthesis. The cysteine producing NAC has proven to be more effective in treating diseases associated with oxidative stress. NAC has been used in the clinic to treat a variety of conditions, including drug toxicity, human immunodeficiency virus, cystic fibrosis and chronic obstructive pulmonary disease 26.
However, NAC is not entirely protective of APAP toxicity. Therefore, other agents should be examined for their effectiveness. Beneficial protective effects for APAP hepatotoxicity have been reported for clofibrate 27, ribose cysteine 28, 29, L-cysteine glutathione-mixed disulfide 30 and 2(RS)-n-propylthiazolidine-4(R)-carboxylic acid 31. The success in using various antioxidants to protect against APAP toxicity is not only for its therapeutic application but because it may shed light on the mechanism of hepatotoxicity. We show here, for the first time, the antioxidative effect of crude water extract of Vetiveria zizanioides against APAP-induced acute hepatotoxicity in mice.
The liver is the target organ for APAP toxicity because this is primarily where the drug is detoxified. Ideal attributes of biomarkers of hepatic damage include added information to serum ALP and AST activities. These include organ specificity for liver and strong correlation with well defined hepatic morphologic changes 32. In this study, initially, the hepatoprotective effect of crude water extract of Vetiveria roots was determined by estimating the serum of ALT, AST and ALP activities along with liver histopathology. The antioxidant effect of the extract was determined by estimating GSH concentration, MDA formation, GPx, GR and GST activities in liver homogenates.
Under normal conditions, APAP is mainly metabolized by undergoing sulfation and glucuronidation 33. APAP is known to cause hepatic damage, with marked elevation of the serum enzymes ALT, AST, and ALP because these enzymes are cytoplasmic and are released into the blood after cellular damage. The animals treated with APAP had significant hepatic damage as elicited by the elevated levels of ALT, AST, and ALP. Significant reduction in the activities of serum ALT, AST and ALP enzymes were observed in the pre and post-treated groups.
The histopathological observations sharing a faster regeneration in hepatic cells in mice seem to suggest the possibility of the plant extract being able to condition those hepatic cells to a state of accelerated regeneration, thus decreasing the leakage of ALT, AST, and ALP into circulation. From this preliminary investigation, it was concluded that the plant extract possesses a hepatoprotective effect against APAP induced hepatotoxicity in mice.
APAP toxicity is generally accepted to be primed by the formation of alkylating intermediate N-acetyl-para-benzoquinoneimine (NAPQI), a metabolite formed during cytochrome P450-catalyzed oxidation of APAP 34, 35. Studies conducted recently stressed the importance of CYP2E1 as the principal enzyme responsible for the formation of NAPQI. NAPQI is a potent electrophilic substance, which causes glutathione depletion and binds covalently to a variety of hepatic cell proteins 36. NAPQI reacts with glutathione spontaneously or catalyzed by GST to form a glutathione adduct which is mainly excreted into bile.
Thus the earliest effect of APAP metabolism is a profound depletion of hepatocellular glutathione which affects both cytosolic and mitochondrial compartments 37. In previous studies, it has been shown that the highest decrease in hepatic GSH concentration is shown 4 h after the administration of a toxic dose of APAP 38, 39, 40. This led to the selection of the single point in time, 4 h, in this study. In our study, there was an 88% decrease in the liver glutathione concentration 4 h after the administration of APAP. The loss of antioxidant capacity of an oxidatively stressed cell is mainly due to a decrease in GSH and an increase in GSSG because glutathione is the most abundant intracellular free thiol.
Thus oxidative stress in-vivo mainly translates to the deficiency of GSH 26. GSH levels are most relevant to acute toxicity. If GSH is depleted, it is known that severe toxicity follows 41. Glutathione transferase, therefore, cannot continuously operate at its maximal potential 42. In this study, the liver cytosolic GST activity was estimated and the decline was parallel to GSH. The decrease in antioxidant defense systems of APAP intoxicated mice renders them more susceptible to hepatotoxicity. The levels of antioxidant enzyme activities in liver homogenates GR, GST and GSH were significantly improved upon pre- or post-treatment with Vetiveria crude extract.
This alleviated the APAP induced hepatotoxicity and thereby the oxidative stress as it can decrease lipid peroxidation and enhance the activities of antioxidant enzymes GR and GST Table 1 and 2, whereas the MDA content was significantly decreased in the APAP treated group. Pre or post treatments with the crude extract of Vetiveria upregulates the GSH concentration and the activity of GST and GR.
It has been found out that peroxynitrite is a critical mediator of AAP hepatotoxicity 43. Both hydrogen peroxide and peroxynitrite can initiate lipid peroxidation 44 which can lead to oncotic necrosis of liver cells. Overexpression of glutathione peroxidase was observed to protect cells against oxidative damage 45.
In this study, GPx activity was markedly increased by APAP treatment. Therefore, it may be suggested that the irreversible loss of GSH as conjugates of NAPQI is remedied by the overexpression of GPx, establishing, at least partially, the capacity to eliminate peroxynitrite and H2O2 by GPx.
Lipid peroxidation is the result of oxidative stress as increased formation of superoxide would lead to hydrogen peroxide formation and lipid peroxidation 4. In the process of lipid peroxidation, malondialdehyde is formed as a catabolic product. Although lipid peroxidation in APAP toxicity is controversial 46, in our study, both Vetiveria pretreated and post-treated groups showed reduced malondialdehyde formation in the liver cytosolic fraction. Similar results have been reported by previous studies 38, 47, 48.
NAC is the current clinical treatment for APAP overdose 26, and it was used as a positive control in this study. NAC reduces APAP induced hepatotoxicity by increasing GSH levels and maintaining thiol status. NAC reduces APAP induced hepatic damage by producing cysteine as a precursor 26. In agreement with this observation in the present study, the GSH concentration of the NAC treated group is similar to the normal control group. Although, the effect Vetiveria extract on liver GSH in APAP toxicity is not equal to NAC, pre-treatment of the extract has a better effect than post-treatment Table 2. This is supported by the result obtained for the animal group treated only with Vetiveria extract, 0.9g/kg each for seven days (Group 2, Table 2). As shown in Table 2, Vetiveria extract is more effective than NAC in regulating the activities of antioxidant enzymes GR, GST, and GPx.
CONCLUSION: Our study indicates that the crude water extract of Vetiveria zizanioides has a significant protective effect on APAP induced hepatotoxicity in mice and this protection may be mediated by preventing the decline of hepatic antioxidant.
ACKNOWLEDGEMENT: The financial assistance provided by the National Science Foundation, Sri Lanka (Research Grant No: RG/2001/M/10) and the gift of acetaminophen by the Sri Lanka Pharmaceutical manufacturing corporation is gratefully acknowledged. We also thank Mr. G.H.J.M. Priyashantha for the technical assistance provided.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Farrell S: Acetaminophen toxicity. Medscape. www.emedicine.medscape.com/article/820200-clinical. 06-02-2012.
- Kaplowitz N: Liver biology and pathobiology. Hepatology 2007; 43(2): S235-S238.
- Jaeschke H, Gores GJ, Cederbaum AI, Pessayre D and Lemasters JJ: Mechanisms of hepatotoxicity. Toxicological Sciences 2002; 65: 166-176.
- James LP, Mayeux PR and Hinson JA: Acetaminophen-induced hepatotoxicity. Drug Metabolism and Disposition 2003; 31: 1499-1506.
- Dambach DM, Durham SK, Laskin JD and Laskin DL: Distinct roles of NF-kappaB, p50 in the regulation of acetaminophen-induced inflammatory mediator production and hepatotoxicity. Toxicology and Applied Pharmacology 2006; 211(2): 157-65.
- Hesham A and El-Beshbishy H: Hepatoprotective effect of green tea (Camellia sinensis) extract against tamoxifen-induced liver injury in rats. Journal of Biochemistry and Molecular Biology 2005; 38(5): 563-570.
- Frei B and Higdon J: Antioxidant activity of tea polyphenols in-vivo: evidence from animal studies. Journal of Nutrition 2003; 133: 3275-3284.
- Jayaweera DMA: Medicinal plants used in Ceylon. Part III. National Science Council of Sri Lanka, Colombo, 1981.
- Kraft P, Bajgrowicz JA, Denis C and Frater G: Odds and trends: recent development in the chemistry of odorants. Angewandte Chemie 2000; 39: 280-3010.
- Martinez J, Rosa PT, Menut C, Leydet A, Brat P, Pallet D and Meireles MA: Valorization of Brazilian Vetiver (Vetiveria zizanioides L. Nahs ex Samill) oil. Journal of Agricultural and Food Chemistry 2001; 52: 6578-6584.
- Zhu BCR, Henderson G, Chen F, Fei H and Laine RA: Evaluation of Vetiver oil and seven insect-active essential oils against Formosan subterranean termite. Journal of Chemical Ecology 2001; 27: 1617-1625.
- Hammer KA, Carson CF and Riley TV: Antimicrobial activity of essential oils and other plant extracts. Journal of Applied Microbiology 1999; 86: 985-990.
- Kim HJ, Chen F, Wang X, Chen HY and Jin Z: Evaluation of antioxidant activity of Vetiver (Vetiveria zizanioides L.) oil and identification of its antioxidant constituents. Journal of Agricultural and Food Chemistry 2005; 53: 7691-7695.
- Dhawan BN and Srimal RC: Acute toxicity and gross effects. In; Laboratory manual for pharmacological evaluation of natural products. United Nations Industrial Development Organization and International Centre for Science and High technology 1998.
- Reitman S and Frankel S: A colorimetric method for the determination of serum levels of glutamic oxaloacetic acid and pyruvic acid transaminases. American Journal of Clinical Pathology 1957; 10: 394-399.
- Jollow DJ, Mitchell JR, Potter WZ, Davis DC, Gillette JR and Brodie BB: Acetaminophen-induced hepatic necrosis, II Role of covalent binding in-vivo. Journal of Pharmacology and Experimental Therapeutics 1973; 187: 195-202.
- Muriel P, Alba N, Perez-Alvarez VM, Shibayama M and Tsutsumi VK: Kupffer cells inhibition prevents hepatic lipd peroxidation and damage induced by carbon tetrachloride. Comparative Biochemistry and Physiology 2001; 130: 219- 226.
- Okawa H, Ohmishi N and Yagi K: Assay for lipid peroxides in animal tissues by the thiobarbituriuc acid reaction. Anals in Biochemistry 1979; 95: 351-358.
- Jodnys-Liebert J, Murias M and Blozyk E: Effect of sesquiterpene lactones on antioxidant enzymes and some drug metabolizing enzymes in the rat liver and kidney. Planta Medica 2000; 66: 199-205.
- Lowry H, Roesenbrough NJ, Farr AL and Randall RJ: Protein measurement with folin phenol reagent. J Biological Chemistry 1951; 193: 263-275.
- Mohandas J, Marshal JJ, Duggin GG, Horvath JS and Tiller DJ: Low activities of glutathione-related enzymes as factors in the urinary bladder cancer. Cancer Research 1984; 44: 5086-5091.
- Habig WH and Jakoby WB: Glutathione S-transferase (rat and human) Methods in Enzymology 1981; 77: 218-231.
- Athar M, Khan WA and Mukthar H: Effect of dietary tannic acid in epidermal, lung and forestomach polycyclic aromatic hydrocarbon metabolism and tumorigenicity in sencar mice. Cancer Research 1989; 49: 5784-5788.
- Ansar S, Iqbal M and Athar M: Nordihydroguairetic acid is a potent inhibitor of ferric nitrilotriacetate-mediated hepatic and renal toxicity and renal tumor promotion in mice. Carcinogenesis 1999; 20(4): 599- 606.
- Janero DR: Malonaldehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radical Biology and Medicine 1990; 9(6): 515-540.
- Atkuri KR, Mantovani JJ, Ehrenberg LA and Ehrenberg LA: N-Acetylcysteine-a safe antidote for cysteine -glutathione deficiency. Current Opinion in Pharmacology 2007; 7: 1-5.
- Chen C, Henning GE, Whiteley HE and Manautou JE: Protection against acetaminophen hepatotoxicity by clofibrate pretreatment: the role of catalase induction. Journal of Biochemical Toxicology 2002; 16: 227-234.
- Lucas AM, Henning G, Dominick PK, Whiteley HE, Roberts JC and Cohen SD: Ribose cysteine protects against acetaminophen-induced hepatic and renal toxicity. Toxicologic Pathology 2000; 28: 697-704.
- Slitt AM, Dominic PK, Roberts JC and Cohen SD: Effect of ribose cysteine pretreatment on hepatic and renal acetaminophen metabolite formation and glutathione depletion. Basic and Clinical Pharmacology and toxicology 2005; 96: 487-494.
- Berkeley LI, Cohen JF, Crankshaw DL, Shirota FN and Nagasawa HT: Hepatoprotection by L-cysteine-glutathione mixed disulfide, a sulfhydryl modified prodrug of glutathione. Journal of Biochemical and Molecular Toxicology 2003; 17: 95-97.
- Srinivasan C, Williams WM, Nagasawa HT and Chen TS: Effects of 2(RS)-n-propylthiazolidine-4(R) - carboxylic acid on extrahepatic sulfhydryl levels in mice treated with acetaminophen. Biochemical Pharmacology 2001; 61: 925-931.
- Ozer J, Ratner M, Shaw M, Bailey W and Shomake S: The current state of serum biomarkers of hepatotoxicity. Toxicology 2008; 245: 194-205.
- Jollow DJ, Thorgeirsson SS, Potter WZ, Hashimoto M and Mitchell DJ: Bromobenzene induced liver necrosis: Protective role of glutathione and evidence for 3, 4 bromobenzene oxides as hepatotoxic metabolite. Pharmacology 1974; 12: 265-275.
- Mitchell JR, Jollow DJ, Potter WZ, Gillette JR and Brodie BB: Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. Journal of Pharmacology and Experimental Therapeutics 1973; 187: 211-217.
- Van de Straat R, Vromans M, Bosman P, de Varies J and Verme-Ulen NP: Cytochrome P450 mediated oxidation of substrates by electron-transfer: the role of oxygen radicals and of 1 and 2- electron oxidation of paracetamol. Chemico-Biological Interactions 1988; 64: 267-280.
- Gerber JG, Macdonald JS, Harbison RD, Villeneuve JP, Wood AJ and Nies AS: Effect of N- acetylcysteine on hepatic covalent binding of paracetamol (acetaminophen). Lancet 1977; 1: 657-658.
- Chen C, Hennig GE and Manautou JE: Hepatobiliary excretion of acetaminophen glutathione conjugate and its derivatives in transport-deficient (TR-) hyperbilirubinemic rats. Drug Metabolism and Disposition 2003; 31: 798-804.
- Hewawasam RP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Protective effect of Asteracantha longifolia extract in mouse liver injury induced by carbon tetrachloride and paracetamol. Journal of Pharmacy and Pharmacology 2003; 55: 1413-1418.
- Meotti FC, Rosa JM, Brocardo PS, Balz D, Waltrick A P, Bagio A, Goulart EC, Dafre AL, Rodrigues AL and Santos AR: Protective effect of crude extract from Wedelia paludos (Asteraceae) on the hepatotoxicity induced by paracetamol in mice. Journal of Pharmacy and Pharmacology 2006; 58: 137-142.
- Terneus MV, Kiningham KK, Carpenter AB, Sulivan SB and Valentovic MA: Comparison of S- adenosyl-L-methionine and N-acetylcysteine protective effects on acetaminophen hepatic toxicity Journal of Pharmacology and Experimental Therapeutics 2007; 320: 99-107.
- Comporti M, Maellaro E, Dell Bello B and Casini AF: Glutathione depletion: its effects on other antioxidant systems and hepatocellular damage. Xenobiotica 1991; 21: 1067-1076.
- Rinaldi R, Eliasson E, Swedmark S and Morgenstern R: Reactive intermediates and the dynamics of glutathione transferases. Drug Metabolism and Disposition 2002; 30(10): 1053-1058.
- Knight TR, Ho YS, Farhood A and Jaeschke H: Peroxynitrite is a critical mediator of acetaminophen hepatotoxicity in mouse livers: Protection by glutathione. Journal of Pharmacology and Experimental Therapeutics 2002; 303: 468-475.
- Radi R, Beckman JS, Bush KM and Freeman BA: Peroxynitrite-induced membrane lipid peroxidation. The cytotoxic potential of superoxide and nitric oxide. Archives of Biochemistry and Biophysics 1991; 288: 481-487.
- Mirault ME, Tremblay A, Beaudoin N and Tremblay M: Overexpression of selano glutathione peroxidase by gene transfer enhances the resistance of T47D human breast cells to clastogenic oxidants. The Journal of Biological Chemistry 1991; 266: 20752-20760.
- Knight TR, Fariss MW, Farhood A and Jaeschke H: Role of lipid peroxidation as a mechanism of liver injury after acetaminophen overdose in mice. Toxicological Sciences 2003; 76(1): 229-36.
- Hewawasam RP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Studies on the Preventive and curative effects of Epaltes divaricata (Family Compositae) extract on carbon tetrachloride-induced hepatotoxicity in mice. Indian Journal of Medical Research 2004; 120: 30-34.
- Hewawasam RP, Jayatilaka KAPW and Pathirana C: Effects of Asparagus falcatus on acetaminophen toxicity in mice: A comparison of antioxidant effect with N-acetyl-cysteine. Journal of Dietary Supplements 2008; 5: 1-19.
How to cite this article:
Hewawasam RP and Jayatilaka KAPW: Antioxidant effect of crude water extract of Vetiveria Zizanioides (Gramineae) in mice with acetaminophen induced hepatotoxicity. Int J Pharmacognosy 2015; 2(1): 11-20. doi: 10.13040/IJPSR.0975-8232.2(1).11-20.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
11-20
1182
2228
English
IJP
R. P. Hewawasam * and K. A. P. W. Jayatilaka
Department of Biochemistry, Faculty of Medicine, University of Ruhuna, Ruhuna, Sri Lanka.
ruwaniph@yahoo.com
19 May 2014
09 December 2014
29 December 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(1).11-20
01 January 2015