ANTIOXIDANT AND HEPATOPROTECTIVE EFFECTS OF METHANOLIC EXTRACT OF ORIGANUM MAJORANA IN CCL4- INDUCED LIVER INJURY IN RATS
HTML Full TextANTIOXIDANT AND HEPATOPROTECTIVE EFFECTS OF METHANOLIC EXTRACT OF ORIGANUM MAJORANA IN CCl4- INDUCED LIVER INJURY IN RATS
K. S. Vishnu Kumar *, M. N. Palaksha, K. Venkatesh, Y. Sandip Kumar and R. R. Nayak
Department of Pharmacology, Sri Sai Aditya Institute of Pharmaceutical Science & Research, Surampalem - 533437, Andhra Pradesh, India.
Objective: The present study was aimed to evaluate the antioxidant and hepatoprotective activity of methanolic extract of Origanum majorana aerial parts against CCl4 induced hepatotoxicity in rats. Materials and Methods: Wistar rats (150-200 g) were divided into five groups each group containing six rats. Group A treated as normal control receives 1% v/v tween-80, p.o for 14 days. Group B received 0.1ml/kg of CCl4 i.p for 10 days which is treated as an experimental control. Group C receives 100 mg/kg of silymarin for 14 days which is treated as standard. Group D and E treated as tests t1 and t2 which receives a methanolic extract of Origanum majorana aerial parts of 200 mg/kg and 400 mg/kg for 14 days respectively. Group C, D, and E were intoxicated with CCl4 (0.1ml/kg i.p) for 10 days 1 h before administration of silymarin and extract. Antioxidant effect of methanolic extract of Origanum majorana was also evaluated by in-vitro antioxidant parameters like hydroxyl and superoxide radicals scavenging activities. The hepatoprotective effect was evaluated by the assessment of biochemical parameters such as SGPT, SGOT, ALP, bilirubin and serum total protein levels and histopathological studies of the liver. Results were analyzed by one-way ANOVA followed by Tukey’s multiple comparison tests. Results: Treatment of animals with methanolic extract of Origanum majorana in doses of 200 mg/kg and 400 mg/kg significantly altered the CCl4 induced changes in the serum and tissue enzyme levels to near normal values. It also improved the liver histopathology profile. And also the extract has hydroxyl and superoxide radical scavenging activities, and their IC50 values were found to be 133.33 µg/ml and 245.09 µg/ml. Conclusion: The results of the study indicate that the methanolic extract of Origanum majorana aerial parts possesses hepatoprotective activity against CCl4 induced hepatotoxicity in rats due to the extract is having antioxidant activity.
| Keywords: |
Carbon tetrachloride, Hepatoprotective, Hepatotoxicity, Origanum majorana, Silymarin
INTRODUCTION: The liver is the second largest organ in the body, and it is the premier chemical factory necessary for survival.
The liver receives a dual blood supply with about 20% of blood coming from the hepatic artery and 80% from the portal circulation. The blood flow to the liver is around 20-25% of the cardiac output. It is the first stop for all nutrients, toxins, and drugs absorbed by the digestive tract.
It also plays a vital role in metabolism and has no. of functions in the body, including glycogen storage, decomposition of red blood cells, plasma protein synthesis and detoxification 1.
Hepatitis is inflammation and necrosis of liver cells. This may be due to chemical and biological contamination of food and water or bad environmental conditions, and malnutrition is the vital factors for the rising liver dysfunction which leads to jaundice 2. Further, the involvement of free radicals such as superoxide anions and hydroxyl radicals and other reactive oxygen species like H2O2 in various diseases has been established.
Toxins, infectious agents, medications and serum inflammatory mediators may result in a diverse range of disease processes, leading to loss of normal histological architecture, reduced cell mass and loss of blood flow. Consequently, functional liver capacity may be lost. The effort has been made to search for hepatoprotective agents. However, no effective therapies are available until now. Therefore, the prevention of liver diseases has a great significance both in theory and in practice 3. Herbal drugs play a major role in the treatment of hepatic disorders in traditional systems of medicine in India.
Origanum majorana is a perennial herb that belongs to the family Lamiaceae, and it is native to southern Europe, North Africa, and Asia Minor. Origanum majorana is used as a home remedy for a chest infection, cough, sore throat, rheumatic pain, nervous disorders, stomach disorders, cardio-vascular diseases, and skin care 4, 5.
Origanum majorana has been reported to exhibit a significant antimicrobial activity 6. Several studies have also shown that ethanolic, aqueous extracts and essential oil of Origanum majorana could protect against liver and kidney damage and genotoxicity induced by lead acetate 7. However little is known about the biologically active compounds of majorana as a medicinal plant. In this study, the aim is to investigate the hepatoprotective activity from the methanolic extract of Origanum majorana aerial parts against CCl4 induced hepatotoxicity in rats.
MATERIALS AND METHODS:
Chemicals: All chemicals and reagents used were of analytical grade. Silymarin tablets micro labs Pvt. Ltd., USA), Carbon tetrachloride, KCl, EDTA, procured from S. D. Fine chemicals. The kits for the estimation of serum glutamic oxaloacetic transaminase (SGOT), serum glutamic pyruvic transaminase (SGPT), alkaline phosphatase (ALP), total bilirubin (TB), direct bilirubin (DB), and total protein were purchased from Coral clinical systems, Verna Goa, India. All solvents and chemicals used were of analytical grade, and all the solutions were freshly prepared.
Plant Collection and Extraction: The aerial parts of Origanum majorana were purchased from local market of Wonder herbals (Kukatpally, Hyderabad) and were identified and authenticated by Dr. K. Madhava Chetty (Assistant Professor, Department of Botany) from Sri Venkateswara University, Tirupathi-517502, where a voucher specimen was deposited.
Aerial parts of Origanum majorana were dried under shade, powdered with a mechanical grinder, and passed through sieve no. 40. The sieved powder was stored in airtight container and kept in room temperature. Dried plant material (500 g) was extracted with 1500 ml of methanol using a Soxhlet extractor (continuous hot percolation) for 72 h at a temperature not exceeding the boiling point of the solvent. The extract was filtered using Whatman filter paper (no.1) and then concentrated in vacuum at 40 °C using a rotary evaporator yielding a waxy material 15.2%. The extract was kept in the dark at 4 °C until tested.
Animals: Healthy Albino rats of wistar strain weighing 150-200 g each were given the standard diet with water ad libitum during the experiment as per the purpose of Control and Supervision of Experiments on Animals (CPCSEA) for laboratory animal facilities 8. The institutional animal ethical committee (Reg. No. 769/2010/CPCSEA) permitted the study.
Acute Toxicity Studies: Acute toxicity studies were performed according to the Organization for Economic Co-operation and Development (OECD) -423 as per annexure 2C guidelines 9. Female Wistar rats were selected by random technique were used. Six rats were divided into 2 groups each containing 3 animals, and they were kept overnight for fasting. The doses given were 300 mg/kg and 2000 mg/kg b.w of methanolic extract of Origanum majorana respectively, and initial body weights were recorded. Individually animals were observed for 4 h for each 30 min to see any clinical symptoms, any change in behavior or mortality. 6 h post dosing again body weights recorded. From the next day onwards the same procedure is repeated up to 14 days, and animal weights were recorded on 8th and 14th day. Oral administration of O. majorana extract of 300 mg/kg and 2000 mg/kg did not show any toxic signs or mortality, and LD50 was found to be more than 2000 mg/kg.
Hepatoprotective Activity: Wistar albino rats weighing 150-200 g were divided into five groups each group containing six animals. The animals in group I served as normal control and received the vehicle (1 ml/kg/day of 1% Tween-80) for 14 days. Group II animals received 0.1ml/kg of CCL4 (E-Merck, Mumbai, India) intraperitoneally (i.p) for 10 days 10. Group III animals received 100 mg/kg of standard drug silymarin (Ranbaxy Lab. Dewas) through oral for 14 days. Group IV and V were treated with a methanolic extract of O. majorana in the doses of 200 mg/kg and 400 mg/kg (as per acute toxicity studies) for 14 days. Group III, IV, and V rats were intoxicated by CCl4 (0.1 ml/kg/day through i.p) for 10days 1hr before administration of the standard drug and extract respectively. On the 14th day, the rats were sacrificed and the individual weights of livers were recorded.
Assessment of Hepatoprotective Activity: All the animals were killed on day 14th day under light ether anesthesia. The blood samples were collected by retro-orbital plexus into sterilized dry centrifuge tubes and allowed to coagulate for 30 min at 30 °C. The clear serum was separated at 2500 rpm for 10 min, and the obtained clear serum is used for estimation of total and direct bilirubin, 11 total protein, 12 serum alanine aminotransaminase (ALT), 13 serum aspartate aminotransferase (AST) 14 and alkaline phosphatase (ALP) 15.
Histopathological Study: The rats were then sacrificed (on the 15th day) under deep ether anesthesia and the liver samples were excised and washed with normal saline. A record of each liver was made, regarding size shape, color, and presence or absence of any nodule. Then, the livers were fixed immediately in 10% formalin solution. A paraffin embedding technique was carried out, and sections were taken at 5-mm thickness, stained with hematoxylin and eosin and examined microscopically for histopathological changes 16.
Antioxidant Activity:
Superoxide Scavenging Activity: Superoxide scavenging activity of the plant extract was determined by McCord and Fridovich method, 1969, 17 which depends on light-induced superoxide generation by riboflavin and the corresponding reduction of nitroblue tetrazolium. 1 ml of different concentrations of plant extract and 1 ml of 6 µM ethylenediaminetetraacetic acid containing NaCl, 0.1 ml of 2 µM riboflavin were transferred to a test tube, and final volume was made up to 3ml using phosphate buffer. Then, the assay tubes were uniformly illuminated with incandescent light (40 Watts) for 15 min, and after that, the optical densities was measured at 560 nm. The percentage inhibition of superoxide production was evaluated by comparing the absorbance values of standard and extract tubes.
Hydroxyl Radical Scavenging Activity: Deoxyribose assay was used to determine the hydroxyl radical scavenging activity in an aqueous medium 18. The reaction mixture containing FeCl3 (104 µM), EDTA (104 µM), H2O2 (1 mM) and 2-deoxy-D-ribose (2.8 mM) were mixed with methanolic extract of Origanum majorana aerial parts at various concentrations (10-250 µg) in 1 ml final reaction volume made with potassium phosphate buffer (20 mM, pH 7.4) and incubated for 1 h at 37 °C. The mixture was heated at 95 °C in a water bath for 15 min followed by the addition of 1 ml each of TCA (2.8 %) and TBA (0.5% TBA in 0.025 M NaOH containing 0.02% BHA).
Finally, the reaction mixture was cooled on ice and centrifuged at 5000 rpm for 15 min. The absorbance of the supernatant was measured at 532 nm. The percentage inhibition of hydroxyl radical was evaluated by comparing the absorbance values of standard and extract tubes.
Statistical Analysis: The results were expressed as mean± SEM of six animals from each group. The data were evaluated by one-way ANOVA followed by Tukey’s multiple comparison tests. A comparison made with the CCL4 control. P-values <0.05 were considered statistically significant.
RESULTS: Hepatoprotective activity: The administration of CCl4 to the animals resulted in a marked increase (P<0.05) in total bilirubin, direct bilirubin, serum amino transaminases (SGOT and SGPT), serum ALP and decrease in serum total protein when compared with Group I (Vehicle control) as shown in Table 1 and 2.
TABLE 1: EFFECT OF METHANOLIC EXTRACT OF O. MAJORANA AERIAL PARTS ON SGPT, SGOT, AND ALP IN CCl4 INDUCED HEPATOTOXIC RATS
| GRPS | Treatment | SGPT (IU/L) | SGOT (IU/L) | ALP (IU/L) |
| A | Normal control | 34.5 ± 2.60 *** | 32 ± 1.93 *** | 127 ± 2.35 ** |
| B | Toxic control | 97.7 ± 5.79 | 120.2 ± 4.94 | 247.3 ± 13.1 |
| C | Standard | 44.5±3.24*** (84%) | 42.7±2.23*** (88%) | 133±2.05** (95%) |
| D | T1 | 68.8± 5.34** (46%) | 86 ± 3.65* (39%) | 188.7±8.51** (49%) |
| E | T2 | 47.7±3.63*** (79%) | 64 ± 3.43** (64%) | 135.7±3.24* (92%) |
TABLE 2: EFFECT OF METHANOLIC EXTRACT OF O. MAJORANA AERIAL PARTS ON TOTAL BILIRUBIN, DIRECT BILIRUBIN, AND TOTAL PROTEIN IN CCl4 INDUCED HEPATOTOXIC RATS
| GR | Treatment | Total Bilirubin (mg/dl) | Direct Bilirubin (mg/dl) | Total Protein (gm/dl) |
| A | Normal control | 0.6 ± 0.04 ** | 0.21 ± 0.01 *** | 7.04 ± 0.09 *** |
| B | Toxic control | 2.18 ± 0.18 | 0.82 ± 0.013 | 3.78 ± 0.08 |
| C | Standard | 0.67±0.034** (96%) | 0.33±0.02*** (80%) | 5.93±0.08*** (66%) |
| D | T1 | 0.97±0.04* (77%) | 0.60±0.01*** (35%) | 4.25± 0.05 * (31%) |
| E | T2 | 0.82±0.044*(86%) | 0.41±0.01*** (67%) | 5.54±0.19** (54%) |
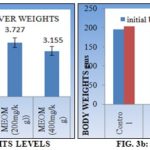
TABLE 3: EFFECT OF METHANOLIC EXTRACT OF O. MAJORANA AERIAL PARTS ON PHYSICAL PARAMETERS LIKE WET LIVER WEIGHT AND BODY WEIGHT IN CCl4 INDUCED HEPATOTOXIC RATS
| GRPS | Treatment | Wet Liver
Weight (gm) |
Body Weight (gm) | |
| Initial | Final | |||
| A | Normal control | 2.73 ± 0.03 *** | 195 ± 4.83 | 204 ± 4.33 ** |
| B | Toxic control | 4.48 ± 0.05 | 197.5± 3.94 | 152.7 ± 2.65 |
| C | Standard | 2.87 ± 0.02 *** (92%) | 203 ± 4.47 | 196.8± 4.67 *** |
| D | T1 | 3.73 ± 0.03 ** (43%) | 200 ± 5.32 | 174.7 ± 7.49 ** |
| E | T2 | 3.15 ± 0.021 *** (76%) | 200 ± 3.78 | 191 ± 3.64 ** |
Values were expressed in mean ± SEM and data was analyzed by one-way ANOVA followed by TUKEY’S multiple comparison tests. Where * represents highly significant at p<0.05, ** represents highly significant at p<0.01, and *** represents very significant at p<0.001. All values were compared with a toxicant. Values in parenthesis indicate percentage of protection.
The oral administration of methanolic extracts of Origanum majorana and silymarin reduced the CCl4 induced increase in the SGOT, SGPT, ALP, direct bilirubin and total bilirubin levels (P<0.05) Fig. 1a, 1b, 1c, 2a, 2b.
The extracts also reversed the depletion of total protein significantly (P<0.05) when compared to CCl4 treated group Fig. 2c and also the administration CCl4 to the animal’s results in increase in wet liver weight and decrease in body weight Table 3 was observed in group II animals but it is seen in reverse condition in extracts and silymarin-treated groups Fig. 3a, 3b.
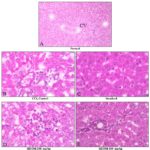
In histopathological studies Fig. 6A, 6B, 6C, 6D, 6E, the liver sections of rats treated with vehicle showed the normal hepatic architecture, Whereas that of CCl4 –treated group showed a total loss of hepatic architecture with intense peripheral central vein necrosis, fatty changes, congestion of sinusoids and apoptosis.
In case of rats treated with silymarin and Origanum majorana methanolic extract 200 mg/kg and 400 mg/kg, showed a normal hepatic architecture was seen with an only moderate accumulation of fatty lobules and mild necrosis, clearly indicating the protection offered by standard drug silymarin and the methanolic extract of O. majorana were given in the figure.
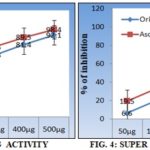
Antioxidant Activity: The in-vitro antioxidant assays showed that the IC50 values were 133.33µg/ml Table 4 and 245.09µg/ml Table 5 for hcavenging activities respectively. Moreover, the results are also comparable with the positive control of the ascorbic acid Fig. 4, 5.
FIG. 1-3: EFFECT OF METHANOLIC EXTRACT OF ORIGANUM MAJORANA ON BIOCHEMICAL AND PHYSICAL PARAMETERS IN CCl4-INDUCED HEPATOTOXIC IN RATS
TABLE 4: EFFECT OF METHANOLIC EXTRACT OF O. MAJORANA AERIAL PARTS ON HYDROXYL RADICAL SCAVENGING ACTIVITY
| Treatment | 50µg | 100µg | 200µg | 300µg | 400µg | 500µg | IC50 |
| O. majoranaa extract | 19.5±0.12 | 37.5±2.60 | 58.2±2.8 | 69.3±1.7 | 81.4±2.1 | 92.1±2.4 | 133.33 |
| Ascorbic acid | 30.3±1.3 | 48.5±0.8 | 66.5±1.4 | 78.5±0.3 | 89.5±0.9 | 98.4±2.8 | 103.09 |
TABLE 5: EFFECT OF METHANOLIC EXTRACT OF O. MAJORANAA AERIAL PARTS ON SUPER OXIDE RADICAL SCAVENGING ACTIVITY
| Treatment | 50µg | 100µg | 200µg | 300µg | 400µg | 500µg | IC50 |
| O. majoranaa extract | 6.6±2.16 | 20.4±3.12 | 31.6±1.2 | 53.5±2.1 | 72.3±3.4 | 85.6±2.12 | 245.09 |
| Ascorbic acid | 19.5±1.2 | 33.6±0.75 | 52.3±1.01 | 66.3±1.6 | 87.4±2.5 | 94.5±1.8 | 148.8 |
FIG. 4 & 5: EFFECTS OF METHANOLIC EXTRACT OF ORIGANUM MAJORANA ON IN-VITRO ANTIOXIDANT PARAMETERS
DISCUSSION:
Hepatoprotective Activity: CCl4 induced hepatic injury is a commonly used model for studying the hepatoprotective effects of drugs or medicinal plant extracts, and the extent of hepatic damage is assessed by the level of released total bilirubin, cytoplasmic alkaline phosphatase & transaminases in circulation. Further, the extent of hepatic damage is assessed by histopathological evaluation 19. The results of the present study undertaken to evaluate the hepatoprotective activity of methanolic extract of Origanum majorana in CCl4 induced liver injury of rats showed that the animals treated with the methanolic extract of Origanum majorana 200 mg/kg and 400 mg/kg b.w significantly reduced the toxic effect of CCl4, similar to the standard silymarin in the levels of liver function serum markers, viz. AST, ALT and ALP, total bilirubin, direct bilirubin and increase in protein synthesis Fig. 1a, 1b, 1c, 2a, 2b, 2c.
The Percentage of protection is greater in the methanolic extract of Origanum majorana 400 mg/kg b.w, which is comparable to the reference drug silymarin (100 mg/kg bw). The results showed that pretreatment with the methanolic extract restored the biochemical parameters, thereby indicating their protection against the injurious effects of CCl4, which may be due to the inhibitory effects on cytochrome P450 resulting in the inhibition of formation of hepatotoxic free radicals 20, 21. Further, histopathological examination of the liver section of the rats treated with toxicant showed intense necrosis and vacuolization.
FIG. 6: HISTOPATHOLOGICAL REPORTS OF RAT-LIVER. Fig. A: Showing normal histological architecture with central vein (CV), While Fig. B: shows the CCL4- induced destruction of architecture in hepatic cells showing fat vacuole and ballooning degeneration and Figure D and E: shows the recovery of CCl4- induced damaged by methanolic extract of Origanum majorana (MEOM) of different doses of 200 and 400 mg/kg respectively showing normal arrangement of hepatocytes, necrosis, mild inflammation, and moderate accumulation fatty vacuoles. And Fig. C: shows the recovery of CCL4- induced damaged by silymarin showing the normal arrangement of hepatocytes absence of necrosis and few fatty vacuoles.
However, the rats treated with silymarin, methanolic extract (at two doses) along with toxicant showed signs of protection against these toxicants to an extent, as evident from the formation of normal hepatic cells and absence of necrosis and vacuoles Fig. 6. Thus, the histological study supports the hepatoprotective activity of the methanolic extract from the toxic effect of CCl4 induced liver damage, which was comparable to silymarin.
Antioxidant Activity: The main characteristic of an antioxidant is its ability to trap free radicals. Highly reactive free radicals and oxygen species are present in biological systems from a wide variety of sources. These free radicals may oxidize nucleic acids, proteins, lipids or DNA and can lead to degenerative disease. Antioxidant compounds like phenolic acids, polyphenols, terpenoids, and flavonoids scavenge free radicals such as peroxide, hydroperoxide or lipid peroxyl and thus inhibit the oxidative mechanisms that lead to degenerative diseases 22.
In the present investigation, different antioxidant assays have been used to evaluate the antioxidant activity of the methanolic extract of Origanum majorana. Hydroxyl (OH.-) and superoxide (O2-) radicals are the most reactive free radical known and can react with everything in living organisms. In the present study, the methanolic extract of Origanum majorana showed significant hydroxyl radical and superoxide radical scavenging activity by their ability to remove hydroxyl and superoxide free radicals due to inhibition of respective mechanisms involved in the formation of radicals Fig. 4, 5. The experimental controls are compared with the standard ascorbic acid, and the IC50 values were found to be 133.33 µg/ml and 245.09 µg/ml of hydroxyl and superoxide radicals scavenging activities.
Thus, the methanolic extract of O. majorana possess free radical scavenging activity under in vitro conditions and could protect the liver tissue against CCl4 induced oxidative stress probably by increasing antioxidant defense activities.
ACKNOWLEDGEMENT: The authors are thankful to Dr. C. Ramesh, Director of SIGMA Institute of Clinical Research and Administration Pvt. Ltd., for providing necessary facilities for this research work. Authors are also thankful to Dr. K. Madhava Chetty, Department of Botany, Sri Venkateswara University, Tirupati, for helping us in authentication of the drug.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Absar AQ, Prakash T, Patil T, Viswanath Swamy AH, Veeran GV and Prabhu K: Hepatoprotective and antioxidant activities of flowers of Calotropis procera (Ait) R.Br in CCl4 induced hepatic damage. Indian J Exp Biol 2007; 45: 304-10.
- Sinha RK and Sinha S: Jaipur: Surabhi Publishers; 201. Ethnobiology; Role of Indigenous and Ethnic Societies in Biodiversity conservation, Human Health Protection and Sustainable Development, 181.
- Bataller R and Brenner DA: Liver fibrosis. J Clin Invest 2005; 115: 209-18.
- Skaria P: Aromatic Plants: Horticulture Science Series by Baby. 01: 216.
- Vagi E, Rapavi E, Hadolin M, Vasarhelyine Peredi K and Balazs A: Phenolic and triterpenoid antioxidants from Origanum majorana herb and extracts obtained with different solvents. Journal of Agricultural and Food Chemistry 2005; 53: 17 21.
- Al-Harbi NO: Effect of marjoram extracts treatment on the cytological and biochemical changes induced by cyclophosphamide in mice. Journal of Medicinal Plants Research 2011; 5: 5479 5485.
- Leeja L and Thoppil JE: Antimicrobial activity of methanol extract of Origanum majorana (Sweet marjoram). J Environ Biol 2007; 28: 145-146.
- El-Ashmawy IM, Amal S and Salama OM: Effects of marjoram volatile oil and grape seed extract on ethanol toxicity in male rats. Basic Clin Pharmacol Toxicol 2007; 101: 320-327.
- CPCSEA guidelines for laboratory animal facility. Committee for control and supervision of experiments on animals. Indian J Pharmacol 2003; 35: 257-74.
- Ecobichon DJ: New York: CRC Press. The Basis of Toxicology Testing 1997; 43-86.
- Jaiprakash B, Aland R, Karadi RV, Savadi RV and Hukkeri VI: Hepatoprotective activity of fruit pulp of Balanites aegyptiaca. Indian Drugs 2003; 40: 296-7.
- Malloy HT and Evelyn KA: The determination of bilirubin with the photoelectric colorimeter. J Biol Chem 1936; 119: 481-90.
- Kingsley GR: The determination of serum total protein, albumin and globulin by biuret reaction. J Biol Chem 1939; 214: 197-200.
- Reitman S and Frankel S: A colorimetric method for the determination of serum glutamic oxaloacetic acid, glutamic pyruvic transaminase. Am J Clin Pathol 1957; 28: 56 63.
- Bessey OA, Lowery OH and Brock MJ: A method for the rapid determination of alkaline phosphatase with five cubic millimeters of serum. J Biol Chem 1964; 164: 321-9.
- Preece A: Manual for histologic technicians. Boston: Little, Brown and Company, Edition 3rd, 1972.
- Ravi Kumar J and Ganga Rao B: Evaluation of in-vitro Antioxidant activity of Spathodea campanulata International Journal of Biological and Pharmaceutical Research 2013; 4(5): 328-332.
- Halliwell B, Grootveld M and Gutteridge JMC: Methods of Biochemical Sciences 1981; 33: 59-90.
- Manjunatha BK, Mankani KL, Vidya SM, Krishna V and Manohara YN: Hepatoprotective activity of Butea superba against carbon tetrachloride-induced hepatic damage in rodents. Pharmacog Mag 2008; 4: S41-5.
- Jain A, Manih S, Lokesh D, Jain A, Rout SP and Gupta VB: Antioxidant and hepatoprotective activity of ethanolic and aqueous extracts of Momordica dioica leaves. J Ethnopharmacol 2008; 115: 61-6.
- Surendra V, Prakash T, Uday RS, Divakar G, Snehal DF and Kotresha D: Hepatoprotective activity of aerial parts of Cynodon dactylon against CCl4 induced in rats. Pharmacog Mag 2008; 4: S195-201.
- Brand-Williams W, Cuvelier ME and Berset C: Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 1995; 28: 25-30.
How to cite this article:
Kumar KSV, Palaksha MN, Venkatesh K, Kumar YS and Nayak RR: Antioxidant and hepatoprotective effects of methanolic extract of Origanum majorana in CCl4-induced liver injury in rats. Int J Pharmacognosy 2014; 1(2): 144-52. doi: link: http://dx.doi.org/10.13040/ IJPSR.0975-8232.IJP.1(2).144-52.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
9
144-152
759
2163
English
IJP
K. S. V. Kumar*, M. N. Palaksha, K. Venkatesh, Y. S. Kumar and R. R. Nayak
Department of Pharmacology, Sri Sai Aditya Institute of Pharmaceutical Science & Research, Surampalem, Andhra Pradesh, India.
vishnukora@gmail.com
14 November 2013
13 January 2014
26 January 2014
http://dx.doi.org/10.13040/IJP.0975-8232.1 (2).144-52
01 February 2014