ANTIOXIDANT AND ANTIMICROBIAL ACTIVITIES OF ROOT LEAVES AND FLOWERS OF CICHORIUM INTYBUS
HTML Full TextANTIOXIDANT AND ANTIMICROBIAL ACTIVITIES OF ROOT LEAVES AND FLOWERS OF CICHORIUM INTYBUS
Ammara Ijaz * 1, Shreez Ahmad 1, Hina Gul 1, Nyla Jabeen 2 and M. Gulfraz 3
Department of Biochemistry 1, PMAS Arid Agriculture University, Rawalpindi, Pakistan.
Department of Biotechnology 2 and Bioinformatics Islamic International University, Islamabad, Pakistan.
Department of Chemistry 3, Comsat Institute of Information Technology, Abbottabad, Pakistan.
ABSTRACT: A study was conducted to assess root, leaves and flowers extracts of Cichorium intybus for their antimicrobial activities (against four bacterial and fungal strains), antioxidants activities (DPPH, ABTS H2O2 and reducing power assays) and cytotoxicities assays. The results indicate remarkable inhibition of plant extracts for the growth of all bacterial and fungal strains as well as appropriate MIC values were observed as compared to standard antibiotics used. The activities of these extracts against four assays like DPPH, H2O2, ABTS, and reducing powder was promising. Whereas the cytotoxic activity of extracts study against brine shrimp was reliable. The phytochemical analysis of the various parts of Cichorium intybus revealed that activities of these extracts might be due to the presence of various secondary metabolites like phenolics and flavonoids those were analyzed with higher quantities from these extracts. Therefore such a study could be useful for the development of new pharmaceuticals that could lead such compounds for the preparation of new medicines required for human and animals disorders.
| Keywords: |
Plant extracts, Natural products, Antimicrobial, Antioxidant activities
INTRODUCTION: The World Health Organization estimated that about three-quarter population relies on plant-based preparations which have been used in their traditional medicinal system and as the basic needs for human primary health care 42. About 25% of the pharmacological drugs were isolated from plants in the developed countries 1, those are being used against many human infections.
A large number of medicinal plants have been investigated to get active secondary metabolites, and recent interest in these substances has been stimulated by potential health benefits arising from the antioxidant activities of these compounds 12.
Secondary metabolites are the chemical components produced by the plants which are classified in different groups due to their divergence in structure, composition, and solubility. These metabolites are remarkably significant and highly considered for their medicinal importance 2. Although flavonoids possess many biochemical properties the best-described property of flavonoids is their capability to act as antioxidants. Natural antioxidants either in the form of raw extracts or their chemical constituents are very effective to prevent the destructive processes caused by oxidative stress 31, 46. Natural antioxidants have a wide range of biochemical activities, including inhibition of ROS generation, direct or indirect scavenging of free radicals, and alteration of intracellular redox potential 41. Antioxidants protect living organisms from the damage caused by uncontrolled production of reactive oxygen species, concomitant lipid peroxidation, protein damage, and DNA strand breakage. An antioxidant, which can quench reactive free radicals, can prevent the oxidation of other molecules and may, therefore, have health-promoting effects in the prevention of degenerative diseases.
The frequency of life-threatening infections has increased worldwide and is becoming an important cause of morbidity and mortality in immune-compromised patients in developed countries. Since few decades, the health benefits of synthetic antibiotics are under threat as most of the commonly used antibiotics become less effective against certain human infections not, only because many of them produce toxic reactions, but also due to the emergence of drug-resistant bacteria. Thus it is essential to investigate newer drugs with lesser resistance. Drugs derived from natural sources play a significant role in the prevention and treatment of human diseases 17, 21. Herbs are widely exploited in traditional medicine, and their curative potentials are well documented in the literature.
Cichorium intybus commonly known as chicory is an important medicinal plant that belongs to the family Asteraceae 42. Plant family Asteraceae consists of about 1000 genera and have more than 25000 species, most of them are used in medicines, edible oils, rubber industry, vegetables and pesticides preparation 2. Significant activities of Chicory roots as Antimicrobial, anti-diabetic, immune enhancement, anti-hepatotoxic, anti-hyperuricemia, and anti-hypertriglyceridemia were reported by Alloush et al. 4
Different studies exposed that water extract of Cichorium intybus L. have provided remarkable effects on low-density lipoprotein (LDL), degradation of the fatty acids of LDL and inhibitory effects on the production of thiobarbituric acid 28. The phytochemical screening reported in the literature indicates the presence of tannins, saponins, flavonoids, terpenoids, cardiac glycosides and anthocyanins in each part of this plant 29.
Therefore, keeping in view the medicines values, Antimicrobial, Antioxidants and Cytotoxic effects of root, leaves and flowers extracts of Cichorium intybus were assessed during present experimental work.
MATERIALS AND METHODS:
Collection of Samples: The root, flowers and leaves samples of Cichorium intybus were collected from areas of Gujrat (Pakistan) in fine plastic bags duly labeled with names and location of collection and were transported to Agriculture lab of Department of Biochemistry PMAS Arid Agriculture Rawalpindi. The samples were identified by expert taxonomist from the Department of Botany and registered as a voucher specimen for future reference.
Preparation of Samples: Samples were washed thoroughly to remove unwanted materials, then shadow and sun-dried followed by oven drying at 60°C for overnight. The dried samples were converted into powdered form and passed through sieves (80 mash). The samples were saved in fine plastic bags and stored at a lower temperature until further uses. Total 50 grams each of root, leaves and flower samples were dissolved in distilled water, methanol, chloroform, and n-hexane and extracted by using soxhlet and rotary evaporator techniques followed by shaking for overnight and then filtrated. The extracts were used for qualitative and quantitative estimation and percentage yield of the extract was calculated and dried extracts were stored in air tight vials for further processes.
Qualitative and Quantitative Estimation of Phytochemicals: Qualitative evaluation of flavonoids, alkaloids, phenols, tannins, and saponins were carried out from various plant extracts by the specific methods described by Harborne (1998)) and AOAC (2003) 5, 20. Whereas for quantitative estimation of plant samples were analyzed for total phenolic, flavonoid and tannins. Total phenolic contents were determined by using the Folin-Ciocalteu reagent method as described by Kim et al. 22
Whereas flavonoids contents were estimated by using the method reported by Hussain et al. 25 Determination of tannins was carried out using Folin-ciocalteu method as reported by Makkar et al. 27
Antioxidant Activity of Plant Extracts: The capacity of the different plant extracts to reduce Fe+3 ions into Fe+2 ions was assessed 8. The scavenging ability of the plant extracts was determined by using 1, 1 diphenyl 1-2 -picrylhydrazyl (DPPH) assay, following method reported by Moon and Shibamoto (2009). Free radical scavenging assays were performed by using 2, 2′-azino-bis-3-ethylbenzthiazoline-6-sulphonic acid (ABTS) 7. Whereas the capacity of plant extracts to scavenge H2O2 was determined by using the procedure described Ruch et al. 35.
Estimation of Antimicrobial Activity of Plant Extracts: The plant extracts were screened to determined antibacterial potential by using agar well diffusion assay against four bacterial strain, Staphylococcus aureus (ATCC 6538), Escherichia coli (ATCC15224), Klebsiella pneumonia (MTCC618) and Bacillus subtilis (ATCC6633) and for comparison standard antibiotics (Cefixime and Roxithromycin) were used in this study 10 and OD was determined at 420 nm with UV/visible spectrophotometer. The MIC was estimated as the lowest concentration of the extracts that blocked the bacterial growth after 24 h of incubation period 15. The antifungal activity of plant extracts was estimated by using agar tube dilution method against four strains of fungus (Aspergillus niger, 0198; Aspergillus flavus, 0064; Aspergillus fumigates, 66 and Fusarium solani, 0291) as reported by Ettebong and Nwafor 15.
Cytotoxic Activity of Plant Extracts:
Brine Shrimps Cytotoxicity Assay: Brine shrimps cytotoxicity assay was carried out to evaluate the cytotoxic effects of plant extracts by using the method reported by 35.
Analysis of the Plant Extracts with HPLC: About 1% of plant extract was prepared in HPLC grade methanol. The samples were sonicated for 10-12 min followed by filtration and adjustment of 200 ml of volume with HPLC grade methanol. An aliquot of 5 ml was filtered through a C18 column and was eluted with 4 ml of methanol. The volume was further increased to 10 ml followed by filtration, and 20 µl of which was injected into HPLC column by adjusting isocratic flow rate of 1 ml/min mobile phase acetonitrile and 0.1% phosphoric acid (36:64) respectively, retention time 7 and absorbance was measured at 200 nm wavelength by using UV/visible detector.
Statistical Analysis: After triplicate analysis of all parameters data was statistically analyzed by using ANOVA as reported by Steel and Torrie (1980) 36.
RESULTS AND DISCUSSIONS: The root, leaves and flower samples of C. Intybus were analyzed for their proximate composition and results are represented in Table 1. Proximate analysis of plants samples gives valuable information regarding the quality of the plant extracts.
TABLE 1: BIOCHEMICAL COMPOSITION (%) OF DIFFERENT PARTS OF C. INTYBUS AFTER ANALYSIS OF 100 g DRY WEIGHT OF SAMPLE
| Sample | Dry matter | Moisture | Crude protein | Crude fat | Crude fiber | Total ash |
| Root | 98.01±0.1 | 42±0.1 | 5.94±0.01 | 17±0.1 | 28.28±0.1 | 7.99±0.1 |
| Leaves | 94.11±0.01 | 17±0.1 | 14.16±0.01 | 19±0.1 | 17.59±0.5 | 19.2±0.5 |
| Flower | 92.1±0.1 | 67±0.1 | 15.6±0.01 | 27±0.1 | 20.2±0.1 | 12.5±0.1 |
Results are mean of 3 values ± Standard deviation
Qualitative analysis of C. intybus extracts indicates the presence of various secondary metabolites like Phenols, flavonoids, alkaloids, tannins, and saponins Table 2.
TABLE 2: PHYTOCHEMICAL COMPOSITION OF DIFFERENT EXTRACTS OF C. INTYBUS
| Extracts | Phenols | Flavonoids | Tannins | Alkaloids | Saponins |
| Root | |||||
| Methanol | + | + | + | + | + |
| Ethanol | + | + | + | + | - |
| n-hexane | + | + | + | + | + |
| Chloroform | + | + | + | + | + |
| Aqueous | + | + | + | + | + |
| Leaves | |||||
| Methanol | + | + | + | - | + |
| Ethanol | ++ | ++ | ++ | + | + |
| n-hexane | + | + | + | - | + |
| Chloroform | + | + | + | - | + |
| Aqueous | + | + | + | - | + |
| Flower | |||||
| Methanol | + | + | + | - | - |
| Ethanol | ++ | ++ | + | + | + |
| n-hexane | + | + | + | - | - |
| Chloroform | + | + | + | - | - |
| Aqueous | + | + | + | - | - |
+ Present; - Absent
The presence of such metabolites indicates the importance of plant extracts; for examples, flavonoids are considered important due to their antioxidant and antimicrobial activities 33. The phenols act as immune enhancer’ anti-inflammatory, anti-clotting, and hormone modulators whereas saponins have anti-hemolytic and cholesterol binding properties. Tannins are soluble polyphenols and have many functions in the living organism, probably due to these reasons, many researchers suggested multiple utilization of C. intybus 19, 40, 44.
Quantification of Flavonoid from Plant Extracts: a Higher amount of flavonoids was found in ethanolic flower extracts of C. intybus extracts (3.162 ± 0.51 mg/g ) followed by ethanolic leaves extracts of C. intybus (2.862 ± 0.15 mg/g) Table 2. Flavonoids have been shown to possess several biological properties many of which may be related, to their antioxidant and free-radical-scavenging ability 14, 32.
The amount of flavonoids found in the present study was lower than the amount reported by other 44 when they analyzed leaves samples of C. intybus by using a different method of analysis, however, amount of flavonoids found in the present study were comparable with results reported by other authors.
TABLE 3: QUANTIFICATION OF IMPORTANT PHYTO CHEMICALS FROM VARIOUS EXTRACTS OF C. INTYBUS
| Extracts
|
Total phenols
(mg GAE/ g dry weight) |
Total flavonoids
(QE mg/g dry weight ) |
Tannins
(mg GAE/g dry weight) |
Yield (%) |
| Root | ||||
| Methanol | 1.139±0.02 | 1.1616±0.06 | 0.187±0.04 | 2.31 |
| Ethanol | 2.681±0.23 | 2.712±0.04 | 1.812±0.05 | 2.09 |
| n-hexane | 0.242±0.07 | 0.816±0.13 | 0.893±0.14 | 1.34 |
| Chloroform | 2.312±0.34 | 2.231±0.07 | 2.748±0.19 | 2.18 |
| Aqueous | 1.082±0.23 | 1.232±0.05 | 0.436±0.05 | 1.04 |
| Leaves | ||||
| Methanol | 2.176±0.08 | 2.862±0.15 | 1.361±0.08 | 1.12 |
| Ethanol | 2.986±0.05 | 2.912±0.06 | 1.481±0.04 | 2.15 |
| n-hexane | 0.817±0.15 | 1.765±0.04 | 1.124±0.05 | 0.58 |
| Chloroform | 2.241±0.08 | 2.714±0.05 | 2.263±0.06 | 1.65 |
| Aqueous | 1.202±0.02 | 1.816±0.03 | 1.932±0.04 | 1.78 |
| Flower | ||||
| Methanol | 1.864±0.07 | 2.781±0.62 | 0.286±0.04 | 1.98 |
| Ethanol | 2.376±0.08 | 3.162±0.51 | 1.391±0.07 | 2.81 |
| n-hexane | 0.163±0.11 | 0.764±0.05 | 0.193±0.02 | 0.89 |
| Chloroform | 1.178±0.06 | 1.863±0.03 | 0.545±0.20 | 1.84 |
| Aqueous | 0.432±0.14 | 1.732±O.01 | 0.302±0.02 | 1.23 |
Values are expressed as mean ± Standard deviation (n=3); % age yield of all three parameters from total extract used
Total Phenolic Contents: The phenolic contents of plant extracts were determined, and results were compared with a standard curve of Gallic acid Table 3. The ethanolic leaves extracts showed a higher amount of total phenol (2.986 ± 0.05mg GAE/g) as compared to other solvents extracts while a lower amount of total phenol was found in n-hexane and aqueous extract of C. intybus Table 3. Phenols are the secondary metabolites and are present in plants in wider range and can scavenge free radicals and are considered as anticancer agents. Amount of total phenols found in the current study were higher than reported by Ashafa et al. 7 after worked on some other plant extracts.
Total Tannin Contents (TTC): According to results higher amount of tannins was found in chloroform root extracts (2.748 ± 0.19 mg GAE/g) followed by chloroform leaves extracts (2.263 ± 0.06 mg GAE/g) of C. intybus. Plant tannins are a distinctive group of polyphenolic polymers of relatively high molecular weight. Tannins prevent the body from free radical-induced RBC hemolysis and involved in decreasing rate of nutrients absorption; those are required to synthesise new substances for body function 11.
Antimicrobial Activity of Plant Extracts The methanol extract of C. intybus exhibited active inhibitory potential against S. aureus, E. coli, K. pneumonia and B. subtilis strains as compared to all other extracts and cefixime and DMSO; however, lower zone of inhibition was observed as compared to roxithromycin (standard) Table 3.
All plant extracts showed a remarkable antibacterial potential against both Grams +ve and Gram-ve strains which indicate that extracts of C. intybus are composed of antimicrobial components (flavonoids, etc.,) that can be used to treat infectious disorders caused by the severe resistant pathogenic microorganisms. The current findings support the results reported by other authors with slight variations 16. Antimicrobial resistance (AMR) is a major threat to global public health. Medicinal plants have long been used as remedies for infectious diseases by native cultures around the world 39 and have the potential for providing effective treatments for antibiotic-resistant infections 9, 18.
TABLE 3: ANTIBACTERIAL ACTIVITIES OF VARIOUS EXTRACTS C. INTYBUS; ZONE OF INHIBITION
| Extracts | S. aureus | E. coli | K. pneumonia | B. subtilis | Roxithromycin | Cefixime | DMSO | |
| Root | ||||||||
| Methanol | 21.6±0.6 | 21.4±0.9 | 19.5±0.3 | 22.6±0.4 | 14.6±0.9 | 13.5±0.3 | 0.0 | |
| n-hexane | 17.2 ±0.5 | 17.3±0.8 | 17.6±0.4 | 16.5±0.7 | 12.3±0.9 | 11.4±0.8 | 0.0 | |
| Chloroform | 18.3±0.6 | 19.2±0.3 | 14.2±0.4 | 15.8±0.5 | 13.6±0.8 | 12.7±0.9 | 0.0 | |
| Aqueous | 12.4±0.8 | 15.6±0.6 | 11.3±0.5 | 11.6±0.3 | 10.2±0.7 | 9.4±0.8 | 0.0 | |
| Leaves | ||||||||
| Methanol | 22.5±0.3 | 20.8±0.4 | 23.5±0.7 | 16.8±0.6 | 14.3±0.7 | 15.8±0.9 | 0.0 | |
| n-hexane | 19.6±0.5 | 19.2±0.9 | 19.2±0.6 | 14.5±0.5 | 14.5±0.7 | 15.3±0.8 | 0.0 | |
| Chloroform | 13.4±0.4 | 15.7±0.8 | 21.6±0.5 | 15.7±0.9 | 13.6±0.7 | 14.8±0.5 | 0.0 | |
| Aqueous | 11.5±0.7 | 13.2±0.7 | 13.5±0.4 | 11.2±0.7 | 9.7 ±0.8 | 8.2±0.3 | 0.0 | |
| Flower | ||||||||
| Methanol | 18.3±0.6 | 23.6±0.5 | 18.3±0.6 | 21.7±0.4 | 15.6±0.6 | 17.9±0.8 | 0.0 | |
| n-hexane | 16.5±0.6 | 21.5±0.1 | 17.6±0.4 | 18.2±0.4 | 14.2±0.5 | 16.8±0.6 | 0.0 | |
| Chloroform | 13.5±0.5 | 18.4±0.4 | 15.4±0.5 | 17.6±0.3 | 14.7±0.6 | 15.4±0.5 | 0.0 | |
| Aqueous | 11.7±0.6 | 12.6±0.5 | 13.2±0.6 | 13.2±0.7 | 12.8±0.6 | 11.2±0.9 | 0.0 | |
Results mean ± SD after triplicate analysis (n=3).
TABLE 4: MINIMUM INHIBITORY CONCENTRATION (µg/ml) FOR VARIOUS BACTERIAL STRAINS
| Extracts | S. aureus | E. coli | K. pneumonia | B. subtilis | Roxithromycin | Cefixime | DMSO |
| Root | |||||||
| Methanol | 0.1±0.6 | 2.1±0.7 | 0.8±0.5 | 1.4±0.6 | 1.6±0.8 | 1.5±0.5 | 0.0 |
| n-hexane | 1.7 ±0.1 | 1.6±0.9 | 0.7±0.4 | 1.6±0.7 | 1.2 ±0.3 | 1.1±0.8 | 0.0 |
| Chloroform | 1.8±0.9 | 1.9±0.5 | 1.2.2±0.4 | 1.5±0.5 | 1.3±0.5 | 1.2±0.5 | 0.0 |
| Aqueous | 1.9±0.3 | 1.6±0.7 | 1.7±0.5 | 1.6±0.3 | 1.2±0.6 | 1.4±0.3 | 0.0 |
| Leaves | |||||||
| Methanol | 0.5±0.6 | 0.8±0.1 | 0.3±0.9 | 1.1±0.4 | 0.2±0.7 | 0.8±0.3 | 0.0 |
| n-hexane | 1.6±0.5 | 1.9±0.8 | 1.9±0.5 | 1.5±0.5 | 1.5±0.9 | 1.1±0.8 | 0.0 |
| Chloroform | 1.4±0.4 | 1.5±0.9 | 1.6±0.7 | 1.4±0.6 | 1.6±0.4 | 0.4±0.6 | 0.0 |
| Aqueous | 1.5±0.7 | 1.3±0.7 | 1.8±0.4 | 1.9±0.3 | 1.7 ±0.8 | 2.2±0.3 | 0.0 |
| Flower | |||||||
| Methanol | 0.3±0.7 | 0.6±0.4 | 1.3±0.6 | 1.7±0.4 | 0.6±0.6 | 0.9±0.2 | 0.0 |
| n-hexane | 1.5±0.5 | 1.5±0.1 | 17.6±0.4 | 18.2±0.4 | 1.2±0.5 | 1.3±0.5 | 0.0 |
| Chloroform | 1.3±0.4 | 1.4±0.4 | 15.4±0.5 | 17.6±0.3 | 1.4.±0.5 | 1.2±0.7 | 0.0 |
| Aqueous | 1.7±0.7 | 1.6±0.1 | 13.2±0.6 | 13.2±0.7 | 1.8±0.2 | 1.8±0.3 | 0.0 |
Results are Means ± SD, (n = 3)
Minimum inhibitory concentration indicates the significant antimicrobial potential of C. Intybus extracts and data obtained, through the determination of MIC of various extracts and antibiotics are presented in Table 4. The results revealed variability in the inhibitory concentrations of each extract for given bacteria. The lowest MIC was observed for methanol extracts perhaps due to its purity or solubility of plant materials in this solvent Table 4. Results found in the present study were comparable to results report by other authors including Guessan et al., 30 and Kaur et al. 25
TABLE 5: ANTIFUNGAL ACTIVITIES OF VARIOUS EXTRACTS OF C. INTYBUS; ZONE OF INHIBITION IN mm
| Extracts
|
Aspergillus
niger |
Fusarium salani | Aspergillus
flavous |
Aspergillus
fumigates |
Terbinafine | DMSO |
| Root | ||||||
| Methanol | 25.8±0.5 | 21.8±0.4 | 18.3±0.3 | 21.5±0.4 | 18.±0.3 | 0.0 |
| n-hexane | 18.3 ±0.5 | 17.2±0.5 | 17.8±0.4 | 15.4±0.8 | 15.3±0.5 | 0.0 |
| Chloroform | 16.3±0.4 | 18.2±0.3 | 16.3±0.4 | 15.8±0.5 | 14.6±0.3 | 0.0 |
| Aqueous | 11.4±0.2 | 9.6±0.3 | 11.6±0.2 | 11.6±0.3 | 10.1±0.6 | 0.0 |
| Leaves | ||||||
| Methanol | 21.5±0.1 | 19.8±0.3 | 21.1±0.5 | 19.6±0.3 | 16.3±0.6 | 0.0 |
| n-hexane | 18.6±0.3 | 17.2±0.9 | 17.2±0.9 | 18.5±0.6 | 15.5±0.4 | 0.0 |
| Chloroform | 16.4±0.2 | 16.1±0.9 | 16.6±0.3 | 15.7±0.9 | 14.6±0.7 | 0.0 |
| Aqueous | 10.5±0.6 | 11.2±0.4 | 11.5±0.4 | 9.2±0.5 | 9.7 ±0.3 | 0.0 |
| Flower | ||||||
| Methanol | 28.1±0.6 | 25.6±0.5 | 19.3±0.5 | 22.5±0.3 | 15.6±0.6 | 0.0 |
| n-hexane | 26.5±0.4 | 21.1±0.6 | 17.3±0.6 | 19.2±0.2 | 16.2±0.4 | 0.0 |
| Chloroform | 23.5±0.5 | 18.4±0.1 | 16.4±0.1 | 16.6±0.4 | 15.7±0.3 | 0.0 |
| Aqueous | 11.7±0.3 | 12.6±0.8 | 11.2±0.5 | 10.2±0.7 | 11.8±0.4 | 0.0 |
Results are Means ± SD, (n = 3).
Data in Table 5 represents the antifungal activity of C. intybus extracts, according to results higher fungal activity was represented by methanolic flower extracts of C. Intybus extracts (28.1±0.6 mm) followed by n-hexane extracts (26.5 ±0.4mm) and lowest zone of inhibition by aqueous leaves extracts (11.7±0.5 mm) Table 5.
Extracts prepared in organic solvents showed activity against fungal strains and our results are comparable to results of plant extracts reported by Fawole et al. (2008) 18, Parekh and Chanda, (2005) 33, who found out that water extracts showed no/poor fungus toxicity than organic solvents, which is also indicated by present study as date given in Table 5.
It was assumed that due to less solubility of the active substance in aqueous media, has shown lower antimicrobial activity whereas it was remarkable in organic solvents.
In-vitro Antioxidant Assay: To check the antioxidant potential of C. intybus extracts, five different assays i.e. DPPH, H2O2, ABTS Phosphomolybdate and reducing power assays were performed, and ascorbic acid was used as positive control.
All solvents extracts (methanol, n-hexane, chloroform, and water) showed a remarkable quantity of antioxidant activity, although the values were quite less than the positive control (ascorbic acid) and standard rutin Table 6, which is indicated by correlation of antioxidants potential of phenolics and flavonoids Table 7.
It was reported by Aqil et al., (2006) 6 that majority of the antioxidant activities of plants extracts are due to the flavones, isoflavones, flavonoids, anthocyanin, coumarin lignans, catechins, and isocatechins. Analysis of plant extracts with HPLC revealed the presence of a reliable quantity of quercetin Table 8, Fig. 2 - 3, that was responsible for the majority of biological activities of these extracts as reported by Javanmardi et al., (2003) 23 and Rathee et al., (2009) 35. Furthermore, antioxidant-based drug formulations are used for the prevention and treatment of complex diseases like atherosclerosis, stroke, diabetes, Alzheimer’s disease and cancer 13. Phenolics act as antioxidants in some ways such as reducing agents, hydrogen donors, free radical scavengers, and singlet oxygen quenchers and, therefore, act as cell saviors 15, 19.
TABLE 6: ANTIOXIDANT POTENTIAL OF VARIOUS C. INTYBUS extracted AT CONCENTRATION OF 100 µg/ml AND IC50 VALUES (µg/ml) OF RADICAL SCAVENGING WHEN ABSORBANCE WAS MEASURED AT 700 nm
| Extracts | DPPH | H2O2 | ABTS | Reducing Power assays | Ascorbic acid | Rutin |
| Root | ||||||
| Methanol | 0.214±0.01 | 3.13±0.05 | 1.32±0.08 | 0.19±0.01 | 1.91±0.8 | 3.77±1.8 |
| n-hexane | 0.311±0.01 | 2.45± 0.06 | 2.25±0.07 | 0.13±0.01 | 1.2 ±0.3 | 1.4±0.8 |
| Chloroform | 0.023±0.01 | 1.92±0.71 | 7.65± 0.15 | 0.16±0.01 | 1.8±0.4 | 1.6±0.5 |
| Aqueous | 3.08±0.01 | 8.13±0.2 | 9.13±0.26 | 0.18±0.05 | 1.7±0.5 | 1.8±0.3 |
| Leaves | ||||||
| Methanol | 0.019±0.00 | 0.85±0.01 | 0.38±0.19 | 0.11±0.01 | 0.4±0.8 | 0.9±0.2 |
| n-hexane | 0.025±0.01 | 1.19±0.81 | 1.91±0.52 | 0.19±0.0 | 1.3±0.5 | 1.1±0.3 |
| Chloroform | 0.023±0.01 | 1.25±0.93 | 1.65±0.74 | 0.09±0.00 | 1.5±0.3 | 1.4±0.5 |
| Aqueous | 0.318±0.01 | 1.43±0.75 | 1.86±0.43 | 0.18±0.01 | 1.6 ±0.7 | 2.2±0.3 |
| Flower | ||||||
| Methanol | 0.016±0.0 | 0.66±0.04 | 1.37±0.65 | 0.19±0.01 | 0.8±0.5 | 0.9±0.1 |
| n-hexane | 0.012±0.01 | 1.57±0.15 | 1.76±0.42 | 0.12±0.03 | 1.7±0.3 | 1.6±0.7 |
| Chloroform | 0.010±0.0 | 1.46±0.04 | 1.54±0.56 | 0.18±0.01 | 1.8.±0.2 | 1.2±0.7 |
| Aqueous | 0.028±0.01 | 4.63±0.12 | 1.82±0.63 | 0.19±0.02 | 2.9±0.2 | 3.8±0.6 |
Results are Means ± SD, (n = 3).
TABLE 7: CORRELATION BETWEEN PHYTOCHEMICALS AND ANTIOXIDANT POTENTIALS OF VARIOUS ASSAYS
| Assays | Phenolic | Flavonoids |
| IC50 DPPH radical scavenging potential | 1.35±0.5 | 1.34±0.6 |
| IC50 of reducing power capacity | 1.16 ±0.5 | 1.17±0.2 |
| IC50 of hydrogen peroxide scavenging potential | 1.51±0.8 | 1.58±0.6 |
| IC50 of ABTS radical scavenging potential | 1.43±0.4 | 0.151±0.01 |
Values are Mean ±SD, (n=3).
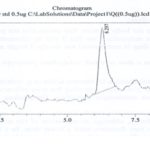
FIG. 1: CHROMATOGRAM OF STANDARD (QUERCETIN)
FIG. 2: CHROMATOGRAM OF FLAVONOID ANALYZED FROM VARIOUS C. INTYBUS EXTRACTS BY USING HPLC
TABLE 8: QUANTIFICATION OF FLAVONOID FROM C. INTYBUS EXTRACTS THROUGH HPLC
| Solvents used | Area | Concentration( mg/g) |
| Methanol | 90200 | 122.627 |
| Ethanol | 89871 | 138.509 |
| n-hexane | 69800 | 100.101 |
Various extractions are showing the presence of flavonoids.
TABLE 9: CYTOTOXICITY SCREENING OF VARIOUS CONCENTRATION (µg/ml) OF C. INTYBUS EXTRACTS
| Extracts | 10 | 100 | 1000 | LD50 |
| Root | ||||
| Methanol | 35.1±0.6 | 42.1±0.6 | 50.8±0.3 | <1000 |
| n-hexane | 34.6 ±0.5 | 41.6±0.5 | 49.7±0.3 | 700 |
| Chloroform | 39.8±0.3 | 46.9±0.3 | 49.2±1.4 | 100 |
| Aqueous | 47.4±0.3 | 51.6±0.7 | 59.7±0.3 | 270 |
| Leaves | ||||
| Methanol | 36.5±0.6 | 41.7±0.1 | 52.3±0.7 | <1000 |
| n-hexane | 31.6±0.5 | 36.9±0.8 | 39.1±0.5 | 665 |
| Chloroform | 33.4±0.5 | 36.5±0.9 | 39.6±0.3 | 100 |
| Aqueous | 51.5±0.7 | 54.3±0.7 | 56.3±0.4 | 260 |
| Flower | ||||
| Methanol | 33.5±0.6 | 35.6±0.5 | 39.3±0.5 | <1000 |
| n-hexane | 31.5±0.5 | 31.9±0.1 | 37.5±0.1 | 668 |
| Chloroform | 39.3±0.4 | 41.5±0.4 | 43.5±0.6 | 1000 |
| Aqueous | 41.7±0.7 | 44.6±0.8 | 43.2±0.5 | 265 |
Values are Mean ±SD, (n=3) and significantly different (P<0.05); positive control are saline sea salt
Brine Shrimps Lethality Assay: Three different dilutions of C. intybus extracts (10,100 and 1000 µg/ml) were made to check brine shrimps cytotoxicity assay. The results revealed the better brine shrimps larvicidal potential and lethality was maximum at the maximum concentration of C. intybus extracts as the lethality of brine shrimps was concentration dependent. It was assumed that plant extracts might be composed of antitumor components in the form of essential phytonutrients. The plant extracts whose value, i.e. LD50 <1000 µg/ml was considered as biologically active while LD50 > 1000 µg/ml was biologically inactive (non -toxic), while higher mortality rate was shown by n-hexane and chloroform extracts of C. intybus Table 9. Results of brine shrimp lethality obtained in the current study were comparable with results reported by Sandeep et al. (2012) 38.
The study revealed that phytochemicals present in C. intybus extracts exhibited wonderful biological properties. The various extracts of C. intybus have shown antibacterial, antifungal, antioxidants, and cytotoxic activities.
It is concluded that the results obtained in the present study are in agreement to a certain degree with the traditional uses of C. intybus extracts against many human ailments. The obtained results could form a good basis for selection of various C. intybus extracts for further investigation in the potential discovery of new natural bioactive compounds.
ACKNOWLEDGEMENT: The financial support provided by the Department of Biochemistry PMAS Arid Agriculture University for this study is highly appreciated.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Verma R., Rawat A, Ganie SA, Rajneesh K, Sharma AR, Mahajan S and Gupta A: In-vitro antibacterial activity of Cichorium intybus against some pathogenic bacteria. British J Pharmaceutical Research 2013; 3(4): 767-775.
- Ahmad MSA and Ashraf M: Essential roles and hazardous effects of nickel in plants. Rev Env Cont Toxicol 2011; 214: 125-167.
- Cook NC and Samman S: Flavonoids- chemistry, metabolism, cardioprotective effects, and dietary sources. Nutritional Biochemistry 1996; 7: 66-76.
- Aletor VA: Allelochemicals in plants and feeding stuffs. Nutritional, biochemicals and physiological aspects in animal-production. Vet Hum Toxicol 1993; 35: 57-67.
- Oluyemi KA, Omotuyi IO, Jimoh OR, Adesanya OA, Saalu CL and Josiah SJ: Erythropoietic and Anti-obesity effects of Garcinia cambogia (Bitter kola) in Wistar Rats. Biotechnol Appl Biochem 2007; 46: 69-72.
- Zengin G, Cakmak YS, Guler GO and Aktumsek A: Antioxidant properties of methanolic extract and fatty acid composition of Centaurea urvillei subsp. hayekiana Wagenitz Rec Nat Prod 2011; 5: 123-132.
- Velioglu YS, Mazza G, Gao L and Oomah BD: Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J Agric Food Chem 1998; 46: 4113-4117.
- Farnsworth NR: Ethnopharmacology and future drug development: The North American experience. J Ethnopharmacol 1993; 38: 145-52.
- Houghton PJ: The role of plants in traditional medicine and current therapy. J AlterComplement Med 1995; 1: 131-43.
- Verma R., Rawat A, Ganie SA, Rajneesh K, Sharma AR, Mahajan S and Gupta A: In-vitro antibacterial activity of Cichorium intybus against some pathogenic bacteria British J Pharmaceutical Research 2013; 3(4): 767-775.
- Alloush GA, Belesky DP, Clapham WM. Forage chicory: a plant resource for nutrient-rich sites. J Agron Crop Sci. 2003; 189: 96–104.
- Milhaud D, Thouvenot E, Heroum C and Escuret E: Prolonged moderate hypothermia in massive hemispheric infarction: clinical experience. J Neurosurg Anesthesiol 2005; 17: 49-53.
- Mehmood N, Zubaır M, Rızwan K, Rasool N, Shahid M and Ahmad VU: Antioxidant, antimicrobial and phytochemical analysis of Cichorium intybus seeds extract and various organic fractions. Iran J Pharm Res 2012; 11(4): 1145-1151. PMCID: PMC3813158
- O.A.C. Official method of analysis, Association of Official Analytical Chemist. EUA 2000.
- Harborne JB: Chapman and Hall Ltd; London: Phytochemical Methods 1973.
- Hussain MA, Mukhtar T, Kayani MZ, Aslam MN and Haque MI: A survey of okra (Abelmoschus esculentus) in the Punjab province of Pakistan for the determination of prevalence, incidence and severity of root-knot disease caused by Meloidogyne spp. Pak J Bot 2012; 44(6): 2071-2075.
- Kaur IP: Ocular preservatives: associated risks and newer options. Cutaneous and Ocular Toxicology 2009; 28(3): 93-103.
- Makkar HPS, Blummel M, Borowy NK and Becker K: Gravimetric determination of tannins and their correlations with chemical and protein precipitation methods. Journal of Science and Food Agriculture 1993; 61: 161-165.
- Yildirim, Oktay M, Bilaloglu V and Turk: Antioxidant assays for plant and food components. J Med Sci 2001; 31: 23-27.
- Ashafa AOT, Sunmonu TO and Afolayan AJ: Toxicological evaluation of aqueous leaf and berry extracts of Phytolacca dioica in male Wistar rats. Food Chem. Toxicol 2010; 48: 1886-1889.
- Rathee P, Chaudhary H, Rathee S, Rathee D, Kumar V and Kohli K: Mechanism of action of flavonoids as anti-inflammatory agents: a review. Inflamm. Allergy Drug Targets 2009; 8(3): 229-235.
- Ruch RJ, Cheng SJ and Klaunig JE: Prevention of cytotoxicity and inhibition of intracellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 1989; 10: 1003-1008.
- Parekh J, Nair R and Chanda S: Preliminary screening of some folklore medicinal plants from western India for potential antimicrobial activity. Indian J Pharmacol2005; 37: 408-409
- Fuselli SR, García de la rosa SB, Eguaras MJ and Fritz R: Chemical composition and antimicrobial activity of citrus essences on honeybee bacterial pathogen paenibacillus larvae, the causal agent of american World J Microbiol Biotechnol 2008a; 24: 2067-2072.
- Tian KT, Bearden WO and Hunter GL: “Consumers’ need for uniqueness: scale development and validation”, Journal of Consumer Research 2001; 28(1): 50-66.
- Dillard CJ and German JB: Phytochemicals: nutraceuticals and human health. J Sci Food Agric 2000; 80: 17441756.
- Okwu DE and Josiah C: Evaluation of the chemical composition of two Nigerian medicinal plants. Afr J Biotech 2006; 5(4): 357-361.
- Yu P, McKinnon JJ and Christensen DA: Improving the nutritional value of oat hulls for ruminant animals with pretreatment of a multienzyme cocktail: in-vitro J Anim Sci 2005; 83(5): 1133-1141.
- Chung KT, Wong TY, Huang YW and Lin Y: Tannins and human health: a review. Crit Rev Food Sci Nutr 1998; 38: 421-464.
- Ettebong E and Nwafor P: In vitro antimicrobial activities of extracts of carpolobia lutea Pak J Pharm Sci 2009; 22(3): 335-338.
- Tambekar DH, Murhekar SM, Dhanorkar DV, Gulhane PB, and Dudhane MN: Quality and safety of street vended fruit juices: a case study of Amravati city, India. Journal of Applied Biosciences 2009; 14: 782-787.
- Baydar H, Sagdic O, Ozkan G and Karadogan T: Antimicrobial activity and composition of essential oils from Origanum, Thymbra and Satureja pecies with commercial mportance in Turkey. Food Control 2004; 15: 169-172.
- Fawole AO, Hunyinbo KI and Fawole OI: Prevalence of Violence against Pregnant Women in Abeokuta, Nigeria. Aust N Z J Obstet Gynaecol 2008; 48(4): 405-14
- N’ Guessan R, Corbel V, Akogbeto M and Rowland M: 2007a. ´ Reduced efficacy of insecticide-treated nets and indoor residual spraying for malaria control in pyrethroid resistance area, Benin Emerg Inf Dis 2012; 13: 199-206.
- Kaur IP: Ocular preservatives: associated risks and newer options. Cutaneous and Ocular Toxicology 2009; 28(3): 93-103.
- Fawole AO, Hunyinbo KI and Fawole OI: Prevalence of Violence against Pregnant Women in Abeokuta, Nigeria. Aust N Z J Obstet Gynaecol 2008; 48(4): 405-14
- Parekh J, Nair R Kaur IP: Ocular preservatives: associated risks and newer options. Cutaneous and Ocular Toxicology 2009; 28(3): 93-103.
- Chanda S: Preliminary screening of some folklore medicinal plants from western India for potential antimicrobial activity. Indian J Pharmacol 2005; 37: 408-409
- Aqil F, Ahmed I Kaur IP: Ocular preservatives: associated risks and newer options. Cutaneous and Ocular Toxicology 2009; 28(3): 93-103.
- Mehmood Z: Antioxidant and free radical scavenging properties of twelve traditionally used Indian medicinal plants. Turk J Biol 2006; 30: 177-183
- Javanmardi J, Stushnoff C, Locke E and Vivanco JM: Antioxidant activity and total phenolic content of Iranian Ocimum accessions. Food Chemistry 2003; 83: 547-550.
- Rathee P, Chaudhary H, Rathee S, Rathee D, Kumar V and Kohli K: Mechanism of action of flavonoids as anti-inflammatory agents: a review. Inflamm Allergy Drug Targets 2009; 8(3): 229-235.
- Devasagayam TPA, Boloor KK and Ramsarma T: Methods for estimating lipid peroxidation: Analysis of merits and demerits (mini review). Indian J Bioche Biophys 2003; 40: 300-8.
- Daly M, Ardelean A, Cha HR, Campbell AC and Fautin DG: A New Species, Adamsia obvolva (Cnidaria: Anthozoa: Actiniaria), from the Gulf of Mexico, and a Discussion of the Taxonomy of Carcinoecium-forming Sea Anemones. Bulletin of Marine Science 2004; 74(2): 385-399.
- Fuselli SR, García de la rosa SB, Eguaras MJ and Fritz R: Chemical composition and antimicrobial activity of citrus essences on honeybee bacterial pathogen paenibacillus larvae, the causal agent of american World J Microbiol Biotechnol 2008a; 24: 2067-2072.
- Sandeep YS, Panigrahi M, Divya GC and Beena DB: Evaluation of in-vitro thrombolytic activity of phytochemicals in Bacopa monnieri Journal of Pharmacy Research 2012; 5(1): 100-101.
How to cite this article:
Ijaz A, Ahamd S, Gul H, Jabeen N and Gulfraz M: Antioxidant and antimicrobial activities of root leaves and flowers of Cichorium intybus. Int J Pharmacognosy 2017; 4(1): 23-32. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(1).23-32.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
23-32
674
1737
English
IJP
A. Ijaz *, S. Ahamd, H. Gul, N. Jabeen and M. Gulfraz
Department of Biochemistry, PMAS Arid Agriculture University, Rawalpindi, Pakistan.
ammaraijaz018@gmail.com
09 November 2016
19 December 2016
26 December 2016
10.13040/IJPSR.0975-8232.IJP.4(1).23-32
01 January 2017