ANTIMICROBIAL SOCKS FORMULATION WITH SILVER NITRATE
HTML Full TextANTIMICROBIAL SOCKS FORMULATION WITH SILVER NITRATE
S. Lakshika 1, N. Prabhu 2*, P. Revathi 1 and S. Manickavasagam 1
Department of Pharmacology 1 and Microbiology 2, Chennai Medical College Hospital and Research Centre (SRM Group), Tiruchirapalli - 621105, Tamil Nadu, India.
ABSTRACT: Antiseptics and Disinfectants are extensively used in the health care industry and also equally in personal hygiene. Among the chemicals, silver has long been known to have antimicrobial activity. Development of new, effective and low-cost antimicrobial textiles has been an objective of this research work due to increasing resistance of microbial pathogens to traditional chemicals. The potential effectiveness of silver socks (SS) fabric as an antimicrobial agent was evaluated in this study. Silver nitrate (10-3M) coated cotton socks significantly reduced the bacterial load of the socks flora among the medical students. The bacterial load is directly proportional to many days and inversely proportional to the concentration of silver nitrate. Pseudomonas species was isolated from four samples in day 3. This study reported that 10-3M silver nitrate solution coated cotton socks were very effective and having bacterial isolates rarely.
| Keywords: |
Antimicrobial socks, silver nitrate, personal hygiene
INTRODUCTION: The antimicrobial activity of silver has been known for centuries1, 2, 3, 4. The knowledge of a broad range of antimicrobial effect of silver has been now implementing including healing of burn wounds, etc. Generally, the antimicrobial mechanism of chemical agents depends on the specific binding with the surface and metabolism of agents into the microorganism. The mechanism of the antimicrobial action of silver ions is closely related to their interaction with thiol groups 5. The interaction of silver ions with thiol groups in enzymes and proteins play an important role in its antimicrobial action 5, 6. An elevated concentration of silver ions in the direct tissue may have reduced the infection rate.
By reviewing various research papers, it is indicated that silver, silver ions and nano compounds of silver could provide a safer alternative to conventional antimicrobial agents which threaten the global medical industry with its resistance 4, 5, 7, 8. The silver coins used to put into milk bottles to keep milk fresh, and water tanks of ships and airplanes that are silvered can render water potable for months. Generally, the silver shrinks the cell and gets dehydrated 9. Due to the outbreak of various infectious diseases and it’s spreading among the communities, the researchers designing newer products daily to meet out such hectic situations. Searching for new antimicrobial products is now fascinating, but the role of pharmaceutical companies in such large scale production is very less. Thus, newer and mutated infectious agents have emerged that leads to multi-organ failure life-threatening conditions 10.
Recently, awareness of general sanitation, contact disease transmission, and personal protection has led to the development of antimicrobial textiles to protect wearers against the personal hygiene and spread of bacterial and other diseases rather than to protect the quality and durability of the textiles 11. Shoes and socks have become necessary accessories of male grooming and executives due to socio-economic advancements and business achievements. Bachelors and students staying in the hostels do not wash their socks regularly due to heavy load of work and laziness. Most of the time, hostel students and representatives share their socks. An interesting observation published in The Hindu, 2007 stated even though people wash their socks regularly, they sometimes forced to wear wet socks due to lack of time to dry them. Wetness, dirtiness, and sweat provide shelter for microorganisms to grow and multiply which leads to bad odor, rashes, athlete’s foot, etc. In diabetic patients, small cut injuries may become a foot ulcer due to the habit of wearing unhygienic socks.
This induced me to take this topic and prove the efficacy of silver nitrate impregnated commercial socks. No proven reports published regarding the bacteriological status of socks and microbiological studies of silver coated socks are not much explored. Hence, the present study was undertaken to prepare silver nitrate solution of 10-3M, to determine the effect against skin flora and also to detect the shelf life of antimicrobial socks in days.
MATERIALS AND METHODS: A prospective observational study was carried out (from July to September 2013) after getting informed consent from the participants and approval from the Institutional Ethical Committee. The study was conducted in a rural teaching hospital. After explaining the study methodology, swabs were taken from the unused and used socks for three days from ten medical students who are attending clinical postings. The inclusion criteria explained to the students are to wear the cotton socks continuously for three days and should not share the socks with their friends. Students who have ulcers, skin lesions, rashes and dermatological infections in their foot and students who take antibiotics regularly were excluded from the study.
Swabs were taken from the socks of participants on day 0, 1, 2 and 3. New cotton socks were dipped in 10-3M silver nitrate solution and dried in hot air oven at 50 ºC until getting completely dry.
The same study group was instructed to wear the socks, and similarly, swabs will be taken on day 0, 1, 2 and 3. The presence of bacteria was quantitatively and qualitatively determined by microbial culture methods. The effect, shelf-life, hygiene of socks were determined by colony count versus time/day. The bacterial load and hygienic practices among the subjects were compared. Antibiotic sensitivity was performed on Muller-Hinton agar using the Kirby-Bauer disk diffusion technique.
RESULTS AND DISCUSSION: In this investigation, swabs were collected from the socks of all the ten subjects before and after usage. As a preliminary study, the swabs from the fresh socks (non silver nitrate dispensed) were considered as control and processed bacteriologically. As a result, very less number of colonies was observed in blood agar (BA), MacConkey agar (MA) and nutrient agar (NA) at 0th day. Whereas, at the end of day 1, day 2 and day 3, more than 300 colonies (too numerous to count-TNTC) were observed. An interesting observation was noted that from samples LPS 2, 3, 5, 6 and 10, colonies of Pseudomonas aeruginosa were grown on all the three days.
TABLE 1: BACTERIAL GROWTH OF 10-3M AgNO3 SOLUTION DIPPED SOCKS
| Sample no. | Number of bacterial colonies | ||||
| Control | Day 0 | Day 1 | Day 2 | Day 3 | |
| LPS 01 | TNTC | NG | NG | NG | NG |
| LPS 02 | TNTC | NG | NG | NG | NG |
| LPS 03 | TNTC | NG | NG | NG | TNTC |
| LPS 04 | TNTC | NG | NG | NG | NG |
| LPS 05 | TNTC | NG | NG | NG | TNTC |
| LPS 06 | TNTC | NG | NG | NG | NG |
| LPS 07 | TNTC | NG | NG | NG | NG |
| LPS 08 | TNTC | NG | NG | NG | TNTC |
| LPS 09 | TNTC | NG | NG | NG | NG |
| LPS 10 | TNTC | NG | NG | NG | TNTC |
TNTC- too numerous to count; NG- no growth.
The main investigation of this study is to identify the antibacterial potential of 10-3M silver nitrate which is dispensed in the cotton socks. Further, the microbiological procedures were followed as mentioned above. The antibiotic sensitivity pattern was also performed. Swabs taken from the cotton socks dipped in the silver nitrate solution in the concentration of 10-3 M showed no growth (too less to count-TLTC) on day 0, day 1 and day 2 in BA, MA and NA. Whereas on the day 3, colonies were observed in four samples (sample no. LPS 3, 5, 8 and 10). Overall, in the sample no. LPS 3 and 5, the colonies of Pseudomonas were identified in the day 3 sample. The detailed observations of the bacterial growth from the cotton socks dipped in 10-3M silver nitrate solution are depicted in Table 1.
The number of samples showed the growth of bacteria before and after exposure to 10-3M silver nitrate was depicted in Fig. 1.
FIG. 1: SAMPLES SHOWED BACTERIAL GROWTH VS. 10-3M AgNO3 SOLUTIONS
The interesting observation in the study was the growth of Pseudomonas and aerobic spore-forming bacteria (ASB). The effect of 10-3M silver nitrate solution controlled the bacterial growth in six subjects whereas the samples LPS 3, 5, 7 and 8 the growth of Pseudomonas was abundant in the day 3 that proved the Pseudomonas is resistant to silver nitrate. From this study, we learned that the concentration of 10-3M silver nitrate solution has wide bactericidal activity. Further, the study required to understand the effect of silver nitrate in lowest concentrations.
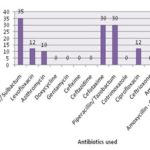
All the Pseudomonas isolated in the study were subjected to antibiotic sensitivity pattern analysis using battery of 14 antibiotics including cefoperazone/sulbactam (CFS), levofloxacin (Le), azithromycin (AZM), doxycycline (Do), gentamycin (G), cefixime (CFM), ceftazidime (CAZ), cefotaxime (CTX), piperacillin/tazobactam (PiT), cotrimoxazole (Cot), ciprofloxacin (Cip), ceftriaxone (CTR), ampicillin (Amp) and amoxicillin clavulanic acid (AMC). The results of the sensitivity pattern showed maximum sensitive to CFS (35mm), CTX (30mm) and PiT (30mm). The Pseudomonas showed resistance to other antibiotics was depicted in Fig. 2. There was a significant reduction in the bacterial load after exposed to silver nitrate solution, and the difference was statistically significant (P<0.01).
FIG. 2: ANTIBIOTIC SUSCEPTIBILITY RESULTS OF ISOLATED PSEUDOMONAS SPECIES
This study was mainly designed to find out the efficacy of silver nitrate solution in known concentration impregnated in the cotton socks. In this study, the bacteria present in the used socks consistently increased when day increases. The Pseudomonas was isolated as a resistant bacteria was quite interesting. The impregnation of silver nitrate solution in the socks control and inhibits the growth of bacteria and act as topical anti-microbial agent 12.
Some studies suggested the network of cotton fabric may provide maximum absorption of silver nitrate and is an effective method for the preparation of antibacterial fabrics 11. The importance of silver ions that enter into cell complex with DNA leads to formulate effective antimicrobial dressing 13. This methodology is likely to be of significant clinical benefit due to convenient, safe and economical means of preparing antimicrobial fabrics. The reduction of bacterial count recorded in this investigation is quite interesting, and no visible bacterial growth was observed.
As a result, this silver-based fabric formulation has been applied to a wide range of products such as burn and traumatic wound dressings, etc. 2, 10, 14.
This antimicrobial socks formulated using silver nitrate solution will be useful for the bachelors and the students who are staying in the hostels and mansions. In our study, the bacterial isolates after exposure to silver nitrate solution got reduced and also found an extension of shelf life for the usage of individuals who did not wash their socks regularly. As per our study to be concerned, we found the shelf life was about 3 days. However, further studies required to find out the extension of the days. The research work will be continued to identify the usage of these antimicrobial socks for various infections states patients including burns, leprosy, etc.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Liau SY, Read DC, Pugh WJ, Furr JR and Russell AD: Interaction of silver nitrate with readily identifiable groups: relationship to the antibacterial action of silver ions. Letter in Applied Microbiology 1997; 25: 279-83.
- Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN and Kim JO: A mechanistic study of the antibacterial effect off silver ions on Escherichia coli and Staphylococcus aureus. Journal of Biomedical and Materials Research 2000; 52: 622-8.
- Kim JS, Kuk E, Yu KN, Kim JH, Park SJ, Lee HJ, Kim SH, Park YK, Park YH, Hwang CY, Kim YK, Lee YS, Jeong DH and Cho MH: Antimicrobial effects of silver nanoparticles. Nanomedicine-Nanotechnology Biology and Medicine 2007; 3: 95-101.
- Kim J, Kwon S and Ostler E: Antimicrobial effect of silver-impregnated cellulose: potential for antimicrobial therapy. Journal of Biological Engineering 2009; 3: 20-8.
- Jung WK, Koo HC, Kim KW, Shin S, Kim SH and Park YH: Antibacterial activity and mechanism of action of the silver ion in Staphylococcus aureus and Escherichia coli. Applied Environmental Microbiology 2008; 74: 2171-8.
- McDonnell G and Russell AD: Antiseptics and infectants: activity, action and Clinical Microbiology Reviews 1999; 12: 147-79.
- Silver S: Bacterial silver resistance: molecular biology and uses and misuses of silver compounds. FEMS Microbiology Review 2003; 27: 341-53.
- Xiu ZM, Zhang QB, Puppala HL, Colvin VL and Alvarez PJJ: Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Letter 2012; 12: 4271-5.
- Guggenbichler JP, Boswald M, Lugauer S and Krall T: A new technology of microdispersed silver in polyurethane induces antimicrobial activity in central venous catheters. Infection 1999; 27: 16-23.
- Ansari MA, Khan HM, Khan AA, Malik A, Sultan A, Shahid M, Shujathullah F and Azam A: Evaluation of the anti bacteriological activity of silver nanoparticles against MSSA and MRSA on isolates from skin infections. Biology and Medicine 2011; 3: 141-6.
- Gupta P, Bajpai M and Bajpai SK: Investigation of antibacterial properties of silver nanoparticle-loaded poly (acrylamide-co-itaconic acid)-grafted cotton fabric. The Journal of Cotton Science 2008; 12: 280-6.
- Percival SL, Bowler PG and Russell D: Bacterial resistance to wound care. Journal of Hospital Infections 2005; 60: 1-7.
- Fox CL and Modak SM: Mechanism of silver sulfadiazine action on burn wound infections. Antimicrobial Agents and Chemotherapy 1974; 5: 582-8.
- Hussmann B, Johann I, Kauther MD and Landgraeber S: Measurement of silver ion concentration of silver-coated megaprostheses: correlation with the clinical outcome. Biomedical Research International 2013; 2013: 763096.
How to cite this article:
Lakshika S, Prabhu N, Revathi P and Manickavasagam S: Antimicrobial socks formulation with silver nitrate. Int J Pharmacognosy 2014; 1(7): 445-48. doi: 10.13040/IJPSR.0975-8232.1(7).445-48.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
445-448
499
2239
English
IJP
S. Lakshika, N. Prabhu *, P. Revathi and S. Manickavasagam
Department of Pharmacology, Chennai Medical College Hospital and Research Centre (SRM Group), Tiruchirapalli, India.
leptoprabhu@gmail.com
13 February 2014
19 May 2014
28 June 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(7).445-48
01 July 2014