ANTIFERTILITY EFFECT OF CAESALPINIA BONDUCELLA (L.) FLEMING IN MALE WISTAR RAT
HTML Full TextANTIFERTILITY EFFECT OF CAESALPINIA BONDUCELLA (L.) FLEMING IN MALE WISTAR RAT
Priyanka Meerwal and Gyan Chand Jain
Department of Zoology, University of Rajasthan, Jaipur - 302004, Rajasthan, India.
ABSTRACT: To evaluate the possible antifertility effect of 50% ethanolic extract of C. bonducella seeds in male rats. Adult, healthy, male Wistar rats were randomly selected and divided into three groups each having 7 rats. Group, I rats were treated with vehicle only and served as control. While group II and Group III rats were orally treated with C. bonducella extract at 200 mg and 400 mg/kg body weight/day, respectively for 60 days. The result of the present study indicated that there was a significant decrease in the relative weights of testes and accessory sex glands in treated rats. The density, motility, and viability of spermatozoa obtained from cauda epididymis were significantly (P<0.01 at 200 mg and P<0.001 at 400 mg) diminished in both treated groups as compared to control group. The serum testosterone level also showed significant (P<0.05 at 200 mg and P<0.01 at 400 mg) decline. The fertility rate and litter size decreased significantly in extract treated rats. Extract treatment also induced alteration in the biochemical milieu of the genital organs. The histopathological study of the tests showed dose-dependent degenerative and atrophic changes. The seminiferous tubules and Leydig cells were shrunk. The number of germ cells and spermatozoa in seminiferous tubules were depleted. Further, a histological study of epididymis also showed degenerative changes in the epithelium and only a few spermatozoa in the lumen of tubules. In conclusion, 50% ethanolic extract of C. bonducella seeds showed the significant antiandrogenic, antispermatogenic effect and consequently declined in fertility of male Wistar rats.
| Keywords: |
Cauda epididymis, C .bonducella, Testis, Testosterone, Spermatozoa
INTRODUCTION: Population explosion is one of the biggest problems all over the World with its inevitable consequences on all development, especially employment, education, housing, health care, sanitation, and environment. The control of fertility in the sense of its limitation especially in developing centuries is the most important and urgent need today.
During the last few decades, a large number of plants have been screened and searched thoroughly for their possible antifertility effects in males to develop male contraceptive agent of plant origin 1, 2.
Caesalpinia bonducella L. Fleming (Family-Fabaceae/Caesalpiniaceae) Commonly known as “Katikaranja,” is a prickly shrub widely distributed all over the world especially in hotter parts of India, Srilanka, and Myanmar. It is a very valuable medicinal plant, utilized in the traditional system of medicine for the treatment of a large number of ailments. The seeds have a bitter taste, are valued in traditional medicine as a tonic, antiperiodic and recommended for treating fever, asthma, and colic.
They are used in the Ayurvedic system of medicine for treating diarrhea, intestinal worms, hydrocoele, liver and spleen diseases, diabetes, leprosy and piles. They are an ingredient of several Ayurvedic preparations. The leaves and bark are used as an emmenagogue, antihelminthic and febrifuge 3. The seeds are traditionally used in the fertility regulation in rural India 4, 5, 6. Sugalis of Andhra Pradesh uses seed powder mixed with Palmyra palm sugar orally for promoting sterility in men 7.
Experimental studies have shown that C. bonducella seeds possess many pharmacological activities, like adaptogenic 8, antioxidant and antitumor 9, antidiabetic 10, immunomodulatory 11, anti-inflammatory 12, antimicrobial 13, antiulcer 14 and anti-filarial 15. Various pharmacological studies of C. bonducella have been reviewed by many workers 16, 17, 18. Ethanolic extract of C. bonducella seeds has also been reported to possess anti-implantation and abortifacient potential in female rats 19. Salunka et al. 20 reported a decrease in ovarian weight and duration of the estrous cycle in female rats by the antiestrogenic activity of alcoholic extract of C. bonducella seeds. Phytochemical analysis of C. bonducella seeds revealed the presence of alkaloids, glycosides, saponins, tannins, triterpenoids 16, 18, 21.
Literature survey reveals that no systematic approach has been made to study the antifertility activity of seeds of this plant in males despite its traditional use in inhibition of male fertility. Therefore, the present study was undertaken to study the antifertility effect of 50% ethanolic extract of C. bonducella seeds in male Wistar rats.
MATERIALS AND METHODS:
Plant Extract: Seeds of C. bonducella were collected from a local area of Smriti Van, Jaipur, Rajasthan, India. The seeds were identified and authenticated at the Herbarium, Department of Botany, University of Rajasthan, Jaipur under the specimen voucher no. (RUBL 211342). The seeds were dried, coarsely powdered and soxhlet with 50% ethanol at 55˚-60˚ C for 35 h. The extract was filtered and concentrated under low temperature and pressure to obtain a dry viscous dark brownish mass. A suspension of the extract was prepared in sterile distilled water. The required extract dose was administered orally with a glass syringe fitted with a feeding needle.
Animals: Colony bred, adult healthy male Wistar rats weighing 170-200 g, were used in the present study. The rats were housed in polypropylene cages and maintained under controlled temperature (23 ± 1ºC) and lighting conditions (12 h light/dark cycle). Standard laboratory chow (Aashirwad Food Industries, Chandigarh, India) and water were provided ad libitum. The experimental study was approved by the Ethical Committee of the Centre for Advanced Studies, Department of Zoology, University of Rajasthan, Jaipur. The Indian National Science Academy (2000) guidelines were followed for maintenance and use of the experimental animals.
Experimental Design: Adult, healthy, colony bred Wistar male rats of proven fertility were divided randomly into three groups of 7 rats each.
Group A: Control rats received 0.5 ml/day of the vehicle, i.e., distilled water.
Group B: Rats were treated with 50% ethanolic extract of C. bonducella seeds at a dose of 200 mg/kg body weight /day, orally, suspended in distilled water for 60 days.
Group C: Rats were treated with 50% ethanolic extract of C. bonducella seeds at a dose of 400 mg/kg body weight/day, orally, suspended in distilled water for 60 days.
Autopsy Schedule: Twenty-four hours after their last dose, the overnight fasted rats were weighed and sacrificed under light ether anesthesia.
Body and Organ Weights: Initial and final body weights of each rat were recorded. The testes, epididymides, vas deferens, seminal vesicle, ventral prostate were dissected out, freed from adherent tissues and blood and weighed to the nearest milligram.
Tissue Biochemistry: Testis, epididymis and seminal vesicle were frozen at -20 ºC for the biochemical estimations. Testis and epididymis were analyzed for total protein 22, glycogen 23, sialic acid 24, and total cholesterol 25. Fructose in seminal vesicle was also estimated according to the method of Mann 26.
Testosterone Hormone: Blood samples were collected from the cardiac puncture and allowed to clot at room temperature. Serum was separated by centrifugation at 2000 rpm for 20 min at 4 ºC and stored at -20 °C for hormone assays. The serum testosterone was measured with the help of chemiluminescence immunoassay.
Sperm Analysis: To determine sperm motility and sperm count, one hundred milligrams of the cauda epididymis was minced with a sharp razor blade in 2 ml of physiological saline (0.9% NaCl 37 ºC). For sperm motility, one drop of evenly mixed sample was applied to a glass slide under a cover glass. The percent motility was determined by counting both motile and immotile spermatozoa in different area 27. Sperm counts were made by a routine procedure using a Neubauer counting chamber, and expressed as million/ml of suspension 28. Sperm vitality was assessed by nigrosin-eosin staining method.
One drop of the 1% aqueous solution of eosin-y and 10% aqueous solution of nigrosin was placed in a microcentrifuge tube. A drop of well-mixed sperm sample was added to it and mixed thoroughly. The mixture was dropped on a glass slide and observed under a light microscope at 400X magnification. The percentage of life (without stain) and dead (red) cells were determined by at least counting 200 cells 29.
Fertility Test: Extract-treated male rats were paired with normal parous females (male: female ratio, 1:2) at 17:00 h daily after 55 days of treatment. The successful mating was confirmed in the forthcoming morning from 56 to 61 days by the presence of vaginal plug and spermatozoa in the vaginal smear. The inseminated females were separated and allowed to deliver at term. The numbers of pups delivered were recorded 27.
Histopathological Examination: For the histological examination, the testis and cauda epididymis were fixed in Bouin’s fixative, washed in water, dehydrated in graded ethanol series, cleared in xylene and embedded in paraffin wax. Section was cut at 5 micrometers and stained with hematoxylin and eosin. The stained slides were observed under a light microscope for histopathological changes. Mean seminiferous tubular diameter was determined with the help of a light microscope equipped with ocular micrometer calibrated with stage micrometer at × 100. The diameter of Leydig cell nucleus was also measured with the ocular help micrometer at × 1000 and expressed as a mean value.
Statistical Analysis: Data are expressed as Mean ± SEM. Statistical difference in the mean was analyzed using one way ANOVA followed by Turkey’s test. P<0.05 was considered as statistically significant.
RESULTS:
Effect on Body and Organ Weight: Although there was a significant gain in the body weight of all the control (P<0.001) and experimental rats (P<0.01) when compared to their respective initial body weight but, the control rats registered more gain in body weight as compared to extract treated rats. Animals exposed to 50% ethanolic extract of C. bonducella seeds at 200 and 400 mg/kg body weight/day doses showed a significant dose-dependent decrease in the relative weights of testes (P<0.05, P<0.001), epididymides (P<0.05, P<0.001), vas deferens (P<0.05, P<0.01), seminal vesicle (P<0.05, P<0.01) and ventral prostate (P<0.05, P<0.01) respectively, when compared with control rats Table 1.
Biochemical Analysis: The total protein and sialic acid contents of the testis and epididymis were decreased significantly in rats of both treated groups [Group B (P<0.05) and Group C (P<0.01)]. However, the concentrations of glycogen and cholesterol were significantly elevated in both testis and epididymis of extract treated rats. A significant dose-dependent decline in seminal vesicle fructose content was also recorded in extract treated rats as compared to control rats Table 2.
Hormonal Assay: A significant decline in serum testosterone level was observed in rats of both treated groups. [Group B (P<0.05) and Group C (P<0.01)] as compared to control rats Table 3.
Sperm Analysis and Fertility Test: The sperm counts and sperm motility were significantly decreased in group B (P<0.01) and group C (P<0.001) animals when compared to control animals. The percentage of viable sperms also reduced significantly in both Group B (P<0.05) and Group C (P<0.001) when compared to Group A (control). The fertility rate and the number of litter size were also declined significantly in extract treated rats Table 3, 4.
TABLE 1: EFFECT OF C. BONDUCELLA SEED EXTRACT TREATMENT ON THE BODY AND ORGAN WEIGHTS OF MALE RATS
| Treatment
|
Body weight
(g) |
Testes | Epididymides | Vas deferens |
Seminal vesicle | Ventral prostate |
|
| Initial | Final | (mg / 100 g b. wt.) | |||||
| Group- A
(Control) |
173.57
±4.72 |
202.14±3.76***
(+16.46%) |
1269.66
±27.85 |
573.76
±8.92 |
158.09
±2.22 |
554.79
±9.91 |
276.89
±4.84 |
| Group- B
C. bonducella (200 mg/kg b. wt.) |
178.57
±3.57 |
197.86±4.48**
(+10.80%) |
1163.74
±25.89 a |
503.00
±15.38a |
143.16
±4.25 a |
511.95
±8.69 a |
243.29
±4.70 a |
| Group- C
C. bonducella (400 mg/kg b. wt.) |
175.71
±3.17 |
191.43±2.61**
(+8.94%) |
1094.71
±21.51c |
472.83
±21.08 c |
139.30
±4.01 b |
495.13
±10.98 b |
222.76
±9.31 b |
Levels of significance: Values represent mean ± SEM of 7 Animals
**- P<0.01; ***- P<0.001 when compared with initial body weight; a- P<0.05; b- P<0.01; c- P<0.001 when compared with control
TABLE 2: EFFECT OF C. BONDUCELLA SEED EXTRACT TREATMENT ON VARIOUS TISSUE BIOCHEMICAL PARAMETERS OF MALE RATS
| Treatments | Total protein
(mg/g) |
Glycogen
(mg/g) |
Sialic acid
(mg/g) |
Total cholesterol (mg/g) | Fructose (mg/g) | ||||
| Testis | Epididymis | Testis | Epididymis | Testis | Epididymis | Testis | Epididymis | Seminal vesicle | |
| Group-A
Control (vehicle) |
209
±6.72 |
226.14
±7.71 |
3.46
±0.11 |
3.24
±0.09 |
5.85
±0.09 |
6.02
±0.10 |
6.19
±0.28 |
6.65
±0.10 |
5.54
±0.17 |
| Group-B
C. bonducella (200 mg/kg b.wt.) |
187.71
±5.69a |
201.14
±4.77a |
4.29
±0.25a |
4.11
±0.24a |
5.39
±0.11a |
5.52
±0.10a |
7.30
±0.23b |
7.16
±0.11a |
4.86
±0.19 a |
| Group -C
C. bonducella (400 mg/kg b. wt.) |
177.57
±3.59b |
192.29
±6.03b |
4.69
±0.27b |
4.25
±0.23b |
5.24
±0.10b |
5.39
±0.09b |
7.92
±0.11c |
7.36
±0.10b |
4.39
±0.15c |
Levels of significance: Values represent mean ± SEM of 7 Animals; a- P<0.05; b- P<0.01; c- P<0.001 when compared with control
TABLE 3: EFFECT OF C. BONDUCELLA SEED EXTRACT TREATMENT ON SERUM TESTOSTERONE, SPERM ANALYSIS AND MORPHOMETRIC PARAMETERS OF MALE RAT
| Treatments | Serum testosterone
(ng/ml) |
Sperm count (Million/ml) |
Sperm
Motility (%) |
Sperm viability
(%) |
Seminiferous tubular diameter
(µ m) |
Leydig cell nuclear diameter
(µ m) |
| Group-A Control (vehicle) |
7.19
±0.19 |
54.57
±1.45 |
75.47
±3.13 |
82.31
±7.16 |
273.43
±3.10 |
7.85
±0.34 |
| Group-B C. bonducella
(200 mg/kg b.wt.) |
6.38
±0.16a |
42.87
±2.58 b |
48.62
±7.18b |
56.77
±4.63 a |
228.57
±11.91 a |
6.71
±0.18 a |
| Group -C
C. bonducella (400 mg/kg b. wt.) |
6.10
±0.28b |
27.80
±1.56c |
33.89
±3.53c |
38.14
±6.36c |
213.57
±10.53b |
6.14
±0.26b |
Levels of significance: Values represent mean ± SEM of 7 Animals; a- P<0.05; b- P<0.01; c- P<0.001 when compared with control
TABLE 4: EFFECT OF C. BONDUCELLA SEED EXTRACT TREATMENT ON FERTILITY RATE AND LITTER SIZE
| Treatments | No. of males | No. of females | No of pregnant females | No. of fetuses delivered | Fertility
% |
| Group-A Control (vehicle) |
7 | 14 | 13/14 | 9.07±0.32 | 92.85 |
| Group-B
C. bonducella (200 mg/kg b. wt.) |
7 | 14 | 9/14 | 5.14±0.91b | 64.29 |
| Group-C
C. bonducella (400 mg/kg b. wt.) |
7 | 14 | 5/14 | 3.29±0.87c | 35.71 |
Levels of significance: a- P<0.05; b- P<0.01; c- P<0.001 when compared with control
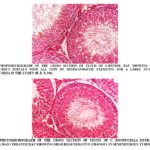
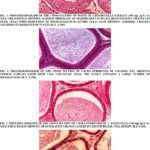
Histopathological Findings in the Testis and Epididymis: The photomicrographs of the testis of control rats showed normal structure of seminiferous tubules surrounded by vascularised dense fibrous connective tissue with prominent Leydig cells. The germinal epithelium in seminiferous tubules exhibited normal shape, size and characteristic arrangement of all successive germ cell types. The lumen was filled with a large number of healthy spermatozoa Fig. 1. The histoarchitecture of the testis treated with two different doses of C. bonducella (200 and 400 mg/kg body weight/day) showed dose-dependent degenerative and atrophic changes. The seminiferous tubules were shrunk, the numbers of germ cells were reduced, and the lumen contains sloughed debris and few spermatozoa. The Leydig cells also showed atrophic changes with reduced size of the nuclei Fig. 2, 3. Histological study of cauda epididymis of control rat showed epididymal tubules lined with tall columnar cells bearing a large number of stereocilia and lumen is full of spermatozoa Fig. 4. The cauda epididymis of extract treated rats showed dose-dependent degenerative changes. The epithelial cells lining the tubules were reduced in height and showed degenerative changes. The lumen contains sperm debris and only a few spermatozoa. The intertubular space was also increased Fig. 5, 6.
DISCUSSION: The findings of the present study showed that the 50% ethanolic seed extract of C. bonducella induced significant antifertility effects in male rats. The assessment of the weights of reproductive organs is one of the most sensitive parameters for the study of the effect of any substance on the male reproductive system 30. In the present study, oral administration of 50% ethanolic extract of C. bonducella seeds for 60 days brought a significant decline in the relative weight of testes, epididymides, vas-deferens, seminal vesicle, and ventral prostate. The weight of the testis largely depends on the mass of developing germ cells, spermatids and spermatozoa 31, 32. The reduction in testicular weight observed in the present study might be due to a reduction in testicular size, spermatogenic arrest, and inhibition of steroid biosynthesis by the Leydig cells. The decrease of relative weight of epididymis may be due to a decline in sperm count or reduced bioavailability of testosterone. It is well recognized that testosterone is crucial for the maintenance of structural integrity and functional activity of accessory sex organs 33, 34. A significant decline in the relative weights of seminal vesicle and ventral prostate could be due to reduced growth and absence of secretion in the lumen as a consequence of androgen depletion 35.
Analysis of sperm parameters viz, sperm density, motility, and viability are indices of spermatogenesis and sperm maturation and provide evidence about the effect on fertility and fecundity 36. Sperm analysis in C. bonducella extracts treated rats exhibited a significant dose depended on the decline in the sperm count, motility and number of viable spermatozoa. Sperm cell count is considered the most sensitive test for spermatogenesis and it is highly correlated with fertility 37. The diminution of epididymal sperm count in extract treated rats may be due to the adverse impact of the extract on spermatogenesis. The epididymis plays an important role in sperm development, maturation, transport and storage where it depends on the luminal environment of epididymis 38. The decline in the percentage of motile and viable spermatozoa in extract treated rats in the present study might have resulted from the alteration in the epididymal milieu due to androgen deficiency and also due to toxic effects on cellular levels 39, 40.
The loss of motility of spermatozoa might also be due to an adverse effect on oxidative/energy metabolism which might disrupt the energy supply for sperm motion 41. These results are in agreement with earlier reports where an extract of C. bonducella seeds has been reported to cause a decline in sperm density 42 and to increase morphological abnormalities in spermatozoa of rats 43.
Treatment with 50% ethanolic extract of C. bonducella seeds brought a significant decline in fertility rate (%) and litter size. This decline in fertility and litter size may be attributed to deterioration of sperm quality, and density 44, 45, since the inadequate concentration, sluggish motile or immotile spermatozoa could not penetrate the cervical mucus and thus failed to fertilize the ova 46, 47.
Testosterone is the primary circulating androgen synthesized and secreted by Leydig cells under the influence of pituitary luteinizing hormone (LH) 48. In the present study, a significant dose-dependent decline in the levels of serum testosterone was observed in rats treated with C. bonducella extract. Histological observation of testis of extract treated rats also supports these findings, as we observed shrinkage and atrophic changes in the Leydig cells. The increased testicular cholesterol content in extract treated rats also support adverse impact on steroidogenesis. The decline in serum testosterone might be due to the inhibitory effect of the plant extract on pituitary LH secretion or through direct action on the Leydig cells resulting in inhibition of steroidogenic activities. C. bonducella seed extract has been reported to possess antiestrogenic effect in female rats due to presence of phytoestrogens 20, which might interfere in estrogen negative feedback by binding to estrogen receptors in the anterior pituitary or hypothalamus and thereby reduce secretion of the gonadotropin-releasing hormone (GnRH), follicle stimulating hormone (FSH) and luteinizing hormone (LH) resulting in the decline of testosterone biosynthesis and secretion in Leydig cells.
It has been reported that protein level is directly correlated with the secretory activity of the testis and epididymis which is dependent on androgen level 49. Oral treatment of rats with C. bonducella extract also induce a biochemical alteration in the milieu of testis and epididymis. The present study indicates a significant reduction in the concentration of protein in testis and epididymis. Protein biosynthesis is a key factor for testicular development and spermatogenesis. Sertoli cells synthesize and secrete a variety of proteins required during spermatogenesis 50. The decline in protein content in testis might be due to disturbances in cellular metabolism, lack of normal level and activity of testicular androgens and decrease in the number of spermatozoa in the testis 51. The decrease in the protein concentration in epididymis possibly affects the internal milieu of the organ and consequently maturation of spermatozoa. A similar decrease in protein contents of the testis and epididymis have been reported due to the treatment of many plant extracts in rats possibly due to antiandrogenic effect 52, 53.
Increase in glycogen content in both testis and epididymis indicates underutilization of glycogen either due to an inhibitory effect on glycogen phosphorylase or glycolytic enzymes 54. Sialic acid is an important constituent of glycoprotein and glycolipids and is essential for the maintenance of the structural integrity of the sperm membrane and sperm maturation 55. The reduction in the sialic acid concentration in testis and epididymis may be correlated with impairment of androgen secretion along with necrotic and atrophic changes in principal cells of the epididymis 53, 56.
Cholesterol is an essential component of mammalian cell membrane and also acts as a precursor for steroid hormone biosynthesis in gonads. A significant increase of testicular cholesterol concentration in the extract treated rats reflects the reduced conversion of cholesterol to androgen or the arrest of steroidogenesis in Leydig cells leading to accumulation of cholesterol in tests 54, 51. Increased in cholesterol concentration in epididymis might result in disturbances in sperm maturation and fertilizing ability of spermatozoa. Cholesterol homeostasis regulation is crucial for post-testicular sperm maturation, and imbalanced cholesterol level may affect these post-testicular events 57. Fructose is an important source of energy for motility of spermatozoa. A significant reduction in fructose content of seminal vesicle reflects impairment of the secretory function of the gland attributed to androgen depletion 58. A similar decrease in seminal vesicle fructose level mediated by the antiandrogenic effect of various plant extracts has been reported earlier by many workers 54, 40.
Histological examination of the testis of 50% ethanolic extract of C. bonducella seeds showed a significant dose-dependent degenerative, atrophic changes and impairment of spermatogenesis. The seminiferous tubules were shrunk and the number of post-meiotic germ cells and spermatozoa in the lumen were significantly reduced. The tiered arrangement of the germ cells was distributed. The Leydig cells were shrunken and showed atrophic changes. These observed histopathological changes in testis possibly occurred as a consequence of the decline in serum testosterone which is essential for normal spermatogenesis as well as for the maintenance of structural morphology and normal physiology of seminiferous tubules 59. The reduction in the number of post-meiotic germ cells in extract treated rats again indicates diminished testosterone level, as testosterone is essential for the post-meotic division during spermatogenesis 60.
According to Sharpe 61, the male reproductive system possesses estrogenic receptor α (ER-α) and β (ER-β). Alcoholic extract of C. bonducella seeds has been reported to induce antiestrogenic effect due to the presence of estrogenic phytoconstituents. 20 Hence, an estrogenic overload may occur as a consequence of the binding of constituents present in the extract to the estrogenic receptors. Many chemicals which bind to estrogen receptors on male genital organs have shown similar degenerative changes and impairment of spermatogenesis by a decline in serum testosterone 31, 62.
Estrogen is likely to affect Leydig cell function through at least two mechanisms, including suppression of LH release by negative feedback and a direct effect on the steroidogenesis 63, 64. Our results are in agreement with previous studies where plant extracts rich in phytoestrogens have shown similar degenerative changes and impairment of spermatogenesis in tests 65, 66.
Administration of 50% ethanolic extract of C. bonducella seeds in rats also caused significant dose-dependent degenerative changes in cauda epididymis. The epithelial cells lining the epididymal tubules were reduced in height, showed degenerative changes and bear short stereocilia. The lumen shared the presence of sperm debris and scanty spermatozoa. The intertubular stroma was also increased. The observed changes in cauda epididymis of extract treated rats may be correlated with deficit androgen supply. These results are in agreement with the previous finding where a similar type of degenerative changes and decline of spermatozoa in lumen have been reported as a consequence of the decrease of androgen supply in rats treated with various plant extracts 67, 68, 69, 70.
CONCLUSION: From the results of the present study it may be concluded that phytoconstituents present in the 50% ethanol extract of C. bonducella seeds by their individual or synergistic activity induced significant antispermatogenic and antiandrogenic effect leading to declining of fertility in male Wistar rats. Further studies at higher doses are in progress.
ACKNOWLEDGMENT: The authors are thankful to Head of the department and coordinator, CAS for providing necessary facilities and also to the University Grant Commission for awarding Rajiv Gandhi National Fellowship to Priyanka Meerwal.
CONFLICT OF INTEREST: Nil
REFERENCES:
- D'Cruz SC, Vaithinathan S, Jubendradass R and Mathur PP: Effects of plants and plant products on the testis. Asian J Androl 2010; 12: 468-479.
- Meerwal P and Jain GC: Male Fertility Regulation with Plant Products: A review. IJPCBS 2015; 5: 146-162.
- Parrotta JA: Healing plants of peninsular India. CABI Publishing 2001; 7: 416.
- Kirtikar KR and Basu BD: Indian medicinal plants, 3rd ed. Allahabad: Lalit Mohan Basu 1946.
- Chopra IC, Handa KL and Kapoor LD: Indigenous drugs of India. Calcutta: Dhur, U.N. and Sons Pvt. Ltd 1958.
- Nadkarni KM: Indian materiamedica. India: Popular Prakashan Pvt. Ltd 1976; 1: 65-71.
- Ram AJ, Raja K, Eswara RK and Raju RRV: Medicinal plant role of sugalis of Gooty forests, Andhra Pradesh. Ethnobotany 2002; 14: 37-42.
- Kannur DM, Hukkeri VI and Akki KS: Adaptogenic activity of Caesalpinia bonduc seed extracts in rats. J Ethnopharmacol 2006; 108: 327-331.
- Archana P, Tandan SK, Chandra S and Lal J: Antipyretic and analgesic activities of Caesalpinia bonducella seed kernel extract. Phytother Res 2005; 19: 376-381.
- Parmeshwar S, Srinivasan KK and Mallikarjuna RC: Oral antidiabetic activities of different extracts of Caesalpinia bonducella seed kernels. Pharm Biol 2002; 40: 590-595.
- Shukla S, Mehta A, John J, Mehta P, Vyas SP and Shukla S: Immunomodulatory activities of the ethanolic extract of Caesalpinia bonducella J Ethnopharmacol 2009; 125: 252-256.
- Kale S, Gajbhiye G and Chaudhari N: Anti-inflammatory effect of petroleum ether extract of Caesalpinia bonduc (L.) Roxb. seed kernel in rats using carrageenan-induced paw edema. Int J Pharm Tech Research 2010; 2: 750-752.
- Saeed MA and Sabir AW: Antibacterial activity of Caesalpinia bonducella Fitoterapia 2001; 72: 807-809.
- Javed AA, Shafique A and Jameel M. Effect of Caesalpinia bonducella on the ulcer and gastric secretions in pylorus ligated rat model. JDDT 2012; 2: 102-104.
- Gaur RL, Sahoo MK, Dixit S, Fatima N, Rastogi S and Kulshreshtha DK: Antifilarial activity of Caesalpinia bonducella against experimental filarial infections. Indian J Med Res 2008; 128: 65-70.
- Moon K, Khadabadi SS, Deokate UA and Deore SL: Caesalpinia bonducella F-An overview. Report and Opinion 2010; 2: 83-89.
- Singh, V., and P.K. Raghav. Review on pharmacological properties of Caesalpinia bonduc Int. J. Med. Arom. Plants 2012; 2: 514-530.
- Shelar PA, Mandavkar YD, Khedkar AS, Thorat MB and Raje VN: Review on pharmacology and phytochemistry of Caesalpinia bonduc. JCPR 2014; 4: 1309-1317.
- Lilaram and Ahmed RN: Effect of ethanolic seed extract of Caesalpinia bonducella on fertility in pregnant female albino rats. APJR 2013; 2: 85-89.
- Salunke KR, Ahmed RN, Marigoudar SR and Lilaram: Effect of graded doses of Caesalpinia bonducella seed extract on ovary and uterus in albino rats. Basic Clin Physiol Pharmacol 2011; 22: 49-53.
- Rajalakshmi P, Vadivel V, Subhashini G and Pugalenthi M: Phytochemical Screening and in-vitro antioxidant activity of Caesalpinia bonducella IJAR 2015; 5: 11.
- Lowry OH, Rosebrough MJ, Farr AL and Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 1951; 193: 256-275.
- Montgomery R: Determination of glycogen. Arch Biochem Biophys 1957; 67: 378-386.
- Warren L: The thiobarbituric acid assay of sialic acids. J Biol Chem 1959; 234: 1971-1975.
- Zlatkis A, Zak B and Boyle AJ: A new method for direct determination of cholesterol. J Lab Clin Med 1953; 41: 486-492.
- Mann T: Biochemistry of the semen and the male reproductive tract. John Wiley and Sons, Inc., London 1964: 391-392.
- Protocol MB-50: A method for examining the effect of the plant extracts administered orally on the fertility of male rats (APF/IP, 9914E). World Health Organization, Geneva, Switzerland 1983.
- Prasad MR, Chinoy NJ and Kadam KM: Changes in succinic dehydrogenase levels in the rat epididymis under normal and altered physiologic conditions. Fertil Steril 1972; 23: 186-190.
- Bjorndahl L, Sodourlund I and Kvist U: Evaluation of the one-step eosin-nigrosin staining technique for human sperm vitality assessment. Hum. Reprod 2003; 18: 813-816.
- Mangelsdorf I, Buschmann J and Orthen B: Some aspects relating to the evaluation of the effects of chemicals on male fertility Regul Toxicol Pharmacol 2003; 37: 356-369.
- Jana K, Jana S and Samanta PK: Effect of chronic exposure to sodium arsenate on hypothalamic-pituitary-testicular activities in adult rats: possible an estrogenic mode of action, Reprod Biol Endocrinol 2006; 4: 9.
- Sellers RS, Morton D, Michael B, Roome N and Johnson JK: Society of Toxicologic Pathology Position Paper: Organ Weight Recommendations for Toxicology Studies. Toxicol Pathol 2007; 35: 751-755.
- Klinefelter GR and Hess RA: Toxicology of the male excurrent ducts and accessory sex glands. In: Reproductive and Developmental Toxicology, Korach KS (ed). Marcel Dekker, New York 1998: 553-591.
- Jarfelt K, Dalgaard M, Hass U, Borch J, Jacobsen H and Ladefoged O: Antiandrogenic effects in male rats perinatally exposed to a mixture of di (2-ethylhexyl) phthalate and di (2-ethylhexyl) adipate. Reprod Toxicol 2005; 19: 505-515.
- Partin AW and Coffey DS: The molecular biology, endocrinology, and physiology of the prostate and seminal vesicles. In Campbell’s Urology (P. C. walsh, A. B. Retik, E. J. D. Vaughan and A. J. wein) WB Philladelphia, Saunders 1998; 3: 1381-1428.
- Foley GL: Overview of male reproductive pathology. Pathol 2001; 29: 49-63.
- El-Kashoury AA, Salama AF, Selim AI and Mohamed RA: Animal model study of reproductive toxicity of the chronic exposure of dicofol. Life Sci J 2009; 6: 1-18.
- Turner TT: De Graaf's Thread: The Human Epididymis. J Androl 2008; 29: 237-250.
- Kulkarni TR, Mateenuddin M, Bodhankar SL and Saharabudhe RA: Reversible anti- fertility effect of lemon seeds (Citrus limonum) in male albino rats. IJPBR 2012; 3: 545-550.
- Ahmed M, Al-Daghri N, Alokail MS and Hussain T: Potential changes in rat spermatogenesis and sperm parameters after inhalation of Boswellia papyrifera and Boswellia carterii Int J Environ Res Public Health 2013; 10: 830-844.
- Rivlin J, Mendel J, Rubinstein S, Etkovitz N and Breitbart H: Role of hydrogen peroxidein sperm capacitation and acrosome reaction. Biol Reprod 2004; 70: 518-522.
- Kanerkar UR, Bhogaonkar PY and Indurwade NH: Antispermatogenic effect of Caesalpinia bonduc (L.) seeds. Int Res J of Science & Engineering 2015; 3: 173-178.
- Peerzade N, Ahmed RN and Marigoudar SR: Morphological changes induced by Caesalpinia bonducella seed extract on rat sperm scanning electron microscope study. J Basic Clin Physiol Pharmacol 2009; 20: 309-313.
- Ramya T, Misro MM, Sinha D and Nandan D: Sperm function and seminal oxidative stress as tools to identify sperm pathologies in infertile men. Fertil Steril 2010; 93: 297-300.
- Dhanapal R, Ratna JV, Gupta M and Sarathchandran I: Preliminary study on antifertility activity of Enicostem maaxillare leaves and Urena lobata root used in Indian traditional folk medicine. Asian Pac J Trop Med 2012; 5: 616-622.
- Singh A and Singh SK: Reversible antifertility effect of aqueous leaf extract of Allamanda cathartica in male laboratory mice. Andrologia 2008; 40: 337-345.
- Bairy L, Paul V and Rao Y: Reproductive toxicity of sodium valproate in male rats. Indian J. Pharmacol 2010; 42: 90-94.
- Soez JM: Leydig cells: endocrine, paracrine, and autocrine regulation. Endocr Rev 1994; 15: 574-6.
- Basha SH, Lalithamma A, Lakshman J and Changamma C: Antifertility effect of Carica Papaya seed extract on hormones in male albino rats. IJBPR 2013; 4: 859-861.
- Weinbauer GF, Luetjens CM, Simoniand M and Nieschlag E: Physiology of Testicular Function. Andrology: 11-59. DOI: 10.1007/978-3-540-78355-8_2, © Springer- Verlag Berlin Heidelberg 2010.
- Olaolu T, Akinwande D and Olaolu AP: Evaluation of aqueous leaf extract of Cissampelos mucronata on testicular function indices in wistar Int J Biochem Res Rev 2015; 5: 233-241.
- Vijaykumar B, Sangamma I, Sharanabasappa A and Patil SB: Antispermatogenic and hormonal effects of Crotolaria juncea seed extract. Asian J Androl 2004; 6: 67-70.
- Jain GC and Ali SM: Effect of ethanolic extract of Cassia alata flowers on reproductive functions of male albino rats. J Exp. Zool 2007; 10: 129-132.
- Mathur N, Jain GC and Pandey G: Effect of Tecoma stans leaves on the reproductive system of male albino rats. Int J Pharmacology 2010; 6: 152-156.
- Rajalakshmi M, Sharma RS, Paland PC and Kapur MM: Sialic acid levels in spermatozoa and luminal fluid of normal and infertile men. Curr Sci 1997; 73: 1012.
- Agrawal M, Sharma P and Kushwaha S: Antifertility efficacy of 50% ethanolic extract of Calendula officinalis in male rats. Int J Pharm. Pharm Sci 2011; 5: 192-196.
- Whitfield M, Pollet‑ Villard X, Levy R, Drevet JR and Saez F: Post-testicular sperm maturation, infertility, and hypercholesterolemia. Asian J Androl 2015; 17: 742-748.
- D'Cruz SC and Mathur PP: Effect of piperine on the epididymis of adult male rats. Asian J Androl 2005; 7: 363-368.
- Sharpe RM, Donachie K and Cooper I: Re-evaluation of the intratesticular level of testosterone required for quantitative maintenance of spermatogenesis in the rat. J Endocrinol 1988; 117: 19-26.
- McLachlan RI, O'Donnell L and Stanton PG, Balourdos G and Frydenberg M: Effects of testosterone plus medroxyprogesterone acetate on semen quality, reproductive hormones, and germ cell populations in normal young men. J Clin Endocrinol Metab 2002; 87: 546-556.
- Sharpe RM: The roles of estrogen in the male. Trends Endocrinol Metab 1998; 9: 371-377.
- Yang ZW, Kong LS, Guo Y, Yin JQ and Mills N: Histological changes of the testis and epididymis in adult rats as a result of Leydig cell destruction after ethane dimethane sulfonate treatment: A morphometric study. Asian J. Androl 2006; 8: 289-299.
- Kaneto M, Kanamori S, Hishikawa A and Kishi K: Epididymal sperm motion as a parameter of male reproductive toxicity: Sperm motion, fertility and histopathology in ethinylestradiol-treated rats. Reprod Toxicol 1999; 13: 279-289.
- Shimomura K, Shimada M, Hagiwara M, Harada S, Kato M and Furuhama K: Insights in to testicular damage induced by ethinylestradiol in rats. Reprod. Toxicol 2005; 20: 157-163.
- Assinder S, Davis R, Fenwick M and Glover A: Adult-only exposure of male rats to a diet of high phytoestrogens content increases apoptosis of meotic and post-meotic germ cells. Reproduction 2007; 133: 11-19.
- Ibrahim AAE: Correlation between fennel-or-anise-oil administration and damage to the testis of adult rats. Egypt J Boil 2008; 10: 62-76.
- Akbarsha MA, Manivannan B, Hamid KS and Vijayan B: Antifertility effect of Andrographis paniculata (Nees) in male albino rat, Indian J Exp Biol 1990; 28: 421-426.
- Kasturi M, Manivannan B, Ahamed RN, Shaikh PD and Pathan KM: Changes in epididymal structure and function of albino rat treated with Azadirachta indica Indian J Exp Biol 1995; 33: 725-729.
- Nusier MK, Bataineh H and Daradka HM: Adverse effects of Rosemary (Rosmarinus officinalis L.) on reproductive function in adult male rats. Exp Biol Med 2007; 232: 809-813.
- Ahmed M, Ahamed R, Aladakatti RH and Ghodesawar MAG: Effect of benzene extract of Ocimum sanctum leaves on cauda epididymal spermatozoa of rats. Iran J Reprod Med 2011; 9: 177-186.
How to cite this article:
Meerwal P and Jain GC: Antifertility effect of Caesalpinia Bonducella (L.) Fleming in Male Wistar rat. Int J Pharmacognosy 2016; 3(6): 265-75. doi: 10.13040/IJPSR.0975-8232.3(6).265-75.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.