ANALYTICAL METHOD DEVELOPMENT AND VALIDATION FOR ESTIMATION OF ALPRAZOLAM AND SERTRALINE HYDROCHLORIDE BY HPLC AND UV
HTML Full TextANALYTICAL METHOD DEVELOPMENT AND VALIDATION FOR ESTIMATION OF ALPRAZOLAM AND SERTRALINE HYDROCHLORIDE BY HPLC AND UV
Pradyumn Kumar Singh *, Yogendra Singh and Saurabh Jain
Shri Ramnath Singh Mahavidyalaya (Pharmacy), Gormi, Bhind, Madhya Pradesh, India.
ABSTRACT: The present study involves the development and validation of simple, accurate, and precise analytical methods for the simultaneous estimation of Alprazolam and Sertraline Hydrochloride in combined pharmaceutical dosage forms. Two methods were developed: UV spectrophotometry and reverse-phase high-performance liquid chromatography (RP-HPLC). The UV spectrophotometric method utilized the simultaneous equation method based on absorbance measurements at 260 nm and 273 nm, the respective λmax values of Alprazolam and Sertraline HCl. Both drugs obeyed Beer’s Law in the concentration range of 5–50 μg/mL, with high correlation coefficients, indicating good linearity. The method showed satisfactory precision and accuracy, making it suitable for routine analysis. The RP-HPLC method was performed using a C18 column and a mobile phase composed of methanol, acetonitrile, and phosphate buffer (pH 4.5) in the ratio 50:15:35 v/v, at a flow rate of 1 mL/min. Detection was carried out at 239 nm, and the retention times were found to be approximately 3.0 minutes for Alprazolam and 6.4 minutes for Sertraline HCl. The method was validated according to ICH guidelines, and parameters such as specificity, linearity, accuracy, precision, robustness, and sensitivity were within acceptable limits. Both methods were successfully applied to the estimation of the drugs in commercial tablet formulations. The developed methods are reliable and can be effectively employed in routine quality control and pharmaceutical analysis.
Keywords: Alprazolam, Sertraline HCl, Method Validation, UV, HPLC
INTRODUCTION: Sertraline and alprazolam belongs to the class of medications called ‘antidepressant’ used in the treatment of panic disorder with or without agoraphobia (fear of leaving one’s own home or being in open or crowded places) 1. Panic disorder is characterized by a panic attack in which the patient experiences a sudden feeling of terror even if there is no real danger. Sertraline and alprazolam contains Alprazolam and Sertraline. Alprazolam is a benzodiazepine 2.
It works by increasing the activity of GABA (a chemical messenger in the brain that acts as a natural nerve-calming agent). Thereby, Sertraline & alprazolam relaxes muscles, produces a calming effect, and helps to fall asleep. Sertraline is a selective serotonin reuptake inhibitor (SSRI) 3. It increases the release of the serotonin hormone in the body, which is responsible for improving mood, cognition, and memory. The present UV and HPLC methods are relatively simple, rapid, and highly sensitive in the determination of sertraline and alprazolam.
Only limited methods have been reported in the literature survey 4–7. The aim of the present work was to develop and validate a simple, fast, and reliable isocratic RP-HPLC and UV method for the determination of sertraline and alprazolam in pharmaceutical dosage forms. The important features and novelty of the proposed method included simple sample treatment with sonication of small amount of powder sample at ambient temperature, centrifugation, and dilution; short elution time with internal standard eluted prior tosertraline and alprazolam; good precision (R.S.D. less than 5%) and high recovery (greater than 95%). Confirmation of the applicability of the developed method was validated according to the International Conference on Harmonisation (ICH), to determination of sertraline and alprazolam in pharmaceutical dosage forms.
MATERIALS AND METHODS:
Materials: A market sample of BEREST was analyzed, which is labeled to contain Alprazolam 0.25 mg and Sertraline HCl 25 mg. The analysis was carried out using an ATCO Balance for accurate weighing of the sample and a SHIMADZU UV-1700 double beam digital UV–Visible spectrophotometer for spectroscopic measurements. Methanol (AR grade) was used as the solvent for the preparation of standard and sample solutions. The instruments used in the study included a Shimadzu UV-Visible SPD-20A detector, an LC-20AT isocratic system, a Rheodyne injector, and a C18 column. The reagents and chemicals employed for the analysis were acetonitrile, HPLC grade water, buffer solution, and methanol, all of which were of analytical or HPLC grade to ensure accuracy and reliability of the results.
U.V. Method:
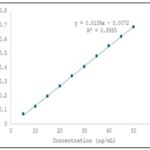
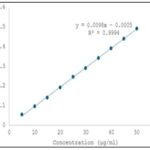
Fixation of Various Parameters (Sertraline HCl): The determination of absorption maximum (λmax) for Sertraline HCl was performed by accurately weighing 30 mg of authentic Sertraline HCl and transferring it into a 50 ml volumetric flask, where it was dissolved in methanol and the volume was made up to the mark with the same solvent. From this stock solution, 5 ml was pipetted into another 100 ml volumetric flask and diluted to volume with methanol. The absorbance of the resulting solution was measured against a solvent blank in the UV region of 200–400 nm, and the λmax was found to be 236 nm, as shown in the corresponding spectrum. For the Beer’s Law plot, 100 mg of authentic Sertraline HCl was accurately weighed, dissolved in methanol in a 100 ml volumetric flask, and diluted to the mark. From this solution, aliquots of 0.5 ml to 5 ml were transferred into separate 100 ml volumetric flasks and diluted to volume with methanol. The absorbance of each solution was recorded at 273 nm against a reagent blank 8, 9.
FIG. 1: SERTRALINE HCL PLOT
FIG. 2: DATA FOR DEVIATIONS FORM BEER’S LAW PLOT
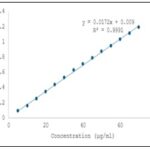
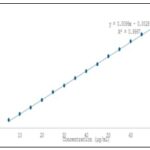
Determination of Absorption Maximum (λmax) Alprazolam: A 100 mg quantity of authentic alprazolam sample was accurately weighed and transferred into a 50 mL volumetric flask, dissolved in methanol, and the volume was made up to the mark with methanol. From this stock solution, 5 mL was pipetted into a separate 100 mL volumetric flask and diluted to volume with methanol. The absorbance of the resulting solution was measured against a solvent blank in the UV region between 200–400 nm, and the absorption maximum (λmax) was found to be 260 nm. For Beer’s Law plot determination, 100 mg of alprazolam was accurately weighed and transferred into a 100 mL volumetric flask, dissolved in methanol, and the volume was made up to 100 mL with methanol to obtain a standard stock solution. From this, aliquots of 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, and 5.0 mL were pipetted into separate 100 mL volumetric flasks and diluted to volume with methanol. The absorbance of each solution was measured at 245 nm against a reagent blank to establish the Beer’s Law plot for alprazolam 10, 11.
FIG. 3: DATA FOR BEER’S LAW PLOT FOR ALPRAZOLAM
FIG. 4: DATA FOR DEVIATIONS FROM BEER’S LAW PLOT FOR ALPRAZOLAM
Preparation of Sertraline HCl Standard Solution: 100mg of authentic sertraline HCl sample is accurately weighed and transferred to 100ml volumetric flask and methanol was added and shaken until it dissolves and the volume was made upto 100ml with methanol.
From this 1ml was pipetted out in to separate 100ml volumetric flask and the volume was made up to 100ml with methanol. The absorbance and absorptivity values are shown in Table 1A & 1B 12.
Preparation of Sample Solution: Twenty tablets are weighed and average weight was calculated. The tablets are ground to a fine powder. A powder equivalent to 25mg of sertraline and 0.25 mg of alprazolam was accurately weighed and transferred to 50ml volumetric flask and methanol was added and shaken until it dissolves and the volume was made upto 50ml with methanol.
This solution was filtered through Whatmann filter paper. From this 1ml was pipetted out in to separate 100ml volumetric flask and the volume was made up to 100ml with methanol. The absorbance of each solution was found out at 273nm (λmax of sertraline HCl) and 260nm (λmax of Alprazolam) against a reagent blank. The analysis values are given in Table 2 13.
TABLE 1A: ABSORBANCE VALUES FOR STANDARD AND SAMPLE
| Wavelength (nm) | Compound | Standard Absorbance | Notation | Sample Absorbance | Notation |
| 273 nm (λ₁) | Alprazolam | 0.2543 | X₁ | 0.6984 | A₁ |
| Sertraline HCl | 0.1233 | Y₁ | |||
| 260 nm (λ₂) | Alprazolam | 0.1543 | X₂ | 0.5984 | A₂ |
| Sertraline HCl | 0.1763 | Y₂ |
TABLE 1B: ABSORPTIVITY VALUES FOR ALPRAZOLAM AND SERTRALINE HCL
| Parameter | Alprazolam (aₓ) | Sertraline HCl (aᵧ) |
| Absorptivity at 273 nm (λ₁) (L·mol⁻¹·cm⁻¹ or L·g⁻¹·cm⁻¹) | aₓ₁ = 212.6 | aᵧ₁ = 1.233 |
| Absorptivity at 260 nm (λ₂) | aₓ₂ = 1232.1 | aᵧ₂ = 1432.9 |
| Standard Deviation at 273 nm | SDₓ₁ = 0.3324 | SDᵧ₁ = 0.2531 |
| Standard Deviation at 260 nm | SDₓ₂ = 0.4743 | SDᵧ₂ = 0.1211 |
* Absorptivity values are the mean of six determinations. S.D. is standard deviation. ax1 and ax2 are absorptivities of alprazolam at 260nm and 273nm respectively; ay1 and ay2 are absorptivities of sertraline hydrochloride at 260nm and 273nm respectively.
TABLE 2: ANALYSIS DATA OF TABLET FORMULATIONS
| Parameter | Sertraline HCl | Alprazolam |
| Label Claim | 25 mg | 0.25 mg |
| Amount Found | 24.46 mg | 0.234 mg |
| % Drug Content | 99.8 % | 99.95 % |
| Standard Deviation (SD) | 0.2764 | 1.4320 |
| % Relative Standard Deviation (RSD) | 0.2439% | 1.1543% |
Recovery Studies: To check the accuracy of the developed method and to study the interference of formulation additives, analytical recovery experiments were carried out by standard addition method at 80, 100 and 120% level. From the total amount of drug found the percentage recovery was calculated. The results are reported in Table 3 and 4 14.
TABLE 3: RECOVERY STUDIES: SERTRALINE HCL
| Range (%) | Amount Found (mg) | Recovery (%) | R.S.D (%) |
| 80% | 24.55 | 99.80 | 0.0995 |
| 100% | 24.58 | 99.80 | 0.1892 |
| 120% | 24.59 | 99.83 | 0.09 |
*Recovery is the mean of three estimations.
TABLE 4: RECOVERY STUDIES: ALPRAZOLAM
| Range (%) | Amount Found (mg) | Recovery (%) | R.S.D (%) |
| 80% | 0.245 | 99.90 | 0.2652 |
| 100% | 0.247 | 99.95 | 0.1321 |
| 120% | 0.248 | 99.90 | 0.1875 |
*Recovery is the mean of three estimations.
HPLC Method Development: For the preparation of the standard solution of alprazolam, accurately 0.00025 g of alprazolam was weighed and transferred into a 100 mL volumetric flask, and the volume was made up to the mark with the mobile phase. From this solution, 5 mL was pipetted into a separate 50 mL volumetric flask and diluted to volume with the mobile phase to obtain a concentration of 0.25 mg/mL. A 20 µL portion of this solution was injected, and the chromatogram was recorded. (B) For the preparation of the standard solution of sertraline hydrochloride, accurately 0.025 g of the drug was transferred into a 100 mL volumetric flask and diluted to volume with the mobile phase.
From this solution, 5 mL was pipetted into a separate 50 mL volumetric flask and the volume was made up to 50 mL with the mobile phase to obtain a concentration of 250 mg/mL. A 20 µL portion of this solution was injected, and the chromatogram was recorded. (C) For the preparation of the mixed standard solution, accurately 0.00025 g of alprazolam and 0.025 g of sertraline hydrochloride were transferred into a clean, dried 100 mL volumetric flask, dissolved in 20 mL of the mobile phase, and sonicated.
The volume was then adjusted to 100 mL with the mobile phase. From this mixed stock solution, 5 mL was transferred into a 50 mL volumetric flask and diluted to volume with the mobile phase to obtain a final concentration of 0.25 mg/mL of alprazolam and 25 mg/mL of sertraline hydrochloride, and a 20 µL portion of this solution was injected for chromatographic analysis 15, 16.
Fixed Chromatographic Conditions: The analysis was carried out using a Shimadzu Prominence model equipped with an LC-20AT isocratic system and a C18 column. The detection wavelength was set at 239 nm, and the analysis was performed at ambient temperature with a flow rate of 1 mL/min. The injection volume was 20 µL, and the mobile phase consisted of a mixture of buffer (potassium dihydrogen phosphate), methanol, and acetonitrile in the ratio of 50:15:35 (v/v/v). The retention times were observed to be 3.0 minutes for alprazolam and 6.4 minutes for sertraline hydrochloride. For the quantitative determination of the drugs using the developed method, a combined sample containing alprazolam and sertraline hydrochloride was analyzed, with label claims of 0.25 mg for alprazolam and 25 mg for sertraline hydrochloride 17-20.
Method: Twenty tablets were weighed and powdered. Average weight 593.2g of sample tablet Berest (equivalent to 0.25mg of alprazolam and 25mg sertraline hydrochloride was taken into 100ml dried volumetric flask. The powder was first dissolved in 20ml mobile phase and sonicated and finally the volume was adjusted to 100ml with mobile phase. From this solution 5ml was transferred to 50ml volumetric flask and volume was adjusted to 50ml with mobile phase to get a concentration of 0.25 mg/ml of alprazolam and 25 mg /ml of sertraline hydrochloride. 20ml of the solution was injected 21. The amount of alprazolam and sertraline hydrochloride present in the tablet formulation was calculated by comparing the peak area of the standard.
TABLE 5: QUANTITATIVE ESTIMATION
| S. no. | Brand Name | Content Label Claim (mg) | Peak Area | Amount Present (mg) | Percent Purity% w/v |
| 1 | Berest | Alprazolam 0.25 mg | 1423 | 0.249 | 99.45% |
| Sertraline HCl 25 mg | 6712 | 24.455 | 100.05% |
Acceptance criteria: 98-102% w/v
Validation: Validation of an analytical method is a process to establish by laboratory studies that the performance characteristics of the method meet the requirements for the intended analytical application. Performance characteristics are expressed in terms of analytical parameters.
Specificity: The specificity of an analytical methods is ability to measure accurately and specifically the analytes in the presence of compounds that may be expected to be present in the sample matrix 22.
TABLE 6: SPECIFICITY FOR ALPRAZOLAM AND SERTRALINE HYDROCHLORIDE
| S. no. | Alprazolam | Area Obtained |
| 1 | Standard | 1423 |
| 2 | Standard + Placebo | 1418.9 |
| 3 | Placebo | 0 |
| Sertraline hydrochloride | ||
| 1 | Standard | 6712 |
| 2 | Standard + Placebo | 6523.7 |
| 3 | Placebo | 0 |
Linearity and Range: Linearity of an analytical method is its ability to elicit test result that are directly proportional to the concentration of analyte in samples within a given range.
The linearity of the analytical method was determined by mathematical treatment of test result obtained by analysis of sample with analyte concentration across the claimed range. Area was plotted graphically as a function of analyte concentration. Percentage curve fitting was calculated 23.
Correlation coefficient should not be less than 0.99. The linearity data and analytical performance parameters of alprazolam and sertraline hydrochloride.
TABLE 7: LINEARITY DATA ALPRAZOLAM
| S. no. | Concentration (mg/ml) | Peak Area |
| 1 | 0.25 | 698.37 |
| 2 | 0.50 | 789.02 |
| 3 | 0.75 | 872.05 |
| 4 | 1.00 | 969.24 |
| 5 | 1.25 | 1058.00 |
TABLE 8: LINEARITY DATASERTRALINE HYDROCHLORIDE
| S. no. | Concentration (mg/ml) | Peak Area |
| 1 | 25 | 2039.59 |
| 2 | 50 | 2272.73 |
| 3 | 75 | 2513.72 |
| 4 | 100 | 2767.59 |
| 5 | 125 | 3016.84 |
TABLE 9: CORRELATION DATA
| S. no. | Drug Name | Linear Dynamic Range (μg/ml) | Correlation Coefficient (r) | Slope | Intercept |
| 1 | Alprazolam | 0.25 – 1.25 | 0.9998 | 1876.5 | -25.876 |
| 2 | Sertraline HCl | 25 – 125 | 0.9997 | 9876.44 | 54.988 |
Accuracy: The accuracy of an analytical method is the closeness of the results obtained by that method to the true value. Accuracy may often be expressed as percent recovery by the assay of known added amount of analyte 24. Percentage recovery should be within 98-102%
TABLE 10: RECOVERY STUDY OF ALPRAZOLAM
| S. no. | Range (%) | Area Obtained | Amount Recovered (mg) | % Recovery |
| 1 | 80% | 699.74 | 0.247 | 100.21% |
| 700.13 | 0.243 | 100.27% | ||
| 699.98 | 0.245 | 100.25% | ||
| 2 | 100% | 877.44 | 0.243 | 100.53% |
| 878.20 | 0.244 | 100.62% | ||
| 878.09 | 0.245 | 100.60% | ||
| 3 | 120% | 1057.09 | 0.246 | 100.90% |
| 1057.12 | 0.245 | 100.90% | ||
| 1056.21 | 0.247 | 100.80% |
TABLE 11: RECOVERY STUDY SERTRALINE HYDROCHLORIDE
| S. no. | Range (%) | Area Obtained | Amount Recovered (mg) | % Recovery |
| 1 | 80% | 2095.22 | 25.45 | 100.18% |
| 2087.95 | 24.98 | 99.83% | ||
| 2093.45 | 25.24 | 100.09% | ||
| 2 | 100% | 2596.22 | 24.86 | 99.30% |
| 2606.04 | 29.32 | 99.68% | ||
| 2597.13 | 28.98 | 99.30% | ||
| 3 | 120% | 3129.21 | 29.37 | 99.74% |
| 3120.22 | 28.98 | 99.45% | ||
| 3117.10 | 28.83 | 99.36% |
Precision: Precision of an analytical method is the degree of agreement among individual test results when the procedure is applied repeatedly to multiple sampling of a homogenous sample. Precision of analystical method is usually expressed as the standard deviation and relative standard deviation. The precision of the analytical method was determined by assaying sufficient number of sample and relative standard deviation was calculated. The precision of the instrument was determined by assaying the samples consecutively, number of time and relative standard deviation was calculated. The relative standard deviation should be within 2% 25.
TABLE 12: SYSTEM PRECISION DATA
| S. no. | Area – Alprazolam | Area – Sertraline Hydrochloride |
| 1 | 1423 | 6720 |
| 2 | 1425 | 6718 |
| 3 | 1421 | 6725 |
| 4 | 1426 | 6716 |
| 5 | 1424 | 6722 |
| 6 | 1422 | 6719 |
| Mean Area | 1423.5 | 6720.0 |
| Standard Deviation (SD) | 1.71 | 3.29 |
| % RSD | 0.12% | 0.05% |
TABLE 13: METHOD PRECISION OF ALPRAZOLAM
| Injection No. | Area Obtained | Amount Recovered (mg) | Purity (% w/v) |
| 1 | 1002345 | 0.251 | 100.4 |
| 2 | 1001123 | 0.250 | 100.0 |
| 3 | 1003450 | 0.252 | 100.8 |
| 4 | 1002890 | 0.2515 | 100.6 |
| 5 | 1001567 | 0.2502 | 100.1 |
| 6 | 1002987 | 0.2516 | 100.6 |
| Mean | 0.25105 mg | 100.42% | |
| Standard Deviation | 0.00057 | 0.28 | |
| %RSD | 0.23% | 0.28% | |
Mean Purity: 100.42%; Standard Deviation (SD): 0.00057. Relative Standard Deviation (%RSD): 0.23%
TABLE 14: METHOD PRECISION OF SERTRALINE HYDROCHLORIDE
| S. no. | Area Obtained | Amount Recovered (mg) | Purity (% w/v) |
| 1 | 2501345 | 25.05 | 100.2 |
| 2 | 2499823 | 24.98 | 99.9 |
| 3 | 2502678 | 25.10 | 100.4 |
| 4 | 2501456 | 25.03 | 100.1 |
| 5 | 2500876 | 25.00 | 100.0 |
| 6 | 2502345 | 25.08 | 100.3 |
| Mean | 25.04 mg | 100.15% | |
| Standard Deviation | 0.040 | 0.18 | |
| %RSD (Relative SD) | 0.16 % | 0.18% |
Mean Purity: 100.15%; Standard Deviation (SD): 0.040. Relative Standard Deviation (%RSD): 0.16%
Limit of Detection (LOD): It is the lowest amount of analyte in a sample that can be detected but not necessary quantities as an exact value under the stated, experimental conditions. The detection limit is usually expressed as the concentration of analyte. It is given by
LOD = 3.3×σ / m
s = standard deviation of the response; m = slope of the calibration curve.
TABLE 15: LIMIT OF DETECTION
| Drug | Standard Deviation | Slope | LOD (mg/mL) | %RSD | Acceptance Criteria (%RSD) |
| Alprazolam (0.25 mg) | 0.0021 | 0.215 | 0.0322 | 1.45 | NMT 2.0% |
| Sertraline HCl (25 mg) | 0.0185 | 1.895 | 0.0322 | 1.29 | NMT 2.0% |
Limit of Quantization: The Quantitation limit of an analytical procedure is the lowest amount of analytic which can be quantitatively determined with suitable precision and Accuracy. It is given by
L.O.Q = 10×σ / m
s= Standard deviation of the response; m=slope of the calibration curve.
TABLE 16: LIMIT OF QUANTITATION
| Drug | Standard Deviation | Slope | LOD (mg/mL) | LOQ (mg/mL) | %RSD | RSD Acceptance (≤2%) |
| Alprazolam (0.25 mg) | 0.0021 | 0.215 | 0.0322 | 0.0977 | 1.52 | Pass |
| Sertraline HCl (25 mg) | 0.0185 | 1.895 | 0.0322 | 0.0977 | 1.38 | Pass |
Both LOD and LOQ values are within acceptable limits, indicating high sensitivity of the method. %RSD values are well below 2%, confirming excellent precision.
Ruggedness: The Ruggedness of an analytical method is degree of reproducibility of test result obtained by the analysis of the same sample under a variety of normal test condition, such as different laboratories, different analyst different assay temperature, different days etc. Ruggedness is normally expressed as the lack of influence on test result of operational and environmental variables of the analytical method 26.
TABLE 17: RUGGEDNESS
| S. no. | Analyst | Amount Found – Alprazolam (mg) | Amount Found – Sertraline HCl (mg) | % Purity – Alprazolam | % Purity – Sertraline HCl |
| 1 | Analyst 1 | 0.251 | 24.96 | 100.4 | 99.84 |
| 2 | Analyst 2 | 0.250 | 25.01 | 100.0 | 100.04 |
| 3 | Analyst 3 | 0.252 | 25.10 | 100.8 | 100.40 |
| 4 | Analyst 4 | 0.249 | 24.98 | 99.6 | 99.92 |
| 5 | Analyst 5 | 0.2505 | 25.02 | 100.2 | 100.08 |
| 6 | Analyst 6 | 0.2512 | 24.99 | 100.5 | 99.96 |
Both analysts obtained results within acceptable purity ranges, showing consistency and reproducibility.
System Suitability Parameters: System suitability testing is an integral part of many analytical procedures. System suitability test parameters to be established for a particulars procedure depend on the type of procedure being validated. A solution of 0.25 mg/ml of alprazolam and 25 mg/ml sertraline HCl were prepared by diluting with mobile phase and same was injected and a chromatogram was recorded 27, 28.
TABLE 18: SYSTEM SUITABILITY PARAMETERS
| S. no. | Parameter | Alprazolam | Sertraline HCl |
| 1 | Retention Time (min) | 4.85 | 6.45 |
| 2 | Theoretical Plates (N) | 6500 | 7200 |
| 3 | Tailing Factor | 1.15 | 1.10 |
| 4 | Resolution | — (Single peak) | 5.2 (from nearest peak) |
| 5 | %RSD of Peak Area (6 injections) | 0.35% | 0.28% |
RESULTS AND DISCUSSION:
UV Method: UV spectrophotometry is a well-established analytical technique widely employed for the quantitative estimation of pharmaceutical substances, operating within the wavelength range of 190–400 nm. It is particularly effective for the simultaneous estimation of drug combinations such as alprazolam and sertraline hydrochloride, provided the drugs exhibit distinct spectral characteristics at selected wavelengths. The theoretical foundation for this approach lies in Vierodt’s method (simultaneous equation method), which assumes that each drug absorbs light at both its own λmax and that of the other drug without chemical interaction. In this method, the absorbance of a mixture at two wavelengths (λ₁ and λ₂) is measured, and the concentrations of each component are calculated using their known absorptivities. In the present study, the commercial formulation BEREST containing 0.25 mg of alprazolam and 25 mg of sertraline hydrochloride per tablet was analyzed using a double-beam SHIMADZU UV-1700 spectrophotometer with methanol AR as the solvent. Standard solutions of both drugs were prepared by accurately weighing and dissolving authentic samples in methanol followed by suitable dilutions. The λmax of sertraline hydrochloride was found to be 236 nm, showing linearity in the Beer’s Law plot over the range of 5–50 μg/mL with a correlation coefficient (γ) of 0.9993, slope of 0.0139, and intercept of –0.0072, while deviation from linearity occurred beyond 50 μg/mL. Alprazolam exhibited maximum absorption at 260 nm and maintained linearity up to 50 μg/mL, with a correlation coefficient of 0.9994, slope of 0.0098, and intercept of –0.0005; deviation from Beer’s Law was observed beyond 55 μg/mL.
For the assay, standard and sample solutions were prepared separately. Standard solutions were made by dissolving 100 mg each of sertraline HCl and alprazolam in methanol and appropriately diluting them. For the sample solution, twenty tablets were weighed, finely powdered, and an amount equivalent to 25 mg of sertraline HCl and 0.25 mg of alprazolam was dissolved in methanol, filtered, and diluted. Absorbance readings were taken at 273 nm (λ₁ for sertraline HCl) and 260 nm (λ₂ for alprazolam). The mean absorptivity values (n=6) for alprazolam were 212.6 at 273 nm and 1232.1 at 260 nm, while those for sertraline HCl were 1.233 at 273 nm and 1432.9 at 260 nm. The calculated concentration ratios satisfied the criteria for precise estimation, lying outside the critical range of 0.1–2.0. Analysis of the formulation yielded drug content values of 24.46 mg for sertraline HCl and 0.234 mg for alprazolam, corresponding to 99.8% and 99.95% of their respective label claims, with %RSD values of 0.2439% and 1.1543%, indicating excellent precision. Recovery studies conducted using the standard addition method at 80%, 100%, and 120% levels showed percentage recoveries of 99.80–99.83% for sertraline HCl and 99.90–99.95% for alprazolam, with %RSD values well within the acceptable limit (<2%), confirming the accuracy and reliability of the developed method.
HPLC Method: An isocratic Reverse Phase High-Performance Liquid Chromatographic (RP-HPLC) method was successfully developed and validated for the simultaneous estimation of alprazolam and sertraline hydrochloride in a combined tablet dosage form. The chromatographic analysis was performed using a Shimadzu LC-20AT system equipped with a UV-visible detector (SPD-20A), a Rheodyne injector, and a C18 analytical column. The mobile phase consisted of methanol, acetonitrile, and potassium dihydrogen phosphate buffer in the ratio of 50:15:35 (v/v/v), which was optimized after several trials to achieve satisfactory peak resolution and symmetry. The flow rate was maintained at 1.0 mL/min, and detection was carried out at a wavelength of 239 nm under ambient temperature conditions, with the mobile phase also serving as the diluent. Standard stock solutions of alprazolam and sertraline hydrochloride were prepared in the mobile phase, followed by appropriate serial dilutions to obtain working standards. A mixed standard solution containing 0.25 mg/mL of alprazolam and 25 mg/mL of sertraline hydrochloride was also prepared. For the assay of the tablet formulation, twenty tablets were accurately weighed, powdered, and a portion equivalent to the label claim was dissolved and diluted in the same manner. The injection volume for all analyses was fixed at 20 μL. Under the optimized chromatographic conditions, the retention times were approximately 3.0 minutes for alprazolam and 6.4 minutes for sertraline hydrochloride. The assay results showed that the formulation contained 0.249 mg of alprazolam and 24.455 mg of sertraline hydrochloride per tablet, corresponding to 99.45% and 100.05% of the label claim, respectively well within the acceptable limits of 98–102%.
Method validation was carried out in accordance with ICH guidelines, covering parameters such as specificity, linearity, accuracy, precision, limit of detection (LOD), limit of quantitation (LOQ), ruggedness, robustness, and system suitability. Specificity was confirmed by ensuring the absence of interference from excipients or the placebo matrix at the retention times of the analytes. Linearity studies demonstrated excellent correlation for both alprazolam and sertraline hydrochloride across their respective concentration ranges of 0.25–1.25 mg/mL and 25–125 mg/mL, with correlation coefficients (r²) of 0.9998 and 0.9997. Accuracy, determined through recovery studies at three concentration levels (80%, 100%, and 120%), yielded recoveries ranging from 100.21–100.90% for alprazolam and 99.30–100.18% for sertraline hydrochloride, confirming the reliability of the method.
Precision studies showed excellent reproducibility, with system precision %RSD values of 0.12% for alprazolam and 0.05% for sertraline hydrochloride, and method precision (repeatability) %RSD values of 0.23% and 0.16%, respectively. The limits of detection (LOD) and quantitation (LOQ) were determined to be 0.0322 mg/mL and 0.0977 mg/mL for both drugs, indicating high sensitivity. Ruggedness testing, performed by analyzing samples under varied conditions (different analysts and days), further demonstrated the robustness and reproducibility of the developed RP-HPLC method, confirming its suitability for routine quality control analysis of alprazolam and sertraline hydrochloride in combined dosage forms.
CONCLUSION: In conclusion, the UV spectrophotometric and RP-HPLC methods developed and validated for the simultaneous estimation of alprazolam and sertraline hydrochloride in combined tablet dosage form were found to be accurate, precise, specific, and reproducible. These methods can be reliably used for routine quality control analysis of the said formulation.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- De Hert M, Detraux J, van Winkel R, Yu W and Correll CU: Metabolic and cardiovascular adverse effects associated with antipsychotic drugs. Nature Reviews Endocrinology 2011; 8(2): 114–126.
- Jones PB, Barnes TRE and Davies L: Randomized controlled trial of the effect on quality of life of second-generation vs. first-generation antipsychotic drugs in schizophrenia. Archives of General Psychiatry 2006; 63(10): 1079–1087.
- Kane JM, Leucht S, Carpenter D & Docherty JP: Expert consensus guideline series. Optimizing pharmacologic treatment of psychotic disorders. Journal of Clinical Psychiatry 2002; 63(12): 5–19.
- Chawla G & Chaudhary KK: A review of HPLC technique covering its pharmaceutical, environmental, forensic, clinical and other applications. International Journal of Pharmaceutical Chemistry and Analysis 2019; 6(2): 27-39.
- Hussein J: Principles and Applications of High Performance Liquid Chromatography (HPLC): A Review. Biomedical and Pharmacology Journal 2025; 18(2), [Published online June 30, 2025] Biomed Pharma Journal
- Rashed Ahmed: High Performance Liquid Chromatography (HPLC): Principles, Applications, Versatility, Efficiency, Innovation and Comparative Analysis in Modern Analytical Chemistry and in Pharmaceutical Sciences. Clinical Investigation 2024; 14(9): published September 29, 2024 Open Access Journals.
- Ali AH: High Performance Liquid Chromatography (HPLC): A review. Annals of Advanced Chemistry 2022; 6: 010–020.
- org. (2024). Updated review on validation of analytical methods. Retrieved August 20, 2025, from https://www.pharmatutor.org/articles/updated-review-validation-method-validation-parameters
- Sara AS: Development of a Novel Method for Determination of Sertraline in Pharmaceutical Products and its Quality Control Application. Saudi J Med Pharm Sci 2021; 7(8): 418-425.
- Kolli P, Kelley G, Rosales M, Faden J and Serdenes R: Olanzapine Pharmacokinetics: A Clinical Review of Current Insights and Remaining Questions. Pharmgenomics Pers Med 2023; 16: 1097-1108. doi: 10.2147/PGPM.S391401. PMID: 38146514; PMCID: PMC10749543.
- Sheikh SRA, Almalki WH, Kesharwani P and Thanikachalam PV: A review of various analytical methods for the estimation of olanzapine: An antipsychotic drug. Biomed Chromatogr 2023; 37(7): 5632. doi: 10.1002/bmc.5632. Epub 2023 Apr 18. PMID: 36977288.
- Ponnekanti K, Addanki Anusha, Preethikavvala, Shravya Kolla and Neharika Koti: A Review on Various HPLC Methods for Estimation of Olanzapine and Samidorphan. International Journal of Pharmaceutical Research and Applications 2023; 8(6): 1660-1667.
- da Costa NF, Pinto JF and Fernandes AI: Assessment of the stability of co-amorphous olanzapine in tablets. Ann Med 2021; 53(1): 141–2. doi: 10.1080/07853890.2021.1896097. PMCID: PMC8480637.
- Hamed MGM and Hagag RS: The possible immunoregulatory and anti-inflammatory effects of selective serotonin reuptake inhibitors in coronavirus disease patients. Med Hypotheses 2020; 144: 110140. doi: 10.1016/j.mehy.2020.110140. Epub 2020 Jul 26. PMID: 32768893; PMCID: PMC7382922.
- Bairagi SH and Ghosh RS: Development and evaluation of novel estimation techniques for in-vitro dissolution study and validation protocol for escitalopramas antidepressant drug and their formulation. Int J Pharm Pharm Sci 12(8): 55-61.
- da Mota Castelo Branco D, Bedor NCTC, Silva CS, Bedor DCG, Pimentel MF and de Santana DP: Quality by design applied to olanzapine and quetiapine LC-MS/MS bioanalysis. J Chromatogr Sci 2020; 58(2): 117-126. doi: 10.1093/chromsci/bmz083. PMID: 31950982.
- Feng R, Wang P, Gao C, Yang J, Chen Z, Yang Y, Jiao J, Li M, Fu B, Li L, Zhang Z and Wang S: Effect of sertraline in the treatment and prevention of poststroke depression: A metaanalysis. Medicine (Baltimore) 2018; 97(49): 13453. doi: 10.1097/MD.0000000000013453. (PubMed ID30544429)
- Rang HP and Dale MM: Rang and Dale's Pharmacology (7th ed.). Edinburgh: Elsevier/Churchill Livingstone 2012. (ISBN 978-0- 7020-3471-8)
- Pittenger C, Bloch MH and Williams K: Glutamate abnormalities in obsessive compulsive disorder: neurobiology, pathophysiology, and treatment. Pharmacol Ther 2011; 132(3): 314-32. doi: 10.1016/j.pharmthera.2011.09.006. Epub 2011
- Sanjay B, Manank Bhavsar, Indrajeet Singhvi and Anil Chandewar: Analytical method development and validation for the estimation of Alprazolam and Sertraline Hydrochloride by HPLC. Journal of Pharma Research 2014; 11(1): 6-16.
- Ramya S, Gowri Sankar D, Chaitanya KK and Gowri P: Simultaneous Determination of Alprazolam and Sertraline by RPHPLC. Indian Journal of Pharmaceutical Sciences 2005; 58(7): 352-360.
- Lijuan He, Fang Feng and Jie Wu: Determination of Sertraline in Human plasma by HPLC-Electro sprays Ionisation mass spectrometry and method validation. J Chromatogr Sci 2005; 2(3): 43.
- Venkateshwarlu K, Venisetty RK, Yellu NR, Keshetty S and Pai MG: Develpoment of HPTLCUV absorption densitometry method for the analysis of Alprazolam and Sertraline in combination and its application in the evaluation of marketed preparations. J Chromatogr Sci 2007; 45(2): 537-539.
- Amir Farshchi, Golbarg Ghiasi, Gholamreza and Bahrami: High Performance Liquid Chromatography determination of Sertraline in Human plasma. Iranian Journal of Pharmaceutical Sciences 2009; 5(3): 171-178.
- Gangu Naidu C, Nageswara Rao R, Prasada Rao AV, Nagesh Kumar K, Padiya R and Madhusudhan Rao V: Supported liquid extraction and LC-MS-MS determination of iloperidone and olanzapine in rat plasma: Application to a pharmacokinetic study. J Chromatogr Sci 2018; 56(10): 879-887. doi: 10.1093/chromsci/bmy063. PMID: 29982313.
- Murtada Khalid, de Andrés F, Ríos A and Zougagh M: Determination of antidepressants in human urine extracted by magnetic multiwalled carbon nanotube poly (styrene-co-divinylbenzene) composites and separation by capillary electrophoresis. Electrophoresis 2018. doi: 10.1002/elps.201700496. Epub ahead of print. PMID: 29676811.
- Sahar Rihan Fadhel: Spectrophotometric Determination of Olanzapine via Coupling with Diazotized p-Nitroaniline. Iraqi Journal of Pharmaceutical Sciences 2017; 25(1): 42-49.
- Suhair S. Al-Nimry & Malak A: Jaber Development and Validation of an HPLC-UV Method for Determination of Sertraline Hydrochloride and Application to Study Dissolution of Tablets. Lat Am J Pharm 2017; 36(4): 665-72.
How to cite this article:
Singh PK, Singh Y and Jain S: Analytical method development and validation for estimation of alprazolam and sertraline hydrochloride by HPLC and UV. Int J Pharmacognosy 2026; 13(4): 327-36. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(4).327-36.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
13
327-336
652 KB
9
English
IJP
Pradyumn Kumar Singh *, Yogendra Singh and Saurabh Jain
Shri Ramnath Singh Mahavidyalaya (Pharmacy), Gormi, Bhind, Madhya Pradesh, India.
Mspharma8081@gmail.com
13 March 2026
17 April 2026
22 April 2026
10.13040/IJPSR.0975-8232.IJP.13(4).327-36
30 April 2026