ALOPECIA AREATA: CURRENT INSIGHTS INTO IMMUNOPATHOGENESIS, DIAGNOSIS, AND EVOLVING THERAPEUTIC STRATEGIES
HTML Full TextALOPECIA AREATA: CURRENT INSIGHTS INTO IMMUNOPATHOGENESIS, DIAGNOSIS, AND EVOLVING THERAPEUTIC STRATEGIES
Tushar Tapadiya, Deepa S. Mandlik, Ravindra Kulkarni and Vaibhav M. Shinde *
Department of Pharmacognosy, Bharati Vidyapeeth (Deemed to be University), Poona College of Pharmacy, Pune, Maharashtra, India.
ABSTRACT: Alopecia areata (AA) is a chronic autoimmune disorder leading to non-scarring hair loss and affecting millions worldwide across all age groups. Despite extensive research, the precise molecular mechanisms remain incompletely understood, arising from complex interactions among immune dysregulation, genetic susceptibility, and environmental triggers. Emerging evidence also implicates lifestyle factors, psychological stress, gut microbiome imbalance, and drug exposures in disease onset and progression. This review synthesizes current knowledge on the immunopathogenesis of AA, integrating insights from T-cell mediated autoimmunity, genetic variations, and epigenetic regulation. Advances in diagnostic strategies, including trichoscopy, histopathology, and severity assessment tools, are critically evaluated with attention to their clinical applicability and limitations. Importantly, the therapeutic landscape of AA is rapidly evolving: beyond traditional approaches such as corticosteroids and minoxidil, recent progress in biologics, JAK inhibitors, regenerative strategies including stem cell and exosome-based therapies, nanotechnology, and microneedle-assisted delivery systems is reshaping treatment paradigms. By bridging mechanistic understanding with therapeutic innovation, this review highlights unmet clinical needs, emphasizes opportunities for precision and regenerative medicine, and outlines future directions toward achieving durable and patient-centered outcomes in AA.
Keywords: Alopecia areata, Autoimmune Disease, Alopecia Pathophysiology, Targeted Therapy, Alopecia Treatment, Hair Loss
INTRODUCTION: Alopecia areata (AA) is an autoimmune disorder in which the immune system mistakenly attacks hair follicles, leading to non-scarring, patchy hair loss on the scalp and other hair-covered areas. AA shows a wide clinical spectrum from small, clearly demarcated bald patches to widespread hair loss that may involve the entire scalp or body 1.
This long-term disorder primarily affects hair follicles and nails, and in certain instances, it may also influence the retinal pigment epithelium 2. AA can appear in many forms, like complete scalp hair loss (alopecia totalis) or total body hair loss (alopecia universalis) 3.
This condition affects people irrespective of their age, gender, or origin. On a worldwide scale, there are approximately 20.9 cases in every 100,000 person-years, with an estimated lifetime risk of about 2% among the general population, with estimates globally ranging from 0.57% to 3.8% 4. Approximately 20% of cases are seen in children, while nearly 60% of adults experience their initial hair loss patches before age 30 5. Multiple research studies have established that AA is associated not only with autoimmune diseases but also it is also linked with several chronic diseases, including thyroiditis and vitiligo. Furthermore, AA may also have connections to chromosomal disorders, notably Down syndrome, as well as polyglandular autoimmune syndrome type 1 6. This condition is closely influenced by immune system activity, with CD8+ T lymphocytes specifically attacking hair follicles 7. Environmental and psychological stresses like tobacco use, poor sleep quality, obesity, and dietary elements like fatty acids and gluten may play a role in triggering AA 8. AA is mostly diagnosed by its common clinical signs, and examinations like trichoscopy and histopathology are useful in patients with AA 9. Treatments for AA may include corticosteroids injected, applied topically, or taken orally as well as minoxidil and specific light-based treatments. However, these approaches may bring about side effects such as unwanted hair growth in certain areas or skin irritation, and there is no guarantee that the hair that regrows during treatment will remain after the therapy is stopped 10. Although this condition is not deadly, it often carries a significant emotional impact. Hair plays a crucial role in human identity, particularly for women, and its loss can profoundly influence self-esteem and body image 11.
This review identifies key gaps in both academic and clinical research surrounding AA by exploring its pathogenesis, diagnostic techniques, and treatment approaches. It first examines the immunologic mechanisms behind AA, along with environmental and epigenetic factors influencing the condition. Next, it outlines current diagnostic tools and severity assessment methods. Additionally, it highlights emerging therapies, including small-molecules, Biologics, and combination therapies. Finally, the review discusses upcoming research directions, accenting the importance of investigating AA pathogenesis and advancing targeted therapies to assist clinicians and dermatologists in developing clear management guidelines.
Pathophysiology: The development of AA is a complex process heavily influenced by the human immune system. A key factor is the involvement of Type IV hypersensitivity reactions driven by T cells which contribute to the disease. Beyond autoimmunity, genetic predisposition and environmental influences, such as stress and viral infections, significantly influence the development and advancement of AA 12.
Immunity: Hair follicles possess a form of specialized immunity, but this is limited to the anagen phase 3. In AA, the immune system attacks hair follicles through two separate pathways. First, it directly attacks the follicles, an action triggered by autoantigens produced by melanocytes within the follicles. Second, it weakens the follicle’s natural protection by increasing MHC class I levels, which makes it easier for CD8+ T cells to spot and attack the hair follicle when autoantigens are present. Upon stimulation, these cells release perforin and granzymes, which work together to damage follicular keratinocytes and Perforin creates pores in target cell membranes, while granzymes trigger apoptosis. This process leads to hair follicle destruction and subsequent hair loss. Some CD8+ T cells later transition into memory T cells, persisting within the body and potentially causing repeated instances of AA 3.
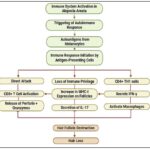
The Th1 and Th17 arms of CD4+ T cells modulate immune activity via IFN-γ and IL-17 release, contributing to inflammatory signalling 13. Macrophage activation by IFN-γ production boosts MHC expression in dendritic cells, aiding T-cell antigen recognition. Likewise, IL-17 from Th17 cells extends and amplifies inflammation. Additionally, AA sees an increase in IL-2 and IL-6 levels, with IL-2 promoting T-cell proliferation and IL-6 playing a key role in B-cell activation and overall inflammatory processes 14. TNF-α is an essential cytokine that stimulates immune cell activation and migration, heightens inflammation, and exacerbates hair follicle damage. The cooperation among cytokines sustains the inflammationin AA. IFN-γ strengthens TNF-α’s activity, further aggravating inflammation. Such disturbances affect the hair cycle by delaying the normal switch from the growing to the resting phase, resulting in early hair loss15.In AA, regulatory T-cell dysfunction diminishes their ability to control effector T-cell activity, leading to immune dysregulation and an excessive autoimmune response 13. Histological analysis of the affected tissue revealed a dense infiltration belonging to CD8+ T lymphocytes, compromising the structural integrity of hair follicles 14. A visual summary of the immune dysregulation and follicular destruction in AA is illustrated in Fig. 1.
FIG. 1: MECHANISTIC OVERVIEW OF IMMUNE-MEDIATED HAIR FOLLICLE DESTRUCTION IN AA
Genetic Factors: AA has a recognized genetic component in its pathophysiology, with incidence rates being notably higher among individuals with a family history of the condition 16. Genome-wide association studies (GWAS) have identified several genetic loci associated with susceptibility. A total of 14 genetic risk loci were found, with HLA-DR emerging as a key etiological factor, showing the strongest correlation with MHC 17. Genome-Wide Association Studies (GWAS) have identified genomic regions linked to AA, including NKG2D, which is connected with natural killer (NK) cell receptors,ULBP3/6, and MICA, which are involved in activating ligands, and CTLA4, which serves as a regulator of T-cell function 18.
MicroRNAs contribute to AA diagnosis by their expression patterns in affected tissues. Recent studies indicate that miR-101 could serve as a potential indicator of disease presence 19. In addition, lesional skin in AA patients exhibits a significant increase in the expression levels of miRNA-155, 146a, and 203. Furthermore, individuals presenting with numerous lesions demonstrate considerably higher miRNA-146a levels than those with just a single lesion 20. A study analysed the association between interleukin-related genes and AA, identifying IL2RA, IL17A, IL12B, and IL23R as key contributors. The study examined various single-nucleotide polymorphisms (SNPs) and found that rs3118470 within IL2RA posed a significant risk factor under dominant and homozygous models. However, SNPs associated with IL17A, IL12B, and IL23R did not exhibit statistically meaningful correlations. The findings suggest that genetic susceptibility to AA may differ across populations, highlighting the importance of further research tailored to various ethnic groups to enhance the accuracy of meta-analyses and improve genetic insights 21.
A whole-exome sequencing and genome-based collapsing study examined 849 AA cases against 15,640 controls. The analysis pinpointed KRT82, a gene responsible for encoding hair-specific type II keratin, as the sole genome-wide significant gene enriched in AA cases, with its expression notably diminished within the follicular and skin of affected individuals. Another AA-related gene, KRTCAP3, is linked to hair keratin, whereas DECR2 plays a role in beta-oxidation and is also strongly associated with the disease 22. Next-generation sequencing has identified CCHCR1 within an AA-linked region of the HLA class I gene cluster. The protein it encodes plays a role in hair shaft structure, suggesting that disrupted keratinization may contribute to AA pathogenesis 23.
JAK-STAT Signalling Pathway: In recent studies, the JAK–STAT pathway has gained attention for its role in AA. This pathway controls immune signalling through cytokine receptors and the JAK-STAT protein family. JAK1 and JAK3 are mainly linked to inflammatory and autoimmune responses, while JAK2 is more involved in red blood cell and platelet production 24. The STAT protein family acts as key transcription factors in the JAK–STAT pathway, binding to DNA and transmitting signals downstream of JAKs. This group includes STAT1 to STAT6, each involved in processes like immune balance, cell growth, and tissue development25. Cytokines such as IFN-γ, IL-2, and IL-15 bind to their receptors on immune or follicular cells, activating JAK1, JAK3, and TYK2 kinases. These kinases then phosphorylate STAT1 and STAT3, which translocate into the nucleus and initiate pro-inflammatory gene expression. This cascade results in CD8⁺ T cell activation, driving follicular inflammation and leading to hair follicle destruction 26.
Environmental Factors:
Smoking and Alcohol: Research has confirmed that smoking elevates the risk of developing AA, with long-term smokers and those who smoke more than five cigarettes daily showing a significantly higher likelihood. Some studies indicate a slight but statistically relevant connection between smoking and the development of AA. The harmful effects of smoking, including free radical formation and elevated pro-inflammatory cytokines, may lead to hair follicle damage and IP. At the same time, immune mechanisms, including Th1, Th2, and Th17, play a critical role in AA's development 27. Alcohol has been shown to elevate the levels of inflammatory cytokines, with TGF-α, IFN-γ, and IL-6, that may worsen inflammatory diseases such as psoriasis and possibly AA. Researchers propose that alcohol consumption may intensify TH17-driven inflammation, potentially triggering AA. Furthermore, moderate alcohol intake can disrupt the body's stress response by altering ACTH and cortisol levels, which may contribute to AA progression 28.
Microbial Imbalance: Research suggests that people with AA tend to have higher levels of Cutibacterium acnes on the scalp, while Staphylococcus epidermidis and S. aureus are present in lower concentrations. This altered microbial composition may contribute to inflammation and autoimmune responses associated with AA 29. A study found that people with severe AA had much lower levels of S. caprae and a sharp rise in the Cutibacterium/S. caprae ratio, suggesting a possible link between bacterial imbalance and disease severity. While the exact role of C. acnes in AA remains uncertain, its presence could be linked to disease progression 30. Another study explored gut microbiota differences between AA universalis patients and healthy individuals. Results showed that a 25% elevation in Parabacteroides distasonis and the Clostridialesvadin BB60 group showed around a 9–11% higher likelihood of developing AU, implying that these bacterial changes could act as predictive markers for the disorder 31.
Role of Psychological Stress in Immune Dysregulation: Emotional stress is widely recognized as a significant factor in the development and progression of AA, with approximately 23% of affected individuals experiencing a major psychological stressor or distressing life events before symptoms initiate or worsen. Stress in AA activates the HPA axis, leading to the release of neuropeptides such as CRH and substance P. These messengers disrupt the hair follicle’s immune privilege, making it easier for the immune system to target it 32. Hair follicle cells accumulate intracellular reactive oxygen species (ROS) under environmental stress and insufficient antioxidant defences, promoting the upregulation of stress-related ligands like MICA. Natural killer cells detect such molecules and respond by releasing IFN-γ 33. Stress triggers both autoimmune responses and programmed cell death in hair follicles. Corticotropin-releasing hormone and substance Ppromote keratinocyte apoptosis through pathways involving tumour necrosis factor-alpha (TNF-α), while simultaneously causing mast cells to release their contents, which leads to neurogenic inflammation. This inflammation speeds up the transition of hair follicles to the catagen stage, leading to premature shedding, causing early hair shedding. Furthermore, substance P worsens the inhibition of hair growth by decreasing activation of the TrkA receptor and increasing signals that promote apoptosis, thereby connecting stress with the development of telogen effluvium and progression of AA 34. A comprehensive Systematic review of 73 studies involving approximately 414,319 participants highlights a significant association between AA and psychological distress. Findings reveal that people with AA are more prone to emotional challenges such as anxiety and depression, often triggered by prior stressful experiences. Many report that the condition negatively affects their quality of life, especially in terms of social engagement and self-esteem. Over time, a significant number may also experience psychiatric issues linked to the psychological burden of the disease 35.
Diet and Obesity: The Mediterranean diet, known for its high levels of omega-3 fatty acids and antioxidants, has been shown to lower systemic inflammation and enhance recovery in conditions such as AA. Vitamin D plays a significant role in immune regulation by inhibiting Th17 cell proliferation, reducing Th1 cell presence, and strengthening T-reg cell function. As an essential immunomodulator, A deficiency in vitamin D has been linked to a higher risk of autoimmune disorders 36. Studies have shown that individuals with AA often have reduced vitamin D levels, which tend to decrease further with increasing disease severity. Recent findings indicate that link between vitamin D deficiency and high CRP, with a greater chance of progression to alopecia universalis 37.
Obesity promotes systemic inflammation, possibly through pathways mediated by interleukin-17, which enhance the probability of Perifollicular inflammation and disrupt immune privilege, ultimately causing hair loss. Research has demonstrated a strong link between high BMI and more severe AA, suggesting that obesity may accelerate disease progression and reduce treatment effectiveness 38. Patients with AA were found to have lower serum adiponectin levels compared to healthy controls, a hormone involved in fat metabolism. Additionally, A reduction in adiponectin levels correlated with increased disease severity, suggesting that adiponectin could representa potential indicator for assessing AA progression in obese individuals 39.
Sleep: Patients with AA tend to have worsened sleep quality, often linked to greater anxiety, emotional distress, and reduced overall well-being. Additionally, AA is closely connected to sleep disorders, as those affected are more likely to develop obstructive sleep apnea and non-apnea insomnia. At the same time, these conditions may also contribute to AA progression 40. Poor sleep quality, assessed through metrics like the Pittsburgh Sleep Quality Index, correlates with higher stress levels and worsening AA symptoms. Inadequate sleep interferes with immune function, promoting inflammation and autoimmune activity. Although the exact pathways are not fully understood, conflicts in the HPA axis, a key stress regulator, appear to contribute 41.
Virus and Immunization: The association between viral illnesses and AA has received increased attention in recent years, particularly in the context of the COVID-19 pandemic. Reports from systematic evaluations have highlighted cases of AA appearing after vaccination, especially following administration of mRNA-based platforms such as those developed by Pfizer and Moderna. Both the viral infection and the immunological response to vaccination can stimulate the secretion of pro-inflammatory mediators like IFN-γ, which is instrumental in damaging hair follicles and disrupting their immune privilege. The elevated levels of IFN-γ and related cytokines post-COVID-19 infection or immunization may accelerate AA progression in individuals with underlying susceptibility 42. AA has also been linked to infections caused by HPV, EBV, HIV, and hepatitis B and C. Additionally, research has documented cases of AA emerging following hepatitis B immunization 43.
Drugs Triggering AA: The data from the FDA’s adverse event reports suggest that monoclonal antibodies, especially adalimumab and dupilumab, are commonly linked to cases of AA 44. A variety of medications, including immunosuppressants (like sulfasalazine), enzyme inducers (such as rifampicin and phenobarbital), retinoids, tyrosine kinase inhibitors, and certain proton pump inhibitors like lansoprazole, have been implicated in the possible induction of AA 45. A documented case of AU developed following treatment with sacubitril/azilsartan, an angiotensin receptor-neprilysin inhibitor prescribed for cardiovascular conditions. The findings indicate that certain cardiovascular therapies might induce alopecia through immune system alterations 46. Another case documented AU following treatment with the B-cell targeting agent rituximab, reinforcing those immunomodulatory drugs, particularly those designed to target specific immune cells, can trigger alopecia in susceptible individuals 47. Chemotherapeutic agents like Docetaxel and capecitabine are also associated with drug-induced alopecia 48. These multifactorial contributors interact with immune mechanisms to initiate and sustain follicular damage, as illustrated in Fig. 2.
FIG. 2: VISUAL SUMMARY OF THE MULTIFACTORIAL TRIGGERS CONTRIBUTING TO THE PATHOGENESIS OF AA
Diagnostic Framework for AA: AA is mostly diagnosed according to clinical findings, supported by additional tools like the hair pull test, dermoscopic evaluation, laboratory investigations, and histological analysis of skin biopsies. However, in the majority of cases, advanced diagnostic techniques are not required.
Clinical Manifestations of AA: In AA, the skin remains unchanged, but affected individuals experience one or more well-defined hair loss patches, typically round or oval, across the scalp; these patches usually range from 1 to 2 centimetres in diameter, though their size may differ. Hair loss may also extend beyond the scalp, affecting periocular hair such as the brows and lashes 49. Sites involved are typically devoid of redness, flaking, or permanent skin damage. The margins of these patches often contain fragile hairs that shed easily, some of which may be broken. The proximal ends of these hairs tend to be thinner and show reduced pigmentation in their lower segments. As AA progresses, patches may enlarge, merge into irregular formations, and potentially develop into AT or AU 50.
Nail involvement is a common issue in AA, affecting around 6–10% of patients as a recognized complication 51. Several nail abnormalities are also seen in AA patients, most notably small punctate depressions (nail pitting) and a coarse, sandpaper-like surface texture. Nail pitting tends to be more common, whereas rough nails are less frequently documented 52. Furthermore, AA patients may exhibit atopic tendencies, autoimmune thyroid disease, or concurrent vitiligo, along with an increased risk of conditions such as diabetes and Down syndrome 53.
Adjunctive Diagnosis of AA:
Hair Pull Test Evaluation: The hair pull test serves as a straightforward and dependable method for identifying AA. Patients are typically advised to avoid washing their hair for 3 to 5 days before the procedure. During the test, a small cluster of approximately 50 to 60 hairs is gently tugged from the scalp using the thumb and forefinger. If more than six hairs are dislodged, the test is deemed positive, indicating a potential diagnosis of patchy AA 54.
Dermoscopy: Dermoscopy is a valuable non-invasive technique used for diagnosing, identifying, and assessing AA. This is marked by distinct features, including “exclamation mark” hairs, where the mid-section narrows while the ends thicken, black dots, and follicular opening irregularities within hair-loss regions. When dermoscopy identifies patterns such as yellow dots, black dots, broken or dystrophic hairs, conical hairs, exclamation point-shaped strands, or short vellus hairs, AA can be diagnosed. Moreover, dermoscopy is useful in differentiating other forms of scalp hair reduction 55.
Histopathological Analysis: Histopathological analysis plays a key role when diagnosing AA is difficult. It typically shows inflammatory infiltration around the hair bulb, mainly composed of lymphocytes, with some eosinophils and mast cells. Moreover, follicular reduction and dystrophic anagen follicles are evident, particularly in patients undergoing AA progression or recovery. Recognizing these structural follicular features helps establish the diagnosis 56.
Genetic Tests: As genomic research progresses, scientists are exploring the use of genetic testing to support AA diagnosis. Certain genes, such as HLA, CTLA-4, and ULBP1, are linked with immune regulation, hair follicle health, and inflammatory responses, indicating their relevance to AA 57. Examining genetic variations could improve the precision of AA diagnosis and assist in predicting patient responses to treatment. Despite advancements, genetic testing has not yet become a standard part of clinical evaluation. Still, it enhances traditional diagnostic approaches by offering valuable insights into AA's genetic predisposition and underlying etiology 58.
Evaluation of AA:
SALT Scores: The Severity of Alopecia Tool (SALT) is a vital clinical metric for assessing the extent of hair loss and tracking disease progression in individuals with AA. The SALT scoring method categorizes the scalp into four regions, each with a defined area percentage 59. It assesses severity by estimating the extent of hair loss in these regions and calculating a cumulative score. Below is a summary of AA severity staging based on the percentage of hair loss and associated scalp region involvement, as shown in Table 1.
TABLE 1: SALT SCALP DISTRIBUTION AND AA SEVERITY
| Scalp Region | Percentage of Total Scalp Area | AA Severity Stage | Percentage of Scalp Hair Loss |
| Top | 40% | S0 | No hair loss |
| Back | 24% | S1 | Less than 25% |
| Left Side | 18% | S2 | 25–49% |
| Right Side | 18% | S3 | 50–74% |
| — | — | S4 | 75–99% |
| — | — | S5 | Complete hair loss |
Under this classification, AA is considered mild when it involves less than 25% of the scalp, intermediate when the extent falls between 25% and 49%, and pronounced when hair loss surpasses 50% 60. The SALT scoring system now incorporates body hair loss (B) and nail involvement (N) assessments to improve diagnostic accuracy. Body hair is categorized as B0 (no loss), B1 (partial loss), and B2 (total loss), while nail involvement is classified as N0 (no changes), N1 (partial changes), and N1a (abnormalities in all 20 nails). These additional classifications improve the assessment process, providing a more comprehensive understanding of the patient’s condition to aid in more effective treatment planning 61.
Other Evaluation Tools: The SALT score is a widely adopted clinical instrument for assessing the severity of AA. In addition to SALT, various other evaluation tools are employed to provide a more comprehensive assessment. Assessment measures include several validated indices that evaluate disease severity, patch visibility, and quality-of-life outcomes in AA. Offer further insights into the disease's impact 62. However, assessment of AA involves considering various dimensions, such as the scope of hair loss, its impact on daily life, treatment progress, disease status, emotional effects, and necessary interventions. By incorporating these aspects, healthcare providers can develop a comprehensive understanding of the patient’s condition, ultimately enabling more targeted and effective treatment strategies.
Treatment Modalities for AA: Hair re-growth is possible in AA, with patchy cases often resolving on their own. Still, recurrence is frequent, and available treatments vary according to the condition’s clinical characteristics. While these therapeutic options help control hair loss and encourage re-growth, they do not address the root cause, and there is no certainty of lasting recovery, as relapses can happen unexpectedly 63. The prolonged persistence of AA increases the likelihood of disease progression, with up to 20% of cases advancing to complete scalp hair loss 64.
Traditional Treatments: Traditional treatment strategies for AA primarily rely on pharmacological methods that regulate immune activity and encourage hair growth. Therapeutic options such as intralesional corticosteroids, systemic and topical steroid preparations, and contact immunotherapy are frequently used, though clinical responses can be highly variable. Furthermore, the potential side effects and risks associated with these treatments can limit their clinical utility. Consequently, while conventional approaches may provide relief, they come with certain drawbacks.
Topical Corticosteroids: The approach involves suppressing local inflammation via topical agents to enhance follicular activity. In early stages, topical steroids effectively address localized bald spots, with their skin absorption enhanced by wrapping the treated area. Class 3 and 4 glucocorticosteroids are commonly used in topical therapy due to their potent intracellular effects. Their action involves genomic and non-genomic processes, with cortisol binding to the glucocorticoid receptor (GR), a widely present cytoplasmic receptor. Once bound, the cortisol-GR complex moves into the nucleus and regulates immune responses by suppressing inflammation and promoting anti-inflammatory cytokine expression through interaction with glucocorticoid responsive elements (GRE). Lower cortisol levels further influence immune regulation by repressing NF-kappa B activity. NF-kappa B is typically activated by IL-1 alongside TNF, playing a crucial role in chronic inflammation 65.
The recurrence rate for topical therapy ranges between 37% and 63%, indicating variability in treatment effectiveness65. While topical treatments are generally less effective for AT and AU, they can still be considered for patients who do not respond to other therapies, particularly when applied under occlusion to enhance absorption 66.
Topical Minoxidil: A popular topical solution, minoxidil helps stimulate hair growth, though it requires consistent application over an extended period to produce noticeable effects. However, studies indicate that its effectiveness in treating AA is limited 67.
This treatment does not target immune-related issues at the local level but instead improves circulation at the hair follicle by promoting angiogenesis, widening blood vessels, stimulating cell proliferation, and regulating potassium channels 68. Therapy has shown effectiveness for AGA in both women and men, but its impact on AA remains uncertain 69. Furthermore, skin sensitivity may occur as a side effect of this drug. Due to its vasodilation properties, caution is advised for patients with heart conditions. Common adverse reactions include mild scalp irritation, often accompanied by itching 70.
Anthralin: Anthralin has been utilized as a therapeutic option for AA, including in paediatric cases 71. The standard approach for anthralin therapy in AA involves short-contact treatment using 0.5–1% anthralin, with application durations varying from a few minutes to overnight 72. The precise mechanism of anthralin in AA remains uncertain. However, researchers suggest that its anti-inflammatory effect may result from the free radicals it produces, which help eliminate infiltrated lymphocytes 73. Anthralin can lead to skin staining and regional lymphadenopathy (LAD), though these effects generally resolve after discontinuation. Patients frequently report itching, burning, or oozing, with some instances involving bullous eruptions. Although these reactions occur frequently, adverse systemic manifestations are rare, rendering anthralin a largely tolerable therapeutic agent 74.
Prostaglandin-Based Topical Agents: Prostaglandins like latanoprost and bimatoprost, commonly used to treat glaucoma, have been associated with eyelash hypertrichosis. As prostaglandin F2-α (PGF2α) analogues, they aid in improving eyelash and eyebrow hypotrichosis and alopecia, likely by prolonging the anagen phase of hair follicles 75. Research indicates that nightly application of 0.005% latanoprost eye formulation to the lid borders resulted in moderate to complete eyelash re-growth for 45% of individuals with eyelash AA, without reported adverse effects 76. In a clinical study, AA patients treated with bimatoprost formulation (0.03%) showed notable hair regrowth, with results surpassing those of mometasone furoate cream (0.1%) 77. Topical latanoprost has proven effective for treating localized patchy AA as a monotherapy or alongside other topical treatments. Additionally, prostaglandin analogues demonstrate a strong safety profile in paediatric and adult AA patients 78.
Contact Immunotherapy: In contact immunotherapy, topical sensitizers such as DPCP and SADBE are used to elicit allergic dermatitis at bald patches, aiming to trigger follicular regeneration 79. When more than 50% of the scalp is affected, but AT is not present, CI is often the preferred treatment due to its minimal side effects 80. To begin contact immunotherapy, patients undergo initial sensitization using a specific agent. Once sensitization is established, the treatment is extended to a broader area, with incremental increases in drug concentration to trigger a manageable allergic reaction. After reaching this stage, the therapy is maintained through weekly sessions 81. Although the exact process behind contact immunotherapy is still uncertain, researchers suggest that hair regrowth may be linked to a shift in inflammatory cells surrounding hair follicles, which relocate to the dermatitis site82.Research shows that contact immunotherapy leads to an average hair regrowth rate of 65.5%, with higher success in patchy AA (74.6%) than in AT/AU (54.5%). Complete regrowth occurs in 32.3% of cases, but relapse rates reach 38.3% during treatment and 49.0% after stopping therapy 83. This therapy is contraindicated for pregnant individuals and those with a history of atopic eczema 84. Potential side effects encompass cervical and occipital lymphadenopathy, disseminated or generalized eczema, along with hypo- or hyperpigmentation 85.
Phototherapy: Phototherapy for AA includes ultraviolet light therapy, ultraviolet B (UVB) laser excimer, and psoralen plus ultraviolet A (PUVA) treatment. UVB therapy at a 308 nm wavelength is more effective than polychromatic narrowband UVB in stimulating T-cell apoptosis in vitro. By inducing apoptosis at the epidermal and dermo epidermal levels, it suppresses T-lymphocyte expansion and downregulates cytokine release 86. UVB therapy has shown a 50% hair regrowth rate, along with a measurable increase in hair diameter 87.
UVA therapy, in contrast, penetrates deeper into the dermis, contributing to tanning but being less absorbed by the epidermis. To enhance the effectiveness of UVA treatment, psoralens compounds that increase skin sensitivity to the light, can be administered either orally (0.1% 8-methoxypsoralen)before UV exposure or deliver topically in an attenuated solution (0.001% 8-methoxypsoralen) 20 minutes before exposure, a method known as PUVA Turban, which helps bypass systemic effects 88. UVA therapy has demonstrated an efficacy rate of 84% in promoting hair regrowth in AA patients. Phototherapy offers promising results, but it is not without risks. Prolonged UV exposure may lead to photoaging, keratotic changes, and a higher incidence of skin cancers 89.
Systemic Therapeutic Approaches:
Systemic Corticosteroids: Oral corticosteroids effectively help control rapidly advancing and widespread AA. However, treatment discontinuation often leads to relapse, and long-term use is associated with significant systemic side effects. A systematic review from 2016 evaluated pulsed corticoid therapy (PCT), a short-term corticosteroid treatment given for a few days each month. Covering 41 studies primarily on intravenous administration, the review found that full recovery was uncommon, but those who responded had a relatively low relapse rate of 17%. As a result, PCT may be a useful option for patients with specific positive prognostic indicators, including multifocal AA, a first occurrence of AA, or cases lasting under two years 90. Combining systemic glucocorticoids with minoxidil therapy has been found to help lower relapse rates, improving treatment outcomes 91.
Methotrexate: Methotrexate has been suggested as a potential treatment for AA. It is recognized for its effectiveness in managing various inflammatory and autoimmune disorders 92. The precise mechanism by which methotrexate manage AA remains unclear. However, evidence suggests that it works by inhibiting the enzyme dihydrofolate reductase, leading to increased adenosine levels and its release into the extracellular space. This process suppresses the accumulation of white blood cells and regulates the activities of monocytes, macrophages, and T cells. Additionally, methotrexate is associated with a reduction in the synthesis of TNF-alpha and interferon-gamma 93. Methotrexate commonly causes digestive-related side effects, such as nausea, vomiting, ulcers in the mouth, and reduced appetite. However, its most severe risk is potential liver damage 94.
Cyclosporine: Cyclosporine functions by inhibiting calcineurin phosphatase, which in turn suppresses the expression of interleukin-2, a key cytokine that contributes to the activation of T-cell-mediated immune responses. While cyclosporine can be administered as a single-agent therapy, its effectiveness is notably enhanced when used in combination with corticosteroids.
Studies report a treatment success rate of 69% for the combination approach, compared to 57% when used alone. Moreover, the likelihood of disease recurrence is considerably lower with combination therapy (36%) than with cyclosporine monotherapy, which shows a relapse rate of 74% 95. Common adverse reactions to the treatment involve digestive discomfort and an increased risk of high blood pressure 96. Due to the limited research, the therapeutic value of cyclosporine in AA remains uncertain, with no strong clinical evidence to confirm or dismiss its efficacy.
Phosphodiesterase Inhibitors: Apremilast, a phosphodiesterase 4 (PDE4) inhibitor, is FDA-approved for use in the therapeutic management of psoriatic arthritis and extensive plaque psoriasis. Emerging clinical evidence supports its potential in managing AA. A retrospective analysis reported substantial hair regrowth in patients with chronic AA who did not respond to conventional treatment options. These individuals exhibited favourable results after receiving oral apremilast therapy 97.
Apremilast functions by increasing intracellular cAMP levels, which boosts IL-10 expression while reducing critical proinflammatory mediators, such as TNF-α, IFN-γ, and IL-17, among others 98. Apremilast is generally associated with gastrointestinal symptoms, mild infections of the upper respiratory tract, and occasional headaches 99. While initial research suggests apremilast may be beneficial for AA, more extensive clinical trials are required to establish its effectiveness with greater certainty.
Biological Therapies:
Inhibitors of Th17-Driven Inflammation: As a human monoclonal antibody, secukinumab is designed to selectively inhibit IL-17A, thereby disrupting the Th17 inflammatory signalling pathway to modulate immune responses. Research indicates that secukinumab led to hair regeneration in individuals affected by both psoriasis and total AA. Yet, once the medication was stopped, hair loss recurred, suggesting that secukinumab could be a promising therapy for AA through its effects on Th17 cytokines 100. Although secukinumab shows theoretical promise in treating AA, its effectiveness has yet to be definitively established.
Since IL-17 is integral to the development of AA, Th17 inhibitors like ixekizumab and brodalumab are also considered promising therapeutic options for managing the condition 101.
Th2 Inflammatory Signalling Pathway Inhibitors: AA is often linked to atopic dermatitis (AD), which is predominantly Th2-skewed. Both conditions share a similar genetic foundation 102. Additionally, AA exhibits phenotypic similarities with atopic dermatitis (AD), including symptoms like pruritus, increased IgE levels, filaggrin mutations, and molecular activity involving Th2, IL-23, and Th1 activation 103.
Dupilumab specifically inhibits the Th2 inflammatory signalling pathway. By attaching to the IL-4 receptor's α-subunit, it inhibits IL-4 and IL-13 signalling, thereby mitigating Th2-driven inflammation and modulating cytokine activity 104. Clinical findings suggest that dupilumab may mitigate the progression of AA in individuals with preexisting atopic dermatitis while simultaneously addressing their AD symptoms 105.
IL-12/IL-23 Inhibitors: Ustekinumab is a fully human IgG1 monoclonal antibody that targets the p40 subunit common to IL-12 and IL-23, thereby modulating Th1 and Th17 cell differentiation 106. Research indicates that ustekinumab therapy resulted in noticeable hair regrowth among patients. The treatment was well tolerated, demonstrating a safety profile comparable to that seen in adults. Common adverse effects observed included respiratory infections, headaches, and localized injection site reactions 107.
Tildrakizumab is a humanized monoclonal antibody targeting the IL-23 pathway by selectively preventing the interaction between its p19 subunit and its receptor, thereby suppressing the secretion of inflammatory mediators 108. A clinical pilot study examined the effects of tildrakizumab on nine adults with AA, including seven cases of AT or AU. Results showed that two participants experienced a modest reduction in SALT scores (11–18 points), whereas the remaining individuals exhibited no noticeable therapeutic response 109. While this study does not offer definitive proof, it indicates that tildrakizumab may hold potential as a therapeutic strategy for AA.
Low‑Dose IL‑2: IL-2 is a crucial cytokine that regulates Treg homeostasis, significantly contributing to immune balance. A reduction in Treg levels may disrupt hair follicular immune privilege (IP), potentially involved in AA development 110. An open pilot study demonstrated that subcutaneous administration following low-intensity IL-2 resulted in a notable elevation in Treg levels at affected skin sites in severe AA cases. This increase correlated with partial hair regrowth and marked clinical improvement in AA 111. Although large-scale, multi-center trials are lacking, current encouraging findings emphasize the need for further investigation into the beneficial potential and underlying mode of action of low-intensity IL-2 in the treatment of AA, given the presently limited data.
Platelet-Rich Plasma: Platelet-rich plasma, a biologically active concentrate rich in platelets, is abundant in bioactive mediators. Its effectiveness in treating AA is attributed to its role in enhancing matrix restoration and stimulating hair structure recovery, primarily via reducing MCP-1 expression and elevating TGF-β levels 112. A review primarily focused on studies examining platelet-rich plasma (PRP) in androgenetic alopecia (AGA), along with three studies on AA. The findings suggest PRP has a generally beneficial effect when administering a minimum of three treatment sessions 113. Another Comparative research indicates that PRP therapy outperforms 5% minoxidil in treating AA 114. Several clinical studies have examined PRP’s effectiveness in treating both AGA and AA, highlighting increasing attention and investigation in this area.
Advanced and Adjunctive Therapies:
Combination Drug Therapy: This approach aims to maximize therapeutic benefits in the shortest possible time while minimizing toxicity and drug resistance risks. An optimal drug combination minimizes toxicity and adverse effects while enhancing synergistic interactions between medications to maximize therapeutic benefits and prevent drug resistance. Low-dose prednisone adjunct to methotrexate improves both efficacy and patient tolerance in treating AT and AU. Additionally, this approach offers a more economical option for managing AA 115.
Another combination of latanoprost with betamethasone and minoxidil shows potential as an effective supplementary therapeutic option. A study of 108 AA patients, both male and female, found that topical latanoprost enhances the efficacy of betamethasone and minoxidil while preserving a favorable safety profile. This combination treatment has been shown to effectively manage patchy alopecia without triggering negative side effects 116.
Role of Exosomes: Extracellular vesicles, including exosomes, are tiny vesicular bodies released by cells into various biofluids, with dimensions generally between 30–150 nm. Laden with bioactive molecules, they are gaining attention for their potential therapeutic uses 117. Exosomes, a specialized type of extracellular vesicle (EVs), can efficiently cross cell membranes and exhibit a reduced likelihood of provoking immune reactions 118. Recent studies highlight the promising role of stem cell-derived exosomes in hair follicle repair and regeneration. Specifically, mesenchymal stem cell-derived exosomes enhance DPC activity by promoting their proliferation and movement in mice. This process triggers the release of essential growth factors like IGF-1 and VEGF, aiding hair restoration 119. Furthermore, integrating exosomes with microneedling (MN) has gained attention as a novel approach for enhancing hair growth. In particular, Microneedle systems with PVA tips and HA bases, enriched with chitosan lactate and ADSC-derived exosomes, show promising therapeutic effects 120. Exosomes show promise as a potential therapeutic modality, indicating their future role in advanced treatment approaches.
Nanotechnology: Nanoparticles (NPs) are artificially engineered microscopic structures designed for various applications. Their distinctive composition grants them a large surface area while ensuring excellent biocompatibility and biodegradability 121. Additionally, NPs improve drug stability, solubility, and concentration, extend drug release, and reduce adverse effects on cellular function and metabolism 122. Interestingly, NPs show an affinity for hair follicle cells, clustering around them, which highlights nanotechnology’s potential in addressing follicle-related conditions such as AA 123. Nanoparticles composed of certain metals effectively reach deep dermal structures and exhibit extended localization 124. Furthermore, NP gels formulated PLGA-based system improve the transport of tofacitinib citrate, leading to greater drug absorption and efficacy 125. Ultimately, nanotechnology emerges as a viable and secure strategy for effectively treating AA.
Microneedle-Based Therapy: Microneedles (MN) provide an innovative way to enhance drug delivery by forming microscopic channels in the stratum corneum, improving the penetration and effectiveness of topical treatments 126. Microneedling has been shown to promote hair regrowth in alopecia by stimulating the wound healing response, leading to the release of platelet-derived growth factor and epidermal growth factors, as well as activating the hair bulge 127. Additionally, microneedling increases the expression of Wnt proteins, which are known to stimulate dermal papilla stem cells and support hair growth 128. Research highlights the positive effects of microneedling (MN) when paired with betamethasone, suggesting its potential as an effective treatment approach 129. While microneedling has been extensively studied for AGA treatment, its potential role in addressing AA remains less explored.
Alternative and Future Therapies:
JAK Inhibitors: JAK inhibitors recently gained attention as a promising therapeutic strategy for managing AA. Several JAK inhibitors are already approved for various inflammatory conditions. Tofacitinib, which targets JAK1 and JAK3, is used for rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis. Baricitinib and Ruxolitinib, both acting on JAK1 and JAK2, are indicated for diseases like rheumatoid arthritis, myelofibrosis, polycythemia vera, and graft-versus-host disease 130. A case report described almost full hair regrowth in three AA patients treated with oral Ruxolitinib. Post-treatment biopsies showed reduced T-cell infiltration around follicles and lower HLA class I and II expression 131. A retrospective study in 11 children with severe AA (aged 6–12) reported that 73% exhibited visible hair regrowth and 64% achieved ≥ 50% SALT improvement after 6 months of tofacitinib treatment. Most adverse effects were mild. The findings align with a broader literature review covering 31 paediatric AA cases, where 87% of patients responded positively to tofacitinib therapy. These outcomes highlight the drug’s potential as a viable and relatively safe option in treatment-resistant paediatric AA 132. Most patients who respond to JAK inhibitors tend to relapse after stopping treatment, typically within 2 to 3 months133. Studies show that JAK inhibitors have demonstrated therapeutic promise in AA and may play a key role in future treatment strategies.
Stem Cell Therapy: Recent research suggests that Mesenchymal Stem Cells (MSCs) could serve as an effective treatment for AA, primarily because of their immunosuppressive functions. They work by reducing pro-inflammatory T cell proliferation and enhancing the presence of controlling T cells 134. Research on IFN-γ-induced AA in mice revealed an increase in the presence of CD4+ and CD8+ T cells. MSC treatment effectively decreased several inflammatory factors and minimized T cell infiltration, suggesting its therapeutic potential in managing AA 135. Using MSC therapy could be an effective strategy for managing and potentially preventing AA.
Gut Microbiota: Recent studies highlight the role of gut microbiota in modulating immune responses and its involvement in AA pathogenesis 136. Research comparing the scalp microbiota between AA patients and healthy individuals shows distinct variations in bacterial populations, particularly Actinobacteria and Firmicutes, suggesting microbial influences on AA 137. Short-chain fatty acids (SCFAs) provide essential energy to gut bacteria and intestinal lining cells while modulating T lymphocyte chromatin structure, thereby enhancing gene expression 138. Their interaction with G protein-coupled receptor 43 (GPR43) is crucial in mitigating inflammation 139. While SCFAs have systemic effects upon absorption, their topical application may be particularly beneficial for AA 140. Moreover, studies suggest topical SCFAs induce tissue plasminogen activator expression, reduce fibrin deposition, and help alleviate chronic sinusitiss 141. These findings indicate that applying short-chain fatty acids (SCFAs) directly to scalp hair follicles could be a promising therapeutic approach for treating AA.
Statins: Statins not only help reduce lipid levels but also play a role in suppressing inflammation and modulating immune responses 142. Research suggests that simvastatin may help reduce inflammation by suppressing NF-κB and JAK/STAT pathways, limiting ROS production, and stimulating the Wnt/β-catenin pathway. Its immune-regulating properties could be further strengthened with ezetimibe 143. A pilot study reported that 12 out of 19 patients with AA, involving 40–70% of the scalp, achieved more than 50% hair regrowth, with no adverse effects observed during the treatment 144. These findings suggest that statins hold promise, but randomized controlled trials (RCTs) with larger sample sizes are needed to confirm their effectiveness.
Antihistamines: Clinical studies suggest that fexofenadine may serve as a suitable adjunct to Diphenylcyclopropenone (DPCP) or Squaric Acid Dibutyl Ester (SADBE) topical immunotherapy for atopic AA by alleviating itching and improving treatment effectiveness 145. Additionally, hair regrowth has been observed following the administration of ebastine 146. Another study suggests that combining antihistamines with topical corticosteroids and superficial cryotherapy enhances hair regrowth in patients compared to those not using antihistamines. Additionally, no serious side effects requiring discontinuation have been observed 147.
Gene Therapy: Alopecia and many other diseases are being explored for treatment through the innovative field of gene therapy 148. Gene therapies typically involve delivering genetic material using modified vectors, either directly into a subject’s epidermal tissue (in-vivo) or indirectly (ex-vivo). In ex-vivo approaches, cells are extracted from the host, genetically modified, and subsequently administered back into the skin 149.
RNA interference-based gene therapy is considered a revolutionary method in individualized treatment. The rapid development of topical siRNA for gene silencing has positioned it as an appealing strategy for drug discovery and therapeutic applications 150. Through the use of siRNA, RNA interference enables highly targeted gene suppression with exceptional specificity 151. A study conducted an in-vivo model of AA explored the use of gelatine microspheres for delivering siRNA to treat AA, demonstrating its effectiveness in suppressing Th1-related genes and promoting hair regrowth. It was observed that Th1 cells were highly present in alopecic regions, with CD4 T lymphocytes predominantly expressing CCR5. Further analysis of cytokine therapy revealed that IL-4 and anti-interferon antibody injections reduced CD8 T cell accumulation and interferon mRNA levels, aiding recovery. The siRNA specifically targeted T-box21 to minimize Th1 cytokine production, while gelatine conjugation enhanced therapeutic efficiency 152.
Herbal Extracts: Herbal remedies have been traditionally used for centuries to promote hair growth and prevent hair loss. Although their safety and efficacy remain uncertain, they are generally believed to cause fewer side effects than conventional treatments 153. Onion has been explored for its therapeutic role in AA. In a clinical trial with 62 participants, 87% of those using topical onion juice showed complete hair regrowth within 8 weeks, compared to only 13% in the control group using plain water 154. These findings suggest that raw onion juice could be beneficial in AA treatment. Similarly, garlic’s antibacterial and vasodilatory effects may support hair regrowth. In a double-blind randomized controlled trial of 40 patients, a 5% garlic gel with 0.1% betamethasone showed greater improvement in hair density and patch size than corticosteroid alone, with no reported side effects. This supports garlic as a potential adjunct in AA therapy 155. Glycyrrhizin, a plant-based glycoside, exhibits immunomodulatory activity and may influence T cell function, including Th17 differentiation. Clinical studies combining it with vitamin B2, with or without peony glucosides, have shown promising regrowth outcomes in both adult and paediatric alopecia areata patients, with comparable efficacy across groups 156.
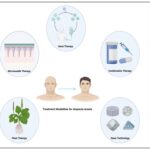
A visual overview of advanced and future-directed therapeutic approaches in AA is presented in Fig. 3.
FIG. 3: THIS FIGURE ILLUSTRATES EMERGING TREATMENT STRATEGIES FOR AA. THESE APPROACHES AIM TO RESTORE IMMUNE BALANCE, STIMULATE FOLLICULAR REGENERATION, AND ENHANCE THERAPEUTIC EFFICACY
Final Insights and Emerging Opportunities: Alopecia areata is a common autoimmune disorder characterized by the abrupt onset of localized hair loss, typically affecting distinct regions of the scalp or body. Even though research on its pathophysiology and available treatments has progressed, several uncertainties remain challenging for effective management. A deeper understanding of AA's underlying mechanisms could enhance clinical treatments, support precision therapy, and facilitate timely detection with more targeted therapies. These developments could have profound medical and social impacts, enhance patient outcomes, and alleviate the overall disease burden. However, current data highlight a considerable gap in the availability of drugs designed specifically for AA, both in development and clinical application, with no definitive cure yet established. Traditional treatments like glucocorticoids, minoxidil, and light therapy face limitations, including inconsistent efficacy, possible side effects, and frequent relapse. Recently, interest in small-molecule drugs and biologic agents has grown due to their promising safety and effectiveness in skin disease management. Among them, Immunotherapeutics such as JAK inhibitors have demonstrated significant potential in AA treatment. However, existing data from small-scale trials and case reports require larger studies to confirm their reliability and safety. Additionally, while key molecular targets associated with AA have been identified, many findings stem from animal models, which may not fully replicate human disease complexity. As a result, further validation is required before these approaches can be translated into viable human treatments.
Emerging therapies for AA, such as stem cell therapy and follicle engineering, offer promising regenerative solutions. Meanwhile, AI-driven drug design incorporating nano-enzymes is opening new doors in treatment innovation. Though these methods remain experimental, they could transform care for patients with difficult-to-treat or severe forms of the condition as research and technology advance. Detecting AA in its early stages is challenging due to dependence on clinical evaluation and the need to identify other causes of the hair loss. The absence of reliable biomarkers makes monitoring even more difficult. While scalp biopsies and dermoscopy can aid diagnosis, their invasive nature restricts widespread use. This underscores the growing demand for non-invasive diagnostic approaches, such as serum biomarkers and advanced imaging methods, to enhance early detection of AA. Progress in gene expression profiling and proteomic technologies has identified potential biomarkers that may aid in predicting disease progression and personalizing treatment strategies. Furthermore, the integration of artificial intelligence, particularly machine learning algorithms for dermoscopicimage analysis, holds significant promise for improving the precision and efficiency of AA diagnosis. The progress of advanced diagnostic tools and targeted therapies holds potential for enhanced disease management in the future. Moreover, Major hurdles include identifying immune triggers, evaluating the chronic safety of treatments, and tailoring therapies to individual genetic and environmental profiles. Ongoing research in these domains is essential to enhance the quality of life for those affected by AA by enabling the development of safer and more effective therapeutic options.
ACKNOWLEDGEMENT: We would like to thank Dr. S. S. Kadam, Chancellor, Bharati Vidyapeeth (Deemed to be University), Dr. A. P. Pawar, Principal, BVDU Poona College of Pharmacy for providing the necessary facilities and support.
Funding: This article did not receive any specific grant from funding agencies.
CONFLICT OF INTEREST: The authors declare no conflict of interest.
REFERENCES:
- Pratt CH, King LE, Messenger AG, Christiano AM and Sundberg JP: Alopecia areata. Nat Rev Dis Primers 2017; 3: 17011.
- Dainichi T, Iwata M and Kaku Y: Alopecia areata: What’s new in the epidemiology, comorbidities, and pathogenesis? J Dermatol Sci 2023; 112(3): 120–7.
- Suchonwanit P, Kositkuljorn C and Pomsoong C: Alopecia Areata: An Autoimmune Disease of Multiple Players. Immunotargets Ther 2021; 10: 299–312.
- Chanprapaph K, Mahasaksiri T, Kositkuljorn C, Leerunyakul K and Suchonwanit P: Prevalence and Risk Factors Associated with the Occurrence of Autoimmune Diseases in Patients with Alopecia Areata. J Inflamm Res 2021; 14: 4881–91.
- Sterkens A, Lambert J and Bervoets A: Alopecia areata: a review on diagnosis, immunological etiopathogenesis and treatment options. Clin Exp Med 2021; 21(2): 215–30.
- Gaurav A, Eang B and Mostaghimi A: Alopecia Areata. JAMA Dermatol 2024; 160(3): 372.
- Lee H, Kim YC and Choi JW: Alopecia areata is not a risk factor for heart diseases: A 10-year retrospective cohort study. PLoS One 2021; 16(5): 0250216.
- Abarca YA, Scott-Emuakpor R, Tirth J, Moroz O, Thomas GP and Yateem D: Alopecia Areata: Understanding the Pathophysiology and Advancements in Treatment Modalities. Cureus 2025; 17(1): 78298.
- Zhou C, Li X, Wang C and Zhang J: Alopecia Areata: an Update on Etiopathogenesis, Diagnosis, and Management. Clin Rev Allergy Immunol 2021; 61(3): 403–23.
- Delamere FM, Sladden MM, Dobbins HM and Leonardi-Bee J: Interventions for alopecia areata. Cochrane Database Syst Rev 2008; (2): 004413.
- Rodgers AR: Why Finding a Treatment for Alopecia Areata Is Important: A Multifaceted Perspective. Jof Invest Dermatology Symposium Proceedings 2018; 19(1): 51–3.
- Trüeb RM and Dias MFRG: Alopecia Areata: a Comprehensive Review of Pathogenesis and Management. Clin Rev Allergy Immunol 2018; 54(1): 68–87.
- Šutić Udović I, Hlača N, Massari LP, Brajac I, Kaštelan M and Vičić M: Deciphering the Complex Immunopathogenesis of Alopecia Areata. Int J Mol Sci 2024; 25(11): 5652.
- Moussa A, Bokhari L and Sinclair RD: Alopecia areata: a review of diagnosis, pathogenesis and the therapeutic landscape. Wound Practice and Res 2022; 30(1): 24–30.
- Olayinka J (Jadé) T and Richmond JM: Immunopathogenesis of alopecia areata. Current Research in Immunology 2021; 2: 7–11.
- Yang S, Yang J, Liu JB, Wang HY, Yang Q and Gao M: The genetic epidemiology of alopecia areata in China. British Journal of Dermatology 2004; 151(1): 16–23.
- Betz RC, Petukhova L, Ripke S, Huang H, Menelaou A and Redler S: Genome-wide meta-analysis in alopecia areata resolves HLA associations and reveals two new susceptibility loci. Nat Commun 2015; 6(1): 5966.
- Petukhova L, Duvic M, Hordinsky M, Norris D, Price V and Shimomura Y: Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature 2010; 466(7302): 113–7.
- Erfan R, Shaker OG, Khalil MAF, Hassan AR, Abu-El-Azayem AK and Samy A: LncRNA NEAT1 and miRNA 101 as potential diagnostic biomarkers in patients with alopecia areata. Noncoding RNA Res 2025; 10: 35–40.
- Mustafa AI, Al ‐refaie AM, El‐Shimi OS, Fawzy E and Sorour NE: Diagnostic implications of MicroRNAs; 155, 146 a, and 203 lesional expression in alopecia areata: A preliminary case‐controlled study. J Cosmet Dermatol 2022; 21(6): 2648–54.
- Tishe ZH, Shawkat S, Popy MN, Ahmed A, Mumu SB and Apu MNH: Interleukin gene polymorphisms and alopecia areata: A systematic review and meta-analysis. Medicine 2024; 103(8): 37300.
- Erjavec SO, Gelfman S, Abdelaziz AR, Lee EY, Monga I and Alkelai A: Whole exome sequencing in Alopecia Areata identifies rare variants in KRT82. Nat Commun 2022; 13(1): 800.
- Oka A, Takagi A, Komiyama E, Yoshihara N, Mano S and Hosomichi K: Alopecia areata susceptibility variant in MHC region impacts expressions of genes contributing to hair keratinization and is involved in hair loss. EBioMedicine 2020; 57: 102810.
- Gao Q, Liang X, Shaikh AS, Zang J, Xu W and Zhang Y: JAK/STAT Signal Transduction: Promising Attractive Targets for Immune, Inflammatory and Hematopoietic Diseases. Current Drug Targets 2018; 19(5): 487–500.
- Aittomäki S and Pesu M: Therapeutic Targeting of the <scp>JAK</scp> / <scp>STAT</scp> Pathway. Basic Clin Pharmacol Toxicol 2014; 114(1): 18–23.
- Dillon KAL: A Comprehensive Literature Review of JAK Inhibitors in Treatment of Alopecia Areata. Clin CosmetInvestig Dermatol 2021; 14: 691–714.
- Khanimov I: Association between smoking and alopecia areata: a systematic review and meta‐analysis. Int J Dermatol 2022; 61(1): 22–4.
- Minokawa Y, Sawada Y and Nakamura M: Lifestyle Factors Involved in the Pathogenesis of Alopecia Areata. Int J Mol Sci 2022; 23(3): 1038.
- Sánchez-Pellicer P, Navarro-Moratalla L, Núñez-Delegido E, Agüera-Santos J and Navarro-López V: How Our Microbiome Influences the Pathogenesis of Alopecia Areata. Genes (Basel) 2022; 13(10): 1860.
- Won EJ, Jang HH, Park H and Kim SJ: A Potential Predictive Role of the Scalp Microbiome Profiling in Patients with Alopecia Areata: Staphylococcus caprae, Corynebacterium, and Cutibacterium Species Microorganisms 2022; 10(5): 864.
- Moreno‐Arrones OM, Serrano‐Villar S, Perez‐Brocal V, Saceda‐Corralo D, Morales‐Raya C and Rodrigues‐Barata R: Analysis of the gut microbiota in alopecia areata: identification of bacterial biomarkers. Journal of the European Academy of Dermatology and Venereology 2020; 34(2): 400–5.
- Torales J, Castaldelli‐Maia JM, Ventriglio A, Almirón‐Santacruz J, Barrios I and O’Higgins M: Alopecia areata: A psychodermatological perspective. J Cosmet Dermatol 2022; 21(6): 2318–23.
- Rajabi F, Abdollahimajd F, Jabalameli N, Nassiri Kashani M and Firooz A: The Immunogenetics of Alopecia areata. In 2022. p 19–59.
- Ahn D, Kim H, Lee B and Hahm DH: Psychological Stress-Induced Pathogenesis of Alopecia Areata: Autoimmune and Apoptotic Pathways. Int J Mol Sci 2023; 24(14): 11711.
- Toussi A, Barton VR, Le ST, Agbai ON and Kiuru M: Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: A systematic review. J Am Acad Dermatol 2021; 85(1): 162–75.
- Pham CT, Romero K, Almohanna HM, Griggs J, Ahmed A and Tosti A: The Role of Diet as an Adjuvant Treatment in Scarring and Nonscarring Alopecia. Skin Appendage Disord 2020; 6(2): 88–96.
- Diotallevi F, Campanati A, Martina E, Radi G, Paolinelli M and Marani A: The Role of Nutrition in Immune-Mediated, Inflammatory Skin Disease: A Narrative Review. Nutrients 2022; 14(3): 591.
- Lee Y Bin and Lee WS: Alopecia Areata and Body Mass Index: A Retrospective Analysis of 257 Cases. Ann Dermatol 2022; 34(4): 305–8.
- Stochmal A, Waśkiel-Burnat A, Chrostowska S, Zaremba M, Rakowska A and Czuwara J: Adiponectin as a novel biomarker of disease severity in alopecia areata. Sci Rep 2021; 11(1): 13809.
- Dai YX, Tai YH, Chen CC, Chang YT, Chen TJ and Chen MH: Bidirectional association between alopecia areata and sleep disorders: a population-based cohort study in Taiwan. Sleep Med 2020; 75: 112–6.
- Park SH, Ji KH, Kim JU, Jang SH, Ahn SW and Hong SM: Evaluation of Sleep Disturbance in Alopecia Areata through Questionnaire: Pittsburgh Sleep Quality Index as a Reasonable Tool. Ann Dermatol 2023; 35(3): 183–9.
- Zhu Y, Ouyang X, Zhang D, Wang X, Wu L and Yu S: Alopecia areata following COVID-19 vaccine: a systematic review. Eur J Med Res 2024; 29(1): 356.
- Fukuyama M, Ito T and Ohyama M: Alopecia areata: Current understanding of the pathophysiology and update on therapeutic approaches, featuring the Japanese Dermatological Association guidelines. J Dermatol 2022; 49(1): 19–36.
- Ravipati A, Pradeep T and Tosti A: A cross‐sectional analysis of medications used by patients reporting alopecia areata on the FDA adverse events reporting system. Int J Dermatol 2024; 63(4): 497–502.
- Alhanshali L, Buontempo M, Shapiro J and Lo Sicco K: Medication-induced hair loss: An update. J Am Acad Dermatol 2023; 89(2): 20–8.
- Teng Y, Fan Y, Shang D, Tao X and Sun D: Alopecia Universalis in an Elderly Chinese Man Induced by Sacubitril/Alisartan, a Novel Angiotensin Receptor-Neprilysin Inhibitor. J Inflamm Res 2023; 16: 3519–22.
- Puiu T, Reimer D and Sokumbi O: Rituximab-Induced alopecia universalis in a patient with bullous pemphigoid. Journal of Drugs in Dermatology 2022; 21(8): 894–5.
- Mounessa J, Caravaglio JV, Domozych R, Chapman S, Dellavalle RP and Dunnick CA: Commonly prescribed medications associated with alopecia. J Am Acad Dermatol 2023; 88(6): 1326-1337.
- Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N and Christiano AM: Alopecia areata. J Am Acad Dermatol 2018; 78(1): 1–12.
- Tosti A: Alopecia Areata and Comorbid Conditions Should We Screen Patients? JAMA Dermatol 2013; 149(7): 794.
- Chelidze K and Lipner SR: Nail changes in alopecia areata: an update and review. Int J Dermatol 2018; 57(7): 776–83.
- Roest Y, Middendorp H, Evers A, Kerkhof P and Pasch M: Nail Involvement in Alopecia Areata: A questionnaire-based survey on clinical signs, impact on quality of life and review of the literature. Acta Dermato Venereologica 2018; 98(2): 212–7.
- Goksin S: Retrospective evaluation of clinical profile and comorbidities in patients with alopecia areata. North Clin Istanb 2022; 9(5): 451–8.
- McDonald KA, Shelley AJ, Colantonio S, Beecker J: Hair pull test: Evidence-based update and revision of guidelines. J Am Acad Dermatol 2017; 76(3): 472–7.
- Mubki T, Rudnicka L, Olszewska M and Shapiro J: Evaluation and diagnosis of the hair loss patient. J Am Acad Dermatol 2014; 71(3): 431.e1-431.e11.
- Sellheyer K and Bergfeld WF: Histopathologic Evaluation of Alopecias. Am J Dermatopathol 2006; 28(3): 236–59.
- Barahmani N, de Andrade M, Slusser JP, Wei Q, Hordinsky M and Price VH: Human Leukocyte Antigen Class II Alleles Are Associated with Risk of Alopecia Areata. Journal of Investigative Dermatology 2008; 128(1): 240–3.
- Zhou B, Chen M, Shang S and Zhao J: Association of CTLA-4 gene polymorphisms and alopecia areata: a systematic review and meta-analysis. Biomarkers 2022; 27(4): 338–48.
- Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J and Canfield D: Alopecia areata investigational assessment guidelines–Part II. J Am Acad Dermatol 2004; 51(3): 440–7.
- King BA, Senna MM, Ohyama M, Tosti A, Sinclair RD and Ball S: Defining severity in alopecia areata: current perspectives and a multidimensional framework. Dermatol Ther (Heidelb) 2022; 12(4): 825–34.
- Ma T, Zhang T, Miao F, Liu J, Zhu Q and Chen Z: Alopecia Areata: Pathogenesis, Diagnosis, and Therapies. MedComm (Beijing) 2025; 6(5): 70182.
- Olsen EA, Roberts J, Sperling L, Tosti A, Shapiro J and McMichael A: Objective outcome measures: Collecting meaningful data on alopecia areata. J Am Acad Dermatol 2018; 79(3): 470-478.e3.
- Alsantali A: Alopecia areata: a new treatment plan. Clin CosmetInvestig Dermatol 2011; 4: 107–15.
- Hammerschmidt M and Mulinari Brenner F: Efficacy and safety of methotrexate in alopecia areata. An Bras Dermatol 2014; 89(5): 729–34.
- Taniguchi K and Karin M: NF-κB, inflammation, immunity and cancer: coming of age. Nat Rev Immunol 2018; 18(5): 309–24.
- Tosti A, Piraccini BM, Pazzaglia M and Vincenzi C: Clobetasol propionate 0.05% under occlusion in the treatment of alopecia totalis/universalis. J Am Acad Dermatol 2003; 49(1): 96–8.
- Harries MJ, Sun J, Paus R and King LE: Management of alopecia areata. BMJ 2010; 341(23-1): 3671-3671.
- Messenger AG and Rundegren J: Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology 2004; 150(2): 186–94.
- Sung CT, Juhasz MLW, Choi FD and Mesinkovska NA: The Efficacy of Topical Minoxidil for Non-Scarring Alopecia: A Systematic Review. J Drugs Dermatol 2019; 18(2): 155–60.
- Kerure A, Ghalla M, Mahajan S, Dhoot D and Barkate H: Real-World Effectiveness, Safety, and Tolerability of Cetosomal Minoxidil 5% Alone and a Fixed Drug Combination of Cetosomal Minoxidil 5% With Finasteride 0.1% in the Management of Androgenetic Alopecia (Inbilt Study). Cureus 2023; 15(7): 41681.
- Durdu M, Özcan D, Baba M and Seçkin D: Efficacy and safety of diphenylcyclopropenone alone or in combination with anthralin in the treatment of chronic extensive alopecia areata: A retrospective case series. J Am Acad Dermatol 2015; 72(4): 640–50.
- Peloquin L and Castelo-Soccio L: Alopecia Areata: An Update on Treatment Options for Children. Pediatric Drugs 2017; 19(5): 411–22.
- Tang L, Cao L, Pelech S, Lui H and Shapiro J: Cytokines and signal transduction pathways mediated by anthralin in alopecia areata-affected dundee experimental balding rats. Journal of Investigative Dermatology Symposium Proceedings 2003; 8(1): 87–90.
- Barton VR, Toussi A, Awasthi S and Kiuru M: Treatment of pediatric alopecia areata: A systematic review. J Am Acad Dermatol 2022; 86(6): 1318–34.
- Tauchi M, Fuchs TA, Kellenberger AJ, Woodward DF, Paus R and Lütjen-Drecoll E: Characterization of an in-vivo model for the study of eyelash biology and trichomegaly: mouse eyelash morphology, development, growth cycle, and anagen prolongation by bimatoprost. British Journal of Dermatology 2010; 162(6): 1186–97.
- Coronel‐Pérez I, Rodríguez‐Rey E and Camacho‐Martínez F: Latanoprost in the treatment of eyelash alopecia in alopecia areata universalis. Journal of the European Academy of Dermatology and Venereology 2010; 24(4): 481–5.
- Zaher H, Gawdat HI, Hegazy RA and Hassan M: Bimatoprost versus Mometasone Furoate in the Treatment of Scalp Alopecia Areata: A Pilot Study. Dermatology 2015; 230(4): 308–13.
- El-Ashmawy AA, El-Maadawy IH and El-Maghraby GM: Efficacy of topical latanoprost versus minoxidil and betamethasone valerate on the treatment of alopecia areata. Journal of Dermatological Treatment 2018; 29(1): 55–64.
- Lee S, Kim BJ, Lee Y Bin and Lee WS: Hair regrowth outcomes of contact immunotherapy for patients with alopecia areata. JAMA Dermatol 2018; 154(10): 1145–51.
- Buckley DA and Du Vivier AWP: The therapeutic use of topical contact sensitizers in benign dermatoses. British Journal of Dermatology 2001; 145(3): 385–405.
- Leong WMS, Mok ZR and Chandran NS: Limited efficacy of diphenylcyclopropenone in the treatment of alopecia areata: Experience from a Tertiary Healthcare Institution in Singapore. Dermatol Ther 2020; 33(6): 14447.
- Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N and Christiano AM: Alopecia areata. J Am Acad Dermatol 2018; 78(1): 15–24.
- Zerbinati N, Esposito C, D’Este E, Calligaro A and Valsecchi R: Topical Immunotherapy of Alopecia Areata: A Large Retrospective Study. Dermatol Ther (Heidelb) 2018; 8(1): 101–10.
- Aghaei S: Topical immunotherapy of severe alopecia areata with diphenylcyclopropenone (DPCP): experience in an Iranian population. BMC Dermatol 2005; 5(1): 6.
- Jang YH, Jung HJ, Moon SY, Lee WJ, Lee SJ and Lee WK: Systematic review and quality analysis of studies on the efficacy of topical diphenylcyclopropenone treatment for alopecia areata. J Am Acad Dermatol 2017; 77(1): 170-172.
- Sigmundsdottir H, Johnston A, Gudjonsson JE and Valdimarsson H: Narrowband–UVB irradiation decreases the production of pro-inflammatory cytokines by stimulated T cells. Arch Dermatol Res 2005; 297(1): 39–42.
- Byun JW, Moon JH, Bang CY, Shin J and Choi GS: Effectiveness of 308-nm excimer laser therapy in treating alopecia areata, determined by examining the treated sides of selected alopecic patches. Dermatology 2015; 231(1): 70–6.
- Grazyna B, Anna W, Matgorzata D and Magdalena P: Effects of psoralen‐uv‐a‐turban in alopecia areata. Skin Med: Dermatology for the Clinician 2006; 5(2): 64–8.
- Kurz B, Berneburg M, Bäumler W and Karrer S: Phototherapy: Theory and practice. JDDG: Journal der Deutschen Dermatologischen Gesellschaft 2023; 21(8): 882–97.
- Shreberk-Hassidim R, Ramot Y, Gilula Z and Zlotogorski A: A systematic review of pulse steroid therapy for alopecia areata. J Am Acad Dermatol 2016; 74(2): 372-374.
- Gupta AK, Carviel J and Abramovits W: Treating alopecia areata: current practices versus new directions. Am J Clin Dermatol 2017; 18(1): 67–75.
- Phan K, Ramachandran V and Sebaratnam DF: Methotrexate for alopecia areata: A systematic review and meta-analysis. J Am Acad Dermatol 2019; 80(1): 120-127.
- Mateos-Haro M, Novoa-Candia M, Sánchez Vanegas G, Correa-Pérez A, Gaetano Gil A and Fernández-García S: Treatments for alopecia areata: a network meta-analysis. Cochrane Database of Systematic Reviews 2023; 2023(10): 013719.
- Hamed KM, Dighriri IM, Baomar AF, Alharthy BT, Alenazi FE and Alali GH: Overview of methotrexate toxicity: a comprehensive literature review. Cureus 2022; 14(9): 29518.
- Nowaczyk J, Makowska K, Rakowska A, Sikora M and Rudnicka L: Cyclosporine with and without systemic corticosteroids in treatment of alopecia areata: a systematic review. Dermatol Ther (Heidelb) 2020; 10(3): 387–99.
- Thorp M, DeMattos A, Bennett W, Barry J and Norman D: The effect of conversion from cyclosporine to tacrolimus on gingival hyperplasia, hirsutism and cholesterol. Transplantation 2000; 69(6): 1218–21.
- Taneja N and Gupta S: Apremilast is efficacious in refractory alopecia areata. Journal of Dermatological Treatment 2020; 31(7): 727–9.
- Chen L, Lei X and Mahnke K: Adenosine and its receptors in the pathogenesis and treatment of inflammatory skin diseases. Int J Mol Sci 2024; 25(11): 5810.
- Langley A and Beecker J: Management of common side effects of apremilast. J Cutan Med Surg 2018; 22(4): 415–21.
- Pagnanelli G, Cavani A, Canzona F and Mazzanti C: Mild therapeutic response of alopecia areata during treatment of psoriasis with secukinumab. European Journal of Dermatology 2020; 30(5): 602–3.
- Loh SH, Moon HN, Lew BL and Sim WY: Role of T helper 17 cells and T regulatory cells in alopecia areata: comparison of lesion and serum cytokine between controls and patients. Journal of the European Academy of Dermatology and Venereology 2018; 32(6): 1028–33.
- Gilhar A, Etzioni A and Paus R: Alopecia Areata. New England Journal of Medicine 2012; 366(16): 1515–25.
- Bakry O, El Shazly RM, Basha M and Mostafa H: Total serum immunoglobulin E in patients with alopecia areata. Indian Dermatol Online J 2014; 5(2): 122–7.
- Penzi LR, Yasuda M, Manatis-Lornell A, Hagigeorges D and Senna MM: Hair Regrowth in a patient with long-standing alopecia totalis and atopic dermatitis treated with dupilumab. JAMA Dermatol 2018; 154(11): 1358–60.
- Harada K, Irisawa R, Ito T, Uchiyama M and Tsuboi R: The effectiveness of dupilumab in patients with alopecia areata who have atopic dermatitis: a case series of seven patients. British Journal of Dermatology 2020; 183(2): 396–7.
- Guttman-Yassky E, Ungar B, Noda S, Suprun M, Shroff A and Dutt R: Extensive alopecia areata is reversed by IL-12/IL-23p40 cytokine antagonism. Journal of Allergy and Clinical Immunology 2016; 137(1): 301–4.
- Yi RC, Moran SK, Gantz HY, Strowd LC and Feldman SR: Biologics and small molecule targeted therapies for pediatric alopecia areata, psoriasis, atopic dermatitis, and hidradenitis suppurativa in the US: A Narrative Review. Children 2024; 11(8): 892.
- Ortolan LS, Kim SR, Crotts S, Liu LY, Craiglow BG and Wambier C: IL-12/IL-23 neutralization is ineffective for alopecia areata in mice and humans. Journal of Allergy and Clinical Immunology 2019; 144(6): 1731-1734.
- Kerkemeyer KLS and Sinclair R: Treatment of chronic alopecia areata with tildrakizumab: an open‐label pilot study. Int J Dermatol 2020; 59(5): 136–7.
- Le Duff F, Bouaziz JD, Fontas E, Ticchioni M, Viguier M and Dereure O: Low-Dose IL-2 for Treating Moderate to Severe Alopecia Areata: A 52-Week Multicenter Prospective Placebo-Controlled Study Assessing its Impact on T Regulatory Cell and NK Cell Populations. Journal of Investigative Dermatology 2021; 141(4): 933-936.
- Castela E, Le Duff F, Butori C, Ticchioni M, Hofman P and Bahadoran P: Effects of Low-Dose Recombinant Interleukin 2 to Promote T-Regulatory Cells in Alopecia Areata. JAMA Dermatol 2014; 150(7): 748–51.
- Almohanna HM, Ahmed AA, Griggs JW and Tosti A: Platelet-rich plasma in the treatment of alopecia areata: a review. Journal of Investigative Dermatology Symposium Proceedings 2020; 20(1): 45–9.
- Badran KW and S and JP: Platelet-rich plasma for hair loss. Facial Plast Surg Clin North Am 2018; 26(4): 469–85.
- El Taieb MA, Ibrahim H, Nada EA and Seif Al-Din M: Platelets rich plasma versus minoxidil 5% in treatment of alopecia areata: A trichoscopic evaluation. Dermatol Ther 2017; 30(1): 12437.
- Joly P, Lafon A, Houivet E, Donnadieu N, Richard MA and Dupuy A: Efficacy of methotrexate alone vs methotrexate plus low-dose prednisone in patients with alopecia areata totalis or universalis. JAMA Dermatol 2023; 159(4): 403–10.
- Ghassemi M, Yazdanian N, Behrangi E, Jafari M and Goodarzi A: Comparison of efficacy, safety and satisfaction of latanoprost versus minoxidil, betamethasone and in combination in patients with alopecia areata: A blinded multiple group randomized controlled trial. Dermatol Ther 2022; 35(12): 15943.
- Kalluri R: The biology and function of exosomes in cancer. J of Clinical Investigation 2016; 126(4): 1208–15.
- Pegtel DM and Gould SJ: Exosomes. Annu Rev Biochem 2019; 88(1): 487–514.
- Rajendran RL, Gangadaran P, Bak SS, Oh JM, Kalimuthu S and Lee HW: Extracellular vesicles derived from MSCs activates dermal papilla cell in-vitro and promotes hair follicle conversion from telogen to anagen in mice. Sci Rep 2017; 7(1): 15560.
- Shi Y, Zhao J, Li H, Yu M, Zhang W and Qin D: A drug‐free, hair follicle cycling regulatable, separable, antibacterial microneedle patch for hair regeneration therapy. Adv Healthc Mater 2022; 11(19): 2200908.
- Chou LYT, Ming K and Chan WCW: Strategies for the intracellular delivery of nanoparticles. Chem Soc Rev 2011; 40(1): 233–45.
- Li X, Peng X, Zoulikha M, Boafo GF, Magar KT and Ju Y: Multifunctional nanoparticle-mediated combining therapy for human diseases. STTT 2024; 9(1): 1.
- Christmann R, Thomas C, Jager N, Raber AS, Loretz B and Schaefer UF: Nanoparticle targeting to scalp hair follicles: new perspectives for a topical therapy for alopecia areata. Journal of Investigative Dermatology 2020; 140(1): 243-246.e5.
- Baroli B, Ennas MG, Loffredo F, Isola M, Pinna R and Arturo López-Quintela M: Penetration of metallic nanoparticles in human full-thickness skin. Journal of Investigative Dermatology 2007; 127(7): 1701–12.
- Kuchukuntla M, Palanivel V and Madhubabu A: Tofacitinib citrate-loaded nanoparticle gel for the treatment of alopecia areata: response surface design, formulation and in-vitro-in-vivo characterization. Recent Adv Drug Deliv Formul 2023; 17(4): 314–31.
- Sivamani RK, Liepmann D and Maibach HI: Microneedles and transdermal applications. Expert Opin Drug Deliv 2007; 4(1): 19–25.
- Dhurat R, Sukesh M, Avhad G, Dandale A, Pal A and Pund P: A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: A pilot study. Int J Trichology 2013; 5(1): 6–11.
- Lee JC, Daniels MA and Roth MZ: Mesotherapy, Microneedling, and Chemical Peels. Clin Plast Surg 2016; 43(3): 583–95.
- Pei D, Chen L, Yao Y, Zeng L and Zhang G: Microneedling combined with compound betamethasone in treatment of severe alopecia areata: A case report. Front Immunol 2022; 13: 939077.
- Pourang A and Mesinkovska NA: New and emerging therapies for alopecia areata. Drugs 2020; 80(7): 635–46.
- Liu LY and King BA: Ruxolitinib for the treatment of severe alopecia areata. J Am Acad Dermatol 2019; 80(2): 566–8.
- Huang J, Li T, Tan Z, Tang Y, Li J and Liu F: Effectiveness of Tofacitinib in Pre-adolescent Alopecia Areata: A Retrospective Case Series and Literature Review. Acta Derm Venereol 2023; 103: 13418.
- Mackay-Wiggan J, Jabbari A, Nguyen N, Cerise JE, Clark C and Ulerio G: Oral ruxolitinib induces hair regrowth in patients with moderate-to-severe alopecia areata. JCI Insight 2016; 1(15): 89790.
- Luque-Campos N, Contreras-López RA, Jose Paredes-Martínez M, Torres MJ, Bahraoui S and Wei M: Mesenchymal stem cells improve rheumatoid arthritis progression by controlling memory T Cell Response. Front Immunol 2019; 10: 798.
- Park SH, Song SW, Lee YJ, Kang H and Kim JE: Mesenchymal stem cell therapy in alopecia areata: visual and molecular evidence from a mouse model. Int J Mol Sci 2024; 25(17): 9236.
- Basmanav FB and Betz RC: Translational impact of omics studies in alopecia areata: recent advances and future perspectives. Expert Rev Clin Immunol 2022; 18(8): 845–57.
- Pinto D, Sorbellini E, Marzani B, Rucco M, Giuliani G and Rinaldi F: Scalp bacterial shift in Alopecia areata. PLoS One 2019; 14(4): 0215206.
- Kanda N, Hoashi T and Saeki H: The Defect in Regulatory T Cells in Psoriasis and Therapeutic Approaches. J Clin Med 2021; 10(17): 3880.
- Maslowski KM, Vieira AT, Ng A, Kranich J, Sierro F and Di Yu: Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 2009; 461(7268): 1282–6.
- Breuer RI, Soergel KH, Lashner BA, Christ ML, Hanauer SB and Vanagunas A: Short chain fatty acid rectal irrigation for left-sided ulcerative colitis: a randomised, placebo controlled trial. Gut 1997; 40(4): 485–91.
- Imoto Y, Kato A, Takabayashi T, Sakashita M, Norton JE and Suh LA: Short‐chain fatty acids induce tissue plasminogen activator in airway epithelial cells via <scp>GPR</scp> 41 & amp; 43. Clinical & Experimental Allergy 2018; 48(5): 544–54.
- Cervantes J, Jimenez JJ, DelCanto GM and Tosti A: Treatment of Alopecia Areata with Simvastatin/Ezetimibe. Journal of Investigative Dermatology Symposium Proceedings 2018; 19(1): 25–31.
- Shin JM, Jung KE, Yim SH, Rao B, Hong D and Seo YJ: Putative therapeutic mechanisms of simvastatin in the treatment of alopecia areata. J Am Acad Dermatol 2021; 84(3): 782–4.
- Lattouf C, Jimenez JJ, Tosti A, Miteva M, Wikramanayake TC and Kittles C: Treatment of alopecia areata with simvastatin/ezetimibe. J Am Acad Dermatol 2015; 72(2): 359–61.
- Inui S, Nakajima T, Toda N and Itami S: Fexofenadine hydrochloride enhances the efficacy of contact immunotherapy for extensive alopecia areata: Retrospective analysis of 121 cases. J Dermatol 2009; 36(6): 323–7.
- Ohyama M, Shimizu A, Tanaka K and Amagai M: Experimental evaluation of ebastine, a second-generation anti-histamine, as a supportive medication for alopecia areata. J Dermatol Sci 2010; 58(2): 154–7.
- Lee Y Bin and Lee WS: Efficacy of antihistamines in combination with topical corticosteroid and superficial cryotherapy for treatment of alopecia areata: A retrospective cohort study. J Am Acad Dermatol 2021; 84(4): 1152–4.
- Kay MA: State-of-the-art gene-based therapies: the road ahead. Nat Rev Genet 2011; 12(5): 316–28.
- Gorell E, Nguyen N, Lane A and Siprashvili Z: Gene Therapy for Skin Diseases. Cold Spring Harb Perspect Med 2014; 4(4): 015149-015149.
- Rosa J, Suzuki I, Kravicz M, Caron A, Pupo AV and Praça FG: Current Non-viral siRNA Delivery Systems as a Promising Treatment of Skin Diseases. Curr Pharm Des 2018; 24(23): 2644–63.
- Hannon GJ and Rossi JJ: Unlocking the potential of the human genome with RNA interference. Nature 2004; 431(7006): 371–8.
- Nakamura M, Jo J ichiro, Tabata Y and Ishikawa O: Controlled Delivery of T-box21 Small Interfering RNA Ameliorates Autoimmune Alopecia (Alopecia Areata) in a C3H/HeJ Mouse Model. Am J Pathol 2008; 172(3): 650–8.
- Semalty M, Semalty A, Joshi GP and Rawat MSM: Hair growth and rejuvenation: An overview. Journal of Dermatological Treatment 2011; 22(3): 123–32.
- Sharquie KE and Al‐Obaidi HK: Onion Juice (Allium cepa L.), A New Topical Treatment for Alopecia Areata. J Dermatol 2002; 29(6): 343–6.
- Hajheydari Z, Jamshidi M, Akbari J and Mohammadpour R: Combination of topical garlic gel and betamethasone valerate cream in the treatment of localized alopecia areata: A double-blind randomized controlled study. Indian J Dermatol Venereol Leprol 2007; 73(1): 29–32.
- Yang D quan, You L ping, Song P hua, Zhang L xin and Bai Y ping: A randomized controlled trial comparing total glucosides of paeony capsule and compound glycyrrhizin tablet for alopecia areata. Chin J Integr Med 2012; 18(8): 621–5.
How to cite this article:
Tapadiya T, Mandlik DS, Kulkarni R and Shinde VM: Alopecia areata: current insights into immunopathogenesis, diagnosis, and evolving therapeutic strategies. Int J Pharmacognosy 2026; 13(3): 132-51. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(3).132-51.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
132-151
794 KB
299
English
IJP
Tushar Tapadiya, Deepa S. Mandlik, Ravindra Kulkarni and Vaibhav M. Shinde *
Department of Pharmacognosy, Bharati Vidyapeeth (Deemed to be University), Poona College of Pharmacy, Pune, Maharashtra, India.
vaibhavshinde847@gmail.com
06 February 2026
16 March 2026
20 March 2026
10.13040/IJPSR.0975-8232.IJP.13(3).132-51
31 March 2026