A REVIEW ON QUALITY BY DESIGN APPROACH (QBD) AND IT’S BENEFITS
HTML Full TextA REVIEW ON QUALITY BY DESIGN APPROACH (QBD) AND IT’S BENEFITS
Ambati Vamsi Krishna *, Paradeshi Mounika, G. Vedavathi Sai and Kannedari Venkatesh
Department of Pharmaceutical Analysis, Chebrolu Hanumaiah Institute of Pharmaceutical Sciences, Chowdavaram, Guntur, Andhra Pradesh, India.
ABSTRACT: The newest approach in pharmaceutical production linked to quality is called Quality by Design (QbD). To guarantee that premium-grade pharmaceuticals are produced, Pharmaceutical Quality by Design, or QbD, is discussed in this article. A description of Quality by Design is provided along with a list of its constituent parts. Every unit activity has its own set of quality metrics and characteristics. The use of Quality by Design and its associated measures can yield significant benefits for pharmaceutical goods. The foundation of pharmaceutical R&D is high-quality drugs and the procedures used in their production. A product's quality cannot be easily verified because this paper just summarises the product's quality profile and the most important components of Quality by Design. Quality by design (QbD) and end-product testing are two ways to compare the quality of various goods. Quality by Design is based on the ICH Guidelines. ICH guidelines apply to the development of medications and the application of quality control methods. The research and manufacturing of pharmaceuticals might profit from Quality by Design (QbD). As the product develops and is designed, it is crucial to determine the desired product performance report under these ideas. The TPP, QTPP, and CQA stand for target product profile, quality target product profile, and critical quality characteristics. to identify and regulate sources of changeability and to understand how key material attributes (CAM) and critical process parameters (CPP) of the raw material affect the CQAs. Quality-based drug development (QbD) can provide valuable insights for the design, development, and manufacturing of pharmaceutical products. The present overview discusses the historical context, fundamentals of the QbD methodology, and regulatory requirements. The method's aim, the experiment's design, and the risk assessment are all explained in depth for the QbD aspects. The ICH Guidelines provide the basis of Quality by Design. The ICH Guidelines Q8, Q9, and Q10 for pharmaceutical quality systems, quality risk management, and pharmaceutical development, respectively, serve as its foundation. It also provides information on applying Quality by Design to the creation and production of medicines.
Keywords: Quality by design, Quality risk management, Pharmaceuticals, ICH Guidelines, Analytical method development, Design space
INTRODUCTION:
Quality: Quality is a key term in Quality by Design. Thus, "standard or suitable for intended use" is what quality is. Such characteristics as identity, strength, and purity are included in this phrase 1.
Quality by Design: -The US-FDA (United States Food and Drug Administration) and the International Council Harmonisation (ICH) have promoted a number of methods for the development of pharmaceutical goods and their subsequent manufacturing 2.
"A systematic approach to development that begins with predefined target and emphasises understanding of products and processes and process control, based sound science and quality risk management" is the definition of "quality by design" (QbD), the name given to this methodology.
Product Quality, Safety, and Efficacy are recognised by the pharmaceutical businesses. By using scientific methods called QbD (Quality by Design), product quality has been rising 3. From product creation to production, scientific methods may provide the necessary and lucid knowledge. By improving productivity and quality, these QbD tools will reduce the risk. These days, the QbD technique has been effectively implemented in the creation of common formulations 4. The US-FDA has published particular QbD guidelines for biotechnology and instant and extended-release medicinal products. The use of ICH quality criteria, including Q8, Q9, Q10, and Q11, is constantly being suggested by regulatory agencies 5.
The creation of a superior product and manufacturing process that consistently produces the intended outcomes is the aim of pharmaceutical development. It is critical to understand that items cannot be checked for quality. Rather than testing the end analytical process findings, quality should be ingrained in the process design. Among the industries with the tightest regulations is the pharmaceutical sector. It continuously creates high-quality human prescription drugs with pharmacotherapeutic benefits to treat a range of diseases 6. A guideline paper for pharmaceutical firms was released by the US Food and Drug Administration [FDA] in 2002. Products should incorporate quality, safety, and efficacy. Quality by Design is the current term for this idea. In a 2004 paper, Janet Woodcock, the head of the Centre for Drug Evaluation and Research, defined pharmaceutical quality as a product free from contamination. They provided the consumer with the therapeutic benefit that the label dependable assured. The QbD methodology is a relatively recent discovery in analytical method development that, when applied properly, may be quite advantageous 7.
The core principle of quality by design is that "quality ought to be integrated, not tested, into the product." The term "design space" refers to the production area of the product, which comprises the workers, supplies, machinery, and manufacturing environment. The design space must be properly defined prior to obtaining regulatory approval 8. One does not consider using design space to be a change, but working outside of it is several parameters are monitored for their influence on the final product's quality when production is carried out outside of the design boundaries. A QbD tool might be derived from the evaluation of each of these criteria and the results reached. All these details are included in the regulatory submission document 9. The formulation of pharmaceutical goods can be developed using information from product development studies. Process variables that emerge during the development phases will be the basis for Quality Risk Management (QRM). Before beginning development studies, the product's Quality Target Product Profiles (QTPPs) must be defined, and evaluation is done to guarantee the desired degree of product quality 10. Design space, specs, and production controls are all included in the QTPP of the product.
Utilising QbD in the Development of Analytical Methods: The pharmaceutical industry is embracing QbD because it enables the development of robust, sturdy, and resilient methods that support ICH principles. This methodology enables ongoing technique improvement. Chromatographic methods such as HPLC are used for impurity determination in pharmaceuticals, method development, and stability research 11. Karl Fisher titration used to calculate the moisture content. Regarding biopharmaceutical procedures. Disintegration investigations. Hyphenated methodology such as LC-MS. sophisticated methods such as capillary electrophoresis, mass spectroscopy and UHPLC. Examining the genotoxic contamination 12.
QbD's Benefits for the Pharmaceutical Industry: There will be a better level of confidence with the established strategy since it is more resilient to condition fluctuations. Deeper understanding of the process is facilitated by it. When the process is transferred from the research division to the quality control division, this approach provides increased transfer effectiveness 13. Design space concepts prevent post-approval modifications, which might cost any organisation a lot of money. It offers a place where the newest approaches may be invented via constant refinement throughout the course of its life cycle 14.
Benefits of QbD: In order to prevent regulatory problems, batch defects must be avoided, and variations must be minimised. Empowerment of technical staff members. a versatile and agile system for fast responses. Increasing production efficiency and decreasing waste and project rejections are goals shared by many companies. Make a scientific knowledge foundation that applies to every product. Work with industry more closely on science-related issues. Verify the consistency of all the data. Put in place a component for risk management and evaluation. Cut down on the time spent assessing the completed product. The speed at which a release date choice may be made 15. QbD is profitable. Get rid of batch errors. Reduce the number of anomalies and expensive inquiries. Prevent issues with regulatory compliance. Learning inside an organisation is a financial investment in the future. Good science is QbD. improved choices about development. Empowering technical personnel 16.
Modern Design Product Quality Steps 17-18:
Development of a Novel Molecular Entity:
- Preclinical study.
- Nonclinical study
- Clinical Study
- Scale up
- Submission for market Approval
Manufacturing:
- Design Space
- Process Analytical Technology
- Real time Quality Control
Control Strategy:
- Risk based decision
- Continuous Improvement
- Product performance
Quality Control in Seven Stages using a Design-Start up Strategy 19:
- Engage a freelance specialist in Quality by design.
- Conduct a gap analysis by having an expert audit your organisation and processes.
- Assemble a simple quality using your entire team in a design workshop.
- Examine the expert's report and suggestions.
- Create a schedule, budget, and implementation strategy.
- Assign the work (or hire someone else to do it).
- As your "Project Assurance" adviser, hold onto the impartial specialist.
Important Elements of QbD:
- These elements of a QbD strategy for advancements that are Now that quality, safety, and efficacy can be connected, the TPP can be improved. The standard qualities of the product are determined as a foundation for product planning and development.
- Qualities of Significance Attributes are characteristics of a material that have to fall under certain accepted bounds, ranges, or distributions.
- In risk assessment, material attributes and CPPs are contrasted with CQAs. Risk assessment instruments such as the FMEA and the bone diagram will be used to calculate the CPPs. The tools that will be utilised for risk management are listed in ICH Q9 20.
- Utilising experiment style (DOEs) allows for the establishment and representation of a significant stylistic relationship between CQAs and CPPs.
- The business's long-term strategy when faced with an unexpected situation, it's crucial to quickly identify and solve the issue.
- Manage product lifetime and continuously enhance quality. The ICH-Q10 standard enhances quality management systems for QbD products throughout their lifespan 21.
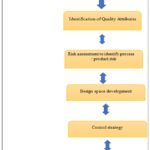
FIG. 1: KEY ELEMENTS OF QBD
Target Product Profile (TPP): The TPP outlines the acceptable appearance of a medicinal product for the purposes of medication development and labelling. TPP outlines the goal, target market, administration path, and other crucial components in addition to the product's high-quality design.
Target Quality Product Profile (TQPP): In the context of product quality, the word TQPP could be considered a logical extension of the term TPP. The information that cannot be passed down from a single generation to the another must be understood and tracked down using the QTPP. To achieve this, a medication product's desired qualities are outlined, taking into account any potential side effects and safety issues. Quantity, strength, identification, instrumentation closure system, and TQPP are examples of the indefinite-quantity type and purity 22. An overview of the medication development programme that focuses mostly on safety and efficacy and is presented in terms of labelling ideas.
- Description
- Clinical Pharmacology
- Indications and Usage
- Contraindications
- Warnings
- Precautions
- Adverse Reactions
- Drug Abuse and Dependence
- Over dosage
- Dosage and Administration
- How Supplied
- Animal Pharmacology and/or Animal
- Toxicology
- Clinical Studies
Critical Quality Attributes (CQA): Certificates of conformity, or CQAs, are used in many different contexts to guarantee a product's efficacy, safety, stability, and quality. To ensure that the final product's quality remains within acceptable parameters, it may also be specified, measured, and monitored. Clinical safety and efficacy are examples of quality qualities, together with the parameter border nearing failure. Another aspect of quality is manufacturing. The criticality risk level may increase if the APT manufacturing process's criticality changes.
Critical Material Attributes (CMAs): When a product cannot meet a QTPP due to a real change in a parameter, it is imperative that the experiment fail. When determining which characteristics are crucial, it's critical to take into account both the degree of modification that one is ready to make and the distinctive qualities of each input material. If the CMAs fall into one or more acceptable ranges, they must satisfy the standards for pharmaceutical substances, excipients, and in-process materials.
Critical Process Parameters (CPP): Any measurable input or output of a process step must be managed in order to achieve the appropriate procedure consistency and product quality. Every item in this read would have an approach parameter. This is how it would operate: Prerequisites and in-process parameters that can significantly affect the final product's yield, purity, and attractiveness are checked 23.
Risk Assessment 24: Speaking of "risk," we mean the possibility as well as the seriousness of harm. A risk-based evaluation of a technique or process may improve its overall quality.
An evaluation of risks is intended to identify the critical components that have an impact on the final product's quality. An evaluation of the risks involved can help to improve communication when dealing with the FDA, trades, R&D/prototype, and multiple production locations. Techniques for evaluating risk include the following 25-26. The ICH guideline Q9 describes a few risk estimation techniques, including:
- Failure Mode Effects Analysis (FMEA).
- Failure Mode, Effects and Criticality Analysis (FMECA).
- Fault Tree Analysis (FTA).
- Hazard Analysis and critical control points (HACCP).
- Hazard Operability Analysis.
- Preliminary Hazard Analysis.
- Risk ranking and filtering.
- Supporting applied mathematics tools.
Design Space: A design space is defined as the "multidimensional combination and interaction of input variables that have been shown to provide assurance of quality, such as material attributes and process parameters." In a design space, one can account for a single unit of operation, many unit operations, or the entire process. According to FDA rules, one does not need to create a formal design space in order to gain product and process expertise.
However, the previously indicated approach can aid in enhancing system understanding and achieving general control. For the new design space, the applicant provides a proposal that has to be authorised by nonsupervisory review. The building of the design space might involve one unit operation, many unit operations, or the whole product formulation process. Although creating a design space is optional according to FDA rules as product and process knowledge may be obtained without one, a technique like this can help achieve more compassionate product quality and overall system management 27.
Utilisation of Design Space: The Connection between crucial quality qualities and process inputs, including variables and parameters. used up until the completion of the process or for one or more unit operations. Both before and after MA can be used. By the applicant's proposal. Not seen as a shift in the design space between. It is necessary for the authorities to validate and evaluate.
Control Strategy: Standards for raw materials, system control, and end-product testing are all part of an all-encompassing production plan that yields high-quality goods. Numerous details on the tools and study techniques are available in this system. Because PATs may be adjusted based on your home's decor, they're a terrific tool for this.
Tools of QbD:
Quality Risk Management: Risk management, according to the FDA, is a strategic safety programme that uses intervention (or) instruments to reduce product risk. Risk to the quality of drugs is assessed, controlled, and reviewed by a systematic procedure that spans the whole product life cycle 44 28. The clinical department, manufacturing operations, sales and marketing, regulatory affairs, and quality unit are all jointly in charge of risk management. In this publication, two key ideas about the use of quality risk management were presented.
- Based on scientific understanding and ultimately connected to patient safety, the risk to quality assessment should be made.
- The formality, thoroughness, and documentation of the quality risk management method should match the level of risk it presents 29.
Process Analytical Technology: An Essential Tool for Quality-Assurance and Design (QAbD): PAT is characterised as "instruments and systems that use real-time measurements, or rapid measurements during processing, of evolving quality and performance attributes of in-process materials to provide information to ensure optimal processing to produce a final product that consistently conforms to established quality and performance standards." PAT is a component of the quality by design (QbD) framework, which provides tools to support quality 30.
Design of Experiment (DoE): The deliberate, planned strategy known as "Design of Experiments" (DoE) is used to determine the relationship between factors influencing a process and its outcome. DoE enables us to set a broad variety of parameters, create and construct models, define responses, evaluate the models, comprehend the results, and eventually make judgements. Single variation studies are generally employed to facilitate multivariate analyses, including those concerning the effects of wavelength, flow rate, and concentration on resolution, retention time, and other variables in HPLC 31. The design of experiment (DoE) is one such structured method that accounts for the impact of CMAs and CPPs on the CQAs of the final dosage form. QbD has garnered a lot of attention since the FDA applied it to the formulation and development of pharmaceutical items 32.
SUMMARY & CONCLUSION: The capacity to quantify TPPs, continual improvement, and a deeper comprehension of goods and procedures are just a few benefits of the QbD methodology. The design of experiment (DoE) is one such structured method that accounts for the impact of CMAs and CPPs on the CQAs of the final dosage form. QbD has garnered a lot of attention since the FDA applied it to the formulation and development of pharmaceutical items. QbD has emerged as a potentially useful scientific instrument for pharmaceutical sector quality assurance. Getting regulatory permission is the pharmaceutical industry's top priority before releasing any product into the market. The proper ATP is only one need for QbD; other requirements include risk assessment, tool selection, and timely completion of the necessary amount of labour. For the pharmaceutical sector to guarantee the quality of its products, analytical method development and validation through QbD is crucial. AQbD leads to a comprehensive grasp of product development and commercial manufacturing. A time and money-saving strategy in design and production is called "quality by design." Biotechnological goods including enzymes, vaccines, monoclonal antibodies, etc. are also extensively covered by QbD. Further regulatory flexibility can be facilitated by this new Quality by Design (QbD) methodology. Quality by Design is also no longer limited to the pharmaceutical industry; it is now a widely used production paradigm.
ACKNOWLEDGEMENT: The authors acknowledge the assistance received from Chebrolu Hanumaiah Institute of Pharmaceutical Sciences management.
Funding: Not Applicable
Ethics Approval and Consent to Participate: Not Applicable
CONFLICT OF INTEREST: We declare that we have no conflict of interest.
REFERENCES:
- Nadpara NP, Thumar R V, Kalola VN and Patel PB: Review Article Quality by Design (Qbd): A Complete Review 2012; 17(2): 20–8.
- Woodcock J: The concept of pharmaceutical quality American Pharmaceutical Review 2004; 7(6): 10–5.
- Chavan SD, Pimpodkar N V, Kadam AS, Gaikwad PS and Pharm PD: Research and reviews: J of Pharmaceutical Quality Assurance Quality by Design. 2015; 1(2): 18–24.
- Roy S: “Quality by Design-Holistic concept of concept of building quality in pharmaceuticals”. Int J Pham Biomed Res 2012; 3: 100-108.
- Lawrence X. Yu: Director of science. “Implementation of Quality by Design”, Question Based Review 2006.
- Pande PP, Sayyad SF, Chavan MJ and Chaudhari SR: Quality by design analytical method development and validation. J Environ Life Sci 2017; 2(2): 39-45.
- Pranali Yeram, Hamrapurkar PD and Padmanabh Mukhedkar: Implementation of quality by design approach to develop and validate stability indicating assay method for simultaneous estimation of sofosbuvir and ledipasvir in bulk drugs and tablet formulation. International Journal of Pharmaceutical Science and Research 2019; 10(1): 180-88.
- Gawade A, Chemate S and Kuchekar A: Pharmaceutical Quality by Design: A New Approach in Product Development. Research and Reviews: Journal of Pharmacy and Pharmaceutical Sciences 2013; 2: 5-11.
- Quality Risk Management. ICH Harmonized Tripartite Guidelines. International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use 2006.
- Pharmaceutical Quality System, ICH Tripartite Guidelines. International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use 2007.
- Delasko JM, Cocchetto D and Burke LB: Target product profile: Beginning drug development with the end in mind, Jan/Feb2005; Issue 1.http://www.fdli.org
- Sangshetti JN, Chitlange SS and Zaheer Z: Quality by Design in Pharmaceuticals, 1st edition, August 2015.
- Jain S: Quality by design (QbD): a comprehensive understanding of implementation and challenges in pharmaceuticals development. IJPPS 2013; 6: 29-35.
- Drakulich A: Critical challenges to implementing QbD: A Q&A with FDA. Pharm. Technol 2009; 33: 90–94.
- Lionberger RA, Lee LS, Lee L, Raw A,Yu LX. Quality by design: Concepts for ANDAs. The AAPS Journal 2008; 10:268–276. https://doi.org/10.1208/s12248-008-9026-7.
- Q8 (R1): Pharmaceutical Development, Revision 1, ICH Harmonized Tripartite Guidelines, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use, 2007.
- Woodcock J, The concept of pharmaceutical quality. American Pharmaceutical Review, 2004, 1–3.
- Food and Drug Administration, Office of Generic Drugs White Paper on Question-based Review: http://www.fda.gov/cder/ OGD/QbR.htm.
- Food and Drug Administration, Guidance for industry, Q6A specifications for new drug substances and products: Chemical substances, 1999.
- Q8 (R1): Pharmaceutical Development, Revision 1, ICH Harmonized Tripartite Guidelines, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use, 2007.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4070262/
- Drakulich A: Critical challenges to implementing QbD: A Q&A with FDA. Pharm. Technol. 2009; 33:90–94.
- Pokharana M, Vaishnav R, Goyal A and Shrivastava A: Stability testing guidelines of pharmaceutical products, Journal of Drug Delivery and Therapeutics 2018; 8(2): 69-175. https://doi.org/10.22270/jddt.v8i2.1564.
- Q9: Quality Risk Management. ICH Harmonized Tripartite Guidelines. International Conference on Harmonization ofTechnical Requirements for Registration of Pharmaceuticals for Human Use, 2006.
- Leuenberger H, Puchkov M, Krausbauer E and Betz G: Manufacturing pharmaceutical granules, is the granulation end-point a myth, Powder Technology 2009; 189: 141–48.
- Miller CE, Chemometrics and NIR: A match made in heaven, Am. Pharm. Rev. Food and Drug Administration CDER, Guidance for industry, Q8 Pharmaceutical Development; 2006; 2: 41–48.
- Vogt FG and Kord AS: Development of quality by design analytical methods. J Pharm Sci 2011; 100: 797-812.
- Nayak AK, Ahmed SA, Beg S, Tabish M and Hasnain MS: Chapter 18: application of quality by design for the development of biopharmaceuticals. In Pharmaceutical Quality by Design 2019; 399-411.
- Meghana Rawal, Amit Singh and Mansoor M: Amiji: Quality-by-Design Concepts to Improve Nanotechnology-Based Drug Development. Pharm Res 2019; 36: 153.
- International Conference on Harmonization (ICH). Guidance for Industry; Q9 quality risk management, ICH harmonized tripartite guideline 2005; 9.
- Gupta Anuj and Neeraj Kumar Fuloria: Short review on quality by design: A new Era of Pharmaceutical drug development. IJDDR 2012; 4(3): 19-26.
- Sharmada S and Sinai Kakodkar: Pharmaceutical Quality-by-Design (QbD): Basic Principles. International Journal of Research Methodology Human 2015; 1(1): 1- 19.
How to cite this article:
Krishna AV, Mounika P, Sai GV and Venkatesh K: A review on quality by design approach (QbD) and it’s benefits. Int J Pharmacognosy 2024; 11(8): 414-20. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(8).414-20.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
414-420
492 KB
488
English
IJP
Ambati Vamsi Krishna *, Paradeshi Mounika, G. Vedavathi Sai and Kannedari Venkatesh
Department of Pharmaceutical Analysis, Chebrolu Hanumaiah Institute of Pharmaceutical Sciences, Chowdavaram, Guntur, Andhra Pradesh, India.
ambativamsi82@gmail.com
28 July 2024
23 August 2024
28 August 2024
10.13040/IJPSR.0975-8232.IJP.11(8).414-20
31 August 2024