A REVIEW ON MODERN EXTRACTION TECHNIQUES OF HERBAL PLANTS
HTML Full TextA REVIEW ON MODERN EXTRACTION TECHNIQUES OF HERBAL PLANTS
Shashank Agrahari * 1, Vipin Kesharwani 2 and Nikhil Kushwaha 3
ARK College of Pharmacy 1, Rakasarai Sarai Akil Kaushambi - 212216, Uttar Pradesh, India.
Department of Pharmacology (PH.D) 2, GLA University Mathura - 281406, Uttar Pradesh, India
Chandra Shekhar Singh College of Pharmacy 3, Koilaha Puramufti Kaushambi - 212203, Uttar Pradesh, India.
ABSTRACT: Plants are naturally gifted for the synthesis of medicinal compounds & extraction is the first crucial step in the preparation of plant formulations. The extraction and characterization of active compounds from medicinal plants have resulted in the discovery of new drugs with high therapeutic value. The new recent extraction techniques with shortened extraction time, reduced solvent consumption, and increased pollution prevention concern and with special care for thermolabile constituents. In this article, the principle behind the operation of various extraction methods, factors influencing method performance, research progress, strength and weakness of different extraction approaches are discussed. The modern techniques for extraction of herbal plant secondary metabolites include Solid-phase micro-extraction, Supercritical-fluid extraction, Pressurized liquid extraction, Accelerated Solvent extraction, High-pressure solvent extraction, Membrane Extraction, The Cold finger method Stir-bar sportive extraction, Ultrasonic extraction, Acid-base extractions, Solid-phase extraction, Microwave-assisted extraction (MAE), Supercritical fluid extraction (SCFE), Pressurized solvent extraction (PSE).
| Keywords: |
Microwave-Assisted Extraction (MAE), Supercritical Fluid Extraction (SFE), Ultrasonication Assisted Extraction (UAE), Accelerated Solvent extraction (ASE), Solid Phase Microextraction (SPME)
INTRODUCTION: For thousands of years, mankind is using plant sources to alleviate or cure illnesses. Plants constitute a source of novel chemical compounds which are of potential use in medicine and other applications. Plants contain many active compounds such as alkaloids, steroids, tannins, glycosides, volatile oils, fixed oils, resins, phenols, and flavonoids deposited in their specific parts such as leaves, flowers, and flowers bark, seeds, fruits, root, etc.
The beneficial medicinal effects of plant materials typically result from the combination of these secondary products 1. The history of plants being used for medicinal purposes is probably as old as the history of mankind.
Extraction and characterization of several active Phyto-compounds from these green factories have given birth to some high activity profile drugs. The potential natural anticancer medicines like vincristine, vinblastine, and taxol can be the best example. Plants are naturally gifted at the synthesis of medicinal compounds. The extraction and characterization of active compounds from medicinal plants have resulted in discovering new drugs with high therapeutic value 2. In 1985 Farnsworth et al. identified 119 secondary plant metabolites which were used as drugs. Out of 255 drugs that are considered basic and essential by the World Health Organization (WHO), 11% are obtained from plants, and a number of synthetic drugs are also obtained from natural precursors. Phytochemicals are known to possess antioxidant 3, antibacterial 4, antifungal 5, antidiabetic 6, anti-inflammatory 7, antiarthritic 8, and radio-protective activity 9 and due to these properties, they are largely used for medicinal purpose.
At present, however, quality and safety-related problems seem to be overshadowing the genuine potential benefits associated with the use of herbal medicine. The problem roots to the lack of high performance, reliable extraction, analytical techniques and methodologies for establishing a standard therapeutic functionality for herbal medicines. Extraction forms the first basic step in medicinal plant research because the preparation of crude extracts from plants is the starting point for the isolation and purification of chemical constituents present in plants.
A key factor in the widespread acceptance of natural or alternative therapies by the international community involves the “modernization” of herbal medicine. In other words, the standardization and quality control of herbal materials by the use of modern science and technology is critical. At present, however, quality-related problems (lack of consistency, safety, and efficacy) seem to be overshadowing the potential genuine health benefits of various herbal products, and a major cause of these problems seems to be related to the lack of simple and reliable analytical techniques and methodologies for the chemical analysis of herbal materials 10.
The phytochemical investigation of a plant may involve the following steps: authentication and extraction of the plant material, separation and isolation of the constituents of interest, characterization of the isolated compounds, and quantitative evaluation 11. Considerable effort has been made by researchers to find efficient extraction methods in order to get high efficiency and efficacy. Efficiency refers to the yield of extraction, whereas efficacy refers to the potency (magnitude of bioactivity / the capacity to produce an effect) of the extract.
For isolation of biological components, extraction from the plant is one of the more sustainable approaches 12. The development of modern sample preparation techniques has significant advantages over conventional methods in terms of reduction in organic solvent consumption and in minimizing sample degradation. They also result in the elimination of undesirable and insoluble components from the extract. The modern methods include microwave-assisted extraction (MAE), ultrasonication-assisted extraction (UAE), supercritical fluid extraction (SFE), solid-phase microextraction (SPME), Soxhwave, etc. Latter is a combination of Soxhlet with microwaves. This combines the rapid heating capacity of microwaves with the simplicity of Soxhlet. Here solvent recovery is also possible, which is not the case in ordinary MAE. However, it has not found widespread use yet 13.
The process of drug extraction can summarily be divided into four essential steps:
- Penetration of the solvent into the drug
- Dissolution of constituents;
- Outward diffusion of the solution from the cells; and
- Separation of dissolved portion and the exhausted drug.
The efficiency of drug extraction depends, therefore, upon the following:
- Nature and properties of the drug and its extractable constituents,
- The particle size of the powdered drug.
- The nature of the solvent; and
- The state of contact between the solvent and the drug particles.
Factors Affecting Choice of an Extraction Process: 14
- Character of the Drugs: Knowledge of the pharmacognosy of the drug to be extracted is essential for the selection of the extraction process that will give the best results. Thus percolation is recommended in the case of hard and tough drugs e.g., Nux vomica, whereas maceration is suitable in case of soft and parenchymatous drugs e.g., gelatin. Similarly maceration is suitable for drugs e.g., Squill, which cannot be easily powdered and unorganized e.g., benzoin.
- Therapeutic Value of the Drug: Maceration is considered suitable if the drug has little therapeutic value e.g., flavours (Lemon), bitters (Gentian) but if the drug has considerable therapeutic value and maximum extraction is required, percolation should be used e.g., Belladonna.
- Stability of the Drug: Continuous extraction has to be avoided if the active constituents of the drug are heat-labile.
- Cost of Drug: In the case of costly drugs e.g., Ginger, percolation is economical, but for cheaper drugs, maceration is good enough. Due consideration has to be given to the cost of the drug and the cost involved in communication for the best possible extraction of the constituents.
- Solvent: Reserved percolation can be used, avoiding continuous extraction if the desired constituents require a solvent other than a bare boiling solvent or an azeotrope.
- Concentration of the Product: Depending on the previous factors, dilute products such as tinctures can be prepared by maceration, or a percolation process can be used for semi-concentrated preparation e.g., concentrated infusions. Concentrated preparations such as liquid extracts or dry extracts are prepared exclusively by percolation. However, continuous extraction can be used if the solvent is suitable and the constituents are heat-stable.
Extraction of Herbal Plants Secondary Metabolite by Traditional Methods Extractions Techniques: 15
(i) Maceration: In this process, the solid ingredient is placed in a stoppered container with 750 ml of the prescribed solvent and allowed to stand for a period of at least 3 days in a warm place with frequent agitation until the soluble matter is dissolved. The mixture is filtered and after most of the liquid has drained. The residue on the filter is washed with sufficient drained, the residue on the filter is washed with a sufficient quantity of the prescribed solvent or solvent mixture, and the filtrates are combined to produce 1000 ml.
(ii) Percolation: The present invention comprises an apparatus and methods for making herbal extracts. The apparatus employs a percolation vessel having a filter and discharge valve. A heater is provided to heat the contents of the vessel to up to about 60 °C. During the cold percolation process. A control is provided to monitor the contents of the vessel and regulate the heater output to obtain and maintain the proper temperature.
A pump draws the output of the vessel and recirculates it to the vessel top so that the vessel effluent passes through the vessel charge. The method entails continuously percolating a solvent through a bed of herbaceous material in the vessel at a temperature between room temperature and about 60 °C. to produce an extract comprising active principles contained in the herbaceous material.
The thus produced extract is highly concentrated and contains a large proportion of the active principles in the raw material. The thus-produced extract can be used in lower unit doses to provide enhanced medicinal treatment.
(iii) Cold pressing: Cold pressing is used to extract the essential oils from citrus rinds such as orange, lemon, grapefruit, and bergamot. The rinds are separated from the fruit, are ground or chopped, and are then pressed. The result is a watery mixture of essential oil and liquid, which will separate given time. It is important to note that oils extracted using this method have are latively short shelf life, so make or purchase only what you will be using within the next six months.
(iv) Digestion: This is a form of maceration in which gentle heat is used during the process of extraction. It is used when the moderately elevated temperature is not objectionable, and the solvent efficiency of the menstruum is increased thereby.
(v) Infusion: An infusion is a dilute solution of the readily soluble constituents of crude drugs. Fresh infusions are prepared by macerating the solids for a short period of time with either cold or boiling water. The USP has not included infusions for some time.
(vi) Decoction: This once-popular process extracts water-soluble and heat-stable constituents from crude drugs by boiling in water for15 min. cooling, and passing sufficient cold water through the drug to produce the required volume.
Different Methods of New Advances Extraction Techniques:
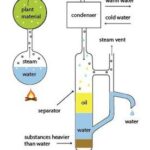
(i) Steam Distillation: To extract the essential oil, the plant material is placed into a still (very similar to a pressure cooker) where pressurized steam passes through the plant material. The heat from the steam causes globules of oil in the plant to burst and the oil then evaporates.
The essential oil vapour and the steam then pass out the top of the still into a water-cooled pipe where the vapours are condensed back to liquids. At this point, the essential oil separates from the water and floats to the top.
Now, this doesn't sound like a particularly complicated process, but did you know that it takes more than 8 million Jasmine flowers to produce just 2 pounds of jasmine oil? No wonder pure essential oils are expensive 16.
FIG. 1: STEAM DISTILLATION
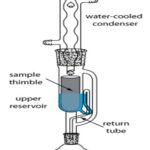
(ii) Soxhlet Extraction: The Soxhlet extraction method integrates the advantages of reflux extraction and percolation, which utilizes the principle of reflux and siphoning to continuously extract the herb with fresh solvent. Soxhlet extraction is an automatic continuous extraction method with high extraction efficiency that requires less time and solvent consumption than maceration or percolation. The high temperature and long extraction time in the Soxhlet extraction will increase the possibilities of thermal degradation 17.
Soxhlet extraction, one of the most frequently used liquid-solid extraction methods developed in the late nineteenth century, is still routinely used for extraction of analytes from several food matrices.
However, among its disadvantage are:
- Its requirement for large volumes of solvent (10-200 ml for 1-100 g of tissue)
- Its use of highly purified polar and non-polar organic solvents. (dichloromethane, hexane-acetone, hexanedichloromethane)
- The long extraction time(18 h) as a result of slow analytic diffusion,
- Desorption from the sample matrix to the extraction fluid
- Dilution of the extracts obtained.
- Generation of dirty extracts that require extensive clean-up and
- Animal and fish tissue are first macerated then ground with sodium sulfate and silica to reduce the water content and rupture cell walls; this method results in higher concentrations of analytic than freeze-drying.
Extraction with non-polar solvents, such as n-alkanes, took a considerable time (> 6 h) and was not as effective as polar solvents, such as dichloromethane 18.
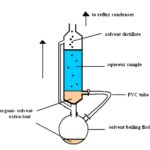
(iii) Solvent Extraction: Solvent extraction is usually used to recover a component from either a solid or liquid. The sample is contacted with a solvent that will dissolve the solutes of interest. Solvent extraction is of major commercial importance to the chemical and biochemical industries, as it is often the most efficient method of separation of valuable products from complex feedstock’s or reaction products. Some extraction techniques in involve partition between two immiscible liquids; others involve either continuous extractions or batch extractions.
Because of environmental concerns, many common liquid/liquid processes have been modified to either utilize benign solvents or move to more frugal processes such as solid-phase extraction. The solvent can be a vapour, supercritical fluid, or liquid, and the sample can be a gas, liquid or solid 19.
FIG. 2: SOXHLET EXTRACTION
FIG. 3: SOLVENT EXTRACTION
(iv) Liquid-solid Extraction: In liquid-solid extraction, a solvent (hydrophilic or hydrophobic, acidic, neutral, or basic) is added to a solid. Insoluble material can be separated by gravity or vacuum filtration, and soluble material is 'extracted' into the solvent. A sequence of solvents of varying polarity or pH can separate complex mixtures into groups. The filtered solution can be used as a solution (for example, to be injected into the GC/MS system), or the solvent can be evaporated to recover the solute(s) in powder or crystalline form 20.
(v) Solid-liquid Extraction: Solid-liquid extraction (leaching) is the process of removing a solute or solutes from a solid by using liquid solvent. Leaching is widely used in chemical industries where mechanical and thermal methods of separations are not possible or practical. Extraction of sugar from sugar beets, oil from oil-bearing seeds, production of a concentrated solution of a valuable solid material are typical industrial examples of leaching. The leaching process can be considered in three parts 21.
- Diffusion of the solvent through the pores of the solid
- Solute dissolves in the solvent.
- Transfer of the solution from porous solid to the main bulk of the solution.
(vi) Liquid-liquid Extraction: If the two phases are immiscible liquids, the technique is called liquid-liquid extraction. Usually, one phase is aqueous (hydrophilic), and the other is a hydrophobic organic solvent. A sequence of extractions with solvents of various solvents can be used to separate, with considerable efficiency, relatively complex mixtures 22.
A New Recent Advance in Extraction Techniques:
Microwave-Assisted Extraction (MAE): Microwave-assisted extraction (MAE) is also an extraction technique based on heating an organic solvent. The principle is rough that a sample and an appropriate solvent (or solvent mixtures) are put in a vessel, which is then pressurized and heated by microwaves. After typically 5 to 20 min the extraction is complete, and the vessels are allowed to cool down before removing the sample/solvent mixture. The solvent must be filtered to remove sample particles prior to the analysis of the extracted components. See figure 5 for a schematic of MAE equipment.
FIG. 4: MICROWAVE-ASSISTED EXTRACTOR
MAE is a more manual technique than PLE as it is performed in batch mode. However, many samples can be processed at the same time. Another feature of MAE is that the heating of the solvent is fast, it goes from inside the sample and outwards, and the heating capability depends on the microwave absorbing properties of the solvent. Polar solvents such as acetone will absorb microwave energy efficiently, as they have molecules with permanent dipole moments that can interact with microwaves.
Non-polar solvents such as hexane will not be heated when exposed to microwaves but can instead be used in immixtures with polar solvents in order to obtain the desired heating properties. Some common solvent mixtures that have been used in MAE are acetonitrile/methanol, hexane/acetone, ethyl acetate/cyclohexane, and isooctane/acetone 23. MAE can be considered as selective methods that favour polar molecules and solvents with high dielectric constant Table 1.
TABLE 1: POLAR MOLECULES AND SOLVENTS WITH HIGH DIELECTRIC CONSTANT
| Solvent | Dielectric constant (200c) |
| Hexane | 1.89 |
| Toluene | 2.4 |
| Dichloromethane | 8.0 |
| Acetone | 20.7 |
| Ethanol | 24.3 |
| Methanol | 32.6 |
| Water | 78.5 |
- Strength and Limitation: This technique reduced extraction time and solvent volume as compared to conventional method (maceration & Soxhlet extraction). Improved recoveries of analytes and reproducibility were observed in MAE method but with caution of using proper conditions to avoid thermal degradation 24.
However, this method is limited to small-molecule phenolic compounds such as phenolic acids (gallic acid and ellagic acid), quacertin, isoflavin, and trans-resveratrol because these molecules were stable under microwave heating conditions up to 100 °C for 20 min. Additional cycles of MAE (e.g., from 2 × 10 s to 3 × 10 s) resulted in a drastic decrease in the yield of phenolics and flavanones, mainly caused by the oxidation of compounds.
Tannins and anthocyanins may not be suitable for MAE as they were potentially subjected to degradation at high temperatures 25.
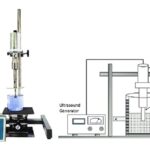
Ultrasound-assisted Extraction (UAE) or Sonication Extraction: UAE involves the use of ultrasound ranging from 20 kHz to 2000 kHz 26.
The mechanic effect of acoustic cavitation from the ultrasound increases the surface contact between solvents and samples and the permeability of cell walls. Physical and chemical properties of the materials subjected to ultrasound are altered and disrupt the plant cell wall, facilitating the release of compounds and enhancing mass transport of the solvents into the plant cells 27. The procedure is simple andrelatively low-cost technology that can be used in both small and large scale of phytochemical extraction.
- Strength and Limitation: The benefits of UAE are mainly a due reduction in extraction time and solvent consumption. However, the use of ultrasound energy more than 20 kHz may have an effect on the active phytochemicals through the formation of free radicals 24, 26.
FIG. 5: UAE
Accelerated solvent extraction (ASE): ASE is an efficient form of liquid solvent extraction compared to maceration and Soxhlet extraction as the method use a minimal amount of solvent. Sample is packed with inert material such as sand in the stainless steel extraction cell to prevent sample from aggregating and block the system tubing 28. Packed ASE cell includes layers of sand sample mixture in between cellulose filter paper and sand layers.
FIG. 6: ASE
This automated extraction technology is able to control temperature and pressure for each individual sample and requires less than an hour for extraction. Similar to other solvent technique, ASE also critically depends on the solvent types. Cyclohexane acetone solution at the ratio of 6:4 v/v with 5-minute heating (50°C) showed to yield the highest bixin from Bixa orellana with 68.16% purity. High recoveries (~94%) of flavonoids from Rheum palmatun were observed using 80% aqueous methanol by ASE, suggesting the suitability of this method for quality control evaluation 29.
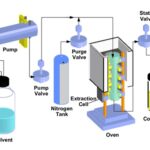
Supercritical Fluid Extraction (SFE): Supercritical fluid (SF) or also called as dense-gas, is a substance that shares the physical properties of both gas and liquid at its critical point. Factors such as temperature and pressure are the determinants that push a substance into its critical region. SF behaves more like gas but has the solvating characteristic of a liquid. An example of SF is CO2 that becomes SF at above 31.1 °C and 7380 kPa. Interest in Supercritical-CO2 (SC-CO2) extraction due to excellent solvent for nonpolar analytes and CO2 is readily available at low cost and has low toxicity. Even though SC-CO2 has poor solubility for polar compounds, modifications such
as adding a small amount of ethanol and methanol enable it to extracts polar compounds. SC-CO2 also produces analytes at concentrate form as CO2 vaporizes at ambient temperature. SC-solvents strength can be easily altered by changing the temperature, pressure or by adding modifiers that lead to reduce extraction time. Optimization of SC-CO2on Wadelia calendulacea achieved its optimum yield at 25 MPa, 25 ºC temperature, 10% modifier concentration, and 90 minute extraction time. A major drawback of this method is the initial cost of the equipment is very high 30.
FIG. 7: SFE
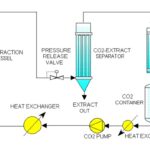
Pressurized Liquid Extraction: The PLE™ (Pressurized Liquid extraction) works similar to Soxhlet extraction, with the exception that during the extraction process, the solvents inside the PLE™ extraction cartridge are near their supercritical region, which has high extraction properties. In the physical region the high temperature makes possible the high solubility and high diffusion rate while the high pressure keeps the solvent below its boiling point. At high pressures and high temperatures, the solvents penetrate the solid samples at a much higher rate permitting a fast and efficient extraction process with minimal solvent usage. To operate the PLE™, 5 to 100 grams of the sample is mixed with sodium sulfate, loaded in the extraction cartridge, and capped with two disposable filtration end fittings. The extraction cartridges are clamped inside the unique and easy to use PLE™ cartridge seal cups. Upon pressing the start key, the HPLC pump dispenses the choice of organic solvents, such as Hexane, DCM, Toluene, etc., into the extraction cartridges. The PLE™ control system then starts the pressurization and heating of the samples. The pressure is maintained at 1500-3000 PSI and at temperature 70-200 ºC degrees. Depending on the method, the extracted solvent containing target analytes is then transferred to the Power-Prep for column clean up or collected in collection vessels 31. For rapid and efficient extraction of analytes from solid matrices such as plant materials, extraction temperature is an important experimental factor because elevated temperatures could lead to significant improvements in the capacity of extraction solvents to dissolve the analytes, in the rates of mass transport, and in the effectiveness of sample wetting and matrix penetration, all of which lead to an overall improvement in the extraction and desorption of analytes from the surface and active sites of solid sample matrices. To achieve all these advantages, however, elevated pressure is needed to maintain the extraction solvents as liquids at high temperatures (usually above their boiling points); this can be accomplished by use of a modern extraction and sampling technique known as pressurized- liquid extraction (PLE) or, more commonly, by its trade name (accelerated solvent extraction).
Fig. 8: PLE
PLE emerged in the mid-nineteen-nineties, but it is surprising to find it has rarely been applied to the extraction/analysis of plant materials. Benthin et al. were among the first to conduct a comprehensive study on the feasibility/usefulness of applying PLE in medicinal herb analysis. In their study PLE extracts from a selection of representative herbs were compared with extracts obtained according to pharmacopoeia monographs; their results indicated that PLE is often superior to other extraction methods currently used in crude herb analysis interns of recovery, extraction time, and solvent consumption (i.e. for all the herbs studied, a significant saving in time and solvents was realized and extraction recoveries of the analytes were equivalent or higher) 31.
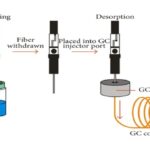
FIG. 9: SPME
Solid-Phase Micro Extraction (SPME): Solid-phase microextraction is the potential answer for many problems encountered in trace analysis. Solid-phase microextraction is a solvent-free equilibrium extraction method that, with proper calibration, can allow quantitative determinations of organic pollutants at very good sensitivity (usually low to mind part per trillion) without the use of any organic solvents. The method can require as little as 30 min for sample preparation, and analytical separations are easily automated and is ideally suited for use in the field. The method, invented in the early nineties by Prof. JanuszPawliszyn from the University of Waterloo in Ontario, Canada, utilized a small segment of fused silica fiber coated with appropriate material and mounted on the syringe-like device for extraction of analyses from various matrices and introduction to a chromatographic system. No solvents are used in the process. Analytic extraction and pre-concentration are combined in a single step. SPME has been commercialized by Supelco, Varian, and Leap Technologies. Since its introduction, it has found numerous applications in the analysis of numerous compounds in a variety of matrices. A miniaturized version of SPE, where the sorbent phase is coated on the outside of a fiber or the inside of a tube, known as SPME, has shown increasing applicability. One review (concentrated on the passive sampling of gas-phase contaminants with SPME 32.
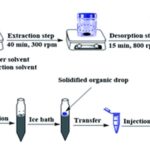
Stir-Bar Sportive Extraction (SBSE): Recently, a novel approach for sample enrichment in an aqueous solution has been covered with a thick layer of polydimethylsiloxane (PDMS) by magnetically stirring the bar in the sample solution; the components are enriched in the PDMS phase.
After this concentration step, analytes are thermally desorbed from the stir bar online with GC-MS; the major advantage of this technique are ease of use, improved sensitivity, high accuracy of analysis, and reduced risk of contamination.
FIG. 10: SBSE
CONCLUSION: Medicinal plant research is aimed at the isolation and identification of naturally occurring substances. Chemical analysis of extracts from plant material will play a central role in the development and modernization of herbal medicine. The majority of extraction procedures for the determination of plant metabolites are developed in such a way that the final extract introduced into the GC and HPLC columns contains only the analytes with all interferences removed. The modern techniques for extraction of herbal plant secondary metabolites include solid phase microextraction, supercritical-fluid extraction, pressurized liquid extraction, micro-wave-assisted extraction, solid-phase extraction, and surfactant-mediated extraction. SFE seems to offer unique advantages in the extraction of medicinal plants, high selectivity, minimum degradation of thermally labile analytes, and elimination of the use of hazardous organic solvents (e.g., use of pure CO2 as the extractant). The main drawbacks of SFE, on the other hand, such as difficulties in extracting polar compounds and high susceptibility to matrix effects, are problematic in the extraction of herbal materials. As plant matrices are highly complex, factors such as the water content and particle size of the matrix and strong analyte–matrix interactions, etc., can severely limit the capacity of SFE to effect high extraction efficiency and rapid kinetics especially for polar analytes.
In recent years, the use of the microwave for the extraction of constituents from plant material has shown tremendous research interest and potential. Conventional techniques for the extraction of active constituents are time and solvent-consuming, thermally unsafe, and the extraction step limits the analysis of numerous constituents in plant material.
MAE has risen rapidly in the last decade, and for most applications, it has proven to be effective in all aspects compared to traditional extraction techniques. The need for the development of existing methods of separation and the introduction of new techniques of high resolution and effectiveness must be seriously felt. In all probability, such developments will give rise to the discovery of new effective compounds from phytopharmaceutical sources. More research is needed to improve the understanding of extraction mechanism, remove technical barriers, improve the design and scale-up of the novel extraction systems for their better industrial applications.
ACKNOWLEDGMENT: Mr. Vipin Kesharwani was an Assitant Professor of my college. ARK College of Pharmacy Kaushambi, and also my Review paper guide. Kesharwani sir gives me Excellent support in this review paper. Mr. Nikhil Kushwaha worked as my senior. He helped me in every step of my unit process and participated in the sequence alignment, and drafted the manuscript with me.
I am Shashank Agrahari carried out all work related to this review paper with the help of my Kesharwani sir and my senior Kushwaha Nikhil, gives good support in this manuscript.
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Tonthubthimthong P, Chuaprasert S, Douglas P and Luewisutthichat W: Supercritical CO2extraction of nimbin from neem seeds an experimental study. J Food Eng 2001; 47: 289-93.
- Romanik G, Gilgenast E, Przyjazny A, Namiesnik J and Kaminski M: Methods of extraction. Article in press 2007: 32-54.
- Wong SK, Lim YY and Chan EW: Antioxidant properties of Hibiscus species variation, altitudinal change costal influence and floral colour change. J Tro For Sci 2009; 21: 307-15.
- Nair R, Kalariya T and Chanda S: Antibacterial activity of some selected Indian medicinal flora. Turk J Biol 2005; 29: 41-47.
- Khan M and Wassilew SW: Natural pesticides from the neem tree and other tropicalplants. (Eds) Schmutterer, H. and Asher, K. R. S., Germany: Digitalverlag GmbH 1987; 645-50.
- Singh N and Gupta M: Effect of ethanolic extract of Syzygium cumuni seed powder onpancreatic islets of alloxendiebetic rats. Indian J Exp Biol 2007; 45: 861-67.
- Kumar A, Krishan MRV, Aravindan P, Jayachandran T, Deecaraman M and Ilavarasan R: Syzygium cumuni Padmanabhan A: Anti-diabetic activity of Syzygium cumini seed and its isolate compounds against streptozotocin induced diabetic rats. J Med Plants Res 2008; 2: 246-49.
- Kumar A, Krishan MRV, Aravindan P, Jayachandran T, Deecaraman M, IlavarasanR, Kumar RM and Padmanabhn A: Anti-inflammatory activity of Syzygium cumini Afr J Biotechnol 2008; 7: 941-43.
- Jagetia GC, Venkatesh P and Baliga MS: Influence of seed extracts of cumini on miceexposed to different doses of γ radiation. J Radiat Res 2005; 46: 59-65.
- Vazquez E, Ciscar R and De la Guardia M: Anal Chim Acta 1997; 344: 241-49.
- Evans WC: General methods associated with the phyto-chemical investigation of herbalproducts. In Trease and Evans Pharmacognosy (15 ed.), New Delhi: Saunders (Elsevier). 2002; 137-48.
- Jadhav D, Rekha BN, Gogate PR and Rathod VK: Extraction of vanillin from vanilla pods: Acomparison study of conventional Soxhlet and ultrasound assisted extraction. J Food Eng 2009; 93: 421-26.
- Kothari V and Seshadri S: Antioxidant activity of seed extract of Annona squamosa and Carica papaya. Nutri Food Sci 2010; 40: 403-08.
- Esteban JL, Martinez-Castro I, Morales R, Fabrellas B and Sanz J: Chromatographic 1996; 43: 63
- Hatano T, Ogawa, N, Kira R, Yasuhara T and Okuda T: Tannins of cornaceous plants. Chem Pharm Bull 1989; 37: 2083-90.
- Li F, Xiong Z, Lu X, Qin F and Li X: Strategy and chromatographic technology of quality control for traditional Chinese medicines. Chinese J Chromatogr 2006; 24: 537-44.
- Wei Q, Yang GW, Wang XJ, Hu XX and Chen L: The study on optimization of Soxhlet extraction process for ursolic acid from Cynomorium. ShipinYanjiu Yu Kaifa. 2013; 34: 7 85–8.
- Luque de Castro MD and Garcia-Ayuso LE: Soxhlet extraction of solid matrices: an outdated technique with apromising innovative future. Anal Chim Acta 1998; 369: 1-10.
- Smith RM: Before the injection- modern methods of sample preparation for Techniques of preparing plantmaterial for chromatographic separation and analysis. J Chromatogr A 2003; 1000: 3-27.
- Zuloaga O, Etxebarria N, Fernandez LA and Madariaga JM: Optimization and comparison of microwave assisted extraction and Soxhlet extraction for the determination of polychlorinated biphenyls in soil samples using an experimental design approach. Talanta 1999; 50: 345-57.
- Sanghi R and Kannamkumarath SS: Comparison of extraction methods by Soxhlet, sonicator and microwave inthe screening of pesticide residues from solid matrices. J. Anal. Chem. 2004; 59:11: 1032-36.
- Karthikeyan S Balasubramanian R and See SW: Optimization and validation of a low temperature microwave assisted extraction method for analysis of polycyclic. 2004: 52; 111-12.
- Williams OJ, Raghavan GSV, Orsat V and Dai: J M. J. Food Biochem 2004; 28: 113-22.
- Kaufmann B and Christen P: Recent extraction techniques for natural products: microwave-assisted extraction and pressurized solvent extraction. Phy Anal 2002; 13: 105-13.
- TrushevaB, Trunkova D and Bankova V: Different extraction methods of biologically active components from propolis: a preliminary study. Chem Cent J 2007; 13.
- Handa SS, Khanuja SPS, Longo G and Rakesh DD: Extraction Technologies for Medicinal and Aromatic Plants, (1stedn), no. 66. Italy: United Nations Industrial Development Organization and the International Centre for Scienceand High Technology 2008; 134-35.
- Dhanani T, Shah S, Gajbhiye NA and Kumar S: Effect of extraction methodson yield, phytochemical constituents and antioxidant activity of Withaniaso mnifera. Arab J Chem 2013; 115-14.
- Rahmalia W, Fabre JF and Mouloungui Z: Effects of Cyclohexane/AcetoneRatio on Bixin Extraction Yield by Accelerated Solvent Extraction Method. Procedia Chem 2015; 14: 455-64.
- Tan, Ji T,Jiang G, Hu F Simultaneous identification and quantification of five flavonoids in the seeds of Rheum palmatum by using accelerated solvent extraction and HPLC-PDA-ESI/MSn. Arab J Chem 2014; 115-18.
- Patil, Sachin BS, Wakte PS, Shinde DB. Optimization of supercriticalfluid extraction and HPLC identification of wedelolactone from Wedelia calendulacea by orthogonal array design. J Adv Res 2013; 5: 629-35.
- Yamabe N, Kang KS, Matsuo Y and Tanaka T: Wedelia calendulacea Yokozawa T: Identification of antidiabetic effect of iridoid glycosidesand low molecular weight polyphenol fractions of Corni Fructus, a constituents of Hachimi-jio-gan,instreptozotocin-induced diabetic rats. Biol. Pharm. Bull. 2007; 30: 1289-96.
- Letellier M, Budzinski H, Charrier L, Capes S and Dorthe AM: Optimization by factorial design of focused microwave assisted extraction of polycyclic aromatic hydrocarbons from marine sediment. J Anal 1999; 364: 228-37.
How to cite this article:
Agrahari S, Kesharwani V and Kushwaha N: A review on modern extraction techniques of herbal plants. Int J Pharmacognosy 2021; 8(5): 177-88. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(5).177-88.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
177-188
952
1342
English
IJP
S. Agrahari *, V. Kesharwani and N. Kushwaha
ARK College of Pharmacy, Rakasarai Sarai Akil Kaushambi, Uttar Pradesh, India.
shashank06091998@gmail.com
31 March 2021
22 May 2021
26 May 2021
10.13040/IJPSR.0975-8232.IJP.8(5).177-88
31 May 2021