A REVIEW ON BUCCAL FILM LOADED WITH LACOSAMIDE FOR THE TREATMENT OF PARTIAL ONSET SEIZURES
HTML Full TextA REVIEW ON BUCCAL FILM LOADED WITH LACOSAMIDE FOR THE TREATMENT OF PARTIAL ONSET SEIZURES
Nilam Panjiyar *, Ankita Pokhriyal, Pranshu Tangri and Arvind Negi
GRD Institute of Management & Technology, Uttarakhand University, Dehradun, Uttarakhand, India.
ABSTRACT: Despite the introduction of new antiepileptic medicines (AEDs) over the past two decades, more than 30% of epilepsy patients still have refractory disease. Combined administration of AEDs or the use of novel AEDs is the most popular therapeutic choice when surgical therapy cannot be provided, even though a tiny number of these resistant individuals may become seizure free when a new AED is added. Lacosamide (LCM), which has novel modes of action and advantageous safety profiles, is the most recent AED to receive approval in Europe and the USA. This article discusses the efficacy and safety of LCM as determined by phase II and III clinical trials, as well as its molecular mechanisms of action and pharmacokinetic characteristics. It is also explored how LCM compares to other AEDs that are currently on the market.
Keywords: Antiepileptic medication, Epilepsy, Partial seizures, New anticonvulsant, lacosamide, Sodium channels, Slow inactivation
INTRODUCTION: Different routes can be used to give medications. The type of drug is usually a factor in the distribution system. Because of its convenience, the oral route of drug administration is considered the safest and most common method. However, if the patient requires a speedy beginning of action or some medications are not suited for oral administration, another delivery method is preferable 1, 2.
Buccal Film Delivery: The buccal route can be used to give medications with a very high first pass metabolism or poor solubility. After being absorbed via the mucosal membrane in the buccal cavity, the medication enters the bloodstream immediately.
Due to the absence of a protracted process of disintegration, dissolution, and first pass metabolism, buccal delivery has a quicker beginning of effect than oral delivery. The buccal route is usually utilized for sustained medication administration. Various polymers, some of which may be mucoadhesive or bioadhesive, are used in this drug delivery system. Tablets, patches, liquids, and semi-solids are delivered via the buccal route 3, 4, 5.
The oral mucosa differs from the skin and the mucous membranes of the digestive system by its shape. The oral mucosa is more permeable than the skin, but less permeable than the gastric and intestinal mucosa. Due to the presence of epithelium cells, the permeability of skin varies. For example, the lining of the gut is made up of only one layer of epithelial cells, whereas the buccal cavity has multiple layers of epithelial cells that prevent medications from passing through. The buccal cavity has many advantages over oral and other routes, despite the fact that the permeability is not so great there. The primary benefit of the buccal administration technique is avoiding the first-pass metabolism. Direct drug circulation to the systemic circulation occurs without passing through the hepatic portal. This route of administration makes it simple to stop the drug's delivery, and it also makes it simple to have sustained delivery. Despite several drawbacks, such as permeability issues, drug retention, and drug dilution from excessive saliva flow, this mode of medication delivery is generally considered to be effective. This may affect how well the medicine is absorbed and absorbed overall 6, 7, 8.
Benefits of Buccal Delivery:
- The key benefit is that it avoids first pass metabolism, making it preferable for medications with considerable first pass metabolism to be administered this way. The medications directly enter the systemic circulation after being absorbed through the mucosal barrier in the cavity.
- The commencement of effect occurs more quickly than with oral and topical routes.
- Convenient for patients.
- Due to first-pass metabolism, there is no medication loss.
- Drugs that may interact with gastrointestinal fluids can be simply delivered buccaly.
- As it is non-invasive in nature, the oral route is safe.
- Poorly permeable medicines can be increased by using a permeation enhancer.
- It is safer to use medications and excipients that are enzyme-prone.
- Pre-systemic drug metabolism is not possible 9, 10.
Drawbacks:
- The administration of drugs via this route is exceedingly challenging for those with extremely poor permeability and high molecular weight.
- Salivation is a biological condition that dilutes medications, which might impact how well they work in the body.
- The membrane could get slightly irritated.
- Use of paedratics is minimal.
- Its surface area, which ranges from 165 to 175 cm2, is quite modest 11, 12.
Limitations:
- During administration, it is forbidden to talk, eat, or drink.
- Limited drug administration occurs because of the reduced area of absorption.
- It's challenging to deliver two or more medications at once.
- It is impossible to give a very low permeability medication.
- Overexposure of the mucosal membrane can reduce its ability to absorb and can be uncomfortable while eating or drinking 12.
Lacosamide: Lacosamide, an antiepileptic medication (AED) with a variety of formulations, received its initial approval in 2008 for use as an adjunctive treatment for partial onset seizures (POS) in adults. In contrast to conventional sodium channel blockers, lacosamide specifically promotes delayed inactivation of sodium channels. This mechanism of action stabilizes hyperexcitable neuronal membranes, inhibits neuronal firing, and reduces long-term channel availability without affecting physiological function. Lacosamide has a well researched and advantageous pharmacokinetic profile, which includes a quick absorption rate, little to no interaction with cytochrome P-450 izoenzymes, and a low risk of drug-drug interactions. Three placebo-controlled, double-blind, randomized studies involving more than 1,300 patients were part of the clinical development of lacosamide. These trials each showed lacosamide's safety and effectiveness as an additional therapy for adults with POS when compared to placebo. Depending on the outcomes of trials examining its use as monotherapy for POS in adults, as a treatment for epilepsy in pediatric patients, and as an adjunctive therapy for uncontrolled primary generalized tonic-clonic seizures in people with idiopathic generalized epilepsy, lacosamide's clinical use may expand.
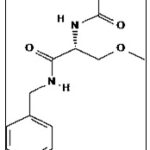
Chemistry: In 1996, researchers at the University of Houston made the discovery of lacosamide. They proposed the possibility that changed amino acids could be therapeutically advantageous in the management of epilepsy. It reduces the frequency and intensity of seizures by acting on the central nervous system (CNS).
Structure:
Molecular Formula: C13H18N2O3
Molecular Weight: 250.294 g/mol
Melting Point: 140-146°C
Nature: Lacosamide is a white to light yellow powder.
Solubility: Sparingly soluble in water and slightly soluble in acetonitrile and ethanol.
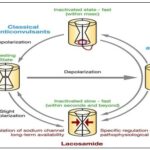
Mechanism of Action: a New way to Target Sodium Channels: The generation of action potentials by voltage-gated sodium channels, which play a crucial role in regulating neuronal excitability, is a well-established fact. The production of seizures in epilepsy is triggered by abnormal and repeated neural firing. The amount of sodium channels that can be activated determines how excitable the brain is. Fast inactivation, which occurs in milliseconds, and delayed inactivation, which occurs in seconds or minutes, are the two mechanisms that control the proportion of sodium channels that are open to activation. Consistent membrane depolarization brought on by repetitive neuronal firing, such as that seen in epilepsy, causes voltage-gated sodium channels to slowly inactivate Fig. 1 13, 14, 15.
FIG. 1: MECHANISM OF ACTION
The first AED discovered to be effective was lacosamide specifically potentiate the slow inactivation of sodium channels. It stabilizes hyperexcitable neuronal membranes and reduces repeated neuronal firing by targeting and enhancing the delayed inactivation of voltage-gated sodium channels (VGSCs). This mechanism differs greatly from that of typical sodium-channel blocking AEDs (SCB), which function by promoting VGSCs fast inactivation (e.g., carbamazepine [CBZ], phenytoin [PHT], lamotrigine [LTG]) 16.
Pharmacological Properties:
Pharmacodynamic Properties: The precise mechanism by which lacosamide exerts its antiepileptic effect in humans remains to be fully elucidated. It appears that the Lacosamide has a dual mode of action that is different from all other available AEDs. It’s mechanism of action is believed to reduce sodium channel over-activity by prolonging the longer lasting resting state (slow inactivation) of the channel, a different action compared with current sodium channel blocking drugs. This means that the lacosamide only affects neurons which are depolarized or active for long periods of time, typical of neurons at the core of an epileptic focus.
Pharmacokinetics Properties Absorption: Bioavailable lacosamide is completely absorbed after oral administration with negligible first- pass effect with a high absolute bioavailability of approximately 100%.
Distribution:
- Lacosamide is less than 15% bound to plasma protein.
- Lacosamide is demethylated to the major metabolite, O-desethyl-lacosamide, which has no known pharmacological activity.
Excreation:
- Lacosamide is primarily (95%) eliminated by renal excretion.
- Less than 0.5% eliminataed through feces.
Elimination Half-life:
- The elimination half-life of Lacosamide is approximately 13hours.
- The major metabolite, Odesethyl-lacosamide, has an elimination half-life of 15 to 23 hours.
Uses:
- It is used in the treatment of neuropathic pain.
- It is used in the treatment of partial onset seizures.
Side Effects:
- Depression
- Confusional State
- Insomnia
- Dizziness
- Headache
- Memory impairment
- Coordination abnormal
- Hyposthesia
- Disturbance in attention
- Vomiting
- Constipation
- Dry mouth
- Diarrhoea
- Fatigue
- Irritability
Special Warning & Precaution For Use:
- Suicidal ideation & behaviour
- Cardiac rhythm & conduction
- Dizziness
Interaction with Other Medicinal Products & Other Form of Interaction:
- Lacosamide should be used with caution in patients treataed with medicinal products known to be associated with PR prolongation (e.g. carbamazepine, lamotrigine, eslicarbazepine, pregabalin) and in patient treated with class I antiarrhythmics.
- Lacosamide does not inhibit or induce CYP2C19 and 3A4 to a clinically relevant extent.
- Caution is recommended in concomitant treatment with strong inhibitors of CYP2C9 (e.g. itraconazole, ketoconazole, ritonavir, clarithromycin), which may lead to increased systemic exposure of lacosamide.
- Interaction trials showed that lacosamide had no effect on the pharmacokinetics of digoxin. There was no clinically relevant interaction between lacosamide and metformin.
- Co-administration of warfarin with lacosamide does not result in clinically relevant change in the pharmacokinetics and pharmacodynamics of warfarin.
CONCLUSION: Lacosamide is being studied for use as a monotherapy and as an adjuvant medication for adult patients experiencing partial-onset seizures. It is now being scientifically tested as a seizure therapy. It differs from other AEDs like carbazepine and phenytoin by having a special sodium channel mechanism. Lacosamide has a favorable pharmacokinetic profile, has no drug-drug interactions, and is offered in a variety of forms, all of which contribute to its reputation for being simple to use. In the treatment of POS, it has demonstrated efficacy and safety profiles through comprehensive research. The success of lacosamide in POS emphasizes the importance of the suggested mode of action, which involves modulation of the delayed sodium channel inactivation as a therapeutic target for epilepsy and persons suffering from this disease. It could result in the development of novel therapeutic alternatives for those suffering from this disease.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Smart JD: Buccal drug delivery. Expert Opinion on Drug Delivery 2005; 2(3): 507–517.
- Vyash and Khar: Control and novel drug delivery 351-58
- Madhusudan Y and Rao AV: Jithan Advances in drug delivery 1: 139-198.
- Pramod S, Vijay S and Chandrkant M: Buccal film : an innovative dosage form designed to improve patient compliance. Int J Pharm Chem Sci 2012; 1(4): 1262–78.
- Hao J and Heng PWS: Buccal delivery systems. Drug Dev Ind Pharm 2003; 29(8): 821–32.
- James swarbrick, Encyclopedia of Pharmaceutical Technology 1: 2664-73.
- Michael J. Rathbone, Ian W. Kellaway, Gilles Pochel and D. Duchene: Modified released drug delivery, Copyright by Marcel Dekker 350-60.
- Roglic G, Unwin N and Bennett PH: The burden of mortality attributable to diabetes: Realistic estimates for the year 2000. Diabetes Care 2005; 28: 2130–5.
- Morrish NJ, Wang SL and Stevens LK: Mortality and causes of death in the WHO multinational study of vascular disease in diabetes. Diabetologia 2001; 44: 14–21.
- Ross R and Harker L: Hyperlipidemia and atherosclerosis. Science 1976; 4258: 1094-100. 10.1126/science.822515
- Brahmankar DM and Jaiswal SB: Biopharmaceutics and pharmacokinetics a treatise, second edition., Vallabhprakashan publications, Delhi 2009; 397-470.
- Lachman L and Lieberman HA: The theory and practice of industrial pharmacy, third edition., Varghese Publishing House, Bombay 1990; 197- 243.
- Beyreuther BK, Freitag J and Heers C: Stohr. Lacosamide: a review of preclinical properties. CNS Drug Rev 2007; 13: 21– 42.
- Oliva M, Berkovic SF & Petrou S: Sodium channels and the neurobiology of epilepsy. Epilepsia 2012; 53: 1849– 1859.
- Kandel ER, Schwartz JH & Jessell TM: Principles of Neural Science, 4th ed. New York: McGraw-Hill 2000.
- Kellinghaus C: Lacosamide as treatment for partial epilepsy: mechanisms of action, pharmacology, effects, and safety. Ther Clin Risk Manag 2009; 5: 757–766. [Taylor &
- Francis Online], [Google Scholar] UCB. Vimpat. In: ®(lacosamide), ed. prescribing information. US: Smyrna, GA; 2011
How to cite this article:
Panjiyar N, Pokhriyal A, Tangri P and Negi A: A review on buccal film loaded with lacosamide for the treatment of partial onset seizures. Int J Pharmacognosy 2024; 11(2): 36-40. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(2).36-40.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
36-40
580 KB
841
English
IJP
Nilam Panjiyar *, Ankita Pokhriyal, Pranshu Tangri and Arvind Negi
GRD Institute of Management & Technology, Uttarakhand University, Dehradun, Uttarakhand, India.
Panjiyarnilam39@gmail.com
23 September 2023
13 February 2024
27 February 202
10.13040/IJPSR.0975-8232.IJP.11(2).36-40
29 February 2024