A PHARMACOLOGICAL REVIEW: PASSIFLORA SPECIES
HTML Full TextA PHARMACOLOGICAL REVIEW: PASSIFLORA SPECIES
Saurabh Tiwari, Shweta Singh *, Shalini Tripathi and Sunil Kumar
Rameshwaram Institute of Technology and Management, Lucknow - 227202, Uttar Pradesh, India.
ABSTRACT: Plants have been the basis of many traditional medicines throughout the world for thousands of years and have continued to provide new remedies to mankind. They are one of the most abundant sources of bioactive compounds. The genus Passiflora L. comprises about 520 species of dicotyledonous plants in the family Passifloraceae. Passiflora incarnata and P. alata, also commonly known as passion flower, are two species of a perennial climbing vine with beautiful exotic flowers and delicious fruit that grow worldwide, preferring subtropical, frost-free climates. They are native to the tropical and semi-tropical United States (Virginia to Florida and as far west as Texas), Mexico, Central American, and from Brazil to Paraguay through northern Argentina. It is used extensively for the treatment of some diseases like anxiety, insomnia, convulsion, sexual dysfunction, cough, and cancer. Passionflower is currently official in the national Pharmacopeia’s of Egypt, France, Germany, and Switzerland, and also monographed in the British Herbal Pharmacopoeia and the British Herbal Compendium, the ESCOP monographs, the Commission E, the German Standard Licenses, the German Homeopathic Pharmacopoeia, and the Homeopathic Pharmacopoeia of the United States. The present article including the detailed exploration of pharmacological properties of P. incarnata is an attempt to provide a direction for further research.
| Keywords: |
Passifloraceae, Passiflora incarnata, Insomnia, Convulsion
INTRODUCTION: The genus Passiflora consists of 500 species that are mostly found in warm and tropical regions. Passiflora comes from the Latin word “Passio” that was first time discovered by Spanish discoverers in 1529 and was described as a symbol for “Passion of Christ” 1, 2. The passion flowers or passion vines (Passiflora) have a genus of about 400 species of flowering plants and the largest in the family of Passifloraceae 3, 4. They are mostly vines, with some being shrubs, and a few species being herbaceous.
The species of this genus are distributed in the warm temperate and tropical regions of the world, but they are much rarer in Asia, Australia, and tropical Africa. The medical utility of very few species of Passiflora has been scientifically studied 5. Passionflower extracts have been classified into several categories of chemical activities like an anxiolytic, spasmolytic, hypnotic, sedative, narcotic and anodyne 6.
These extracts are part of a treatment that has successfully treated outpatients with adjustment disorder and anxious mood 7. Many species have been found to contain beta-carboline harmala alkaloids with anti-depressant properties. The flower and fruit have only traces of these chemicals, but the leaves and the roots are often more potent and have been used to enhance the effects of mind-altering drugs.
Once dried, the leaves can also be smoked. Passiflora quadrangularris is used by traditional healers for snake bites. Snake bites cause blood clotting and eventually burst blood vessels around the bite; this is known as hemorrhaging 8.
Classification:
Botanical source : Passiflora incarnata L.
Family : Passifloraceae
Genus : Passiflora L.
Species : Passiflora incarnata L.
Identifying Characteristics: The stems wiry; three-lobed leaves, serrate; pale pink flowers 5-7 cm across; fruits ovoids or globose, 3-5 cm long Fig. 1-3 9.
Bioactive Compounds: The chemical content of Passiflora species is also not well delineated. Investigators have differed on whether its sedative effects are due to indole alkaloids such as harmane, harmaline, and harmol; flavonoids such as apigenin, luteolin, and scopoletin; or an isolated trisubstituted benzoflavone.
Also, recently it was determined that Passiflora contains more gamma-aminobutyric acid (GABA) than 20 other plants examined 10. One of six alkaloids isolated from P. incarnata has been called "passiflorine," and is believed by some to be the plant's active compound, although the Agricultural Research Service's web site describes passiflorine as inactive. The Chemical Abstract Service's database's only similar entry is "passiflorine," a steroid-like molecule found in P. edulis stems and leaves that is not an alkaloid. Passionflower extracts consist of fresh or dried aerial parts of P. incarnata or P. alata, collected during the flowering and fruiting period. Botanical identity is confirmed by thin-layer chromatography, microscopic and macroscopic examination, and organoleptic evaluation.
Extracts contain 0.825% apigenin and luteolin glycosides, vitexin, isovitexin and their C glycosides, kaempferol, quercetin, and rutin; indole alkaloids (0.01%), mainly harman, harmaline, harmine; coumarin derivatives; cyanogenic glycosides (gynocardin); amino acids (including GABA); fatty acids (linoleic and linolenic); gum; maltol; phytosterols (stigmasterol); sugars (sucrose); and a trace of volatile oil 11, 12, 13, 14, 15, 16.
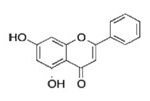
Chrysin: C15H10O4 (5, 7-dihydroxy-2-phenyl-(9CI)
Chrysin is a naturally occurring flavone chemically extracted from the blue passion flower (Passiflora caerulea). Chrysin acts as an aromatase inhibitor supplement to bodybuilders and athletes. It has been shown to induce an anti-inflammatory effect, most likely by inhibition of COX-2 expression via IL-6 signaling 17.
In rodent in-vivo studies, chrysin was found anxiolytic 18, 19. In herbal medicine, it is recommended as a remedy for anxiety, but there are no controlled data in humans’ available 20. Chrysin exhibited an anxiolytic effect, which was slowed by an increase in locomotor activity in rats when injected at 1 mg/kg. This effect was linked to GABA benzodiazepine receptors in the brain because the anxiolytic effect was blocked by an injection of Flumazenil, which is a benzodiazepine antagonist 21. Chrysin and apigenin have been shown to inhibit the growth of breast carcinoma cells 22, human thyroid cancer cells 23 and human prostate tumors 24. Apigenin is considered anti-mutagenic because it reduces the effects of mutagens in rats 25.
Benzoflavone:
The β-Naphthoflavone, also known as 5, 6-benzoflavone, is a potent agonist of the aryl hydrocarbon receptor and an inducer of detoxification enzymes as cytochromes P450 (CYPs) and uridine 5'-diphosphate glucuronosyl-transferases (UGTs) 26. β -Naphthoflavone is a putative chemopreventive agent 27.
Harmala Alkaloids: C13H12N2O (7-Methoxy-1-methyl-9H-pyrido [3, 4-b] indole)
The Passiflora family contains small amounts of harmala alkaloids, harmane (passaflorine), and possibly harmine (telepathine), harmaline, harmol, and harmalol. The presence of the last four in P. incarnata is disputed 28 because they are contained in only very small amounts (0.01% or less) 29. Furthermore, they have been identified as stimulants and monoamine oxidase inhibitors 30, 31, 32 which would give antidepressant rather than sedative effects. Wild rue (Peganum harmala) which contains significant amounts of these substances (and after which they were named) is used therapeutically as a stimulant rather than a sedative. The harmala alkaloid which is the active principle in Passiflora might also be a cause for concern for kidney toxicity, as these substances are toxic to the kidneys 33.
Extracts of the aerial parts of P. incamata L. contain the beta-carbolines: harman, hamun, hannalin, harmol, and harmalol, along with an aroma compound, maltol 34. Beta-carbolines, like those of P. incamata L., induce voluntary ethanol intake in rats 35. Some people may be interested in the fact that harman has been identified in beer; wine 36 and cigarette smoke 37. Beta-carbolines have been found to prevent neuron damage to the brain mitochondria of dopamine-induced mice by acting as an antioxidant and scavenging hydroxyl radicals 38. Harman and related compounds are mutagenic and have become more mutagenic after nitrosarion occurs in the acidic conditions of the stomach. Harman acts as a vasorelaxant (something that reduces inflammation or edema); it functions by releasing GABA, serotonin and noradrenaline 39.
Glycosides:
Orientin: Glycosides are molecules in which a sugar is bound to a non-carbohydrate moiety, usually a small organic molecule. Glycosides play numerous essential roles in living organisms. Many plants store chemicals in the form of inactive glycosides which can be activated by enzyme hydrolysis 40.
Leaf and stem material of P. edulis contain the new cyanogenic glycosides (2R)–α-allopyranosyloxy-2-phenylacetonitrile and (2S)–α-Dallopyranosyloxy-2-phenylacetonitrile, along with smaller amounts of (2R)–prunasin, (2S)-sambunigrin. Many different types of glycosides are present in passion flowers such as apigenin, homoorientin, 7-isoorientin, isoschaftoside, isovitexin, kaempferol, lucenin, luteolin, norientin, passiflorine (named after the genus), quercetin, rutin, saponaretin, saponarin, shaftoside, vicenin, and vitexin.
In some cases, this glycoside occurs with simple β-D-glucopyranosides: tetraphyllin A, deidaclin, tetraphyllin B, volkenin, epivolkenin and taraktophyllin. P. citrine contains passicapsin, a rare glycoside with the 2, 6- dideoxy- β -D-xylo-hexopyranosyl moiety, while P. herbertiana contains tetraphyllin A, deidaclin, epivolkenin and taraktophyllin, P. discophora tetraphyllin B and volkenin, and P. xviolacea tetraphyllin B 41. Some other glycosides present in Passiflora are the hydrocarbon nonacosane and the anthocyanidin pelargonidin-3-diglycoside 42.
Passiflora morifolia extracts contain the cyanohydrins glycoside and linamarin 43. Linamarin causes an increase of lactic acid and total cholesterol in the liver and brain in addition to the depletion of brain phospholipids in rabbits 44.
Isoorientin: (Luteolin-8-C-glucoside)
Orientin is a flavone, a chemical flavonoid-like compound found in the passion flower, the Açai palm, and Anadenanthera peregrina. Orientin is also reported to be in millets. Isoorientin (or homoorientin) is the luteolin-6-C glucoside. It can be isolated from the passion flower, Vitex negundo, the Açaí palm and Swertia japonica 45.
Other Organic Compounds: Passionflower contains many alkaloids, flavonoids as well as many organic compounds such as organic acids. This genus is rich in formic, butyric, linoleic, linolenic, malic, myristic, oleic and palmitic acids as well as phenolic compounds, and the amino acid α-alanine. Some species contain esters such as ethyl butyrate, ethyl caproate, n-hexyl butyrate and n-hexyl caproate which give the fruits their flavor and appetizing smell. Sugars, contained mainly in the fruit, are mostly d-fructose, d-glucose and raffinose. Among enzymes, Passiflora was found to be rich in catalase, pectin methylesterase and phenolase. Apart from glycosides, phenols, and alkaloids, various miscellaneous phytoconstituents which were also reported to be in P. edulis include, Edulans I and II 46 and pectins 47. The pectin fractions contain mainly sugars (83 - 85%, w/w). However, non-sugar components such as nitrogen-containing material (3 -8%, w/w) and ash (5 - 7%, w/w) are also present in these fractions 48.
Pharmacology:
Cannabinoids Reversal: The newly reported benzoflavone (BZF) moiety from the plant P. incarnata (Linn) has been evaluated in light of traditional reports on the use of this plant in breaking down cannabis addiction. In the modern or allopathic system of therapeutics, there has been no suitable remedy to combat the severe withdrawal effects of various cannabis products, including marihuana, marijuana, bhang, hashish, ganja, etc., the world-wide consumption of which has attained alarming proportions, especially among the younger generation. It has been reported that the BZF of P. incarnata, when administered concurrently with cannabinoids, prevented the development of tolerance and dependence of cannabinoids in mice. Even an acute administration of the BZF significantly blocked the expression of withdrawal effects in cannabinoid dependence. So these studies suggested that the BZF may have a beneficial role in cannabinoids reversal 49.
Nicotine Reversal: Some of the pharmacological studies on the BZF moiety also confirmed that the BZF moiety isolated from P. incarnata was very effective in countering the menace of addiction‑prone substance nicotine in laboratory animals. In light of various reports mentioning the usefulness of P. incarnata in tobacco addiction, studies have been performed by using the bioactive BZF moiety isolated from the aerial parts of P. incarnata. So these studies, although preliminary, suggested that the BZF may have value in treating nicotine addiction 50.
Alcohol Withdrawal: A BZF moiety has been reported recently to be responsible for the multifarious CNS effects of P. incarnata Linn. In the light of the established usefulness of the BZF moiety in counteracting the withdrawal effects of substances like cannabinoids and nicotine by the authors, the bioactive BZF moiety has been tested in mice treated with an addictive dose of ethyl alcohol, to evaluate its effectiveness in countering alcohol dependence. The chronic administration of P. incarnata with alcohol had better preventive effects than the single acute treatment with P. incarnata in alcohol‑dependent mice. These results suggested that the treatment of P. incarnata extract could be used as a safe and alternative drug for alcohol withdrawal 51.
Anticonvulsant Activity: The current treatment of epilepsy with modern antiepileptic drugs (AEDs) is associated with side effects, dose-related and chronic toxicity, and teratogenic effects, and approximately 30% of the patients continue to have seizures with current AEDs therapy. Natural products from folk remedies have contributed significantly in the discovery of modern drugs and can be an alternative source for the discovery of AEDs with novel structures and better safety and efficacy profiles. Evidence for the anticonvulsant activity of P. incarnata in the clonic seizure of the pentylenetetrazole model has been tested. As the protective effects of P. incarnata in clonic seizure, it suggests that it could be useful for the treatment of an absent seizure. Furthermore, the important role of the benzodiazepine receptor in the effects of P. incarnata should be considered 52.
Antimicrobial Activity: In Passiflora species, many of the chemical components of passion flower (passicol) have antimicrobial activity 53, 54, 55. The ethanol leaf extracts exhibited variable degrees of antibacterial activity against P. putida, V. Cholera and moderate activity were noted in S. flexneri and S. pyogenes respectively. The acetone extracts exhibited strong to moderate activity against V. cholerae followed by P. putida, S. flexneri and S. pyogenes. The ethanol fruit extracts showed moderate activity against the bacterial pathogens namely V. cholerae, P. putida, S. pyogenes, and S. flexneri. Among the two parts tested, the leaf extracts exhibited better antibacterial activity than the fruits 56. The earlier reports focused on the antibacterial properties of Passiflora species by different methods. Antibacterial activity of Passiflora which has got activity against Pseudomonas tetrandra, Escherichia coli, Bacillus subtilis, and Pseudomonas aeruginosa.
Antioxidant Activity: P. nitida leaf and P. palmeri stem extracts were characterized by a high antioxidant power that correlates with high catechin and odiphenol contents and shows antimicrobial activity. However, P. foetida leaf extracts, which also show high antimicrobial activity, have low antioxidant power and low amounts of o-diphenol and catechin. P. tenuifila leaves show very high amounts of flavones and total phenols, but intermediate levels of antioxidant activity, probably due to the lower contribution of o-diphenols and gallocatechins relative to the phenol content 57.
The antioxidant activity of leaf and stem extracts of P. edulis was determined using the 1, 1-diphenyl- 2-picrylhydrazyl (DPPH) free radical scavenging assay 58. DPPH offers a convenient and accurate method for titrating the oxidizable groups of natural or synthetic anti-oxidants 59. The crude extracts (leaf and stem) of P. edulis were mixed with 95% methanol to prepare the stock solution (10 mg/100 mL).
Anti-inflammatory Activity: The aqueous leaves extract of Passiflora species exhibited potent anti-inflammatory action in the experimental model in-vivo 60. The aqueous leaves extract of P. edulis possess a significant anti-inflammatory activity on mice 61. The systemic administration of P. edulis exhibited pronounced anti-inflammatory actions, characterized by inhibition of leukocyte influx to the pleural cavity and associated with marked blockade of myeloperoxidase, nitric oxide, TNF-α and IL-1α levels in the acute model of inflammation caused by intrapleural injection of mice. In one experiment, P. edulis was more effective in suppressing the TNF-α and IL-1α levels than dexamethasone 62. P. edulis therefore may be a source of new therapeutic candidates with a spectrum of activity similar to the current anti-inflammatory steroids such as dexamethasone.
Anti-Tumor Activity: Fruit’s decoction of different Passiflora species has been evaluated for the inhibition of the activity of gelatinase matrix metalloproteinases (MMP-2 and MMP-9). Two metalloproteases were involved in the tumor invasion, metastasis, and angiogenesis. Water extract of P. edulis, at different concentrations, was inhibited by the enzymes 63.
Congestive Heart Failure: An extract containing passion flower and hawthorn has been studied as a possible treatment for shortness of breath and difficult use of exercise in patients with congestive heart failure. Although the results are promising, the effects of passion flower alone are unclear. The high-quality human research of passion flower alone compared to prescription drugs used for this condition is needed before a strong recommendation can be made 64.
Clinical Applications: Allergies few reports of the use of passion flower products on allergic reactions, asthma, irritated sinuses, skin rashes, and skin blood vessel inflammation (vasculitis) have been reported in the available literature. It is believed that some reactions may have been caused by impurities in combination products, not by passionflower itself 65.
Side Effects and Warnings: Passion flower is generally considered to be a safe herb with few reported serious side effects. In cases of side effects, the products being used have rarely been tested for contamination, which may have been the cause. Cyanide poisoning has been associated with passiflora fruit, but this has not been proven in human studies. Rapid heart rhythm, nausea, and vomiting have been reported. Side effects may also include drowsiness /sedation and mental slowness. Patients should be cautious when driving or operating heavy machinery.
Passionflower may theoretically increase the risk of bleeding and affect blood tests that measure blood clotting 66. There is a reported case of liver failure and death of a patient taking a preparation of passion flower with kava. Caution should be applied in taking any kava-containing products, as kava has been associated with liver damage. It has been suggested that the cause of the liver damage is less likely related to the presence of passion flower.
Uses:
Traditional Uses: The uses here are based on tradition or scientific theories of Passiflora species. Some of these conditions are potentially serious and should be evaluated by a qualified healthcare provider. These traditional uses includes alcohol withdrawal, antibacterial, anti-seizure, anti-spasm, aphrodisiac, asthma, attention deficit hyperactivity disorder (ADHD), burns (skin), cancer, chronic pain, cough, drug addiction, Epstein-Barr virus, fungal infections, gastrointestinal discomfort (nervous stomach), Helicobacter pylori infection, hemorrhoids, high blood pressure, menopausal symptoms (hot flashes), nerve pain, pain (general), skin inflammation, tension and wrinkle prevention 67.
Industrial Uses: Some species of Passiflora are cultivated outside their natural range because of their beautiful flowers. P. incarnate L. commonly used in many herbal remedies is well known for its sedative properties, while several other species are cultivated for the production of fruit juice (P. edulis, P. quadrangularis, P. ligularis) 68. Passicol can also be produced from fruit rinds of the purple passion fruit, which are waste products from the manufacture of passion fruit juice.
The resulting rich juice, which has been called a natural concentrate, can be sweetened and diluted with water or other juices (especially orange or pineapple), to make cold drinks. In South Africa, passion fruit juice is blended with milk and alginate; in Australia, the pulp is added to yogurt.
CONCLUSION: Species of Passiflora are commonly found throughout the world. These studies place this indigenous drug as a novel candidate for bio-prospection and drug development for the treatment of such diseases as anxiety, insomnia, convulsion, sexual dysfunction, cough, cancer, and postmenopausal syndrome. The medicinal applications of this plant and countless possibilities for investigation remain in relatively newer areas of its function.
A comprehensive account of the chemical constituents is given in this review. Various types of preparations, extracts and individual compounds derived from this species have been found to possess a broad spectrum of pharmacological effects on several organs such as the brain, blood, cardiovascular and nervous systems as well as on different biochemical processes and physiological functions including proteosynthesis, work capacity, reproduction, and sexual function. Hence, phytochemicals and minerals of these plants will enable to exploit its therapeutic use.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kinghorn GR: Passion, stigma, and STI. Sex Transm Inf 2001; 77: 370-
- Dhawan K, Dhawan S and Sharma A: Passiflora: A review update. J Ethnopharmacol 2004; 94: 1-23.
- Montanher A, Zucolotto SM, Schenkel E and Frode T. Evidence of anti-inflammatory effects of Passiflora edulis in an inflammation model. J Ethnopharmacol 2007; 109: 281-288.
- Beninca J, Montanher A, Zucolotto S, Schenkel E and Frode T: Evaluation of the antiinflammatory efficacy of Passiflora edulis. Food Chem 2007; 104: 1097-1105.
- Akhondzadeh S, Naghavi HR, Vazirian M, Shayeganpour A, Rashidi H and Khani M: Passionflower in the treatment of generalized anxiety: a pilot double-blind, randomized controlled trial with oxazepam. J Clin Pharm Therapeut 2001; 26-5: 363-367.
- Ozarko G: Passiflora. www.icon.com.au. 2001.
- Broutin M, Bugerol T, Guitton B and Broutin E: A combination of plant extracts in the treatment of outpatients with adjustment disorder with anxious mood: controlled study versus placebo. Fundamental Clin Pharmacol 1997; 11(2): 127-132.
- Ingale AG and Hivrale AU: Pharmacological studies of Passiflora and their bioactive compounds. African Journal of Plant Science 2010; 4(10): 417-426.
- The Wealth of India. A Dictionary of Indian Raw Materials and Industrial Products. CSIR 7: 278-9.
- Carratu B, Boniglia C, Giammarioli S and Mosca M: Free amino acids in botanicals and botanical preparations. Journal of Food Science 2008; 73(5): C323-C328.
- Bradley PR: British Herbal Compendium, Bournemouth: British Herbal Medicine Association 1992, 1: 23
- Bruneton: J. Pharmacognosy, Phytochemistry, Medicinal Plants. Paris: Lavoisier Publishing 1995.
- 'Passiflorae herba.' Monographs on the Medicinal Uses of Plant Drugs. Exeter, U.K.: European Scientific Cooperative on Phytotherapy 1997.
- Newall CA, Anderson LA and Phillipson JD: Herbal Medicines: A Guide for Health-Care Professionals. London: The Pharmaceutical Press 1996.
- Wichtl M and Bisset NG: Herbal Drugs and Phytopharmaceuticals. Stuttgart: Medpharm Scientific Publishers 1994.
- Leung AY and Foster S: Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics, 2nd New York: John Wiley & Sons, Inc 1996.
- Woo KJ, Jeong YJ and Park-kwon TK: Chrysin induced apoptosis is mediated through caspase activation and akt inactivation in U937 leukemia cell. Biochem Biophys Res Commun 2004; 325-334: 1215-1222.
- Brown E, Hurd NS, McCall S and Ceremuga TE: Evaluation of the anxiolytic effects of chrysin a Passiflora incarnata extract in the laboratory rat. AANA J 2007; 75(5): 333-337.
- Wolfman C, Viola H, Paladini A, Dajas F and Medina JH: Possible anxiolytic effects of chrysin a central benzodiazepine receptor ligand isolated from Passiflora coerulea. Pharmacol Biochem Behav 1994; 47-1: 1-4.
- Balch PA: Prescription for herbal healing: an easy to use A to Z reference to hundreds of common disorders and their herbal remedies. Avery, New York, 2002; ISBN 0-89529-869-4.
- Zand RS, Jenkins DJ and Diamandis EP: Steroid hormone activity of flavonoids and related compounds. Breast Cancer Res. Treatments 2000; 62(1): 35-49.
- Yin F, Giuliano AE, Law RE and Van Herie AJ: Apigenin inhibits growth and induces G2/M arrest by modulating cyclin CDK regulators and ERK MAP kinase activation in breast carcinoma cells. Anticancer Res 2001; 21-1A: 413-420.
- Yin F, Giuliano AE and Van Hearle AJ: Growth inhibitory effects of flavonoids in human thyroid cancer cell lines. Thyroid 1999; 9(4): 369-376.
- Knowles LM, Zigrossi DA, Tauber RA, Hightower C and Milner JA: Flavonoids suppress androgen-independent human prostate tumor proliferation. Nutr Cancer 2000; 38-1: 116-122.
- Nagasugi T, Nakashima M and Komai K: Antimutagens in gaiyou (Artemisia argyi levl. et vant.) J Agric Food Chem 2000; 48(8): 3256-3266.
- Chlouchi A, Girard C, Bonet A, Viollon AC, Heyd B, Mantion G, Martin H and Richert L: Effect of chrysin and natural coumarins on UGT1A1 and 1A6 activities in rat and human hepatocytes in primary culture. Planta Med 2007; 73(8): 742-747.
- Izzotti A, Bagnasco M, Cartiglia C, Longobardi, M, Camoirano A, Tampa E, Lubet RA and De FS: Modulation of multigene expression and proteome profiles by chemopreventive agents. Mutat. Res 2005; 11-59(11-2): 212-223.
- Bennati E: New Encyclopaedia of Botanical Drugs and Preparations Saffron Walden. Boll Chim Farm 1968; 107-716.
- The Lawrence Review of Natural Products Facts and Comparisons, Herbal Diuretics (Monograph), 1989: 1-2.
- Fernandez D, Arriba A, Lizcano JM, Balsa MD and Unzeta M: Inhibition of monoamine oxidase from bovine retina by betacarbolines. J Pharm Pharmacol 1994; 46-10: 809-13.
- Rommelspacher H, May T and Salewski B: Harman (1- methyl-beta-carboline) is a natural inhibitor of monoamine oxidase type A in rats. Eur J Pharmacol 1994; 252(1): 51-59.
- Ergene E and Schoener EP: Effects of harmane (1-methyl-betacarboline) on neurons in the nucleus accumbens of the rat. Pharmacol Biochem Behav 1993; 44(4): 951-957.
- Hagiwara A, Sano M, Asakawa E, Tanaka H, Hasegawa R and Ito N: Enhancing effects of harman and norharman on induction of preneoplastic and neoplastic kidney lesions in rats initiated with NethyI-N-hydroxyethyl nitrosamine. Jpn J Cancer Res 1992; 83-9: 949-955.
- Soulimani R, Younos C, Jannouni S, Bousta D, Misslin R and Mortier F: Behavioural effects of Passiflora incaranata and its indole alkaloid and flavonoid derivatives and maltol in the mouse. J Ethnophannacol 1997; 57-61: 11-20.
- Baumn SS, Hill R and Rommelspacher H: Harman induced changes of extracellular concentrations of neurotransmitters in the nucleus accumbens of rats. Eur J Pharmacol 1996; 1-2: 75-82.
- Bosin TR and Faull KF: Harman in alcoholic beverages: pharmacological and lexicological implications Alcohol. Clin Exp Res 1988; 12-5: 679-682.
- Totsuka Y, Ushiyama H, Ishihara J, Sinha R, Goto S, Sugimara T and Wakabayashi K: Quantification of the comutagenic beta-carbolines nonharman and Harman in cigarette smoke condensates and cooked foods. Cancer Lett 1999; 143-152: 139-143.
- Lee KG and Shibamoto T: Antioxidant properties of aroma compounds isolated from soybeans and mung beans. Journal of Agricultural and Food Chemistry 2000; 48(9): 4290-4293.
- Dolzhenko AT and Komissarov IV: Characteristics of the presynaptic action of the barman and its derivatives compared to benzodiazepine tranquilizers. Farmakol Toksikol 1987; 50(2): 13-16.
- Brito AM: Synthesis and characterization of glycosides. Springer 2007. ISBN 978-0-387-26251-2.
- Jaroszewski JW and Olafsdottir ES: Cyanohydrin glycosides of Passiflora: Distributions pattern a saturated cyclopentane derivative from guatemalensis and formation of pseudocyanogenic α-hydroxy amides as isolation artifacts. Phytochem 2002; 59-65: 501-511.
- Duke JA: Phytochemical and Ethnobotanical Databases –Passiflora spp. 2008 http://www.ars-grin.gov/duke.
- Jaroszewski JW, Rasmussen AB, Rasmussen HB, Olson CE and Jorgensen LB: Biosynthesis of cyanohydrin glucosides from unnatural nitriles in intact Passiflora morifolia and Tunera angustifolia. Phytochem 1996; 42-43: 649-654.
- Padmaja G and Panikkar KR: Intermediary metabolic changes in rabbits administered linamarin or potassium cyanide. Indian Journal of Experimental Biology 1989; 27-37: 635-639.
- Linda D, Lloyd W and Rooney LW: Sorghum and millet phenols and antioxydants. J Cereal Sci 2006; 44: 236-251.
- Dhawan K, Dhawan S and Sharma A: Passiflora: a review update. J Ethnopharmacol 2004; 94: 1-23.
- Pinheiro ER, Silva IMDA, Gonzaga LV, Amante ER, Reinaldo FT, Ferreira, MMC and Amboni RDMC: Optimization of extraction of high ester pectin from passion fruit peel (Passiflora edulis flavicarpa) with citric acid by using response surface methodology. Bioresour. Technol 2008; 99-113: 5561-5566.
- Yapo BM and Koffi L: Dietary fiber components in yellow passion fruit rind a potential fiber source. J Agric Food Chem 2008; 56-14: 5880-5883.
- Dhawan K, Kumar S and Sharma A: Reversal of cannabinoids (delta 9‑THC) by the benzoflavone moiety from methanol extract of Passiflora incarnata Linn in mice: A possible therapy for cannabinoid addiction. J Pharm Pharmacol 2002; 54: 875-81.
- Dhawan K, Kumar S and Sharma A: Nicotine reversal effects of the benzoflavone moiety from Passiflora incarnata Linn in mice. Addict Biol 2002; 7: 435-41.
- Dhawan K, Kumar and Sharma A: Suppression of alcohol‑cessation oriented hyper-anxiety by the benzoflavone moiety of Passiflora incarnata in mice. J Ethnopharmacol 2002; 81: 239-44.
- Nassiri-Asl M, Shariati-Rad F and Zamansoltani F: Anticonvulsant effects of arial parts of Passiflora incarnata extract in mice: Involvement of benzodiazepine and opioid receptors. BMC Complement Altern Med 2007; 7: 26.
- Nicolls JM: Antifungal activity in Passiflora Ann. Bot. (London). 1970; 34: 229-337.
- Birner J and Nicoll JM: Passicol an antibacterial and antifungal agent produced by Passiflora plant species: preparation and physicochemical characteristics. Antimicrob. Agent Chemother 1973; 3: 105-109.
- Nicolls JM, Birner J and Forsell P: Passicol an antibacterial and antifungal agent produced by Passiflora plant species: qualitative and quantitative range of activity. Antimicrob. Agents Chemother 1973; 3: 110-117.
- Afolayan AJ and Meyer JJM: The antimicrobial activity of 3, 5, 7-trihydroxy flavones isolated from the shoots of Helichrysum aureonitens. J Ethnopharmacol 1997; 57: 177-181.
- Bendini A, Cerretani L, Pizzolante L and Toschi TG: Phenol content related to antioxidant and antimicrobial activities of Passiflora Extracts. Eur. Food Res Technol 2006; 223: 102-109.
- Blois MS: Antioxidant determinations by the use of a stable free radica Nature 1958; 26: 1199-1200.
- Cao G, Sofic E and Prior RL: Antioxidant and prooxidant behavior of flavonoids: structure-activity Free radical Biol Med 1997; 22: 759-760.
- Beninca J, Montanher A, Zucolotto S, Schenkel E and Frode T: Evaluation of the AntiInflammatory Efficacy of Passiflora Edulis. Food Chemistry 2007; 104: 1097-1105.
- Vargas AJ, Geremias DS, Provensi G, Fornari PE, Reginatto FH, Gosmann G, Schenkel EP and Frode TS: Passiflora alata and Passiflora edulis spray dried aqueous extracts inhibit Inflammation in the mouse model of pleurisy. Fitoterapia 2007; 78: 112-119.
- Montanher A, Zucolotto SM, Schenkel E and Frode T: Evidence of anti-inflammatory effects of Passiflora edulis in an inflammation model. J Ethnopharmacol 2007; 109: 281-288.
- Puricelli L, Aicab ID, Sartorb L, Garbisa S and Caniato R: Preliminary evaluation of inhibition of matrix metalloprotease MMP-2 and MMP-9 by Passiflora edulis and foetida aqueous extracts. Fitoterapia 2003; 74: 302-304.
- Capasso A and Sorrentino L: Pharmacological studies on the sedative and hypnotic effect of Kava kava and Passiflora extracts combination. Phytomed 2005; 12(1-2): 39-45.
- Giavina-Bianchi PF, Castro FF and Machado ML: Occupational respiratory allergic disease induced by Passiflora alata and Rhamnus purshiana. Ann. Allergy Asthma Immunol 1997; 79-5: 449-454.
- Kapadia GJ, Azuine MA and Tokuda H: Inhibitory effect of herbal remedies on 12-O-tetradecanoyl phorbol-13-acetate-promoted Epstein Barr virus early antigen activation. Pharmacol Res 2002; 45-53: 213-220.
- Dhawan K, Kumar S and Sharma A: Reversal of cannabinoids (delta9-THC) by the benzoflavone moiety from methanol extract of Passiflora incarnata Linneaus in mice: a possible therapy for cannabinoid addiction. J. Pharm. Pharmacol 2002; 54(6): 875-881.
- Bendini A, Cerretani L, Pizzolante L and Toschi TG: Phenol content related to antioxidant and antimicrobial activities of Passiflora Extracts. Eur Food Res Technol 2006; 223: 102-109.
How to cite this article:
Tiwari S, Singh S, Tripathi S and Kumar S: A pharmacological review: Passiflora species. Int J Pharmacognosy 2016; 3(1): 10-18. doi: 10.13040/IJPSR.0975-8232.3(1).10-18.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
10-18
599
2376
English
IJP
S. Tiwari, S. Singh*, S. Tripathi and S. Kumar
Rameshwaram Institute of Technology and Management, Lucknow, Uttar Pradesh, India.
singh.shweta252@gmail.com
26 October 2015
26 November 2015
29 November 2015
10.13040/IJPSR.0975-8232.IJP.3(1).10-18
31 January 2016