A DESCRIPTIVE REVIEW ON RAPHANUS SATIVUS LINN.
HTML Full TextA DESCRIPTIVE REVIEW ON RAPHANUS SATIVUS LINN.
Sujeet Pratap Singh * and Vikas Verma
Department of Pharmacy, Apex University, Jaipur, Rajasthan, India.
ABSTRACT: Raphanus sativus (radish), a member of the Brassicaceae family, is a widely cultivated and consumed root vegetable across the globe. Its nutritional and medicinal significance has been extensively documented through various studies. Traditionally, extracts from both aerial and underground parts of radishes have been employed in folk medicine to manage a range of ailments, including digestive disorders, urinary infections, hepatic inflammation, cardiovascular complications, and ulcers. The therapeutic potential of radish is primarily attributed to its rich composition of bioactive secondary metabolites, such as glucosinolates, polyphenols, flavonoids, and isothiocyanates, which confer antioxidant, anti-inflammatory, antidiabetic, hepatoprotective, and anticancer activities. This review emphasizes the impact of radish extract administration in pathological conditions, including cancer, diabetes, liver dysfunction, and oxidative stress. Additionally, it provides a detailed analysis of the molecular mechanisms by which radish-derived compounds regulate critical drug targets associated with various cancers and metabolic disorders. By modulating signaling pathways, apoptotic proteins, and detoxification enzymes, these bioactive constituents demonstrate considerable potential in preventing and managing chronic diseases, highlighting radish as a promising candidate for both functional foods and therapeutic applications in modern medicine.
Keywords: Radish, Raphanus sativus, Brassicaceae, Phytochemistry, Pharmacology
INTRODUCTION: Raphanus sativus linn, the scientific name for radish, is a member of the Brassicaceae family. Mooli is the popular name for this annual plant that is eaten as a vegetable. In numerous states, radish is referred to by a variety of names, including Mullangi, Moolika, and Mooli. Originally from Europe and Asia, Raphanus sativus L 1. It grows between 190 to 1240 meters above sea level in temperate areas. Its roots are thick, varied in size, shape, and colour, and it stands between 30 and 90 cm tall.
They have a strong flavour and are edible. In essence, one of the classic Japanese delicacies is the takuan, or salted radish roots, which are consumed in Japan in quantities of over 500,000 tonnes annually. The distinctive yellow hue of the salted radish roots develops during preservation. This species is often used to treat respiratory and hepatic conditions 2.
The efficacy of its extracts in treating microbial illness, as stated in traditional medicine, is validated by their antibiotic activity and temporal persistence. Salmonella thyphosa, Pseudomonas aeruginosa, and Bacillus subtilis were all susceptible to the antibacterial action of the root's juice. The aqueous and ethanolic extracts demonstrated efficacy against Candida albicans and Streptococcus mutans. The whole plant's aqueous extract has antimicrobial action against Staphylococcus epidermidis and Sarcinia lutea 2. The metabolites that R. sativus produces are included in this review based on their structural classifications 3.
Taxonomical Classification 4:
Kingdom: Plantae-plantes, Planta, Vegetal, plants
Subkingdom: Viridiplantae-Green plants
Infrakingdom: Streptophyta-land plants
Super division: Embryophyta
Division: Tracheophyta vascular plants, tracheophytes
Subdivision: Spermatophytina-spermatophytes, seed plants, phanérogames
Class: Magnoliopsida
Superorder: Rosanae
Order: Brassicales
Family: Brassicaceae
Genus: Raphanus L.-R. sativus
Species: Raphanus sativus L
Vernacular Name:
Hindi: (Mūlī)
English: Radish
Sanskrit: (Mūlaka)
Bengali: (Mulo)
Tamil: (Mullangi)
Telugu: (Mullangi)
Kannada: (Moolangi)
Malayalam: (Mullangi)
Marathi: (Mula)
Gujarati: (Mūḷā)
Punjabi: (Mūlī)
Urdu: (Mooli)
Odia: (Mūlā)
Assamese: (Mula)
Nepali: (Mula)
Botanical Characterization: A few distinctive physical traits of R. sativus may aid in the plant/fruit's identification. Radish, or R. sativus, is a member of the Brassicaceae family. Radish plants increase every year or every two years, especially during the winter. It may be found all across the world and is often cultivated in the highlands from March to August. Radish plants' exterior characteristics include differences in fruit type, flower petal colour, floral symmetry, and leaf type and arrangement 5.
It has been noted that R. sativus blooms were formerly symmetrical. The petals of R. sativus flowers are pink, white, and pale red in colour. Leaves come in two varieties. A leaf that is lobed or unlobed but not divided into leaflets is referred to as a simple leaf, whereas a compound leaf is composed of two or more distinct leaflets. One leaf per node along the stem was once the alternate leaf configuration. There are lobes and a feature resembling teeth on the leaf blade's edge. When mature, the fruit of R. sativus does not break into pieces, although it was once dry and weighed between 10 and 250 mm. The farmers usually cultivate the radish in the season of winter as it needs a clear sunlight and low temperature for better growth 6.
FIG. 1: LEAVES, ROOT AND SEEDS OF RAPHANUS SATIVUS
Habit and Habitat: The plant's habitat is between 190 to 1240 meters above sea level, and it is primarily found in temperate regions. Sowing takes place from March to July in mountainous areas and from September to March in the northern plains. The stems can be simple or branching, and the fleshy, enlarged tap roots come in a variety of sizes, shapes, and colours 1, 3, 8.
Distribution: It began to spread from southern China to other parts of Japan and other Asian nations. It is an essential food crop found to be widely grown in Korea, South east Asia, Japan, etc. It has been discovered to have a number of therapeutic uses, including the treatment of gynaecological diseases, jaundice, and urinary tract infections 9. Brazil produces around 9,000 t of radish annually 10. In parts of the United States and Canada, oil seed cultivars are readily available. They are derived from wild and farmed species variants 8.
History: During pre-roman times, it was domesticated in European countries. Since ancient times, this plant has been regarded as a significant food source in Egypt. The species' older varieties are often biennials, meaning they need to be vernalised or treated with cold for a while before they may begin to produce seeds and bloom. The same is true for bigger species, such as daikon, winter radishes, and black Spanish. Modern variants are spherical and icicle-shaped, and they come in pink, red, white, and violet hues. A minimum number of cold treatments are needed to develop them from seeds, and it takes 30 to 40 days for a fully grown radish to mature 11, 12, 13.
Traditional Uses: Raphanus sativus (R. sativus) is a multi-purpose plant with significant nutritional and medicinal uses.
- It is an edible plant; the leaves and fruits are generally cooked and sometimes eaten raw.
- sativus possesses astringent and diuretic properties, which is why it is traditionally used to increase bile flow.
- Radish juice has long been used as a home remedy for cough, rheumatic arthritis, and gallbladder stones 12.
- Raw radishes are commonly consumed in salads, especially in European diets, and are also used in various traditional dishes.
- In the Middle East, radish juice is consumed for its perceived health benefits.
- In Unani, Greeko-Arab, and Indian folk medicine, sativus is used to treat:
- Jaundice
- Gallstones
- Liver disorders
- Rectal disorders
- Indigestion and other gastric pains
- The leaves, often discarded, contain 10 times more vitamin C than the roots, providing higher antioxidant activity.
- In Yemenite folk medicine, radish juice is used to help eliminate kidney stones 13.
- The leaves and roots are used in the treatment of:
- Asthma
- Chest tightness
- Intestinal parasitic infections
- The root exhibits multiple pharmacological properties, including:
- Antiscorbutic
- Antispasmodic
- Astringent
- Cholagogue
- Digestive effects
- sativus contains raphanin, a compound with antibacterial and antifungal properties.
- Raphanin inhibits the growth of pathogens such as:
- Staphylococcus aureus
- Escherichia coli
- Streptococci
- Pneumococci
- The plant has demonstrated anti-tumor activity.
- Consumption of sativus enhances adiponectin production, a hormone that helps protect against insulin resistance.
- sativus is rich in antioxidants and minerals, particularly calcium and potassium.
- These nutrients help to:
- Lower high blood pressure
- Reduce the risk of heart disease
- sativus is a good source of natural nitrates, which improve blood circulation.
- The plant contains glucosinolates (sulfur-containing compounds) that:
- Protect cells from genetic mutations
- Inhibit tumor cell growth
- Reduce the risk of cancer development 13, 14
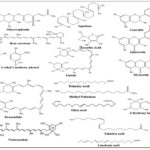
Chemical Constituents: In Asian nations, particularly China, Japan, and Korea, Raphanus sativus is a staple vegetable 15. Alkaloids and nitrogen compounds, coumarins, enzymes, gibberellins, glucosinolates, oil seed compounds, organic acids, phenolic compounds, sulphur compounds, flavonoids, saponins, carbohydrates, proteins, amino acids, tannins, brassinosteroids, and polyphenols are the main chemical components of Raphanus sativus 16, 17. With a biological value of 76.6 and a digestibility coefficient of 73.5%, Raphanus sativus leaves are a great source of protein. Their origin is mentioned below
- Biochemical substances such as methins, sapogenins, levon
- Enzymes such as phosphatase, catalase
- Histaminergic component and a weak spasmolytic
- Amino acids such as lysine, methionin etc.
- Polyphenolics such as protocatechuic acid, vanillic acid
- Antibacterial substances such as Sulphoraphene and raphanin
- Free radical scavengers etc 18.
Factors such as climate, soil type, genotype, seasonal variation, processing methods, extraction, and quantification techniques significantly influence enzyme activity and stability, thereby affecting the hydrolysis of glucosinolates. Raphanus sativus seeds and leaves contain raphanin, an important bioactive constituent. The plant also contains glucoraphanin, glucobrassicin, methoxyglucobrassicin, and glucoraphasatin, which are distributed in both the skin and flesh of the vegetable 19–21.
FIG. 2: LIST OF CHEMICAL CONSTITUENTS
The peels of Raphanus sativus L. var. niger contain numerous important phytoconstituents, including tannins, saponins, flavonoids, phlobatannins, anthraquinones, carbohydrates, reducing sugars, steroids, phytosterols, alkaloids, amino acids, terpenoids, cardiac glycosides, and chalcones, which contribute to their medicinal value 22. Taste-determining factors such as glucosinolate content, soluble carbohydrates, and myrosinase activity are also present. Additionally, the vegetable contains proximate nutrients like protein, dietary fiber, carbohydrates, and antioxidants 22. Compounds such as isothiocyanates and dithiolthiones help reduce cancer risk 24. Medicinal plants serve as valuable resources for drug development, cultural practices, nutrition, and therapeutic applications worldwide 25.
Pharmacological Activity:
Antioxidant Effects of Radishes: The roots and leaves of radish possess significant nutritional value and diverse secondary metabolites with strong antioxidant properties. Compared to roots, leaves contain higher levels of protein, calcium, and ascorbic acid, while total phenolic content is nearly two-fold higher, correlating with enhanced free radical scavenging activity 26. Distinct polyphenols are distributed in different tissues; roots are rich in pyrogallol (free form) and vanillic acid (bound form), whereas leaves contain epicatechin (free form) and coumaric acid (bound form). Notably, flavonoid levels in leaves are four times higher than in roots. Flavonoids, major polyphenolic compounds with multiple hydroxyl groups, exhibit strong free radical scavenging potential 27. Consequently, radish leaves represent an excellent source of bioactive antioxidant compounds. In vitro studies by Wang et al. 28 demonstrated both antioxidant and prooxidant activities of red radish extracts rich in anthocyanins, particularly acylated pelargonidin derivatives. These compounds showed concentration-dependent scavenging of ABTS⁺ radicals and strong reducing power. Ferric ion–reducing and metal-chelation assays further confirmed antioxidant potential. Prooxidant activity, assessed via plasmid DNA damage assays, depended on concentration and reaction conditions. Anthocyanins also exhibit chemoprotective effects by activating phase II enzymes and promoting apoptosis 29–36.
Anticancer Effects of R. sativus: According to Rampal et al. 37, isothiocyanates demonstrate strong anticancer potential through multiple mechanisms of pharmaceutical importance. These include modulation of phase I and phase II detoxification enzymes, induction of programmed cell death (apoptosis), and inhibition of uncontrolled cell cycle progression in cancer cells. Radish extracts, rich in isothiocyanates, have therefore gained attention for their protective and therapeutic roles against various cancers, highlighting their relevance in cancer prevention and complementary treatment strategies.
Breast Cancer: The aerial extract of radish has been shown to induce significant cytotoxicity in the breast cancer cell line MDA-MB-231 by modulating the ErbB–Akt signaling pathway 38. Epidermal growth factor receptor (EGFR), a key oncogene in breast cancer, consists of ErbB1–ErbB4 proteins, and their overexpression is strongly associated with tumor progression 39, 40. Ligand binding to EGFR promotes receptor dimerization and tyrosine kinase autophosphorylation, activating downstream signaling pathways responsible for cell proliferation and survival 41, 42. Treatment with radish aerial extract significantly downregulated the mRNA and protein expression of ErbB2 and ErbB3, thereby inhibiting EGFR-mediated signaling 38. Furthermore, EGFR activation stimulates the PI3K/Akt pathway, which enhances tumorigenesis by promoting cell survival and inhibiting apoptosis 43–46. Radish extract reduced Akt expression in a dose-dependent manner, increasing antitumor activity 38. Additionally, sulforaphene, an active isothiocyanate from radish, reduced SKBR-3 cell viability, induced G2/M arrest, disrupted cytoskeletal organization, and triggered apoptosis with minimal toxicity to normal cells.
Colon Cancer: In general, cancer cells exhibit elevated basal levels of reactive oxygen species (ROS), making them more susceptible to ROS-targeted therapeutic strategies with higher selectivity. Administration of sulforaphane and sulforaphene selectively induced cancer cell death while sparing normal cells, likely due to efficient antioxidant defense mechanisms in healthy cells. Sulforaphane activates both intrinsic and extrinsic apoptotic pathways; in the intrinsic pathway, it modulates mitochondrial membrane proteins, increases proapoptotic protein expression, reduces antiapoptotic proteins, and triggers caspase cascade activation 50. In the extrinsic pathway, sulforaphane enhances apoptosis through induction of TNF-related apoptosis-inducing ligand (TRAIL) and suppression of ERK and Akt signaling 51–53. Sulforaphene exhibits stronger antimutagenic effects than sulforaphane 54. Both compounds possess electrophilic centers that interact with nucleophilic cellular targets such as glutathione and Keap1 cysteine residues, leading to Nrf2 stabilization 55. Additionally, these bioactive compounds stimulate phase II detoxifying enzymes, enhancing carcinogen elimination.
Cervical, Lung and Prostate Cancer: The chemopreventive effects of radish extracts have been evaluated in cervical (HeLa), lung (A549), prostate (PC-3), and breast (MCF-7) cancer cell lines by Beevi et al. 56, revealing the molecular mechanisms of radish-mediated apoptosis. Hexane extracts from radish roots contain isothiocyanates (ITCs) such as MTBITC, erucin, 4-methylpentyl isothiocyanate, 4-pentenyl isothiocyanate, and sulforaphene, which induced apoptosis in both p53-deficient and proficient cells, indicating p53-independent signaling. Apoptosis involved Bcl-2 family regulation and caspase-3 activation, selectively targeting cancer cells while sparing normal cells 57. Radish extract treatment caused cell detachment, inhibition of elongation, shrinkage, and DNA fragmentation. Gene expression analysis confirmed modulation of apoptotic pathways across diverse cancer cell types. In-vivo, sulforaphene inhibited tumor growth in Balb/C mice with lung cancer by suppressing PI3K–Akt signaling, reducing PTEN expression, and blocking Akt phosphorylation 58. These findings highlight the anticancer potential of radish-derived isothiocyanates in both cellular and animal models.
Liver Cancer: The extract of Spanish black radishes significantly inhibited HepG2 cell proliferation by modulating the phase I and phase II detoxification systems 59. Its anticancer activity is attributed to glucosinolates, particularly glucoraphasatin and 4-methylthio-3-butenyl isothiocyanate. The crude extract enhanced phase II detoxification enzymes, including quinone reductase, heme oxygenase-1, and thioredoxin reductase-1, while increasing mRNA levels of phase I enzymes such as CYP1A1, CYP1A2, and CYP1B1. Radish extracts activated these enzymes via the AhR and Nrf2 pathways. Although phase I activation may generate reactive intermediates, the concurrent induction of phase II enzymes ensures effective detoxification and reduces toxicity 60.
Hepatoprotective Effects: Bioactive compounds in radish roots and sprouts, including indole-3-carbinol, 3-[ethoxy-(methylthio) methyl]-2-pyrrolidinethione, and 3-(E)-(methylthio)-methylene-2-pyrrolidinethione, have been shown to reduce the severity of fatty liver disease in mouse models, while black radish extracts alleviated carbon tetrachloride (CCl4)-induced liver injury in rats 61. Fresh radish juice prevented CCl4-induced hepatotoxicity by inhibiting lipid peroxidation, replenishing non-protein sulfhydryl (NP-SH) levels, and enhancing liver detoxification. Phytochemical analysis revealed hepatoprotective sulfur compounds, phenols, and terpenoids. Radish enzyme extracts protected HepG2 cells and rats against tacrine- and CCl4-induced hepatotoxicity by maintaining membrane integrity, reducing GOT and GPT leakage, and lowering serum triglycerides and total cholesterol 62, demonstrating significant hepatoprotective potential.
Antidiabetic Effects of Radish: The traditional use of radish extracts for digestive ailments suggests the presence of phytochemicals with antidiabetic properties. Water-soluble radish extracts exhibit hypoglycemic effects due to insulin-like polyphenols or glucose-inhibiting compounds 63, 64. The antidiabetic activity involves regulation of glucose-related hormones, reduction of diabetes-induced oxidative stress, and balancing glucose uptake and absorption. Radish extracts enhance adiponectin synthesis, improving insulin sensitivity, promoting lipid oxidation, and aiding weight management 65, 66. Adiponectin activates its receptors (ADIPOR1/2) and PPARγ, regulating genes involved in inflammation, oxidative stress, gluconeogenesis, and glucose uptake 67, 68. It also phosphorylates ACC2 to increase fatty acid oxidation and modulates antioxidant enzymes such as superoxide dismutase (SOD), mitigating ROS levels 69. These mechanisms collectively contribute to radish’s antidiabetic potential.
CONCLUSION: The discussion above clearly highlights the multi-disciplinary properties of Raphanus sativus (R. sativus), demonstrating its significant nutritional, medicinal, and pharmacological potential. Being a widely available vegetable throughout almost all seasons, R. sativus provides accessible health benefits across diverse populations. Every part of the plant, including the roots, leaves, and fruits, contains bioactive phytochemicals such as isothiocyanates, polyphenols, flavonoids, and sulfur-containing compounds, each contributing to its therapeutic properties. These compounds exhibit antioxidant, anticancer, hepatoprotective, antidiabetic, and cardioprotective effects, making R. sativus highly potent against various lifestyle-related and chronic diseases. Moreover, its widespread availability and rich phytochemical profile offer an excellent foundation for future research, promoting the development of novel treatments for life-threatening diseases and expanding its application in modern medicine and functional foods.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Paredes SD: Etnobotánica Mexicana: Plantaspopularmenteempleadasenel Estado de Michocánenel tratamiento de enfermedades hepaticas y vesiculares. Tesis Lic. México D.F. Facultad de Ciencias. UNAM 1984.
- Caceres A: Screening on antimicrobial activity of plants popular in Guatemala for the treatment of dermatomucosal diseases. J. Ethnopharm 1987; 20: 223–237.
- Marquardt P: N-methylphenethylamine in vegetables. Arzneimittelforschung 1976; 26: 201–203.
- Tang D, Dong Y, Ren H, Li L and He C: A review of phytochemistry, metabolite changes, and medicinal uses of the common food mungbean and its sprouts (Vigna radiata). Chem Cent J 2014; 8: 4. doi:10.1186/1752-153X-8-4, PMID 24438453
- Abdou IA, Abou-Zeid AA, El-Sherbeeny MR and Abou-El-Gheat ZH: Antimicrobial activities of Allium sativum, Allium cepa, Raphanus sativus, Capsicum frutescens, Eruca sativa, Allium kurrat on bacteria. Qual Plant Mater Veg 1972; 22: 29-35. doi: 10.1007/BF01099735
- Lim S, Ahn JC, Lee EJ and Kim J: Antiproliferation effect of sulforaphane isolated from radish (Raphanus sativus) seeds on A549 cells. Appl Biol Chem 2020; 63: 1-8.
- Yoichi T, Yohichi H, Shigeru Y and Naoto S: Structure of L-arabino-galactan contained glycoproteins from radish leaves. Carbohydr Res 1984; 134: 215–218.
- Tsumuraya Y, Nakamura K, Hashimoto Y and Yamamoto S: Immunological properties of arabinogalactan proteins from leaves of Cruciferous plants. ABC 1984; 48: 2915–2917.
- Keishiro W, Oka O and Hirozo MH: Ferredoxin isoproteins and their variation during growth of higher plants. Physiol Veg 1985; 23: 679–686.
- Polya GM, Chandra S and Condron R: Purification and sequencing of radish seed calmodulin antagonists phosphorylated by calcium-dependent protein kinase. Plant Physiol 1993; 101: 545–551.
- Yinghua Z, Zhonghan Y and Zongxun C: Studies on stigma pellicle glycoproteins of Raphanus sativus ZhiwuXuebao 1983; 25: 544–550.
- Raphanus sativus; n.d. Useful Tropical Plants. Available from: https://tropical.theferns.info/viewtropical.php?id=Raphanus+sativus [Lastaccessed on 2022 May 09].
- Banihani SA: Radish (Raphanus sativus) and diabetes. Nutrients 2017; 9: 1014. doi: 10.3390/nu9091014, PMID 28906451
- Information about Nutrients and Herbs - Uses, Benefits. Planet Ayurveda; 2019. Available from: https://www.planetayurveda.com/R.sativus [Last accessed on 2022 May 09].
- Terras FR, Schoofs HM, DeBolle MF, Van Leuven F, Rees SB, Vanderleyden J, Cammue BP and Broekaert WF: Analysis of two novel classes of plant antifungal proteins from Radish (Raphanus sativus) seeds. J Biol Chem 1992; 267: 15301–15309.
- Hans MR, Cornelis PW, Martinus SW, Lolke S, Broekaert WF, Bronwen RS and Martinus VW: Antifungal protein fragment-derived peptides and their agricultural, therapeutic, or preservative uses. UK PCT. Appl. Wo 1997; 97(21): 815.
- Stoehr H and Herrmann K: Phenolic acids of vegetables. III. Hydroxycinnamic and hydroxybenzoic acids of root vegetables. Z. Lebensm Unters Forsch 1975; 159, 219–224.
- Matile P: The Lytic Compartment of Plant Cells. Springer-Verlag, New York 1975; 56–63.
- Faye L, Mouatassim B and Ghorbel A: Cell wall and cytoplasmic isozymes of radish-β-fructosidase have different N-linked oligosaccharides. Plant Physiol 1986; 80: 27–23.
- Tamura G, Iwasawa T, Masada M and Fukushima K: Some properties of cysteine synthase from radish roots. Agric Biol Chem 1976; 40: 637–638.
- Sekimata M, Ogura K, Tsumuraya Y, Hashimoto Y and Yamamoto S: A β-galactosidase from radish (Raphanus sativus) seeds. Plant Physiol 1989; 90: 567–574.
- Shigeo A and Yuhei M: β-Amylase of the Japanese radish and soybeans. TampakushitsuKakusan, Koso, Bessatsu 1976; 434–436.
- Yuhei M: Preliminary crystallographic data for a basic peroxidase from Japanese radish. Mem Res Inst Food Sci Kyoto Univ 1973; 36: 9–11.
- Dahlbender B and Strack D: Purification and properties of 1-(hydroxycinnamoyl)-glucose: 1-(hydroxycinnamoyl)-glucose hydroxycinnamoyl- transferase from radish seedlings. Phytochemistry 1986; 25: 1043–1046.
- Vitoria AP, Lea PJ and Azevedo RA: Antioxidant enzymes response to cadmium in radish tissue. Phytochemistry 2001; 57: 701–710.
- Goyeneche R, Roura S, Ponce A, Vega-Galvez A, Quispe-Fuentes I, Uribe E and Di Scala K: Chemical characterization and antioxidant capacity of red radish (Raphanus sativus) leaves and roots. J Funct Foods 2015; 16: 256–264. [CrossRef]
- Grassi D, Desideri G and Ferri C: Flavonoids: Antioxidants against atherosclerosis. Nutrients 2010; 2: 889–902.
- Wang LS, Sun XD, Cao Y, Wang L, Li FJ and Wang YF: Antioxidant and prooxidant properties of acylatedpelargonidin derivatives extracted from red radish (Raphanus sativus niger, Brassicaceae). Food Chem Toxicol 2010; 48: 2712–2718. [CrossRef] [PubMed]
- Castaneda-Ovando A, de Lourdes Pacheco-Hernández M, Páez-Hernández ME, Rodríguez JA and Galán-Vidal CA: Chemical studies of anthocyanins: A review. Food Chem 2009; 113: 859–871. [CrossRef]
- Kong JM, Chia LS, Goh NK, Chia TF and Brouillard R: Analysis and biological activities of anthocyanins. Phytochemistry 2003; 64: 923–933. [CrossRef]
- Mazza G and Miniati E: Anthocyanins in Fruits, Vegetables and Grains; CRC Press: Boca Raton, FL, USA 2018.
- Bagchi D, Sen CK, Bagchi M and Atalay M: Antiangiogenic, antioxidant and anticarcinogenic properties of a novel anthocyanin-rich berry extract formula. Biochemistry 2004; 69, 75–80. [PubMed]
- Renis M, Calandra L, Scifo C, Tomasello B, Cardile V, Vanella L, Bei R, Fauci LL and Galvano F: Response of cell cycle/stress-related protein expression and DNA damage upon treatment of CaCO2 cellswith anthocyanins. Br J Nutr 2008; 100: 27–35. [CrossRef]
- Shih PH, Yeh CT and Yen GC: Anthocyanins induce the activation of phase II enzymes through theantioxidant response element pathway against oxidative stress-induced apoptosis. J Agric Food Chem 2007; 55: 9427–9435. [CrossRef]
- Wang LS and Stoner GD: Anthocyanins and their role in cancer prevention. Cancer Lett 2008; 269: 281–290.[CrossRef]
- Feng R, Ni HM, Wang SY, Tourkova IL, Shurin MR, Harada H and Yin XM: Cyanidin-3-rutinoside, anatural polyphenol antioxidant, selectively kills leukemic cells by induction of oxidative stress. J Biol Chem 2007; 282: 13468–13476. [CrossRef]
- Rampal G, Khanna N, Thind TS, Arora S and Vig AP: Role of isothiocyanates as anticancer agents andtheir contributing molecular and cellular mechanisms. Med. Chem. Drug Discov 2012; 3: 79–93.
- Kim WK, Kim JH, Jeong DH, Chun YH, Kim SH, Cho KJ and Chang MJ: Radish (Raphanus sativusleaf) ethanol extract inhibits protein and mRNA expression of ErbB2 and ErbB3 in MDA-MB-231 human breast cancer cells. Nutr Res Pract 2011; 5: 288–293. [CrossRef] [PubMed]
- Slamon DJ and Clark GM: Amplification of c-erbB-2 and Aggressive Human Breast Tumors?: Response Science 1988; 240: 1796–1798. [CrossRef]
- Takeuchi K and Ito F: EGF receptor in relation to tumor development: Molecular basis of responsiveness of cancer cells to EGFR-targeting tyrosine kinase inhibitors. FEBS J 2010; 277: 316–326. [CrossRef]
- Burgess AW, Cho HS, Eigenbrot C, Ferguson KM, Garrett TP, Leahy DJ, Lemmon MA, Sliwkowski MX, Ward CW and Yokoyama S: An open-and-shut case? Recent insights into the activation of EGF/ErbB receptors. Mol Cell 2003; 12: 541–552. [CrossRef]
- Citri A and Yarden Y: EGF–ERBB signalling: Towards the systems level. Nat Rev Mol Cell Biol 2006; 7: 505.
- Janmaat ML, Kruyt FA, Rodriguez JA and Giaccone G: Response to epidermal growth factor receptor inhibitors in non-small cell lung cancer cells: Limited antiproliferative effects and absence of apoptosis associated with persistent activity of extracellular signal-regulated kinase or Akt kinase pathways. Clin.Cancer Res 2003; 9: 2316–2326. [PubMed]
- Yuan ZQ, Sun M, Feldman RI, Wang G, Ma XL, Jiang C, Coppola D, Nicosia SV and Cheng JQ: Frequent activation of AKT2 and induction of apoptosis by inhibition of phosphoinositide-3-OH kinase/Aktpathway in human ovarian cancer. Oncogene 2000; 19: 2324. [CrossRef] [PubMed]
- Bellacosa A, De Feo D, Godwin AK, Bell DW, Cheng JQ, Altomare DA, Wan M, Dubeau L Scambia G and Masciullo V: Molecular alterations of the AKT2 oncogene in ovarian and breast carcinomas. Int J Cancer 1995; 64: 280–285. [CrossRef] [PubMed]
- Sen P, Mukherjee S, Ray D and Raha S: Involvement of the Akt/PKB signaling pathway with disease processes. Mol Cell Biochem 2003; 253: 241–246. [CrossRef] [PubMed]
- Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E, Frisch S and Reed JC: Regulation of cell death protease caspase-9 by phosphorylation. Science 1998; 282: 1318–1321. [CrossRef]
- Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J and Greenberg ME: Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999; 96: 857–868. [CrossRef]
- Tang ED, Nuñez G, Barr FG and Guan KL: Negative regulation of the for khead transcription factor FKHRby Akt J Biol Chem 1999; 274: 16741–16746. [CrossRef]
- Juge N, Mithen RF and Traka M: Molecular basis for chemoprevention by sulforaphane: A comprehensive review. Cell Mol Life Sci 2007; 64: 1105. [CrossRef] [PubMed]
- Clarke JD, Dashwood RH and Ho E: Multi-targeted prevention of cancer by sulforaphane. Cancer Lett 2008; 269: 291–304. [CrossRef] [PubMed]
- Ahn YH, Hwang Y, Liu H, Wang XJ, Zhang Y, Stephenson KK, Boronina TN, Cole RN, Dinkova-Kostova AT and Talalay P: Electrophilic tuning of the chemoprotective natural productsul for a phane. Proc Natl Acad Sci USA 2010; 2010. [CrossRef] [PubMed]
- Jin CY, Moon DO, Lee JD, Heo MS, Choi YH, Lee CM, Park YM and Kim GY: Sulforaphane sensitizestumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis through downregulation of ERK and Akt in lung adenocarcinoma A549 cells. Carcinogenesis 2006; 28: 1058–1066. [CrossRef] [PubMed]
- Kaur IP: Inhibition of cooked food-induced mutagenesis by dietary constituents: Comparison of two natural isothiocyanates. Food Chem 2009; 112: 977–981.
- Papi A, Orlandi M, Bartolini G, Barillari J, Iori R, Paolini M, Ferroni F, Fumo MG, Pedulli GF and Valgimigli L: Cytotoxic and antioxidant activity of 4-methylthio-3-butenyl isothiocyanate from Raphanus sativus (Kaiware Daikon) sprouts. J Agric Food Chem 2008; 56: 875–883. [CrossRef] [PubMed]
- Salah-Abbes JB, Abbes S, Houas Z, Abdel-Wahhab MA and Oueslati R: Zearalenone induces immunotoxicity in mice: Possible protective effects of radish extract (Raphanus sativus). J Pharm. Pharmacol 2008; 60: 761–770. [CrossRef] [PubMed]
- Beevi SS, Mangamoori LN, Subathra M and Edula JR: Hexane extract of Raphanus sativus roots inhibits cell proliferation and induces apoptosis in human cancer cells by modulating genes related to apoptoticpathway. Plant Food Hum Nutr 2010; 65: 200–209. [CrossRef] [PubMed]
- Yang M, Wang H, Zhou M, Liu W, Kuang P, Liang H and Yuan Q: The natural compound sulforaphene, as a novel anticancer reagent, targeting PI3K-AKT signaling pathway in lung cancer. Oncotarget 2016; 7: 76656. [CrossRef]
- Hanlon PR, Webber DM and Barnes DM: Aqueous extract from Spanish black radish (Raphanus sativusVar. niger) induces detoxification enzymes in the HepG2 human hepatoma cell line. J Agric Food Chem 2007; 55: 6439–6446. [CrossRef]
- Rampal G, Khanna N, Thind TS, Arora S and Vig AP: Role of isothiocyanates as anticancer agents and their contributing molecular and cellular mechanisms. Med. Chem. Drug Discov 2012; 3: 79–93.
- Ahn M, Kim J, Hong S, Kim J, Ko H, Lee NH, Kim GO and Shin T: Black Radish (Raphanus sativusvar. niger) Extract mediates its hepatoprotective effect on carbon tetrachloride-induced hepatic injury byattenuating oxidative stress. J Med Food 2018; 21: 866–875. [CrossRef] [PubMed]
- Lee SW, Yang KM, Kim JK, Nam BH, Lee CM, Jeong MH, Seo SY, Kim GY and Jo WS: Effects of white radish (Raphanus sativus) enzyme extract on hepatotoxicity. Toxicol Res 2012; 28: 165. [CrossRef][PubMed]
- Taniguchi H, Muroi R, Kobayashi-Hattori K, Uda Y, Oishi Y and Takita T: Differing effects of water-soluble and fat-soluble extracts from Japanese radish (Raphanus sativus) sprouts on carbohydrate and lipid metabolism in normal and streptozotocin-induced diabetic rats. J Nutr Sci Vitaminol 2007; 53: 261–266.
- Broadhurst CL, Polansky MM and Anderson RA: Insulin-like biological activity of culinary and medicinal plant aqueous extracts in-vitro. J Agric Food Chem 2000; 48: 849–852.
- Taniguchi H, Kobayashi-Hattori K, Tenmyo C, Kamei T, Uda Y, Sugita-Konishi Y, Oishi Y and Takita T: Effect of Japanese radish (Raphanus sativus) sprout (Kaiware-daikon) on carbohydrate and lipid metabolismsin normal and streptozotocin-induced diabetic rats. Phytother Res 2006; 20: 274–278. [CrossRef]
- Okada Y, Okada M and Sagesaka Y: Screening of dried plant seed extracts for adiponectin production activity and tumor necrosis factor-alpha inhibitory activity on 3T3-L1 adipocytes. Plant Food Hum Nutr 2010; 65: 225–232. [CrossRef]
- Ghoshal K and Bhattacharyya M: Adiponectin: Probe of the molecular paradigm associating diabetes and obesity. World J Diabetes 2015; 6: 151. [CrossRef]
- Fruebis J, Tsao TS, Javorschi S, Ebbets-Reed D, Erickson MRS, Yen FT, Bihain BE and Lodish HF: Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidationin muscle and causes weight loss in mice. Proc Natl Acad. Sci USA 2001; 98: 2005–2010.
- Deepa SS and Dong LQ: APPL1: Role in adiponectin signaling and beyond. Am J Physiol.-Endoc Metab 2009; 296: 22–36. [CrossRef]
How to cite this article:
Singh SP and Verma V: A descriptive review on Raphanus sativus Linn. Int J Pharmacognosy 2026; 13(3): 175-83. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(3).175-83.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.