A COMPREHENSIVE APPROACH ON DEVELOPMENT OF GENETICALLY STABLE PLANTS
HTML Full TextA COMPREHENSIVE APPROACH ON DEVELOPMENT OF GENETICALLY STABLE PLANTS
Vrundamohan, M. L. Lal Prasanth and A. Anu Jagajith *
Dr. Moopens College of Pharmacy, Wayanad, Kerala, India.
ABSTRACT: Pharmaceutical demand for enhanced secondary metabolite production is increased today and for meeting deficiency of secondary metabolite can be achieved by plant tissue culture. Healthy and disease-free plants can be propagated by this technique. Also endangered plants can be preserved or conserved by this technique. Micropropagation of plants of medical importance and multiplication of high yielding plants is achieved by using plant tissue culture and they are adapted to local atmospheric condition by biotechnological approach. Core principles used in plant tissue culture like Totipotency, Aseptic environment, Nutrient media. Plant tissue culture is also used in Micropropagation, Genetic engineering, Germplasma conservation, Virus elimination etc. Advantages of plant tissue culture include Disease -free plants, Faster multiplication, Year-round production, True-to type offspring, Preservation of genetic diversity. The challenges of plant tissue culture are maintaining sterility and preventing contamination requires techniques.
Keywords: Aseptic culture, Explant, In-vitro, Media, Micropropagation, Regeneration, Totipotency, Callus, Embryo rescue, Organogenesis, Somatic embryogenesis, Plant breeding
INTRODUCTION: The introduction of a comprehensive approach to genetically stable plants involves utilising advanced genetic techniques to develop crops with traits that remain stable across generations. This approach encompasses rigorous testing, precise genetic modification, and regulatory oversight to ensure the safety and stability, aim to create crops that consistency deliver desired characteristics while minimizing the risk of unintended effects or genetic drift. This approach holds promise for enhancing food security, sustainability, and resilience in agriculture. Tissue culture is the growth of tissues or cells separate from the organism.
This is typically facilitated via use of a liquid, semi -solid, or solid growth medium, such as broth or agar. Tissue culture commonly refers to the culture of animal cells and tissues, while the more specific term plant tissue culture is being named for the plants. Plant tissue culture is a kind of plant cultivation “in-vitro” or in the lab using a nutrient growth medium under aseptic conditions by cultivating seeds or different parts of plants (embryos, organs, tissues, single cells, and protoplasts).
Totipotency: Cell’s inherent ability to develop into whole plant or plant organs in in-vitro condition is known as totipotency. Not all plant cells are totipotent, however, there are a sufficient number of totipotent cells in the plant (e.g., in the pith). The cell theory (Schwann 1839) gave birth to the concept of totipotency. Single cells give rise to whole plant or organism. Manifestation of variation from single cell to morphologically and functionally different organs is known as differentiation. The process of mature cell revering to the meristematic state and forming undifferentiated callus tissue is termed as dedifferentiation. Then ability of these differentiated cells to be cultured indefinitely on fully defined medium and their capacity to regenerate into whole plant is known as redifferentiation. Reason of redifferentiation is presence of sufficient number of inactive genes that are able to express only under adequate culture condition 1.
Cellular totipotency = Dedifferentiation + Redifferentiation.
Thus, cellular totipotency is an important attribute of plant cells. It is of interest in basic as well as applied areas of plant science 2.
FIG. 1: TOTIPOTENCY
Explant: Detached portion of cell or tissue from plant organ to start in-vitro cultures is known as explant. Age and type of explants play vital role in formation of callus. Choice of explant is always based on desired research. Explant can be obtained from bud, roots, nodal segments, apical meristem, seed, embryo, anthers, pollens and or protoplast cells 1.
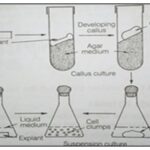
Callus: It is mass of actively dividing undifferentiated cells produced by plant tissue explants. Callus can be re suspended in liquid media to create a suspension culture of single totipotent cells or differentiated in to plant with the appropriate manipulations of culture conditions 1, 2.
Plant Regeneration:
- The process of growing an entire plant from a single cell or group of cells.
- Regeneration is possible because plant can be made totipotent using Hormones.
- Differentiated tissue: stems, leaves, roots, etc.
- Undifferentiated (embryonic) cells are totipotent: Can become a whole new plant differentiating in to a whole new plant 2.
The plant can be regenerated:
Organogenesis: It refers to the formation of organs from the cultured explants. The shoots buds or monopolar structures are formed by manipulating the ratio of cytokinin to auxins in the cultures 2.
Somatic Embryogenesis: In somatic embryogenesis, the totipotent cells may undergo embryogenic pathway to form somatic embryos which are grown to regenerate into complete plants 3.
Types:
Meristem Culture:
Explant: Shoot apical with few primordial leaves.
Media: Explant size, temperature (30-40 C) and culture media.
Meristem culture involves the development of an already existing shoot meristem and subsequently, the generation of adventitious roots from the developed shoots. For example, removal of virus: Heat treatment.
Uses: Clonal propagation, production of virus free germplasm, mass production of desirable genotypes, cryo -preservation (cold storage) or in-vitro conservation of germplasm 5.
FIG. 2: MERISTEM CULTURE
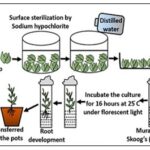
Root Culture: Root cultures can be established in vitro from explants of the root tip of either primary or lateral roots and can be cultured on fairly simple media. The growth of roots in-vitro is potentially unlimited, as roots are indeterminate organs. Although the establishment of root cultures was one of the first achievements of modern plant tissue culture, not widely used in plant transformation studies 5.
FIG. 3: ROOT CULTURE
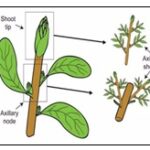
Shoot Tip Culture: Shoot tip which are larger than shoot apex employed to establish meristem culture having several leaf primordia. E.g., Artemisia annua plant contains artemisinin mainly from the leaves of the plant and its concentration varies from 0.01 to 0.05 % D W in the Chinese varieties. The callus and cell cultures of Artemisia annua did not synthesize any artemisinin, recently up to 0.165% of artemisinin has been achieved in shoot culture growing on MS media. Shoot meristem cultures are potential alternatives to the more commonly used methods for cereal regeneration. Uses: soma clonal variation and transformation of plants in disease free plants e.g., sweet potato, Cauliflower, Rhubarb 5.
FIG. 4: SHOOT TIP CULTURE
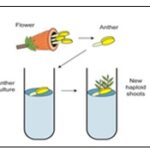
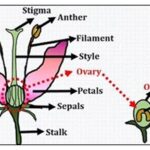
Anther Culture: Haploid plants may be obtained by placing anthers or isolated pollen grains on a suitable culture medium; this constitutes anther and pollen culture, respectively. Often, the capacity for haploid production declines with the age of donor plants. Flower buds of the appropriate developmental stage are collected, surface sterilized, and their anthers are excised and placed horizon-tally on culture medium. Flower buds with small anthers, may themselves be cultured and, in avoid injury to anthers since it may induce callus formation from anther walls. Alternatively, pollen grains may be separated from anthers and cultured on a suitable medium 6.
FIG. 5: ANTHER CULTURE
Ovary Culture: Culture of unfertilized ovaries to obtain haploid plants from egg cell or other haploid plants from egg cell or other haploid cells of the embryo sac is called ovary culture, and the process is termed as gynogenesis. The first report of gynogenesis was by San Noam in 1976 in case of barely. About 0.2-6% of the cultured Culture ovaries show gynogenesis and one or two, rarely up to 8, plantlets originate from each ovary. Ovary culture has mainly two limitations: i) So, far it has been successful only in less than two dozen species, and ii) The frequency of responding ovaries (1-5%) and the number of plantlets/ ovary (1-2) is quite low. Therefore, anther culture is preferred over ovary culture; only in those cases where anther culture fails, e.g., sugar beet, and for male sterile lines, ovary culture assumes significance. Uses: It is preferred in increasing efficiency of germination of seeds that are difficult to germinate in-vivo. Precocious germination by application of plant growth regulators is possible with seed culture 8.
Uses: Production of clean seedlings for explants or meristem culture.
FIG. 6: OVARY CULTURE
Embryo Culture: In this culture explants are egg, zygote, pro-embryo or mature embryo. Sometimes embryo or mature seeds may also be used for embryo culture. Young embryos need to be isolated with their suspensors intact as it provides gibberellins to the developing embryo.
Uses: Overcoming seed dormancy and self-sterility of seeds of seeds, due to embryo rescue interspecific or intergeneric hybridization is possible useful where endosperm development is poor, shortening of shortening of breeding cycle, production of haploid plants, production of homozygous diploid lines through chromosome doubling thus reducing the time required to produce inbred lines, uncovering mutations or recessive phenotypes, overcoming embryo abortion due to incompatibility barriers.
FIG. 7: EMBRYO CULTURE
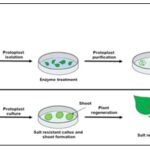
Protoplast Culture: Protoplasts are plant cells without cell wall. Protoplasts are most commonly isolated from either leaf mesophyll cells or cell suspension, although other sources can be used to advantage. Two general approaches to removing the cell wall (a difficult task without damaging the protoplast) can be taken- mechanical or enzymatic isolation.
Mechanical isolation, although possible, often results in low yields, poor quality and poor performance in culture due to substances released from damaged cells. Enzymatic isolation is usually carried in a simple salt solution with a high osmoticum, plus the cells wall degrading enzymes. It is usual to use a mix of both cellulase and pectinase enzymes, which must be of high quality and purity.
Protoplasts are fragile and easily damaged, and therefore must be cultured carefully, liquid medium is not agitated and a high osmotic potential is maintained, at least in the initial stages. The liquid medium must be shallow enough to allow aeration in the absence of agitation. Protoplasts can be plated out on to solid medium and callus produced. Whole plants can be regenerated by organogenesis from this callus. Protoplasts are ideal targets for transformation by a variety of means 9.
FIG. 8: PROTOPLAST CULTURE
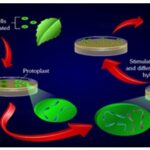
Protoplast Fusion: A number of strategies have been used to induce fusion between protoplasts of different strains/ species; of these the following three have been relatively more successful. Protoplasts of desired strains/ species are mixed in almost equal proportion; generally, they are mixed while still suspended in the enzyme mixture. The protoplast mixture is then subjected to a high pH (10.5) and high calcium concentration (50 m mol 1-1) at 37°C for about 30minutes (high pH, high ca2+ treatment). This technique is quite suitable for some species, while for some others it may be toxic. Polyethylene glycol (PEG) induced protoplast fusion is the most commonly used as it induces reproducible high frequency fusion accompanied with low toxicity to most cell types. The protoplast mixture is treated with 28-50% PEG (MW 1500-6000) for 15-30 min, followed by gradual washing of the protoplasts to remove PEG; protoplast fusion occurs during the washing. During the washing process, PEG molecules may pull out the plasmalemma components bound to them. This would disturb plasmalemma organization and may lead to the fusion of protoplasts. A more selective and less drastic approach is the electro fusion technique, which utilises low voltage (65-80 V cm-1) electric current pulses to align the protoplasts in a single raw like a pearl-chain. The high voltage creates transient disturbances in the organization of plasmalemma, which leads to the fusion of neighbouring protoplasts. The entire operation is carried out manually in specially designed equipment, called electroporator.
Uses: Combining genomes to produce somatic hybrids, asymmetric hybrids, or cybrids (hybrids of mitochondrion genome with nuclear genome), production of organelle recombinants, transfer of cytoplasmic male sterility.
FIG. 9: PROTOPLAST FUSION
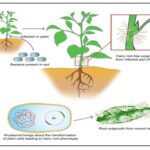
Hairy Root Culture: Hair root is a plant disease caused by Agrobacterium rhizogenes Conn., a Gram-negative soil bacterium and is capable of infecting a wide range of plant species, causing crown gall diseases. It has natural transformation abilities. In this culture hairy roots are produced. Hairy roots are highly branched hair like structured root. Plant tissue- infected with Agrobacterium rhizogenes - this infection causes formation of hairy root. Root tip, shoot or leaf piece is used as explant and transferred in to nutrient media to develop callus. This callus is then infected with bacteria. This bacterium contains Ri plasmid. Due to infection segment of Ri plasmid get transferred in to callus. This segment gets integrate along with generate material of callus and callus cell get transformed and produces hairy root 10.
FIG. 10: HAIRY ROOT CULTURE
Aseptic Condition: Complete aseptic conditions are required for tissue culture. The, tissues, equipment’s, culture media, and the room should be free from microorganisms. Usually dry heat, wet heat, ultrafiltration and chemicals are used for the sterilisation process. The equipment is sterilised by the dry heat method in an autoclave. Wet heat sterilisation used for glass wares involves autoclaving at 1210c and 15lb pressure for 15 minutes. Ultrafiltration is used for the sterilisation of liquid media which are unstable at high temperature. The working area and the instruments are sterilised by chemicals such as alcohol 12.
Surface Sterilisation of Material: Materials such as seed, fruit, stem, leaf, etc need to be subjected to surface sterilisation, to prevent microbial contamination since, they usually carry microorganisms on a normal basis. The following sterilisation agents are used:
- 9-10% calcium hypochlorite for 5-30minutes
- 2% sodium hypochlorite solution for 5-30 minutes. The materials need to be washed
- 10-12% of hydrogen peroxide solution for 5-15minutes.
- 1-2% bromine water, for 2-10 minutes.
- 1% solution of chlorine water, mercuric chloride, silver nitrate or antibiotics, etc, can also be used.
- Absolute alcohol is used for hard tissues 15.
Sterilisation of Equipment: Chromic acid sulphuric acid mixture, hydrochloric acid, nitric acid, strong detergent solution, alcohol, incubator or autoclave etc are used for this purpose.
- Glass wares are also sterilised by dipping overnight in a strong detergent solution and then washing with hot water followed by cold sterile water. To overcome the corrosive nature of the chromic acid- sulphuric acid mixture, detergent solutions are used 16.
- Autoclave can also use to sterilise equipment’s, usually at 1210c for 20-30 minutes at 15lb pressure. De- mineralised water must be used in an autoclave. Aluminium foil is not suitable for wrapping, as it is impermeable to steam. Unwaxed papers are used for the purpose.
- UV light can also use for sterilisation purpose.
Sterilisation of Culture Medium: The culture medium is sterilised by autoclaving at 15lb pressure at 1210c for 15-30 minutes. The autoclaving time depends upon the volume of the culture volume. e.g., 75ml of liquid per vial requires a period of 20 minutes; 25-5000ml/vial requires 25 minutes and 1000ml/vial needs 30 minutes. After sterilisation, the medium should be used within 14 days. The gibberellins, and other chemicals used in culture media, undergo degradation at high temperature during autoclaving. Antibiotics are added to the medium to prevent the growth of the microorganism.eg., potassium benzyl penicillin (100micro gram/ml) which prevents the growth of gram +ve bacteria, streptomycin sulphate(100microgram/ml) which prevents the growth of gram -ve bacteria and gentamycin (100microgram/ml) which prevents the growth of growth both gram +ve and gram -ve bacteria. Usually, a large quantity of distilled water required for plant tissue culture. The distilled water may also be sterilised by autoclaving for an hour in a Pyrex glass flask provided with a non- absorbent plug fitted to its mouth 19.
Sterilisation of Environment: The tissue culture room should be sterilised by the fumigation method. The incubation chamber is sterilised with sterilised with UV rays before inoculation. The UV germicidal lamp is turned on about two hours before initiation and should be turned off before using the chamber. The chamber is kept free from microorganism by a continuous flow of sterilised air. The surface of the inoculation chamber must be wiped with ethyl alcohol or isopropanol (70% or 80%). Before inoculating the tissue into the culture vessel, the neck off the vessel is quickly flamed. Its plug is rapidly removed, the tissue inoculated and the plug immediately replaced. Before initiating the inoculation work, hands must be washed thoroughly with soap and hot water for several minutes and the skin wiped with paper towels. Before certain operations, such as removed seed coats by hands, the hands and fingers maybe dipped in aeration.
Proper or Optimum Aeration: Aeration is required for the cell to grow. Tissues which are cultured on semisolid media do not require any special method for aeration, but tissues which are grown in suspension cultures, requires special devices for aeration. The aeration for submerged cultures can be provided by following methods:
- By placing the culture vessel with the liquid medium on an automatic shaker.
- Filter paper bridge method can also be used i.e.; the two ends of the filter paper are dipped in a medium and the middle horizontal portion on which the tissue is placed remains above the level of the medium.
- Aeration can be also be provided by passing sterilised air through the medium and by stirring the medium.
The culture vessels are closed with non- adsorbent cotton covered in cheese cloth. This process allows proper aeration but prevents the entry of micro- organisms 19.
Equipments: Culture vessels: Usually borosilicate glass vessels are preferred, since they can be easily sterilised and their mouths flamed during transfer processes. The culture vessels include test tubes, conical flasks, bottles special flat tubes.
- Different types of glass wares like measuring cylinders, beakers, funnels, petri dishes, graduated pipette, conical flask etc. are required for the preparation of nutrient media.
- Non- absorbent cotton plug, screw cap or polyurethane foam is required to close the mouth of the culture vessel.
- Spirit lamps are required to sterilise the mouth of the culture vessels.
- Scissors, scalpels and forceps are needed for explant preparation from excised plant parts and for their transfer.
- An autoclave is required for sterilisation the nutrient media and glass wares, scissors, forceps, etc., prior to use.
- An inoculator is also required for maintaining the temperature conditions to facilitate the culture of callus and its subsequent maintenance.
- A shaker is also needed for the suspension of cell culture.
- An inoculating chamber is a must, to conduct all aseptic transfers. The inner surface of the chamber is sterilised by wiping out with ethyl alcohol and allowing sterile air to pass through it.
- A sensitive balance is required to weigh the chemicals and nutrient elements.
- A refrigerator is needed for storing chemicals like growth hormones, , and stock solutions.
- A pH meter for adjusting the pH of the media, and other instruments like magnetic stirrer and centrifuge are also needed.
Hands lens, dissecting microscope, compound microscope with microphotographic facilities, and shelves for keeping the reagents, and also labels, marking pencils etc 17.
Suitable Nutrient Medium 22: The isolated plant cells and tissues require a basal medium for their growth. The medium generally contains inorganic salt of major and minor elements, carbon source, growth hormones and vitamins. The composition of tissue culture media involves the following ingredients:
Inorganic Substance: Macro - elements (in mmol/l) like Na, K, P, Ca, Mg and S and micro elements (in mmol/l) like Ba, Zn, Cu, Mo, Cl, Ni, Al etc are used. Ammonium nitrate or any ammonium salt serves as the nitrogen source. KCl, KNO3, or KHPO4 is the potassium supplement while CaCl2.2H2O, Ca (NO3).4H2O or its anhydrous form is used as the calcium source. Magnesium and sulphur are added in the form of magnesium sulphate. Phosphorous is obtained from NaH2PO4.H2O or KH2PO4.
Organic Substance: Sugar, usually 2-4% of sucrose or glucose is used.
Vitamins and Amino Acid: Thiamine hydrochloride (0.1-1mg/1), pyridoxine(0.5mg/l), nicotinic acid (0.5mg/l), folic acid (0.5mg/l), pantothenic acid(0.1mg/l) and thiamine (vit B1). The stock solution of all these substances at 100 times the required concentration is prepared and stored in a refrigerator. Since, the vitamins are heat sensitive, they are filter sterilised and added with a syringe or pipette to the warmun solidified autoclaved medium. The presence of amino acids like arginine, aspartic acid, glutamic acid, glutamine or methionine is beneficial.
Growth Hormones 24, 23: Growth hormones play a very important role in callus culture. Auxins and cytokinin’s promote cell division, cell elongation, cell differentiation and organ formation.
IAA (indole-3-acetic acid,1-50mg/l), NAA (naphthalene acetic acid,0.1-10mg/l) and 2,4-D (2,4-dichlorophenoxy acetic acid, 0.05-0.5mg/l) are used as auxin source. Kinetin (0.01-10mg/l), zeatin and benzyl adenine are used as cytokinin. Coconut water, yeast extract, casein hydrolysate, malt extract is also added to supplement the media when chemically defined media do not produce desired results.
Carbon Source: Activated charcoal (the adsorbent) is also used in the used in the media because of the following reasons:
- Removes unwanted substances or contaminants from the media.
- Controls the supply of endogenous growth hormones.
- Darkens the supporting matrix.
- Adsorbs the secondary products secreted by the culture tissue.
- De- mineralises or double -distilled water.
- Agar is used as a solidifying or gelling agent, usually at a concentration of 0.6-1% agar 28.
Respiration and Metabolism: Studies of respiration of growing callus provides means of separating between normal and disease growth. Enzymes levels provide idea regarding composition of media and growth parameters of developing callus. Changes in the composition of the medium alter different phases of metabolism that control cell enlargement. For e.g., Changes in media causes increase or decrease in respiration and ascorbic acid oxidase activity in cultured tobacco pith parenchyma 33.
Proliferation and Organ Function: Tissue culture allow to study different types of proliferation such as sections may proliferated as a layer over cut surface; proliferation may be from preexisting cambia; many times, sections develop roots or bud meristems, parenchymatous cultures may develop vascular bund; out-standing efforts have been made to clarify cell growth, cell division, callus formation and organ differentiation in terms of growth substances and balances of common metabolites.
Biotransformation: The phenomenon of precursor feeding leading to its subsequent conversion into other compounds by living cells through functional group modification is called as biotransformation. This process can be done by using microorganism or plant cell suspension, hairy root culture and immobilized cells.
The advantage of biotransformation by plant cell system over non-biochemical reaction system includes: selectivity, specificity, stereo selectivity etc. wide range of reaction such as glycosylation, glycosyl esterification, hydroxylation, isomerization, methylation, demethylation, dehydrogenation etc. Example: Peganum harmala cell culture converted geranyl acetate to geraniol and linalyl acetate to linalool and alpha terpineol 35.
Production of Secondary Metabolites: Plant tissue culture techniques offer an excellent alternative through cell culture, organ culture, endophytes, genetic manipulations and hairy root culture is also becoming very popular method of producing secondary metabolites from plant roots. E.g., cardiac glycosides, morphine and codeine etc 34.
Cloning: Cloning is based on single cells separated from tissues and cultured in a manner to allow separate recovery of the cell mass derived from them.
Mutant Selection: It is an important tool for crop improvement. Cells are subjected to mutagenic treatments and subsequently mutants are selected. Selection of mutant cells is usually performed by addition of toxic substance to cells followed by isolation of resistant cells. In this manner cell lines of potato resistant to 5-methyl-tryptophan were selected and these cultures permitted accumulation of free phenyl alanine, tryptophan and tyrosine. Induction of polyploidy introduction of genetic variability 37.
Micro Propagation: It is common method being utilize both at research and commercial level. It can be employed for the mass production of plant including nursery stock species, ornamental plants, vegetables and field crop.
Clonal Propagation: It involves propagation through technique of cell, tissue or organ culture. Advantage: rapid multiplication of superior clones, maintenance of genetic uniformity, multiplication of sexually derived sterile hybrids. In this technique shoot tips or auxiliary buds are utilised for propagation on culture media without intervention of callus phase.
Embryo Rescue: In many plants normal fertilization occurs but ovule fails to develop into a mature seed. In such cases, fertilised egg or immature embryo is removed and cultured to generate hybrids with few new characters. This is called embryo rescue.
Germ Plasm Storage: Generally, most of the plants are stored in the form of seeds or plantlets. But some seeds fail to grow in to plantlets. This problem can be overcome by raising identical clones in form of seeds, buds, protoplasts, shoot tips. This material is then stored in a minimal medium and with low light intensity and temperature or cryopreserving (storage at low temperature using liquid nitrogen) without affecting the viability. This is called germplasm storage which is then used to develop whole plant.
Single Cell Culture of Higher Plants: Due to tissue culture technique, it is possible to isolate and grow single cell of higher plant. This single cell method enables to evaluate additional chemical, morphological, genetical and pathological similarities and differences. Even clarification and location of virus infection can be studied by single cell. E.g., Tobaccco plant 40.
Challenges and Future Prospects
Challenges: The most common challenges in tissue culture include contamination, vitrification, browning of media, recalcitrance, soma clonal variation and loss of totipotency.
Contamination: Tissue culture requires a completely sterilised or aseptic environment. But, any negligence in maintaining the proper environment may lead to contamination of cultures. It will affect the time, money, and effort that you have put into culturing plants. The most common microorganisms affecting cultures include fungi, bacteria, Molds, and yeasts. In some cases, they are introduced from an outside source but in others, they might be residing inside associated with explant tissues, as endophytes.
Prevention:
- The culture room should be clean and the culture space should be organized, equipped with laminar flow, and sterilised with alcohol before working.
- The consumables, culture vessels, media and water should be sterilised.
- The explants should be properly surface sterilised.
- Use (ppm) plant preservative mixture like products to prevent contamination from the cultured plants.
Vitrification: It is a physiological disturbance or malformation occurring in tissue culture plants and affecting their proper growth and development. In this case, the plants have glassy appearances and thick, enlarged, and brittle leaves and stems. The main causes of vitrification in tissue culture plants are believed to the factors triggering oxidative stress. It includes:
- High salt concentration.
- High relative humidity.
- Low light intensity.
- Gas accumulation in the atmosphere of the jar.
- Length of time intervals between subcultures.
- The number of subcultures, concentration, and type of gelling agent.
- The type of explant used.
- The concentrations of microelements.
- Hormonal imbalances (high level of cytokinin’s).
Prevention:
- Increase carbohydrate level in the medium.
- Change light intensity.
- Modify concentration of agar.
- Reduce humidity in the culture container.
- In some cases, an increase in calcium concentration has been observed to reduce the problem.
Browning of Media and Explant: Tissue browning is the result of the accumulation and oxidation of phenolic compounds in culture tissue. It reduces the rate of cell division and regeneration capacity of explants that cause either their death or null responses in culture media. The phenomenon can be majorly observed in woody plant species.
Prevention:
- Use activated charcoal in the media to avoid the accumulation of inhibitory phenolic compounds.
- Use antioxidants and citric acid in the media to prevent the oxidation of phenolic compounds.
- Subculture the explants after a period of time to avoid the accumulation of the browning compounds.
- Use a sharp edge knife to cut the explants and keep them in water for a while, which act as the best solvent for secondary compounds present in tissues.
- In some cases, explants placed in vertical position also help against browning conditions.
Recalcitrance: Recalcitrance is defined as the inability of plant cells and tissues to respond in an in vitro culture environment. Tissue recalcitrance is generated because of the reaction of free radicals with the macromolecules of cells, causing cellular dysfunction.
Prevention:
- Select juvenile parts of the plants as explant.
- Rejuvenate parts of desired plants using cytokinin spray for tissue culture.
Soma Clonal Variation: It is the phenotypic or genotypic changes in the cultured plant. It occurs due to change in chromosome structure and number; and DNA sequence. The phenomenon is termed genetic heterogeneity.
Prevention:
- Avoid long term cultures.
- Prevent use of 2,4-D in culture media.
- Use auxiliary shoot induction systems.
- Keep the number of subculturing plants at a minimum.
- Re initiate cultures from new plants.
Acclimatization: Acclimatization is the adaptability of tissue cultured plants when brought to the natural environment or in the greenhouse. You must know, plants cultured in a completely artificial environment go under shock and die if not acclimated properly.
Prevention:
- Optimize the hardening conditions for plants such as mineral nutrition in media.
- Establish a photoautotrophic system.
- Gradually adapt the plant in ex-vivo Like, gradually adjusting the percentage, humidity for in-vitro plants when transferred to ex-vitro conditions.
Future Prospects: The promise of biotechnology may be realized by successfully inserting horticulturally genes in to the genome of desirable genotypes. This is been achieved by parasexual hybridization (protoplast fusion).
In near future the following can help the further development of tissue culture techniques:
- Determination of mechanism, frequency and utility of soma clonal variants.
- New technologies can be undertaken for further understanding plant physiology and genetics.
- Discovery of new chemicals used in tissue culture.
- Efficient computer controlled, flow-through systems will be tailored to meet environmental and medium requirements from explants to finished plantlets (Kozai et al., numerous citations).
- Mass production of plant constituents (secondary production of pharmaceutical, flavouring, other uses).
- Tissue culture plants (TCPs) may have increased branching and flowering, greater vigour and higher yield, many due to possibility of elimination of diseases.
Centers Conducting Plant Tissue Culture:
Glaxo Smith Kline (GSK): GlaxoSmithKline is a global pharmaceutical company that also has a significant plant tissue culture business. The company uses plant tissue culture to produce vaccines, medicines, and other products.
Mosa Meat: Mosa Meat is a Dutch company that is developing cultured meat, which is meat that is produced in a laboratory without the need to slaughter animals. The company uses plant tissue culture to produce the scaffolding for the cultured meat.
Laboratories Pierre Fabre: Laboratories Pierre Fabre is a French pharmaceutical company that also has a significant plant tissue culture business. The company uses plant tissue culture to produce phytomedicines, which are medicines derived from plants.
Spiderwort: Spiderwort is an American biotechnology company that is developing a new type of plant tissue culture technology that is more efficient and less expensive than traditional methods.
Phytotechnology Laboratories: Phytotechnology Laboratories is an American company that provides plant tissue culture services to the pharmaceutical, agricultural, and cosmetic industries.
Ricetec, inc.: RiceTec, Inc. is an American company that develops and markets high-yielding rice varieties using plant tissue culture.
Segra: Segra is a plant biotechnology company that specializes in industrial-scale cannabis plant micropropagation also known as “plant tissue culture”.
Plant Mill: Plantmill is a biotechnology company located in Thrissur, Kerala, India. It uses tissue culture to propagate disease-free plants.
Green Clones Naturals PVT LTD: Green Clones Naturals PVT LTD is a biotechnology company located in Kochi, India. It specializes in plant tissue culture for a variety of crops, including flowers, fruits, and vegetables.
Marketing Product from Plant Tissue Culture:
Agriculture: PTC is used to produce large numbers of plants that are genetically identical to the parent plant. This is important for farmers who want to ensure that their crops are consistent in quality and yield.
Horticulture: PTC is used to propagate ornamental plants, such as flowers and shrubs. This allows growers to produce plants with desirable traits, such as disease resistance and compact growth habits.
Forestry: PTC is used to propagate trees for reforestation and forestry applications. This allows foresters to plant trees that are genetically resistant to pests and diseases.
Pharmaceuticals: PTC is used to produce medicinal plants. This allows pharmaceutical companies to produce a consistent supply of high-quality medicinal plants.
Cosmetics: PTC is used to produce plants that are used in cosmetics. This allows cosmetic companies to produce a consistent supply of high-quality plant-based ingredients.
Bananas: PTC is used to produce bananas that are resistant to diseases, such as black Sigatoka.
Apples: PTC is used to produce apples that are disease-resistant, have a long shelf life, and have good flavour.
Oranges: PTC is used to produce oranges that are seedless, have a high juice content, and are easy to peels.
Roses: PTC is used to produce roses that are disease-resistant, have long stems, and have beautiful flowers.
Vanilla: PTC is used to produce vanilla plants that are high-yielding and produce high-quality vanilla beans
CONCLUSION: Plant tissue culture is the science of growing plant cells, tissues or organs in a controlled environment. It is a powerful tool that has revolutionized the field of plant science. It has enabled the rapid propagation of plants, the production of disease-free plants, and the development of new plant varieties. Plant tissue culture is also used for the production of secondary metabolites, such as pharmaceuticals and cosmetics. Tissue culture plays a critical role in the genetic transformation of a plant species. It is an important tool for the continuous production of active compounds including secondary metabolites and engineered molecules. Plant tissue and cell culture describes the sterile growth and multiplication of plant cells, tissue in vitro. Plant cells cultured with nutrient media in an artificial environment can be clonally propagated at scale, quickly produce mature plant. Plant tissue culture offers a powerful tool for rapid propagation, disease eradication, germplasm preservation and plant improvement. Its versality and precision make it a valuable asset in agriculture, horticulture and research addressing existing challenges will further unlock its potential for sustainable and efficient plant product.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Scragg AH: Plant cell culture. J Bioethanol 1992; 26(1): 243–432.
- Pulianmackal AJ, Kareem AVK, Durgaprasad K, Trivedi ZB and Prasad K: Competence and regulatory interactions during regeneration in plants. Front Plant Sci 2014; 5(4): 1–16
- George EF and Sherrington PD: Plant propagation by tissue culture: handbook and directory of commercial laboratories. Eversley 1984.
- Shimomura K, Yoshimatsu K, Jaziri M and Ishimaru K: Traditional medicinal plant genetic resources and biotechnology applications: Plant Biotechnology and Plant Genetic resources for Sustainability and Productivity 1997; 295–316.
- Preil W: Micropropagation of ornamental plants. Plant Tissue Culture, 100 years since Gottlieb Haberlandt 2003; 115–133.
- Pierik RLM: Commercial Aspects of Micropropagation. Hortic - New Technol Appl 1991; 141–153.
- O’Riordain F: Directory of European plant tissue culture laboratories, 1996-97. Vol. 822, Brussels: Commission of the European Communities 1999; 36-54.
- Chebet DK, Okeno JA and Mathenge P: Biotechnological approaches to improve horticultural crop production in Kenya. Acta Hortic 2003; 625: 473-479.
- Tal B, Roken JS and Goldberg I: Factors affecting growth and product formation in plant cells grown in continuous culture. Plant cell Rep 2: 219-222.
- Hansen G and Wright MS: Recent advances in the transformation of plants. Trends Plant Sci 4: 226-231.
- Akin-Idowu PE, Ibitoye DO and Ademoyegun OT: Tissue culture as plant production technique for plant horticultural crop. Afr J Biotechnol 2009; 8(16): 3782-3788.
- Ammirato PV: Embryogenesis. Handbook of plant cell culture. Event, D., sharp, W.R., ammirato, P.V. and Yamada, Y.(Eds.) Maccmillan, New York 1983; 82-123.
- Bajaj YPS: Somatic Embryogenesis and Synthetic Seed, Biotechnology in Agriculture and Forestry, Springer-Verleg, Berlin 1995; 30.
- Bhojwani SS and Razdan MK:Plant Tissue Culture: Theory and Practice, Elsevier Science Pub, Amsterdam, 1983; 4.
- Cyr DR, Black M and Bewley JD: Seed substitutes from the laboratory, Seed Technology and its Biological Basis.(Eds.).Sheffield Acad. Press, Sheffield 2000; 326-372.
- Cutter EG: Recent experiment studies of the shoot apex and shoot morphogenesis, Bot. REV 31: 7-113.
- George EF: Plant propagation by tissue culture. Part 1, the Technology, Exegetics Ltd, Edington 1993; 7.
- Guri AZ and Patel KN: Composition and method to prevent microbial contamination of plant tissue culture media, U.S. Patent No. 5,750.420.8)
- Hanning GE and Conger BV: Factors influencing somatic embryogenesis from cultured leaf segment of Dactylis glomerata. J plant Physiol 1986; 123: 23-29.
- Leifert C and Cassells AC: Microbial hazards in plant tissue and cell cultures. In-vitro Cellular and Development Biology 2001; 37: 133-138.
- Debergh PC and Zimmerman RH: Micropropagation, Technology and Application, Kluwer Academic Pubishers 1991; 11.
- Donnelly DJ and Vidaver WE: Glossary of plant Tissue Culture, Timber Press, Porland 1988; 12.
- Kyte LJ and Kleyn: Plants from Test Tubes: An Introduction to Micropropagation. Ed 3rd Timber Press 1996; 13.
- Smith RH: Plantt tissue culture-techniques and experiments. Academic Press 1992; 14.
- Freshney RI and Wiley L: Culture of Animal cells: A Manual of Basic Techniques 1987; 15.
- Misawa M and Fiechter A: “Adv. In biochem. Eng. /Biotech, Springer -Verlag, Berlin, Heidelberg, New York, Tokyo 1985; 73: 16.
- Trigiano RN and Dennis JG: Plant tissue culture concepts and laboratory exercises. CRC Press 1996; 17.
- Barz W and Wagner F: “Plant Tissue Culture and its Bio-technological application”, Springer -Verlag, Berlin Heidelberg 1977; 250: 18.
- Martin SM and Staba EJ: In “plant tissue culture as a source of biochemicals”, CRCOress, Florida, USA 1989; 151-164.
- Tanaka H: Biotechnol Bioeng, Florida, USA 1983; 23-24: 5920.
- Dostoevsky F and Irodov I: “Essential of plant Biotechnology”, San Puerto publication, 2nd Edition 1123-1131.
- Bannerman RH: Traditional medicine in modern health care. World Health Forum 1982; 3(1): 8–133.
- Sarker KP, Islam A, Islam R, Hoque A and Joarder I: In-vitro propagation of Rauvolfia serpentina Benth. ex Kurz. through tissue culture. Planta Med 1996; 62: 358–3594.
- Vieira RF and Skorupa LA: Brazilian medicinal plants gene bank. Acta Hortic 1993; 30: 51–585.
- Farnsworth NR: Biological and phytochemical screening of plants. J Pharm Sci 1966; 55(3): 225–2766.
- Balandrin MJ and Klocke JA: Medicinal, aromatic and industrial materials from plants. In:Bajaj YPS (ed) Biotechnology in agriculture and forestry, vol 4. Springer, Heidelberg 1988; 1–367.
- Kumar S, Hassan SA, Dwived S, Kukreja AK, Sharma A, Singh AK, Sharma S and Tewari R: Proceedings of the national seminar on the frontiers of research and development inmedicinal plants. J Med Aromatic Plants Sci 2000; 22(4): 461–4638.
- Cheetham PSJ: Biotransformations: new routes to food ingredients. Chem Ind 1995; 7: 265–26855.
- Scragg AH: The production of aromas by plant cell cultures. In: Schepier T (ed) Advance biochemical and engineering biotechnology. Springer, Berlin 1997; 56.
- Krings U and Berger RG: Biotechnological production of flavours and fragrances. Appl Microb Biotechnol 1998; 49: 1–857.
- Verma P, Mathur AK, Srivastava A and Mathur A: Emerging trends in research on spatialand temporal organization of terpenoid indole alkaloid pathway in Catharanthus roseus: literature update. Protoplasma 2012; 249(2): 255–26858.
How to cite this article:
Vrundamohan, Prasanth MLL and Jagajith AA: A comprehensive approach on development of genetically stable plants. Int J Pharmacognosy 2024; 11(4): 131-43. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.11(4).131-43.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.