SIMPLE METHOD OF ISOLATION OF LOGANIN FROM NUX VOMICA FRUITS AND ITS QUANTIFICATION BY HPLC
HTML Full TextSIMPLE METHOD OF ISOLATION OF LOGANIN FROM NUX VOMICA FRUITS AND ITS QUANTIFICATION BY HPLC
S. P. Patil * and K. S. Laddha
Medicinal and Natural Product Research Laboratory, Department of Pharmaceutical Sciences and Technology, Institute of Chemical Technology, Matunga, Mumbai - 400019, Maharashtra, India.
ABSTRACT: Loganin is an iridoid glycoside, a key phytoconstituent in Strychnos plants having a crucial role in the biosynthesis of indole, monoterpene alkaloids and other natural products. It is a bitter iridoid glycoside with numerous activities like anti-inflammatory, anti-oxidant, glucose-lowering and anti-diabetic nephropathy activities. The objective of this study was to develop a simple method for isolation of loganin from fruit pulp of Nux vomica and quantify by HPLC. The isolated compound was characterized by TLC, UV, IR, MS, and NMR spectral analysis and purity were confirmed by HPLC analysis. Quantification of loganin was done from fruit, seeds and pulp extract using HPLC. A simple solvent partitioning method was used for the extraction and isolation process. This isolation method developed involved easy solvent-solvent extraction technique followed by recrystallization to purify the compound.
| Keywords: |
Loganin, Strychnos, Loganiaceae, Iridoid, HPLC
INTRODUCTION: Loganin is a simple monoterpenoid iridoid glycoside obtained in highest concentration from fruit pulp of Nux vomica. It is used as a precursor in the biosynthesis of indole alkaloids and seco iridoids 1, 2. It has a wide range of pharmacological activities like anti-microbial, hepatoprotective, anti-tumor, immune-stimulating, anti-inflammatory, leishmanicidal and cytotoxic activities 3. Loganin as well as its chemical analogs that have been semi-synthesized show activity against human cancer cells 4. It is named after Loganiaceae having first isolated from Nux vomica seeds. It also occurs in Aistania boonei (Apocynaceae), Derfontainia spinosa (Collumelliaceae), Cornus officinalis (Cornaceae), Vinca rosea (Apocynaceae), Menyanthes trifoliata (Menyanthaceae) 5.
Loganin is frequently isolated by column chromatography which is tedious and time-consuming method. Another patented method is reported in which concentrated butanolic extract is refrigerated overnight to obtain a white precipitate of loganin, but this method is least reproducible and not feasible in case of plant material having very low concentrations of loganin. Synthesis of loganin is also accomplished by photochemical cycloaddition of 2-formyl malonaldehyde acid methyl ester to the tetrahydropyronyl ether of 3-cyclopentenol 6.
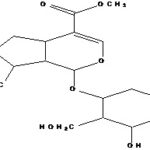
FIG. 1: STRUCTURE OF LOGANIN
MATERIALS AND METHOD:
Plant Material: Fruits of Nux vomica (kuchla) were procured from Goa (Mavlinge farm) which was further authenticated, and ICT/MNPRL/NV/01 voucher specimen was deposited in Medicinal Natural Products Laboratory, Department of Pharmaceutical Sciences and Technology, Institute of Chemical Technology, Matunga, Mumbai.
Chemicals: All the chemicals used for extraction and isolation were of Laboratory reagent grade and obtained from S.D. fine, India. All the solvents used for HPLC analysis were of Analytical reagent grade and obtained from Rankem Limited, India.
Instrumentation: UV spectrum was recorded on Jasco V-530 spectrophotometer. IR spectra were recorded on a Shimadzu instrument, HPLC analysis was performed with a Jasco (C18 HiQ sil column-250 mm × 4.6 mm i.d), Detector: Jasco UV-1575. Mass spectrum was recorded on Micro-mass Q-TOF MS Mass spectrometer. HNMR spectra were recorded on a JOEL 400-MHz instrument with an internal standard of Tetramethylsilane (TMS).
Experimental:
Method of Isolation: Nux vomica fruits were peeled, seeds were separated, and pulp was removed (100g). The pulp was extracted with methanol (500 ml) for 6 h using Soxhlet extractor. Methanolic extract was filtered, concentrated and suspended in water. This was then partitioned three times with each of the solvents; petroleum ether, chloroform, and ethyl acetate respectively. Ethyl acetate layer was concentrated, precipitated using petroleum ether (0.81g). This precipitate obtained was recrystallized with ethanol to give pure loganin (0.56g). TLC and HPLC studies were carried out to determine the purity of the sample while the structure was elucidated and confirmed by UV, IR, MS, and 1HNMR spectral analysis.
Quantification of Loganin in the Pulp, Seeds, and Peel of Strychnos Fruits by HPLC:
Loganin Standard Solution Preparation: Stock solution of Loganin was prepared in HPLC grade methanol at a concentration of 1 mg/ml. Working solutions of 25, 50, 100, 150 and 200 µg/mL were prepared in methanol. The calibration plot was then made for concentration (µg/mL) versus peak area. The linear equation from the standard plot was used to determine concentration of Loganin in test samples.
Sample Solution Preparation: Continuous hot solvent extraction of fruit pulp was done using a Soxhlet apparatus for 2 h at 50 ºC using solvent drug ratio 10:1 (ml/g). The solvent from the extract was evaporated to obtain a residue. 10mg of the residue was weighed and dissolved in HPLC grade methanol to make up volume up to 10 mL. The solutions were then filtered through a 0.2 µm membrane filter, and 20 µL of the resulting solution was subjected to HPLC analysis. The final concentration of loganin in the extracts was calculated by using the linear equation for the calibration curve.
Chromatographic Conditions: HPLC analysis of the extracts was performed on HPLC Jasco, PU-1580, UV detector Jasco UV -1575, column C18 HiQ sil column-250 mm × 4.6 mm i.d. Samples were injected by using a Rheodyne injector fitted with a 20 µL fixed loop. Standard and sample solutions were filtered before injection. Determination of Loganin was carried out with the mobile phase composed of acetonitrile: water with 0.5% OPA (16:84) at wavelength 236 nm at a flow rate of 1.0 mL/min 7.
RESULTS AND DISCUSSION: Studies of loganin have reported that its structure plays an important role in the biosynthesis of secoiridoids and indole alkaloids. The objective of the study was to develop a rapid and convenient method for isolation of loganin from Nux vomica fruits. A literature survey has revealed that efforts have been made previously to isolate loganin from pulp and seeds of Nux vomica fruits which involved column chromatography, however, the yield obtained was very low and it was tedious and time-consuming 8. The advantage of current method is to isolate loganin to obtain better yield, purity and in the shorter time. For extraction and isolation process, a simple solvent partitioning method was used. Partitioning with petroleum ether and chloroform removed all non polar and fatty substances from the extract, whereas ethyl acetate being polar extracted loganin leaving the water-soluble impurities in an aqueous layer. After concentrating this layer, precipitation of crude loganin was done using petroleum ether. As loganin is insoluble in petroleum ether, it precipitates out quickly, for further purification recrystallization was done with ethanol since it is less soluble in absolute alcohol.
Thin layer chromatography was performed on pre-coated silica gel plates using Ethyl acetate: methanol: water (77:15:8 v/v) as mobile phase 9. A single band was seen at Rf 0.45 at 254nm. The melting point was found to be in the range of 221-223 ºC. The UV/Vis maxima were found to be at 237 nm in methanol which is identical with reported literature. IR spectrum of isolated compound showed characteristic peaks at 1600 cm-1 (C=C), 1714 cm-1 (Carbonyl), 3400cm-1(Hydroxyl). The 1H NMR analysis (400MHz, CDCl3) exhibited δ 1.08 (d, 3H, CH3) and 3.72(s, 3H, COOCH3). [M+Na]+ was found to be 413.17 and base peak observed at 802.76. The fragmentation pattern of mass spectroscopy and 1HNMR data of isolated compound was found in accordance with reported values in literature 10, 11.
FIG. 2: TLC ANALYSIS OF METHANOLIC EXTRACT NUX VOMICA FRUITS
Track 1: Isolated Loganin
Track 2: methanolic extract of the pulp of Nux vomica fruits.
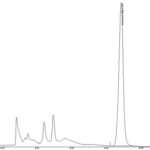
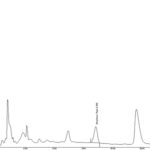
FIG. 3: HPLC CHROMATOGRAM OF ISOLATED LOGANIN
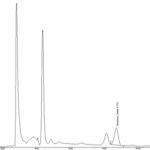
FIG. 4: HPLC CHROMATOGRAM OF PULP EXTRACT OF NUX-VOMICA FRUITS
FIG. 5: HPLC CHROMATOGRAM OF SEED EXTRACT OF NUX-VOMICA FRUITS
FIG. 6: HPLC CHROMATOGRAM OF PEEL EXTRACT OF NUX-VOMICA FRUITS
TABLE 1: PERCENTAGE OF LOGANIN ESTIMATED FROM STRYCHNOS FRUIT PULP, SEEDS AND PEELS
| S. no, | Sample | Percentage of Loganin (%) |
| 1 | Pulp | 1.25 |
| 2 | Seeds | 0.18 |
| 3 | Peel | 0.14 |
CONCLUSION: This isolation method developed involved easy solvent-solvent extraction technique followed by recrystallization to purify the compound. The reported method of isolation has shown good reproducibility in terms of purity and yield and can be further optimized for large scale production.
ACKNOWLEDGEMENT: The authors are grateful to the University Grant Commission, New Delhi, India, for providing financial assistance.
CONFLICT OF INTEREST: We declare that we have no conflict of interest.
REFERENCES:
- Berger A, Kostyan MK, Klose SI, Gastegger M, Lorbeer E, Brecker L and Schinner J: Loganin and secologanin derived tryptamine – iridoid alkaloids from Palicourea crocea and Palicourea padifolia (Rubiaceae). Phytochemistry 2015; 116: 162-169.
- Inoue K, Tanahashi T, Inouye H, Kuwajima H and Takaishq K: Intermediacy of 6-hydroxyloganin in the ring cleavage course of loganin to secologanin. Phytochemistry 1989; 28: 2971-2979.
- Tseng YT, Chen CS, Jong YJ, Chang FR and Lo YC: Loganin possesses neuroprotective properties, restores SMN protein and activates protein synthesis positive regulator Akt/mTOR in experimental models of spinal muscular atrophy. Pharmacological Research 2016; 111: 58-75.
- Khan M, Garg A, Srivastava SK and Darokar MP: A cytotoxic agent from Strychnos nux-vomica and biological evaluation of its modified analogues. Medicinal Chemistry Research 2012; 21: 2975.
- Inouye T, Yoshida S and Tobita M: Studies on monoterpene glucosides- chemical correlation between asperuloside and loganin. Tetrahedron letters 1970; 26: 3905-3915.
- Buchi G, John A, Carlson JE, Powell J and Tietze LF: Total synthesis of loganin. Journal of American Chemical Society 1973; 95: 540-545.
- Li X, Wang Q, Zhang L, Xu L and Yin W: HPLC study of tissue distribution of loganin in rats. Biomedical Chromatography 2006; 20: 1087-1092.
- Lee K, Sung SH, Kim SH, Jang YP, Oh TH and Kim YC: Cognitive-enhancing activity of Loganin isolated from Cornus officinalis in scopolamine-induced amnesic mice. Archives of Pharmacal Research 2009; 32: 677-683.
- Wagner H and Bladt S: Plant Drug Analysis, A thin layer chromatography Atlas. Springer, Second edition 2004.
- http://mona.fiehnlab.ucdavis.edu/spectra/display/TY000062.
- Calis I, Lahloub MF and Sticher O: Loganin, Loganic Acid and Periclymenoside, a new biosidic ester Iridoid Glucoside from Loniceva peviclymenum (Caprifoliaceae). Helvetica Chimica Acta 1984; 67: 160-165.
How to cite this article:
Patil SP and Laddha KS: Simple method of isolation of loganin from Nux vomica fruits and its quantification by HPLC. Int J Pharmacognosy 2017; 4(1): 39-42. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(1).39-42.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
39-42
527
1738
English
IJP
S. P. Patil * and K. S. Laddha
Medicinal and Natural Product Research Laboratory, Department of Pharmaceutical Sciences and Technology, Institute of Chemical Technology, Matunga, Mumbai, Maharashtra, India,
p.patilsapna@gmail.com
16 November 2016
17 December 2016
26 December 2016
10.13040/IJPSR.0975-8232.IJP.4(1).39-42
01 January 2017