PHARMACOGNOSTICAL STANDARDIZATION OF KAILASPATI
HTML Full TextPHARMACOGNOSTICAL STANDARDIZATION OF KAILASPATI
Y. V. Ushir * 1 and P. G. Morankar 2
S.M.B.T. Institute of D. Pharmacy 1, Dhamangaon, Nashik - 422403, Maharashtra, India.
S.M.B.T. College of Pharmacy 2, Dhamangaon, Nashik - 422403, Maharashtra, India.
ABSTRACT: Pharmacognostical parameters for the leaves including petiole of Kailaspati was studied with the aim of drawing the Pharmacognostical standards for this species. Macroscopical and microscopical characters of leaves and petiole, powder characteristics studies, fluorescence studies and thin layer chromatographic studies of the leaves powder included. The study also deals with the preliminary phytochemical screening of the leaves and petioles of Kailaspati with various extracts such as petroleum ether to water. Glandular, non-glandular trichomes and starch grains were identified. The qualitative analysis of some secondary metabolites, to ascertain medicinal claims of this widely used medicinal plant. The results showed that the moderate presence of oils, alkaloids, glycosides, flavonoids, ketosteroids, amino acids, phenols, and triterpenoids. The study includes pharmacognostical standardization of Kailaspati with petiole first time.
| Keywords: |
Kailaspati, Macroscopy, Microscopy, Pharmacognosy, Phytochemical
INTRODUCTION: Kailaspati botanical name Couroupita guianensis Aubl. Is grown in Indian gardens as an ornamental tree for its beautiful flowers. It is known as ‘Cannonball’ tree in English and ‘Kailaspati’ in Hindi, and it belongs to the family Lecythidaceace. Couroupita guianensis is a large deciduous evergreen tree growing to a height of 20 meters. It is a medicinal plant which is endowed with curative properties including antifungal, antibiotic, antiseptic, analgesic, antimalarial, stomachache, toothache, scabies, gastritis, bleeding piles, dysentery, and scorpion poison 1.
The fresh fruit pulp is used in the preparation of cooling medicinal drink, and various parts are useful in skin disease 2, 3. The leaf has been found to show antioxidant activity, anthelmintic activity, immunomodulator, and anti-nociceptive activity 4. The pulp of the fruit of the cannonball tree is rubbed on the infected skin of the dog. It is claimed that when the dog licks its skin, this medicine will also work internally 5. The flowers are used to cure cold, intestinal gas formation and stomach ache6. Barks are used to treat hypertension, tumors, pain, and inflammatory process 7. Different extracts of the flower have been screened for immunomodulatory activity 8, 9. In flowers, mainly eugenol, linalool, and stigmasterol were identified. Leaves of C. guianensis are widely used as an analgesics medicine by the Brazilian rural population 10, 11.
This study is intended to establish, conventional pharmacognostical and modern pharmacognostical parameters of leaves of the plant. These will be used as diagnostic features in the identification, evaluation and monograph preparation of the plant.
MATERIALS AND METHODS:
Procurement of Plant Material: Fresh leaves C. guianensis was collected from Nashik road (Dist. Nashik), Maharashtra, India in Aug-Sep 2012. Plant sample was authenticated by Ministry of Environment and Forest, Botanical Survey of India; Pune (Voucher specimen number MOPCOG1).
Preparation of Plant Material: Fresh mature leaves are stored in formalin solution, and powder of leaves of C. guianensis was prepared by passing through sieve # 44 and kept in airtight container.
Chemicals and Instruments: Photomicroscope (OLYMPUS Pvt. Ltd., New Delhi; Model- CH 2OiBIMF) provided ‘3V-MICRO’ video attachment eyepiece device (Version 8) with 10x, eyepiece (12 megapixels) with cells tracking function and 4x digital zoom camera was used. Solvents and reagents were procured from Loba Chemicals, Mumbai, India.
Macroscopical Examinations: The macro- morphological features of the plant leaves were observed under a magnifying lens and simple microscope 12.
Microscopical Examinations: Fresh leaves and herbaceous petiole of the species were studied using transverse sections. The different parts of leaf-like lamina and midrib were studied according to the methods of Brain and Turner 13. For the microscopical studies, cross sections were prepared and stained as per the procedure of K. R. Khandelwal 14. The different lens of photo-microscope as, OLYMPUS iNEA 5X, 10X/0.2; India, and 100X/1.25 oil India were used for capturing the photographs.
Histochemical Studies: The dilute iodine solution, Dragendroffs reagent, dilute ferric chloride solution, Phloroglucinol + HCl, etc. The reagent treated a hard section of the plant tissue was observed and a microscope to detect the presence of histochemical components.
Powder Microscopy: A little quantity of leaves powder was taken onto a microscopic slide, 1-2 drops of 0.1% w/v phloroglucinol solution and a drop of concentrated hydrochloric acid were added and covered with a coverslip. The slide preparation was mounted in glycerol and examined under a microscope. The presence of starch grain was detected by the formation of blue color on the addition of 2-3 drops of 0.01 M iodine solution 15. The characteristic structures and cell components were observed and their photographs were taken using photomicrography.
Physical Evaluation: The moisture content of the powdered determined based on the loss of drying method16. The ash values were determined, to find out about the physiological state and level of extraneous matter. Extractive values were determined according to the official methods prescribed in Ayurvedic Pharmacopoeia 17.
Phytochemical Investigation: The successive extractive values carry out as per the procedure of C. K. Kokate 18, 19.
Inorganic Elements and Fluorescence Analysis: Total ash of the drug was subjected to testing different inorganic constituents 20, 21. Fluorescence analysis of successive leaf extract was done by the standard method of Chase and Pratt 22. The behavior of drug powder with various chemicals was carried out as per Rathee 23.
TLC Finger Print Profile: Thin layer chromatography of the methanolic and ethanolic extract was studied, and Rf values were determined 24.
RESULTS AND DISCUSSION:
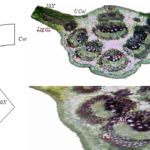
Macroscopic and Microscopic Examination: Macroscopically the fresh leaf of C. arborea is 14.5-18.0-21.6 cm in length and 3.85-5.34-7.90 cm in width and petiole 0.3-0.8-1.7 cm in length. The leaf is simple, glabrous, broadly obovate, acuminate apex with slightly toothed or entire, (tertiary veins subparallel, ± at right angles to midrib) margin and shiny dark green Fig. 1.
Trans-sections of Leaves: Lamina has a structure of dorsiventral type of leaf. The lamina shows upper and lower epidermises comprise uniseriate, polygonal to rectangle cells. The cuticle thickness is approximately the same on both epidermises in C. guianensis. There are covering and noncovering trichomes on both epidermises.
The palisade with a single layer of regular, long, columnar cells, beneath which, 2 to 3 layered mass of closely packed cells filled with chloroplast was present. Mesophyll is traversed by a large number of veins and is represented by groups of few spiral vessels.
Midrib shows concavo-convex outline in the basal and middle region. 3-5 layered collenchymas located below upper epidermises and continuous 2 to 3 layer collenchymas observed above lower epidermis. The vascular bundles are surrounded by a parenchymatic bundle sheath and spongy parenchyma cells. Scattered collateral vascular bundle as 5 numbers above the lower epidermis, 2-4 number below the upper epidermis and 2 numbers in middle surface were observed. The collateral vascular bundle is prominent, occupying the central portion of the midrib.
FIG. 1: MACROSCOPY OF C. GUIANENSIS
FIG. 2: TRANSVERSE SECTION OF C. GUIANENSIS
Petiole: Petiole anatomy of numerous arcuate or annular bundles in arcs. It shows two prominent grooves towards the upper side whereas lower side is round. The epidermis is composed of a single layer of cells. Few trichomes are observed on the lower epidermal cell which is identical with that of the leaf. Two to three layers of collenchymatous cells are found around the epidermal cell.
FIG. 3: TRANSVERSE SECTION OF PETIOLE OF C. GUIANENSIS
Eight to nine collateral vascular bundles are present in the ground tissue. The xylem is found towards the upper side and phloem lies towards the lower side.
The remaining portion of the ground tissue is composed of parenchymatous cells Fig. 3.
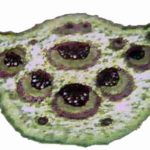
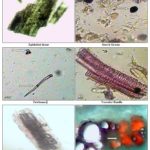
Powder Characteristics: The powder was green in color, on microscopical examination, the powder showed anisocytic stomata, epidermal cell, simple starch grains, glandular and non-glandular trichomes, lignified annular xylem vessel, lignified tissues and spongy parenchyma with veinlet Fig. 4.
FIG. 4: POWDER CHARACTERISTICS OF C. GUIANENSIS LEAVES
Histochemical Studies: The counter idea about the presence of phytoconstituent is obtained through this study like phenolic compound in palisade as indicated by the brownish black stain on ferric chloride solution treatment Table 1.
TABLE 1: HISTOCHEMICAL STUDIES OF C. GUIANENSIS
| Reagent | Phytoconstituent | Histological zone in leaves | Inference |
| Phloroglucinol + HCl (1:1) | Lignin | Vascular bundle | + |
| Aniline Sulphate + H2SO4 | Lignin | Vascular bundle | + |
| Iodine Solution | Starch | Vascular bundle, lamina | + |
| Sudan III Solution | Oil globules | Vascular bundle | + |
| Aqs. FeCl3 Solution | Phenolics | Pallisade cells | + |
| Dragendroff’s reagent | Alkaloid | Lamina | + |
| Libermann-Burchardlt reagent | Steroids | Lamina | + |
| Millon’s Reagent | Proteins | Midrib region | - |
+ Positive; - negative
Physical Evaluation: The moisture content seems to be lower than necessary to support the growth of microbes to bring any change in the composition of the drugs. Physical constant as ash value of the drug gives an idea of the earthy matter or the inorganic composition and other impurities present along with the drug. Extractive values are useful for the determination of exhausted or adulterated drugs Table 2.
TABLE 2: PHYSICOCHEMICAL PARAMETERS OF C. GUIANESIS
| Parameter | % w/w Avg. ± S. D. |
| Ash Values | |
| Total | 08.77 ± 0.063 |
| Acid - insoluble | 02.85 ± 0.103 |
| Water – soluble | 05.84 ± 0.091 |
| Extractive Values | |
| Pet. Ether Soluble (40-60o) | 01.85 ± 0.017 |
| Ethanol Soluble (95%) | 13.57 ± 0.202 |
| Water Soluble | 08.43 ± 0.095 |
| Moisture content | 08.43 ± 0.130 |
Phytochemical Investigation: Revealed the presence of primary and secondary metabolites as oils, alkaloids, glycosides, flavonoids, steroids, amino acids, phenolic substances, and triterpenoids.
Inorganic Elements and Fluorescence Analysis: Various inorganic elements present in the plant are Na+, K+, Fe++, Cl- and NO2.
In fluorescence analysis revealed that the powdered leaves of were treated various chemical reagents to give different colors Table 3.
The fluorescence color is specific for each compound. A nonfluorescent compound may fluoresce if mixed with impurities that are fluorescent.
TABLE 3: FLUORESCENCE ANALYSIS OF C. GUIANESIS
| Powdered drug | Visible/Daylight | The short UV light (254nm) | The long UV light (365nm) |
| Powder | Dark Green | Greenish Black | Greenish |
| Powder + 1N HCl | Greenish brown | Light green | Golden yellow |
| Powder + 50% H2SO4 | Reddish | Brownish | Blackish |
| Powder + 1N NaOH | Reddish | Yellowish green | Brown |
| Powder + 1N NaOH (alcoholic) | Greenish | Light green | Blackish |
TLC Fingerprint Profile: Thin layer chromatography of the chloroform and ethanolic extracts was carried out using Chloroform: Glacial acetic acid: Methanol: Water (64:32:12:8) and Toluene: Ethyl acetate: Formic acid (7:3:1) as mobile phase respectively and the Rf were recorded in Table 4.
TABLE 4: TLC FINGERPRINT FOR C. GUIANESIS
| Mobile phase | Extract | Number of spots and their Rf value |
| Chloroform: Methanol (8:0.6)
Detection- 10 % Alcoholic H2SO4 |
Chloroform
|
0.03, 0.06, 0.35, 0.81 and 0.89 |
| Toluene: Ethyl acetate: Formic acid (7:3:1)
Detection- 365nm |
Ethanolic
|
0.03, 0.08, 0.55, 0.71, 0.87 and 0.93
|
CONCLUSION: These data and parameters have been investigated for C. guianesis to lay down standards which could be useful to find the authenticity of this traditional medicinal system plant. These investigations may be useful to supplement existing information about distinguishing from substitutes and adulterants. In other words, the pharmacognostic features examined in the present study may serve as a tool for validation of the raw material and standardization of its formulations at herbal industrial level in the forthcoming days.
ACKNOWLEDGEMENT: The author(s) are thankful to Principal Dr. A. S. Dhake and management of S.M.B.T. College of Pharmacy, Dhamangaon (Nashik; India) for providing facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Manimegalai S and Rakkimuthu G: Phytochemical screening of stem of Couroupita guianensis. Inter J. Pharma Sci Res 2012; 3(11): 4434-4437.
- Geetha M, Saluja AK, Shankar MB and Mehta RS: Analgesic and anti-inflammatory activity of Couroupita guianensis J Nat Rem 2004; 4(1): 52-55.
- Aruna EA and Laddha KS: Scientific Abstract, 53rd Indian Pharmaceutical Congress 2001: 212.
- Mariana MG, Sidnei OB, Catharina EF, Ricardo MK, Maria EM, Fábio SM and Patrícia DF: Antinociceptive activity of fractions from Couroupita guianensis Aubl leaves. J Ethnopharm 2010; 127(2): 407-413.
- Lens C, Harper T, George K and Bridgwater E: Medicinal and ethnoveterinary remedies of hunters in Trinidad, BMC Complementary and Alternative medicine 2001; 1-10.
- Umachigi PS, Jayaveera KN, Kumar CKA and Kumar GS: Pharmacol online 2007; 3: 269-281.
- Sanz BJ, Campos-de-la CJ, Epiquien RMA and Canigueral S: A first survey on the medicinal plants of the Chazuta valley (Peruvian Amazon). J Ethnopharmacol 2009; 122: 333-362.
- Pradhan D, Panda PK and Tripathi G: Evaluation of immunomodulatory activity of methanolic extract of Couroupita guianensis Flowers in rats. Nat Prod Rad 2009; 8: 37-42.
- Khan MR, Kihara M and Omoloso AD: Antibiotic activity of Couroupita guianensis. J Herbs Spices and Med Plants 2003; 10: 95-108.
- Desal T, Golatakar SG, Rane JB, Ambaye RY and Kamath VR: Larvicidal property of Couroupita guianensis Ind Drugs 2003; 40: 484-486.
- Rane JB, Vahanwala SJ, Golatkar SG, Ambaye RY and Khadse BG: Chemical examination of the flowers of Couroupita guianensis Indian J Pharm Sci 2001; 63: 72-73.
- Tyler V, Brady L and Robbers J: Pharmacognosy. K. M. Varghese Company, India 1977: 103-141.
- Brain KR and Turner TD: The practical Evaluation of Phytopharmaceuticals. Wright-Scientechnica Bristol 1975: 81-86.
- Khandelwal KR: Practical Pharmacognosy techniques and experiments. Nirali Prakashan, Pune, Edition 13, 2005: 130-149.
- Thitikornpong W, Phadungcharoen T and Sukrong S: Pharmacognostic evaluations of Lagerstroemia speciosa J Med Plant Res 2011; 5(8): 1330-1337.
- WHO, Geneva. Quality Control methods for medicinal plant material. A.I.T.B.S. Publishers and Distributors, New Delhi 2002: 31.
- Anonymous, The Ayurvedic Pharmacopoeia of India. Government of India, Ministry of Health and Family Welfare, Controller of Publication, New Delhi, Part-I, Vol. I, 1985: 24-28, 143-44.
- Kokate CK: Handbook of Practical Pharmacognosy. Vallabh Prakashan, New Delhi, Edition 4, 1994: 58-136
- Harborne JB: Phytochemical methods. Chapman and Hall, London, Edition 3, 1984: 88, 203.
- Anonymous, The Ayurvedic Pharmacopoeia of India. Government of India, Ministry of Health and Family Welfare, Controller of Publication, New Delhi, Part-I, Vol. II, 1996: 390, A-100.
- Anonymous, Indian Herbal Pharmacopoeia. Indian Drug Manufacturer’s Association, Mumbai 2002: 1-11.
- Chase CR and Pratt RJ: Fluorescence of powdered vegetable drugs with particular reference to development of a system of identification. Am Pharm Assoc 1949; 38: 324-31.
- Rathee S: Pharmacognostical & Phytochemical evaluation of stem of Capparis decidua (Forsk) Edgew. Pharmacognosy Journal 2009; 2: 75-81.
- Wagner H and Bladt S: Plant Drug analysis: A Thin Layer Chromatography Atlas. 2nd Springer-Verlag Berlin Heidelberg, New Delhi, Edition 2, 1996: 305-7.
How to cite this article:
Ushir YV and Morankar PG: Pharmacognostical standardization of kailaspati. Int J Pharmacognosy 2014; 1(5): 336-42. doi: 10.13040/IJPSR.0975-8232.1(5).336-42.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.