ISOLATION AND CHARACTERIZATION OF β-SITOSTEROL FROM TEPHROSIA VILLOSA PERS
HTML Full TextISOLATION AND CHARACTERIZATION OF β-SITOSTEROL FROM TEPHROSIA VILLOSA PERS
Sufiyan Ahmad * 1, Mohib Khan 2, Rashid Akhtar 3, Md. Imran 3 and N. V. Deore 1
Gangamai College of Pharmacy 1, Nagaon, Dhule - 424005, Maharashtra, India.
Oriental College of Pharmacy 2, Sanpada, Navi Mumbai - 400705, Maharashtra, India.
K.B.H.S.S. Trust’s Institute of Pharmacy 3, Malegaon Camp, Nasik- 423105, Maharashtra, India.
ABSTRACT: Natural products and herbal remedies used in traditional folklore medicine have been the source of many medically beneficial drugs because they elicit fewer side effects, relatively cheap, affordable and claimed to be effective. However, in order to make these remedies acceptable to modern medicine, there is a need to scientifically evaluate them to identify the active principles and to understand their mechanism of action. Tephrosia villosa Pers. (Fabaceae) is a medicinal plant widely used as a folk medicine in India. The present study deals with the isolation and partial purification of bioactive compounds from the crude methanol extracts of the leaves of Tephrosia villosa Pers. The quantification and the identification of compounds in the crude extract and active bands isolated by preparative TLC were accomplished using spectroscopic analysis. The most important compounds β-sitosterol identified in the crude extract appreciable amounts may account for its various biological activities.
| Keywords: |
Isolation, Leaves, β-Sitosterol, Plant extraction, Tephrosia villosa
INTRODUCTION: In many of the developing countries the use of plants drugs is increasing because modern life – saving drugs are beyond the reach of three quarters of the third world population, although many such countries spend 40 – 50% of their total health budget on drugs because of natural origin and lesser side effect or dissatisfaction with result of synthetic drugs 1. Standardization of plant-based medicine is a difficult task; because plants synthesize not only single compounds but it may vary even up to hundreds of compounds may be present in the plant. Hence, it is difficult to standardize herbal medicines as compared to other medicines.
Correct identification and quality assurance of the starting material is, therefore, an essential prerequisite to ensure reproducible quality of herbal medicine, which contributes to its safety and efficacy 2, 3. The quality and quantity of safety and efficacy information on traditional medicines are not sufficient to meet the criteria to support its use worldwide. The reason behind the lack of research data is not only due to health policies but also due to the lack of methodologies for the evaluation of herbal medicines. The plants possess many active therapeutically active chemical constituents associated with many inert substances such as cellulose, lignin and coloring agents, etc. The active constituents are extracted from plants and purified for their pharmacological utility.
So, the quality control of herbal drugs is important for their active chemical constituents in the modern system of medicine. To meet the new thrust of inquisitiveness, standardization of herbal drug is mandatory 4-8. Tephrosia villosa Pers. has many medicinally active compounds in it hence, the focus of this paper is on the analytical methodologies, which include the extraction, isolation, and characterization of active ingredients in leaves of Tephrosia villosa Plant. β-sitosterol is reported to exhibit a spectrum of pharmacological activities against various disease conditions. These include conditions such as inflammation, arthritis, diabetes, cardiovascular ailments, renal disorder, hepatic toxicity, microbial infections and cancer 9. The available literature suggests that β-Sitosterol is a nontoxic agent and does not cause any systemic toxicity in animals at doses ranging from 30 to 2000 mg/kg 10.
MATERIALS AND METHODS:
Plant Material: The plant specimens for the proposed study were collected from the Salem Dist. (T.N.) and authenticated by Dr. Marimuthu, Arts, science college Attur, Tamil Nadu (India). After drying 2kg leaves and stems of Tephrosia villosa milled and pass from sieve # 40. The milled powder material kept in airtight container up to further use. The collected, cleaned and powdered leaves of Tephrosia villosa Pers. was used for the extraction purpose. 200 gm of powdered material was evenly packed in the Soxhlet apparatus. It was then extracted successively with various solvents of increasing polarity 11-14.
Qualitative Estimations: Preliminary phyto-chemical screening of extracts 15-16. The above extracts obtained from the leaves were subjected for the various chemical test for the identification of active phytoconstituents groups by following standard procedure.
Thin Layer Chromatography and Preparative TLC: For Thin Layer Chromatography and preparative TLC analysis, the method used was taken from quality standards of Indian medicinal plants ICMR (volume 5, 2008) for β-sitosterol. Improvements were made to the sample preparation and standard preparation.
Identification of Separated Compound: Pinch of the sample was added in a clean and dry test tube and dissolved in chloroform. Acetic anhydride (1 ml) was added in the test tube. Few drops of sulphuric acid solution were added from the wall of the test tube; solution shows violet color indicates the presence of triterpenoids.
RESULTS AND DISCUSSION: For thin layer chromatography, improvements were made to the sample preparation 0.25 gm of Tephrosia villosa methanolic extract was diluted with 10ml methanol and standard preparation dissolve 10 mg of β-sitosterol (available from total herb solution) in 10 ml of methanol. TLC plates developed with toluene: ethyl acetate (80: 20) showed a violet spot at an Rf value of 0.57 Fig. 1 when sprayed with 1% anisaldehyde-sulfuric acid reagent, heat to 105ºC for 15 min.
The spot coinciding with the standard was marked and then scraped from the plate and scraped silica was sonicated in methanol for 15 min then the solution was filtered and β-Sitosterol was obtained by evaporating methanol. Further, analysis was done for the isolated compound. By repetitive preparative TLC, the β-sitosterol separated was about 25 mg 17.
FIG. 1: TLC OF EXTRACT WITH STANDARD β-SITOSTEROL
Isolation of β-sitosterol was done using Preparative Thin Layer Chromatography (Prep TLC): Prep TLC is an alternative technique to column chromatography much faster than classic Column Chromatography. For sample preparation for preparative TLC methanolic extract, 25 g was partitioned with 50 ml portions of petroleum ether to separate the nonpolar terpenoids.
The petroleum ether extract was concentrated and used for isolating β-sitosterol by preparative thin layer chromatography. Silica gel 60F254 pre-coated TLC plates (Merck) 20 × 20 cm developed with toluene: ethyl acetate (80: 20) showed a violet spot at an Rf value of 0.57 Fig. 2 when sprayed with 1% anisaldehyde-sulfuric acid reagent, heat to 105 ºC for 15 min.
FIG. 2: TLC OF STANDARD β-SITOSTEROL
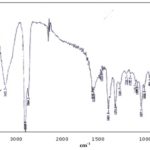
IR spectrum was taken as KBr pellets on Perkin-Elmer IR spectrometer. 3426.3 (stretching, O-H), 2936.9, 2864.2 (stretching, C-H), 1648, 1636 (stretching, C=C) results were showed in Fig. 3 and Table 1.
TABLE 1: FUNCTIONAL GROUP RANGES FOR IR SPECTRUM OF COMPOUND
| S.
No. |
Functional group | Range | |
| Actual | Observed | ||
| 1 | OH | 3200-3600 | 3426.3 |
| 2 | C-H Strech | 2850-2970 | 2936.9, 2864.2 |
| 3 | C=C Strech | 1500-1680 | 1648, 1636 |
| 4 | C-H Bend | 1340-1470 | 1461.8 |
| 5 | C-O | 1050-1300 | 1056.1 |
FIG. 3: IR SPECTRUM OF COMPOUND
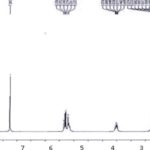
H1 NMR-spectra of Compound H1 NMR (CDCl3, 400 MHz): H- 3-3.52 (1H, m), H-6-5.34 (1H, m), H-18-1.14 (3H,s), H-19-1.26 (3H, s), H-21-0.91 (3H, s), H-26-1.01(3H, s), H-27-0.97 (3H,s), H-29-0.93 (3H, s) Fig. 4 and 5 and Table 2.
High-Performance Liquid Chromatography (HPLC) for β-sitosterol: The data obtained from the chemical test, physical tests and spectral studies of the isolated compound is matching well with that of reported for β-sitosterol and by which the isolated compound is characterized as β-sitosterol Fig. 6.
FIG. 4: H1 NMR SPECTRA OF COMPOUND RECORDED IN CDCl3, 400 MHz
TABLE 2: 1H NMR AND 13C NMR SPECTRA FOR COMPOUND
| Position | δH NMR | δC NMR |
| 1 | 31.9 | |
| 2 | 36.1 | |
| 3 | 3.52 (1H,m) | 71.8 |
| 4 | 42.2 | |
| 5 | 140.7 | |
| 6 | 5.34 (1H,m) | 121.6 |
| 7 | 31.9 | |
| 8 | 28.2 | |
| 9 | 42.3 | |
| 10 | 39.7 | |
| 11 | 21.0 | |
| 12 | 31.8 | |
| 13 | 42.3 | |
| 14 | 45.8 | |
| 15 | 20.2 | |
| 16 | 21.0 | |
| 17 | 50.1 | |
| 18 | 1.14 (1H,s) | 20.2 |
| 19 | 1.26 (3H,s) | 19.4 |
| 20 | 30.2 | |
| 21 | 0.91 (3H,s) | 18.9 |
| 22 | 33.9 | |
| 23 | 28.9 | |
| 24 | 45.8 | |
| 25 | 29.1 | |
| 26 | 1.01 (3H,s) | 19.8 |
| 27 | 0.97 (3H,s) | 19.8 |
| 28 | 23.0 | |
| 29 | 0.93 (3H,s) | 11.9 |
FIG. 5: C13 NMR SPECTRA OF COMPOUND RECORDED IN CDCl3, 400 MHz
FIG. 6: HPLC FOR β-SITOSTEROL
CONCLUSION: Isolation of β-sitosterol was done using preparative Thin Layer Chromatography, the chemical formula for β-sitosterol C29H50O, synonym 22, 23-Dihydrostigmasterol, Stigmast-5-en-3-ol, β-sitosterin, description colourless crystals, solubility Freely Soluble in chloroform, petroleum ether and solubilized only upon sonication and heating in methanol, melting point 147-146°C, purity 90.36%. β-sitosterol IR (KBr) 3426.3 (stretching, O-H), 2936.9,2864.2 (stretching, C-H), 1648, 1636 (stretching, C=C) results were showed in Fig. 3 and Table 1. Mass (m/z): 415.13 Fig. 7 and Table 3. H1 NMR (CDCl3, 400 MHz) H- 3-3.52 (1H, m), H-6-5.34 (1H, m), H-18-1.14 (3H,s), H-19-1.26 (3H, s), H-21-0.91 (3H, s), H-26-1.01(3H, s), H-27-0.97 (3H,s), H-29-0.93 (3H, s). HPLC puro sphere RP 18 column, mobile phase Methanol: water: Acetic acid (70:30:1), flow rate 1ml/min, detector UV/VIS, λmax 205nm and retention time 15 min. Estimation of β-sitosterol in Tephrosia villosa Pers. leaves chromatographic technique column Microsorb-MV 100-5 C18 250 X 4.6 mm, Mobile Phase acetonitrile: methanol: glacial acetic acid (80: 20:0.01%), flow rate 1ml/min, detection 205 nm, injection volume 20 µL and run time 3.5 min. The percentage of β-sitosterol was found in Tephrosia villosa Pers. leaves 2.08 %.
FIG. 7: MASS SPECTRA OF COMPOUND
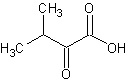
FIG. 8: CHEMICAL STRUCTURE OF β- SITOSTEROL
TABLE 3: MOLECULAR WEIGHT OF COMPOUND
| S.
no. |
Molecular weight | |
| Actual | Observed | |
| 1 | 414.70 | 415.13 |
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- WHO: General Guidelines for Methodologies on Research and Evaluation of Traditional Medicine. World Health Organization, Geneva, 2000.
- Joshi K, Chavan P, Warude D and Patwardhan B: Molecular markers in herbal drug technology. Current Science 2004; 87: 159-165.
- Chanda S, Nagani K and Parekh J: Assessment of quality of Manilkara hexandra (Roxb.) Dubard leaf (Sapotaceae): Pharmacognostical and physicochemical profile. Pharmacognosy Journal 2010; 2: 520-524.
- Chaudhury RR: Herbal medicine for human health. World Health Organization Geneva. CBS publishers and distributors LTD, New Delhi 1999.
- Kokate CK, Purohit AP and Gokhale SB: Pharmacognosy. Nirali Prakashan, Edition 31st, 2005: 97-131.
- Raina MK: Quality control of herbal and herbo-mineral formulations. Indian Journal of Natural Products 2003; 19: 11-15.
- Raven PH, Evert RF and Eichhorn SE: Biology of Plants, Freeman, New York, Edition 6th, 1999.
- Yan XJ, Zhou JJ, Xie GR and Milne GWA: Traditional Chinese Medicines: Molecular Structures. Natural Sources and Applications, Aldershot, Ashgate 1999.
- Chaturvedi PK, Bhui K and Shukla Y: Lupeol, Connotations for chemoprevention. Cancer Lett 2008; 263: 1-13.
- Murtaza I, Saleem M, Adhami VM, Hafeez BB and Mukhtar H: Suppression of cFLIP by lupeol, a dietary triterpene, is sufficient to overcome resistance to TRAIL-mediated apoptosis in chemoresistant human pancreatic cancer cells. Cancer Res 2009; 69: 1156-1165.
- Trease and Evans: Pharmacognosy. ELBS Publication, New Delhi, Edition 15th, 2005: 138.
- Harborne JB: Phytochemical methods. A guide to modern technique of plant analysis; reprint published by Chapman and Hall Ltd., 1976: 4-6.
- Mukherjee PK: Quality Control of herbal drugs. An approach to the evaluation of botanicals, Business horizons pharmaceutical publisher, New Delhi, Edition 1st, 2002: 389-398.
- Cooper and Gunn: Tutorial pharmacy, J. B. publishers, New Delhi, 2004: 259.
- Kokate CK: Practical Pharmacognosy. Vallabh Prakashan 1994: 107-111.
- Brain K and Turner TD: The practical evaluation of phytopharmaceuticals. Wright Scienteania Bristol 1983; 103-106.
- Agarwal RB and Rangar VD: Anti-inflammatory and antiarthritic activities of lupeol and 19a–H lupeol isolated from Strobilanthus callosus and Strobilanthus ixiocephala roots. Indian Journal of Pharmacology 2003; 35: 384-387.
How to cite this article:
Ahmad S, Khan M, Akhtar R, Imran M and Deore NV: Isolation and characterization of β-sitosterol from Tephrosia villosa Pers. Int J Pharmacognosy 2016; 3(9): 400-04. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(9).400-04.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
400-404
680
1213
English
IJP
S. Ahmad *, M. Khan, Rashid Akhtar, M. Imran and N. V. Deore
Gangamai college of Pharmacy, Nagaon, Dist. Dhule, Maharashtra, India
sufimpharm@rediffmail.com
09 August 2016
16 September 2016
26 September 2016
10.13040/IJPSR.0975-8232.IJP.3(9).400-04
30 September 2016