IN-SITU RAFT FORMING SYSTEM: A REVIEW
HTML Full TextIN-SITU RAFT FORMING SYSTEM: A REVIEW
S. S. Raut * and H. A. Shinde
NGSPM’s College of Pharmacy, Anjaneri, Nashik - 422213, Maharashtra, India.
ABSTRACT: Oral delivery of the drug is the preferable drug delivery due to the ease of administration, patient compliance, and flexibility in the formulations. In recent era, various technologies have been made in research and development of controlled release oral drug delivery system to overcome various physiological difficulties such as variation in gastric retention and emptying time. Conventional oral dosage forms pose low bioavailability problems due to their rapid gastric transition from the stomach, especially in case of drugs which are less soluble at alkaline pH of the intestine and locally acting drugs in the stomach get rapidly emptied. So, frequency of dose administration in such cases is increased. Gastro retentive drug delivery system is facing many challenges which can be overcome by upcoming newly emerging approach, i.e., raft forming system. The present study provides valuable information and highlights advances in this raft forming system. Different types of smart polymers used for their formulation have also been summarized. The review focuses on the mechanism, formulation, development, and system optimization aspects of the raft forming system and also highlight parameters which may lead to response variations in altered physiological conditions are discussed as well.
| Keywords: |
Raft-forming system, Gastro retentive drug delivery system, Floating drug delivery system, Gastrointestinal track, Gastric residence time, Altered physiological conditions
INTRODUCTION: Raft forming system is one of the floating drug delivery systems. Floating drug delivery system is retained in the stomach and is useful for drugs that are poorly soluble or unstable in intestinal fluids. Floating drug delivery system (FDDS) have a bulk density less than gastric fluids and so remain buoyant in the stomach without affecting the gastric emptying rate for a prolonged period. While the system is floating on the gastric contents, the drug is released slowly at the desired rate from the system. After the release of drug, the residual system is emptied from the stomach. This results in increased gastric residence time and better control of fluctuations in plasma drug concentration 1.
In raft forming systems have been widely investigated as vehicles for prolonged drug delivery. This interest has been sparked by the advantages shown by raft forming polymeric delivery systems such as ease of administration and reduced frequency of administration, improved patient compliance, and comfort. Raft formation occurs due to one or a combination of different stimuli like pH change, temperature modulation, and solvent exchange.
So, raft forming system via different route such as oral, nasal, ophthalmic, etc can be formulated. Various natural and synthetic polymers such as gellan gum, alginic acid, xyloglucan, pectin, chitosan, poly (DL-lactic acid), poly (DL-lactide-co-glycolide) and polycaprolactone are used for formulation development of raft-forming drug delivery systems. Gastro retentive raft forming system helps to increase the bioavailability of drug compared to the conventional liquid dosage form. The raft formed from raft forming system, being lighter than gastric fluids, floats over the stomach contents or adhere to gastric mucosa due to the presence of bioadhesive nature of the polymer and produce gastric retention of dosage form and increase gastric residence time resulting in prolonged drug delivery in the gastrointestinal tract.
This review attempts to discuss stomach specific raft forming system in detail including formulation factors to be considered in the development of the raft-forming system. Also, different types of smart polymers, their mechanisms of gel formation from the sol form 2, 3. Raft-forming systems have received much attention for the delivery of antacids and drug delivery for gastrointestinal infections and disorders. The mechanism involved in the raft formation includes the formation of viscous cohesive gel in contact with gastric fluids, wherein each portion of the liquid swells forming a continuous layer called a raft. This raft floats on gastric fluids because of low bulk density created by the formation of CO2.
FIG. 1: SCHEMATIC REPRESENTATION OF RAFT FORMING SYSTEM
Usually, the system contains a gel-forming agent and alkaline bicarbonates or carbonates responsible for the formation of CO2 to make the system less dense and float on the gastric fluids an antacid Raft forming floating system. The system contains gel-forming agent (e.g., alginic bicarbonate, calcium carbonate, mannitol, and a sweetener. These ingredients were granulated, and citric acid was added to the granules. The formulation produces effervescence and aerates the raft formed, making it float acid), sodium bicarbonate and acid neutralizer, which forms a foaming sodium alginate gel (raft) when in contact with gastric fluids.
The raft thus formed floats on the gastric fluids and prevents the reflux of the gastric contents (i.e., gastric acid) into the esophagus by acting as a barrier between the stomach and esophagus. A patent assigned to Reckitt and Colman Products Ltd. describes a raft forming formulation for the treatment of Helicobacter pylori (H. Pylori) infections in the GIT 2, 3.
Basic Anatomy of Stomach and its Physiology of Stomach: During past 4 decades, the idea of gastro retention is known to researchers and is popularly cultured Davis, in 1968, 1st described the concept of floating drug delivery system. To understand the approaches for gastro retention it is necessary to overview gastric physiology and gastric motility.
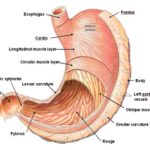
Anatomy: Human stomach has a resting volume of 25-50 ml, which can distend up to 1500 ml following a meal. The stomach is a J-shaped organ. It is located in the upper left-hand portion of the abdomen, just below the diaphragm. It occupies a portion of the epigastric and left the hypochondriac region. The main function of the stomach is to store the food temporarily, grind it and then release it slowly into the duodenum. Since the drugs are absorbed in the upper small intestine, it will be beneficial to develop the dosage forms that reside in that region.
Anatomically stomach is divided into 3 parts:
Fundus: The superior part of the stomach, this lies above the imaginary horizontal plane passing through the cardiac orifice.
Body: The body or corpus is the central part of the stomach. This lies between the fundus and the antrum, and it is the largest part of the stomach.
Antrum: This lies in the imaginary transpyloric plane and to the right of the angular notch (incisura angularis). It joins the pyloric canal on its right. The main function of fundus and body is storage whereas that of the antrum is mixing or grinding. The fundus also exerts a steady pressure on the gastric contents, pressing them towards the distal stomach Fig. 2. To pass through the pyloric valve into the small intestine, particles should be of the order of 1-2 mm. The antrum does this grinding. The stomach has a limitation of short residence time 23, 24, 25, 26.
Physiology: The physiology and disease state of the stomach has a direct effect on the design of controlled drug delivery system because the drug is absorbed from and enters into the site of action. Factors such as pH, nature, and volume of gastric secretions and gastric mucosa play an important role in drug release and absorption 24.
FIG. 2: ANATOMY OF HUMAN STOMACH 23
pH: The stomach has acidic pH due to the secretion of HCl, favors the absorption of acidic drugs if they are soluble in gastric fluids since they are unionized in large extent in such a pH. The pH of the stomach in the fasted condition is about 1.5 to 2, and in fed conditions, it is usually 2 to 6. A large volume of water administered with oral dosage form changes the pH of the stomach to the pH of water initially 24, 28.
Gastric Mucosa: The surface of the gastric mucosa is a layer of simple columnar epithelial cells called surface mucous cells. The mucosa contains a lamina propria and a muscularis mucosa.
TABLE 1: ANATOMICAL DIFFERENCE BETWEEN DIFFERENT REGIONS OF THE GIT 24, 25
| Particulars | Stomach | Small intestine | Large intestine | Rectum |
| pH range | 1-3 | 5-7.5 | 7.9-8.0 | 7.5-8.0 |
| Length (cm) | 20 | 285 | 110 | 20 |
| Diameter (cm) | 15 | 2.5 | 5 | 2.5 |
| Surface area (m2) | 0.1-0.2 | 200 | 0.15 | 0.02 |
| Blood flow (L/min) | 0.15 | 1.0 | 0.02 | - |
| Transit time h | 1-5 | 3-6 | 6-12 | 6-12 |
Stomach lumen and mucous neck cells are secrets mucus. Parietal cells produce intrinsic factor and hydrochloric acid. The mucous, parietal and chief cells from stomach secrets 2000 - 3000 ml gastric juice per day. Gastric glands include a type of endocrine cell, the G cell, which stimulates several aspects of gastric activity 23.
Gastric Secretion: Acids, pepsin, gastrin, mucus, and some other enzymes are the secretions of the stomach. Normal adults produce a basal secretion up to 60 ml with approximately 4 mM of hydrogen ions every hour. Other potent stimulators of gastric acid are the hormones like gastrin, peptides, amino acids and gastric distension 23.
Gastrointestinal Transit Time: Food content remains in each segment of the gastrointestinal tract for different periods. The time a dosage form takes to traverse the stomach is usually termed the ‘gastric emptying rate’ Table 2. Since most of the drugs are absorbed from the upper part of the intestine, the total effective time for the drug absorption is 3-8 h 24.
TABLE 2: TRANSIT TIME OF FOOD IN EACH SEGMENT OF THE GASTROINTESTINAL TRACT 24
| Segment | Liquid | Solid |
| Stomach | 10-30 min | 1-3 h |
| Duodenum | <60 sec | <60 sec |
| Jejunum and ileum | 3 h ± 1.5 h | 4 h ± 1.5 h |
| Colon | - | 20-25 h |
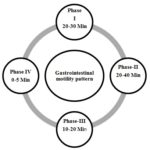
Gastric Motility: Gastric motility is also a key factor in stomach specific drug delivery. Thorough knowledge of motility is prerequisite for developing a retentive form of the drug. Gastric motility differs in fasting and fed states. In fasting states, an Inter-digestive myoelectric motor complex (IMMC), a 2 h. cycle of peristalsis is generated which progresses to ileocecal junction. It consists of 4 phases.
FIG. 3: SCHEMATIC REPRESENTATION OF THE INTERDIGESTIVE MOTILITY PATTERN 4, 25
Phase I: Also called quiescent period with rare low amplitude contractions, lasting for 30- 60 min.
Phase II: It comprises of intermediate amplitude contractions with bile secretion, lasting for 20 - 40 min.
Phase III: Also called Housekeeper waves, it forms of very high amplitude contractions offering maximum pyloric opening and efficient evacuation of stomach contents. It lasts for 10 - 20 min with a frequency of 4-5/min.
Phase IV: Transitional phase between phase III and I of two consecutive cycles. It lasts for less than 5 min.
In fed states, motility is induced 5-10 min after ingestion and persists as long as food remains in the stomach, typically 3-4 h. Activity is the same as phase II of IMMC. Gastro retentivity of the drug was required to increase the bioavailability of drug and to reduce the undesirable effects caused by exposure of drug to other regions of GIT 3, 23, 24, 26.
Factor Affecting on RFS:
Factors Related to Dosage Form:
Size of the Dosage Form: To allow the dosage form to pass through the pyloric valve into the small intestine the particle size should be in the range of 1 to 2 mm. In most cases, the larger the dosage form, the greater will be the GRT.
Due to the larger size of the dosage form, it could not quickly pass through the pyloric antrum into the intestine. Small-size tablets leave the stomach during the digestive phase while the large-sized tablets are emptied during the housekeeping waves 2, 3, 4, 9.
The shape of Dosage Form: Ring-shaped and tetrahedron-shaped devices have a better gastric residence time as compared to other shapes 2, 3, 4, 9.
The density of Dosage Form: Dosage forms having a density lower than the gastric contents can float to the surface, while high-density systems sink to the bottom of the stomach. Both positions may isolate the dosage system from the pylorus. A density of b1.0 g/cm3 is required to exhibit floating property. However, the floating tendency of the dosage form usually decreases as a function of time, as the dosage form gets immersed into the fluid, as a result of the development of hydrodynamic equilibrium 2, 3, 4, 9.
Food Intake and its Nature:
Fed and Unfed State: Under fasting conditions, the gastrointestinal motility is characterized by periods of strong motor activity or MMC that occurs every 1.5 to 2 h. The MMC sweeps the undigested material from the stomach, and if the timing of administration of the formulation coincides with that of the MMC, the GRT of the unit can be expected to be very short. However, in the fed state, MMC is delayed, and GRT is considerably longer 1, 2, 9.
Food Intake and Nature of Food: Food intake, viscosity, and volume of food, caloric value, and frequency of feeding have a profound effect on the gastric retention of dosage forms. The presence or absence of food in the gastrointestinal tract influences the gastric retention time of the dosage form. Usually, the presence of food in the gastrointestinal tract improves the gastric retention time of the dosage form and thus, the absorption of drugs increases by allowing its stay at the absorption site for a longer period 1, 2, 3.
Calorie Content: The rate of gastric emptying primarily depends on the caloric contents of the ingested meal. It does not differ for proteins, fats, carbohydrates as long as their caloric content is the same. Generally, an increase in acidity, osmolarity, and caloric value slows down gastric emptying. GRT can be increased between 4 and 10 h with a meal that is high in proteins and fats 9.
Frequency of Feed: The GRT can increase by over 400 min when successive meals are given compared with a single meal due to the low frequency of MMC 2, 3.
Patient-Related Factors:
Gender: Gastric emptying rate may differ in male and female. Generally, the gastric emptying in women was slower than in men 1, 2, 3, 4.
Age: Elderly people, especially those over 70 years have a longer gastroretentive time. Thus gastric emptying time is slowed down 1, 3, 4.
Posture: The effect of posture on gastric residence time, found no significant difference in the mean gastric residence time for individuals in the upright, ambulatory and supine state. In the upright position, the raft forming systems floated to the top of the gastric contents and remained for a longer time, showing prolonged gastric residence time. But the non-floating units settled to the lower part of the stomach and underwent faster emptying as a result of peristaltic contractions, and the floating units remained away from the pylorus. However, in the supine position, the floating units are emptied faster than the non-floating units of similar size 4, 7.
Concomitant Drug Administration: Administration of drugs with impact on gastrointestinal transit time for example drugs acting as anticholinergic agents (e.g., atropine, propantheline), opiates (e.g., codeine) and prokinetic agents (e.g., metoclopramide, cisapride) can alter gastro retention of oral dosage forms. Anticholinergics like atropine and propantheline increase gastric residence time. Drugs like metoclopramide and cisapride decrease gastric residence time 7, 10.
Disease State: In gastric ulcer, diabetes, and hypothyroidism there is an increase in gastric residence time. In the case of hyperthyroidism and duodenal ulcers, there is a decrease in gastric residence time 3, 4.
The volume of the GI Fluid: The resting volume of the stomach is 25 to 50 ml. The volume of liquids administered affects the gastric emptying time. When the volume is large, the emptying is faster. Fluids took at body temperature leave the stomach faster than colder or warmer fluids 9.
Effect of Gastrointestinal Fluid: On comparison of the floating and non-floating units, it was concluded that regardless of their sizes the floating units remained buoyant on the gastric contents throughout their residence in the GIT, while the non-floating units sink and remained in the lower part of the stomach. Floating units away from the gastro-duodenal junction were protected from the peristaltic waves during the digestive phase while non-floating forms stayed close to the pylorus and were subjected to propelling and retro spelling waves of the digestive phase 7, 10.
pH: Variation of gastric pH occurs because of a number of physiological factors and patient-related factors like diseases, diet, disease, presence of gases, age, pathological conditions, drugs, as well as intra- and inter-subject variation. This variation in pH may significantly influence the performance of orally administered drugs.
About 20% of the elderly people exhibit either diminished (hypochlorhydria) or no gastric acid secretion (achlorhydria). Pathological conditions such as pernicious anemia and AIDS may significantly reduce gastric acid secretion leading to elevated gastric pH.
Also, drugs like H2 receptor antagonists and proton pump inhibitors significantly reduce gastric acid secretion. These things affect the mechanism of the raft formation in the individuals. It means that the formation of raft varies according to the stomach pH of the patient at the time of administration of the formulation 1, 2, 8, 9.
The Design of the Raft Forming System: The formulation of the raft forming system depends on the physicochemical properties of the drug molecule, the diseased condition for which treatment is required, the patient population and the marketing preference. Physicochemical factors include molecular weight, lipophilicity, and molecular charge; an anatomical and physiological factor includes membrane transport and pH of tissue fluid; formulation factors include pH, gelation temperature, viscosity, osmolarity, and spreadability. To achieve the gastric retention of the dosage form, the dosage form must be able to satisfy the following criteria. They are as follows:
- The drug should be released slowly from the system.
- The dosage form must be able to withstand the force exerted by peristaltic waves in the stomach and the constant contractions, grinding and churning moments.
- Should maintain specific gravity lower than gastric contents (1.004-1.01 g/cm3).
- The dosage form must remain in the stomach for a prolonged period.
- Better patient compliance.
- Easy for administration for the patient.
- After the release of the drug, the device should be easily evacuated from the stomach 2, 3, 4, 8.
Ingredients used in the Formulation of the Raft Forming System: An appropriate candidate should be selected for the formulation of controlled release gastro-retentive formulation. Various ingredients used in the formulation of such system are gel-forming agent and alkaline bicarbonates or carbonates which are responsible for the formation of a less dense system which floats on the gastric fluids 9.
Drugs Selection Criteria for the Raft Forming System: Raft forming systems have received much attention for the delivery of antacids and drug delivery for gastrointestinal infections and disorders. The raft forming system is the potential approach for heartburn and esophagitis. This system is suitable for acid soluble drugs that are poorly soluble or unstable in intestinal fluids. Various drugs that can be used for the raft forming system are summarized in Table 3 with their category.
TABLE 3: DRUGS USED FOR THE RAFT FORMING SYSTEM 1, 12, 13
| S. no | Category | Drugs |
| 1 | Antacids | Aluminum hydroxide, aluminum phosphate, magnesium silicate, magnesium hydroxide,
calcium carbonate |
| 2 | H2 receptor antagonist Proton pump inhibitor | Cimetidine, ranitidine, loxatidine, famotidine, nizatidine, Omeprazole, lansoprazole, pantoprazole, rabeprazole, esomeprazole |
| 3 | Anti-cholinergic | Oxyphenonium, propantheline, telezepine, pirenzepine |
| 4 | Anti-helicobacter
pylori drugs |
Amoxicillin, clarithromycin, tetracycline,
metronidazole, tinidazole, colloidal bismuth |
Thus the criteria of the drug to be considered for the selection of the drug for gastro retention are as follows:
- Drugs acting locally in the stomach, g. Antacids and drugs for H. Pylori viz., Misoprostol
- Drugs that are primarily absorbed in the stomach, g. Amoxicillin
- Drugs those are poorly soluble at alkaline pH, g. Furosemide, Diazepam, Verapamil, etc.
- Drugs with a narrow window of absorption, g. Cyclosporine, Methotrexate, Levodopa, etc.
- Drugs which are absorbed rapidly from the GI tract. g. Metronidazole, tetracycline 1, 12, 13, 16, 17, 18.
The Criteria of the Drug that are not suitable for Gastric Retention are:
- Drugs that have very limited acid solubility.
- Drugs that suffer instability in the gastric environment.
- Drugs intended for selective release in the colon.
Polymer Used for Formulation: Various polymers are employed in floating drug delivery systems to target the delivery of the drug to a specific region in the gastrointestinal tract, i.e. stomach. Various natural and synthetic polymers are used in the formulation of the raft forming drug delivery system. A natural polymer such as alginic acid, guar gum, gellan gum, xyloglucan, pectin, chitosan, etc. and a synthetic polymer such as poly (DL-lactic acid), poly (DL-lactide-co-glycolide) and poly-caprolactone, HPMC, etc. are used for formulation development of the raft forming drug delivery system.
A polymer used for in situ gels should have the following characteristics:
- It should be biocompatible.
- It should have pseudoplastic behavior.
- The polymer should be capable of increasing the viscosity with increasing the shear rate 9, 12.
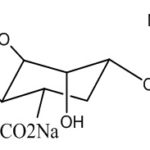
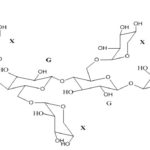
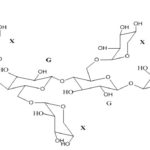
Sodium alginate: Sodium alginate is a widely used polymer of natural origin. Chemically, it is alginic acid salt, consisting of -L-glucuronic acid and -D-mannuronic acid residues connected by 1,4-glycosidic linkages. The solution of alginates in water form firm gels in the presence of di-or trivalent ions (e.g., calcium and magnesium ions). Alginates salts, specifically, sodium alginate is mostly used for the preparation of the gel-forming solution, for delivery of the drugs and proteins. Alginate salts are considered most favorable because of biodegradable and nontoxic nature, with additional bioadhesive property. Sodium alginate is a salt of alginic acid - a linear block copolymer polysaccharide consisting of β- D-mannuronic acid and α-L-glucuronic acid residues joined by 1,4-glycosidic linkages. Aqueous solutions of alginates form firm gels on the addition of di- and trivalent metal ions. The results indicated that the alginates form compact structures when the ionic radical of the cation are lower. Sodium alginate has been employed in the preparation of gels for the delivery of biomolecules such as drugs, peptides, and proteins.
FIG. 4: CHEMICAL STRUCTURE OF SODIUM ALGINATE
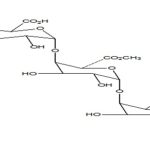
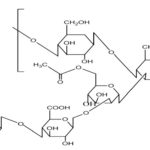
Pectin: These are plant origin anionic characteristics can be divided into two polysaccharides isolated from the cell wall of most plants and consist of -(1-4) -D-galacturonic acid residues. Pectin undergoes gel formation in the presence of the medium; a stiff gel is produced. The gelling capacity divalent ions (e.g., Ca) which causes cross-linking of the is determined on the 2+ basis of stiffness and time galacturonic acid units (ionic cross-linking) and also in the period for which gel remains, as such. Presence of the H+ ions (pH-dependent gelling).
FIG. 5: CHEMICAL STRUCTURE OF PECTIN
Pectin is a complex polysaccharide comprising mainly esterified D-galacturonic acid residues in an a-(1-4) chain. The acid groups along the chain are largely esterified with methoxy groups in the natural product. The hydroxyl groups may also be acetylated. Pectin gelatine types: high-methoxy and low-methoxy gelation. Gelation of high methoxy pectin usually occurs at pH<3.5. Low-methoxy pectin is gelled with calcium ions and is not dependent on the presence of acid or high solids content.
Gellan gum: Gellan gum (FDA approved) secreted by the Sphingomonas elodea (Pseudomonas elodea) and chemically is anionic deacetylated polysaccharide with repeating tetrasaccharide units composed of -D-glucuronic acid (1 unit), -L-rhamnose (1 unit) and -D-glucuronic acid (2 units) residues. Gellan gum undergoes gel formation due to change in temperature or due to the presence of cations (e.g., Na+ K+, Ca2+). Gellan gum is an anionic deacetylatedexocellular polysaccharide secreted by Pseudomonas elodea with a tetra- saccharide repeating unit of one α-L- rhamnose, one β-D-glucuronic acid, and two β-D-glucuronic acid residues. It is a water-soluble polysaccharide. It forms a gel via formation of double helices, followed by their ionic cross-linking.
FIG. 6: CHEMICAL STRUCTURE OF GELLAN GUM
Xyloglucan: It is a plant-based polysaccharide obtained from seeds of tamarind. Chemically, this polysaccharide composed of a chain of (1-4)- -D-glucan having (1-6)-D xylose units as branches which have partial (1-2)- - D - galactoxylose substitution. Xyloglucan, itself, does not undergo gel formation but dilute solutions partly degraded by galactosidase exhibit gelling properties on heating (temperature dependent gel formation). Besides the use in oral drug delivery, it is also being used for ocular and rectal drug delivery.
Xyloglucan has shown a very low gelation time of up to a few minutes. Xyloglucan is a polysaccharide derived from tamarind seeds and is composed of a (1-4)-β-D- glucan backbone chain, which has (1-6)-α- D xylose branches that are partially substituted by (1-2)-β-D-galactoxylose. Xyloglucan is composed of heptasaccharide, octasaccharide, and nonasaccharide oligamers, which differ in the number of galactose side chains. Although xyloglucan itself does not gel, dilute solutions of xyloglucan which has been partially degraded by galactosidase exhibit a thermally reversible sol-gel transition on heating.
FIG.7: CHEMICAL STRUCTURE OF XYLOGLUCAN
Xanthan gum: Xanthan gum is a high molecular weight extracellular polysaccharide seeds and is composed of a (1-4)-β-D- glucan backbone chain, which has (1-6)-α- D xylose branches that are partially substituted by (1-2)-β-D-galactoxylose. Xyloglucan is composed of heptasaccharide, octa-saccharide, and nonasaccharideoligamers, which differ in the number of galactose side chains. Although xyloglucan itself does not gel, dilute solutions of xyloglucan which has been partially degraded by galactosidase exhibit a thermally reversible sol-gel transition on heating.
FIG. 8: CHEMICAL STRUCTURE OF XANTHAN GUM
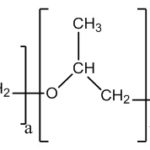
Pluronic F-127: Poloxamers or pluronic (marketed by BASF Corporation) is the series of commercially available difunctional triblock copolymers of non-ionic nature. They comprise of a central block of relatively hydrophobic polypropylene oxide surrounded on both sides by the blocks of relatively hydrophilic polyethylene oxide. Due to the PEO/PPO ration of 2:1, when these molecules are immersed into the aqueous solvents, they form micellar structures above critical micellar concentration.
FIG. 9: CHEMICAL STRUCTURE OF PLURONIC F-127
They are regarded as PEOPPO- PEO copolymers. Chemically they are Oxirane, methyl-, polymer with oxirane or α-Hydro-ω- hydroxypoly (oxyethylene) a poly (oxypropylene) b poly (oxyethylene) a block copolymer. The pluronic triblock copolymers are available in various grades differing in molecular weights and physical forms. Depending upon the physical designation for the grades are assigned, like F for flakes, P for paste, L for liquid. Pluronics or Poloxamers also undergo in situ gelations by temperature change.
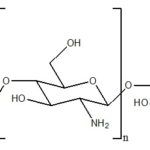
Chitosan: Chitosan is a biodegradable, thermosensitive, polycationic polymer obtained by alkaline deacetylation of chitin, a natural component of shrimp and crab shell. Chitosan is a biocompatible pH dependent cationic polymer, which remains dissolved in aqueous solutions up to a pH of 6.2. Neutralization of chitosan aqueous solution to a pH exceeding 6.2 leads to the formation of a hydrated gel-like precipitate. The pH gelling cationic polysaccharides solution are transformed into thermally sensitive pH dependent gel-forming aqueous solutions, without any chemical modification or cross-linking by addition of polyol salts bearing a single anionic head such as glycerol, sorbitol, fructose or glucose phosphate salts to chitosan aqueous solution.
FIG. 10: CHEMICAL STRUCTURE OF CHITOSAN
Carbopol: Carbopol is a well-known pH-dependent polymer, which stays in solution form at acidic pH but forms a low viscosity gel at alkaline pH. HPMC is used in combination with carbapol to impart the viscosity to carbopol solution while reducing the acidity of the solution. Various water triblock copolymers consisting of poly (oxyethylene) and poly (oxypropylene) units that undergo changes in solubility with the change in environment temperature. Pluronic ™ F 127. A 25-40% aqueous solution of this material will gel at about body temperature, and drug release from such a gel occurs over up to one week 1, 10, 12, 15, 29.
FIG. 11: CHEMICAL STRUCTURE OF CARBOPOL
Different Approaches for RFS System: There are four broadly defined mechanisms used for triggering the in raft formation of biomaterials: Physiological stimuli (e.g., temperature and pH), physical changes in biomaterials (e.g., solvent exchange and swelling) chemical reactions (e.g., enzymatic, chemical and photoinitiated polymerization).
Raft Formation Based on Physical Mechanism:
Swelling: Raft formation may also occur when absorbs water from the surrounding environment and expand to occur desired space. One such substance is myverol 18-99 (glycerol mono-oleate), which is polar lipid that swells in water to form lyotropic liquid crystalline phase structures. It has some bioadhesive properties and can be degraded in-vivo by enzymatic action 2, 3.
Diffusion: This method involves the diffusion of solvent from polymer solution into surrounding tissue and results in precipitation or solidification of the polymer matrix. N- Methylpyrrolidone (NMP) is a useful solvent for such system 2, 3.
Raft Formation Based on Physiological Stimuli:
Thermally Trigged System: Temperature sensitive hydrogels are probably the most commonly studied class of environment-sensitive polymer systems in drug delivery research. The use of biomaterial whose transitions from sol-gel is triggered by an increase in temperature is an attractive way to approach in-situ formation. The ideal critical temperature range for such system is ambient and physiologic temperature, such that clinical manipulation is facilitated and no external source of heat other than that of the body is required for trigger gelation. A useful system should be tailorable to account for small differences in local temperatures, such as might be encountered in appendages at the surface of the skin or in the oral cavity.
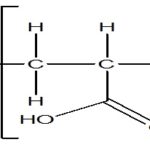
Three main strategies exist in the engineering of the thermoresponsive sol-gel polymeric system. For convenience, temperature-sensitive hydrogels are classified into negatively thermosensitive, positively thermosensitive, and thermally reversible gels. Negative temperature-sensitive hydrogels have a lower critical solution temperature (LCST) and contract upon heating above the LCST. Polymers with low critical temperature (LCST) transition between ambient and physiologic temperature is used for this purpose. One of the most extensively investigated polymers that exhibit useful LCST transition is poly(N- isopropylacrylamide) (PNIP AAm). PNIPAAM is a water-soluble polymer at its low LCST, but hydrophobic above LCST, which result on precipitation of PNIPAAM from the solution at the LCST.
A positive temperature-sensitive hydrogel has an upper critical solution temperature (UCST), such hydrogel contracts upon cooling below the UCST. Polymer networks of poly (acrylic acid) (PAA) and polyacrylamide (PAAm) or poly (acrylamide-co-butyl methacrylate) have positive temperature dependence of swelling. The most commonly used thermoreversible gels are these prepared from poly (ethylene oxide) -bpoly (propylene oxide) -b-poly (ethylene oxide) (Pluronics®, Tetronics®, poloxamer 19, 20, 21.
pH-Dependent Gelling: Another formation of Raft is based on Change in pH. Certain polymers such as poly-vinyl acetal diethylaminoacetate (AEA), Mixtures of poly (methacrylic acid) (PMA) and poly (ethylene glycol) (PEG) shows change from sol to gel with the change of pH. The polymers with a large number of ionizable groups are known as polyelectrolytes. Swelling of hydrogel increases as the external pH increases in the case of weakly acidic (anionic) groups, but decreases if the polymer contains weakly basic (cationic) groups 22.
Raft Formation Based on Chemical Reactions:
Ionic Crosslinking: Polymers may undergo a phase transition in the presence of various ions. Some of the polysaccharides fall into the class of ion-sensitive ones. Gellan gum commercially available as Gelrite ® is an anionic polysaccharide that undergoes in situ gelling in the presence of mono- and divalent cation, including Ca2+, Mg2+, K+, and Na+. Gelation of the low-methoxy pectin can be caused by divalent cations, especially Ca2+. Likewise, alginic acid undergoes gelation in the presence of divalent/ polyvalent cations, e.g. Ca2+ due to the interaction with a guluronic acid block in alginate chain 1, 8, 9.
Enzymatic Cross-linking: Raft formation catalyzed by natural enzymes has not been investigated widely but seems to have some advantages over chemical and photochemical approaches. For example, an enzymatic process operates efficiently under physiologic conditions without the need for potentially harmful chemicals such as monomers and initiators.
Intelligent stimuli-responsive delivery systems using hydrogels that can release insulin have been investigated. Cationic pH sensitive polymers containing immobilized insulin and glucose oxidase can swell in response to blood glucose level releasing the entrapped insulin in a pulsatile fashion. Adjusting the amount of enzyme also provides a convenient mechanism for controlling the rate of gel formation, which allows the mixtures to be injected before gel formation 5, 6, 7.
Photo-polymerization: A solution of monomers such as acrylate or other polymerizable functional groups and initiator such as 2,2 dimethoxy-2-phenyl acetophenone, camphor Quinone and ethyl erosin can be injected into a tissues site and the application of electromagnetic radiation used to form gel designed readily to be degraded by chemical or enzymatic processes or can be designed for long term persistence in-vivo.
Typically long wavelength ultraviolet and visible wavelengths are used. A photopolymerizable, biodegradable hydrogels as a tissue contacting material 5, 6, 7.
Advantages of Floating Raft Forming System:
- Raft-forming system forms a low-density viscous layer on gastric contents and hence provides more effective surface area than a tablet. These lead to more drug release and improve bioavailability.
- Floating obtained faster than the other floating dosage form.
- Improve patient compliance by making a once a day therapy.
- Improve therapeutic efficacy.
- Easy to administer to a patient.
- It increases the contact time of drug at the site of maximum absorption (stomach).
- It provides advantages such as the delivery of drugs with narrow absorption in the small intestinal region.
- Reduction in plasma level fluctuation.
- Target stomach specific drug delivery system like pylori-induced gastric ulcer 2, 4, 8, 14.
Limitation of Floating Raft Forming System:
- These systems are formulated in the form of solution which is more susceptible to stability problems. These are due to chemical degradation (oxidation, hydrolysis, etc.) or microbial degradation.
- The formulation must be stored properly because if the formulation is not stored properly it may cause stability problem. This is due to change in the pH of the system on prolonged storage or on storing inappropriate temperature conditions.
- Exposure of certain polymer to radiations (e.g. UV, Visible, electromagnetic, ) induces the formation of gel within the package 2, 4, 8, 14, 28.
Evaluation Parameter of RFS:
In-vitro Evaluation:
Physical Appearance: The clarity of formulated solution and Raftl was determined by visual inspection under black and white background 3, 10.
Viscosity Measurement of RFS: Viscosity of the raft forming solution was determined with a Brookfield viscometer (Model No Brookfield DVE -LV viscometer Version 10.0) using a 20 ml aliquot of the sample. Measurements were performed using appropriate spindle number and the temperature was maintained at 25 ± 1 °C. All measurements were made in triplicate 3, 9.
pH: The pH was measured of RFS using a calibrated digital pH meter at 25 °C. All measurements of pH are made in triplicate 3, 10.
Buoyancy Study: It was determined to measure the time taken by the dosage form to float on the top of the dissolution medium after it is placed in the medium. The test is usually performed in SGF (simulated gastric fluid, 0.1N HCl) which was maintained at 37 °C.
The time between the introduction of dosage form and its buoyancy on the simulated gastric fluid and the time during which the dosage form remains buoyant were measured. The time for which the dosage form continuously floats on the dissolution media is floating time. The time taken for the dosage form to emerge on the surface of the medium is called Floating Lag Time (FLT) or Buoyancy Lag Time (BLT) and the total duration of time by which dosage form remains buoyant is called Total Floating Time (TFT) 3, 9.
Gel Strength: The gel strength apparatus was fabricated in house using a measuring cylindrical of 1.2 cm radius and a bore of 0.1 mm at its base. A needle 2 cm in length was used to which nylon threads were tied. Solution (10 ml) is taken in the cylinder with temporarily sealed bore followed by addition of 50 ml 0.1 N HCl for raft formation. After raft formation, the HCl is drained off by opening bore leaving the Raftl mass formed. The needle was rested on to the surface of the raft. At the free end of the thread, a lightweight pan is attached to which the weight was added. The gel/raft strength is reported in terms of weight required to pass the needle probe through the formed raft mass. The gel/raft strength is calculated using this formula 3, 9, 10.
Gel Strength= M × g / a ……………. (1)
Where, M = Weight at which needle passes through the formed gel mass, g = Gravitational force, a = Area of surface.
Measurement of Water Uptake by the Raft: The water uptakes by the raft of the selected formulations of sodium alginate were determined by a simple method. In this study raft formed in 40 ml of 0.1 N HCl (pH 1.2) was used. From each formulation, the raft portion from the 0.1 N HCl was separated, and the excess HCl solution was blotted out with a tissue paper. The initial weight of the raft taken was weighed and to this raft10 ml of distilled water was added, and after every 30 min of the interval water was decanted and the weight of the Raft was recorded, and the difference in the weight was calculated and reported 7, 38.
% water uptake = W2 –W1/ W1 × 100 ……… (2)
Where, W1 = initial weight of gel (10 ml), W2 = weight of swollen matrix after 16 h.
Drug Content: Accurately, 1 ml of RFS was added to an appropriate amount of 0.1N HCl to yield a solution containing the strength of 1000 μg/ml. From that 10 μg/ml solution was prepared by diluting stock solution. Determine drug content using the above stock solution 17.
In-vitro Drug Release Study: The release rate of the drug was determined using USP apparatus 2 at 50 rpm. This speed slow enough to avoid breaking of raft formulation and is maintaining mild agitation condition exist in-vivo. The dissolution medium used is 900 ml of simulated gastric fluid (0.1 N HCl), and the temperature is maintained at 37 °C. A sample is withdrawn at every 60 min time interval. The sample is analyzed, and the % cumulative release is calculated 9, 17.
In-vivo Evaluation:
Radiology and Scintigraphy: It involves the use of radio-opaque markers. X‐ray/Gamma Scintigraphy helps to locate dosage form in the gastrointestinal tract (GIT); thus one can predict and correlate the gastric emptying time and the passage of dosage form in the GIT. Barium sulfate is widely used as Radio Opaque Marker. Here the inclusion of a radio‐opaque material, i.e. BaSO4 into a solid dosage form enables it to be visualized by X-rays at different intervals to determine gastric retention. Similarly, the inclusion of γ‐emission of radionuclide in a formulation allows indirect external observation using a scintiscanner.
In the case of γ‐scintigraphy, the γ‐rays emitted by the radionuclide are focused on a camera, which helps to monitor the location of the dosage form in the GIT. 99Tc is widely used as the emitting material 29, 36.
Gastroscopy: Gastroscopy is peroral endoscopy used with fiber optics or video systems. Gastroscopy is used to inspect visually the effect of dosage form for prolongation in the stomach. It can also give a detailed evaluation of the gastroretentive drug delivery system 37.
Magnetic Marker Monitoring: In this technique, the dosage form is magnetically marked with incorporating iron powder inside the dosage form. Image of the dosage form can be taken by very sensitive bio-magnetic measurement equipment. Advantage of this method is that it is radiationless and thus not too much hazardous 29, 37.
13C Octanoic Acid Breath Test: 13C octanoic acid is incorporated into the gastroretentive drug delivery system, and the system is introduced in the stomach. In the stomach due to a chemical reaction, octanoic acid liberates CO2 gas which comes out in a breath. The important carbon atom which will come in CO2 is replaced with 13C isotope.
So the time up to which 13CO2 gas is observed in breath can be considered as the gastric retention time of the dosage form. As the dosage form moves to the intestine, there is no reaction and no CO2 release. So this method is cheaper than the other 37.
Patented Formulations of RFS: Patent Formulations are as follows:
TABLE 4: PATENTED FORMULATIONS 30, 31, 32, 33, 34
| S. no. | US Patent | Formulations |
| 1 | US20120009275 | In-situ forming hydro gel wound dressing containing antimicrobial agents |
| 2 | US20050063980 | Gastric raft composition |
| 3 | US5360793 | Rafting antacid formulation |
| 4 | US20020119941,35 | In -situ gel formation
of pectin |
| 5 | US20110082221 | In -situ gelling system as sustained delivery for eye |
Marketed Formulation of RFS: Various marketed preparation of RFS is as follows.
TABLE 5: MARKETED FORMULATION OF RFS 9, 25
| S. no. | Brand name | Drug |
| 1 | Liquid Gaviscon | Aluminium hydroxide and magnesium stearate |
| 2 | Topalkan | Aluminum hydroxide and magnesium stearate |
| 3 | Conviron | Ferrus sulphate |
CONCLUSION: Development of an efficient gastro-retentive system for stomach specific drug delivery is a real challenge. So, to produce the desired gastro retention various approaches have been employed, out of which a floating drug delivery system has emerged as the most promising technique. RFS is one of the approaches of floating drug delivery system which undergo raft formation in acidic stomach conditions and provide stomach specific release of drug for a longer duration while being buoyant on the gastric fluid surface.
As the system remains in the stomach for longer duration local action of the drug due to prolonged contact time to gastric mucosa is increased. This leads to less frequent dosing and improved efficiency of treatment. Raft-forming system is not only helpful for sustained drug delivery but also convenient for pediatric and geriatric patients. This system is helpful as an alternative of the oral solid dosage form with the advantages of the liquid dosage form. Sustained and prolonged release of the drug, good stability and bioavailability characteristics make the raft forming system very suitable candidate for gastric retention of the drug. Thus the raft forming system promises to be the potential approach for gastric retention drug delivery system. Development of RFS provides the opportunity of a line extension for the marketplace; wide range of drugs.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Shah P, Bhimani B and Patel U: Brief review of polymers use in raft forming system. Inter J of Pharmaceutical Research & Bioscience 2014; 3(5): 437-48.
- Doshi N: In situ gel: a novel approach of gastro retentive drug delivery. Asian Journal of Pharmaceutical Sciences and Research 2013; 3(3).
- Vishvadeep P, Akshay P, Henil P, Jay P, Ravi P and Upendra P: A review on in-situ gelling system: Novel approach for stomach specific drug delivery. International Journal of Pharmaceutical Research and Bioscience 2014; 3(2): 466-480.
- Bhavsar DN, Varde NM, Sini S, Shah VH and Upadhyay UM: Advances in GRDDS: Raft forming system a review. Journal of Drug Delivery and Therapeutics 2012; 2(5): 123-28.
- Chaturved S, Kumari P, Singh S and Agrawal V: Approaches to increase the gastric residence time: floating drug delivery systems- a review. Asian J Pharm Clin Res 2013; 6(3): 1-9.
- Ramya DD, Abhirami M, Brindha R, Gomathi S and Vedhahari BN: In-situ gelling system - a potential tool for improving therapeutic effects of drugs. International J of Pharmacy and Pharmaceutical Sciences 2013; 5(3): 27-30.
- Lovenish B, Pramod KS and Rishabha M: A short review on gastro-retentive formulations for stomach specific drug delivery: special emphasis on floating in-situ gel systems. African J of Basic and Applied Sci 2011; 3(6): 300-312.
- Samar ES: Floating systems for oral controlled release drug delivery. PhD Theses, Pharmazie der Freien Universität Berlin, Juli 2010; 1-95.
- Vipul D, Prajapati, Girish K, Tohra AK and Bhumi SZ: Raft forming system-an upcoming approach of gastro-retentive drug delivery system. Elsevier, Journal of Controlled Release 2013; 168: 151-165.
- Kalia N, Nirmala and Harikumar SL: In-situ gelling system: A review. Journal of Drug Delivery and Therapeutics 2014; 4(4): 93-103.
- Vinod K, Santhosh V, Anbuazaghan S, David B, Padmashri A and Sandhya S: Approaches for gastrotentive drug delivery systems. International J of Applied Biology and Pharmaceutical Technology 2010; 1(2): 589-601.
- Singh A, Dubey V and Arora V: Role of natural polymers used in floating drug delivery system. Journal of Pharmaceutical and Scientific Innovation 2012; 11-15.
- Nirmal HB, Bakliwal SP and Pawar SP: In-situ gel: new trends in controlled and sustained drug delivery system. Inter J of Pharm Tech Research 2010; 2(2): 1398-1408.
- Nikita D: Floating drug delivery system. Journal of Current Pharmaceutical Research 2011; 7(1): 6-20.
- Shreeraj S, Pratik U, Darsh P and Jinal S: In-situ gel: A Novel approach of gastroretentive drug delivery. Asian J of Biomedical and Pharmaceutical Sci 2012; 2(8): 01-08.
- Jayswal B, Yadav V, Patel K, Patel B and Patel P: Formulation and evaluation of floating in-situ gel based gastro retentive drug delivery of cimetidine. International J for Pharmaceutical Res Scholars 2012; 1(2); 327-337.
- Sanket C, Dron M, Ravi P, Jay P, Rahul D and Sunita C: Formulation and evaluation of floatable in-situ gel for stomach-specific drug delivery of ofloxacin. American Journal of Advanced Drug Delivery 2013; 1(3): 285-299.
- Rishad R, Chhagan N, Dashrath M and Nurudin P: Development of a novel floating in-situ gelling system for stomach specific drug delivery of the narrow absorption window drug baclofen. Iranian Journal of Pharmaceutical Research 2010; 9(4): 359-368.
- Sapra P, Patel D, Soniwala M and Chavda J: Development and optimization of in-situ periodontal gel containing levofloxacin for the treatment of periodontal diseases. J of Scientific and Innovative Research 2013; 2(3): 608-27.
- Asija R, Patel N, Shah S, Asija S and Barupal A: Sustain ophthalmic delivery of levofloxacin from a pH-triggered in-situ gelling system. International Research Journal of Pharmacy 2012; 3(4): 273-276.
- Akhani J and Modi C: Formulation development and evaluation of in-situ gel for vaginal drug delivery of the antifungal drug. An Inter J of Pharmaceutical Sciences, Pharma Science Monitor 2014; S-1, 5(2): 343-364,
- Patel D, Patel, DK and Patel C: Formulation and evaluation of floating oral in-situ gelling system of amoxicillin. International Scholarly Research Network, Pharmaceutics 2011; 276250.
- Tortora GH and Derrickson B: Principle of anatomy and physiology, Wiley International, Edition 11th, 902-940.
- Brahmankar DM and Jaiswal SB: Biopharmaceutics and pharmacokinetics- A Treatise. Vallabh Prakashan, Edition 2nd, 2009; 63-71.
- Jain NK: Progress in controlled and drug delivery system. CBS Publishers and Distributers Pvt., Ltd., 76-97.
- Ross and Wilson: Anatomy and physiology in heath and illness, Edition 12th, 294-308.
- Singh S, Singh J, Muttu MS, Balasubramaniam J and Mishra B: Gastroretentive drug delivery system of metoclopride hydrochloride: Formulation and in-vitro Current Drug Delivery 2007; 4: 269-275.
- Sathish D, Himabhindu S, Shravan K and Rao Y: Floating drug delivery system for prolonged gastric residence time: A review. Current Drug Delivery 2011; 8: 494-510.
- Caragh S, Pillay V, Choonara YE and Toit LC: Current Drug Delivery 2009; 6: 451-460.
- Gillian E and Ronald P: An alginate, pectin and an antacid, such as sodium or potassium bicarbonate; gastrointestinal reflux disease, US 20050063980 A1, 2004.
- Bruktawit T: In-situ forming hydrogel wound dressings containing antimicrobial agents US 20120009275 A1, 2011.
- William J: Rafting antacid formulation, US5360793, 1994.
- Yawei N and Knneth M: In-situ gel formation of pectin, US 20020119941, 2002.
- Gillian E and Ronald P: Gastric raft composition, US 20050063980, 2005.
- Claire H and Stephane J: In-situ gelling systems as sustained delivery for the front of the eye, US20110082221, 2011.
- Rathee P, Jain M, Garg A, Nanda A and Hooda A: Gastrointestinal mucoadhesive drug delivery system: a review. J Pharm Res 2011; 4: 1448-1453.
- Soni R, Patel A, Patel R, Patel M, Patel K and Patel N: Gastroretentive drug delivery systems: a review. Int J Pharma World Res 2011; 2: 1-24.
- Panwar P, Chaurasiya D, Jain G and Sheorey R: Formulation and evaluation of oral floatable in-situ gel of diltiazem HCl. International Journal of Novel Drug Delivery Technology, 2012.
How to cite this article:
Raut SS and Shinde HA: In-situ raft forming system: a review. Int J Pharmacognosy 2018; 5(6): 337-49. doi link: http://dx.doi.org/ 10.13040/IJPSR.0975-8232.IJP.5(6).337-49.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.