SCREENING OF AERVA LANATA LINN. FOR PHARMACOGNOSTIC AND ANTIMICROBIAL ACTIVITY
HTML Full TextSCREENING OF AERVA LANATA LINN. FOR PHARMACOGNOSTIC AND ANTIMICROBIAL ACTIVITY
Anita and A. Retna Malar *
Department of Chemistry and Research Centre, Scott Christian College (Autonomous), Nagercoil - 629001, Tamil Nadu, India.
ABSTRACT: Aerva lanata has been used in traditional medicine for many years and used for the treatment of urinary bladder and kidney stones. The aim was to study the pharmacognostic characters and antimicrobial activity of important medicinal plant Aerva lanata (Amaranthaceae). The physicochemical, phytochemical and fluorescence analysis of powdered aerial and root parts of Aerva lanata was performed according to the standardization procedures recommended by the World Health Organization. The physicochemical parameters such as total ash, acid insoluble ash, water-soluble ash reveal that the plant has less amount of carbonates, silicates and are less affected by contamination and inorganic compounds. The various colors obtained by fluorescence were analyzed, and the moisture content of aerial and root parts were found to be 37.5%, 35.33% respectively. The preliminary phytochemical screening showed the presence of alkaloids, saponins, terpenoids, flavonoids, proteins, glycosides, coumarins, carbohydrate, steroids, tannins, phenols and phytosterols. The antimicrobial activity performed of Aerva lanata on Pseudomonas aeruginosa, Klebsiella pneumonia, Bacillus substilis, Streptococcus mutants, Escherichia coli, Staphylococcus aureus showed maximum activity against gram-negative bacteria on aerial parts of Aerva lanata. Thus the results of the present study can serve as a valuable source for the identification of this plant and their active components in future investigations.
| Keywords: |
Pharmacognostic, Antimicrobial, Moisture, Fluorescence, Phytochemical, Pharmacognosy
INTRODUCTION: The traditional herbal products are heterogeneous. They impose several challenges like quality control, quality assurance, and regulatory process. Most of the herbal product in the market today has not been subjected to the drug approval process to demonstrate their safety and effectiveness 1.
The therapeutic efficacy of medicinal plants depends upon the quality and quantity of chemical constituents. The misuse of herbal medicine starts with wrong identification 2. Pharmacognostic studies of medicinal plants can solve all these problems. Pharmacognostic studies ensure plant identity, lay down standardization parameters which help and prevent adulteration. This ensures reproducible quality of herbal products 3. Standardization is a system to ensure that every medicine has the correct amount of constituents and will induce its therapeutic effect 4. Determination of ash residues and active components like saponin, alkaloids play a significant role in the standardization of the indigenous crude drugs 5. The extract obtained after standardization may be used as a medicinal agent as such in the form of tinctures or fluid extracts. These medicinal agents contain complex mixtures of many medicinal plant metabolites, such as alkaloids, glycosides, terpenoids, flavonoids 6.
Antimicrobial screening of plant extracts and phytochemicals represents a starting point for antimicrobial drug discovery 7. According to Sumitra Chanda, ash values are used to determine the quality and purity of the crude drug. It indicates the presence of various impurities like carbonate, oxalate, and silicate. The water-soluble ash is used to estimate the amount of inorganic compound present in drugs. The insoluble acid ash consists mainly of silica and indicate contamination with earthy material. The moisture content of drugs should be at a minimal level to discourage the growth of bacteria, yeast or fungi during storage. Fluorescence analysis is an important parameter for pharmacognostic evaluation of crude drugs 3. Thus, the present work was done on Aerva lanata which deals with the standardization of medicinal plant; antimicrobial activity has been performed. The objective of the present study was:
- To study the pharmacognostical features of medicinal plant Aerva lanata.
- To carry out the phytochemical screening tests to various extracts of Aerva lanata.
- To ensure the antibacterial activity of chloroform, methanol aerial and root extracts of Aerva lanata.
MATERIALS AND METHODS:
Collection of Plants: The medicinal plant Aerva lanata belonging to the family Amaranthaceae has been collected in and around Nagercoil area, Kanya Kumari district, Tamil Nadu during February 2012.
Identification of Plants: The collected plants were identified using standard keys, and voucher specimens of these plants were submitted at the herbarium of Scott Christian College (Autonomous), Nagercoil. The voucher number is SCCN 3267.
Pharmacognostic Standardization Parameters: The powdered plants chosen for the present study were subjected to pharmacognostic standardization parameters such as physico-chemical analysis, moisture content, fluorescence analysis, and phytochemical analysis.
Physico-Chemical Analysis: The powdered aerial and root parts of Aerva lanata were subjected to physicochemical analysis. Total ash, Acid insoluble ash, Water soluble ash, is done based on standard methods.
i) Determination of Total Ash: Total ash was determined by weighing 2 gm of air-dried sample in silica dish and incinerated at a temperature not exceeding 450 °C until free from carbon and then was cooled and weighed 8, 9.
Total ash % = Weight of total ash / Weight of sample taken × 100
ii) Determination of Acid Insoluble Ash: The total ash was boiled with 25 ml of 2M dilute HCl for five minutes. The insoluble matter was collected on ashless filter paper and was washed with hot water, ignited, cooled in a desiccator and weighed. The percentage of acid insoluble ash was calculated concerning the air-dried sample 10.
% Acid insoluble ash value = Weight of acid insoluble ash / Weight of sample taken × 100
iii) Determination of Water-Soluble Ash: The total ash was boiled with 25 ml of water for five minutes. The insoluble matter was collected on ashless filter paper and was washed with hot water, ignited for fifteen minutes at a temperature not exceeding 450 °C. The weight of insoluble matter was subtracted from the weight of total ash. The difference in weight represents the water-soluble ash. The percentage of water-soluble ash was calculated concerning the air-dried sample 11, 12.
% Water soluble ash value = Weight of water soluble ash / Weight of sample taken × 100
iv) Determination of Moisture Content: 10g of plant parts were weighed separately in pre-weighed glass Petri dishes and dried in hot air oven (80 -95 °C) for 3 h. The dishes were then transferred to desiccators, cooled to room temperature, and the weight was recorded. The drying was continued until the final weight became stable. The weight differences were noted, and the moisture content was expressed in percentage weight of fresh plant parts 13.
Moisture content % = Initial weight – final weight/ Initial weight × 100
Fluorescence Analysis: The fluorescence analysis of Aerva lanata was done by placing powdered aerial and root parts on a slide and observing the color changes under UV and visible lights after treating with several drops of specific reagents such as methanol, hexane, chloroform, ethanol, acetone, acetic acid, propanol, acetonitrile, ethyl acetate, and water. The development of color was noted within 1-2 min to avoid drying and resultant color change 14, 15.
Preliminary Phytochemical Screening Tests: The shade dried parts of aerial and root parts of Aerva lanata were extracted in a soxhlet apparatus for 24 h using different solvents in the order of polarity like hexane, chloroform, ethyl acetate, acetone, methanol, water. The extracted solvents were distilled by vacuum distillation under reduced pressure to separate the solvents from the extracts. The crude extracts obtained from soxhlet extraction were subjected to preliminary phytochemical screening. Phytochemical screening involves testing the different extracts of Aerva lanata for various phytochemicals by qualitative analysis, which gives a general idea regarding the nature of constituents present in crude extracts 16. The qualitative phytochemical test carried for various extracts were given below.
A) Determination of Alkaloids: About 3 ml of concentrated extract was taken in a test tube, and 1 ml of HCl was added to the mixture and heated gently for 20 min and then cooled and filtered. The filtrate was used for following test
Wagner’s Test: The filtrate was treated with Wagner’s reagent (solution of iodine in potassium iodide); formation of reddish brown precipitate indicates the presence of alkaloid 17.
Mayer’s Test: A few ml of filtrate is treated with 4-5 drops of Mayer's reagent (Potassium mercuric iodide) Formation of a yellow or white colored precipitate or turbidity indicates the presence of alkaloids 18.
B) Test for Saponins:
Foam Test: 5 ml of the extracts were mixed with 20 ml of distilled water then agitated in a graduated cylinder for about 15 min formation of foam indicates saponin 18.
Froth Test: Exactly 0.5 g of the extract was dissolved in distilled water in a test tube. Frothing which persisted on warming was taken as preliminary evidence for saponins 19.
C) Test for Phytosterols:
Libermann Burchard’s Test: Extracts were treated with chloroform and filtered. The filtrates were treated with a few drops of acetic anhydride, boiled and cooled. Concentrated sulphuric acid was added. The formation of a brown ring at the junction indicates the presence of phytosterols 20.
D) Test for Proteins:
Xanthoproteic Test: The extracts were treated with a few drops of concentrated nitric acid. Formation of yellow color indicates the presence of proteins 21.
E) Test for Triterpenes:
Salkowski Test: Extracts were treated with a few drops of concentrated sulphuric acid were added to the chloroform solution, shaken and allowed to stand, the appearance of golden yellow color indicates the presence of triterpenes 21.
F) Test for Diterpenes:
Copper Acetate Test: The extract was dissolved in water and treated with 10 drops of copper acetate solution; the formation of emerald green color indicates the presence of diterpenes 19.
G) Test for Glycosides:
Glycoside Test: 0.5 mg of the extract was dissolved in 1 ml of water, and then the aqueous NaOH solution was added. Formation of yellow color indicates the presence of glycosides 22.
Concentrate H2SO4 Test: To 5 ml extract, add 2 ml glacial acetic acid, one drop 5% FeCl3 and concentrated H2SO4. Brown ring appears indicates the presence of glycosides 23.
H) Test for Tannins:
Ferric Chloride Test: To the extracts add a few drops of 1% neutral ferric chloride solution were added, the formation of a blackish blue color indicates the presence of tannins 21.
I) Test for Steroids:
Salkowski’s Test: 5 ml of the extract was dissolved in 2 ml of chloroform, and an equal volume of concentrated sulphuric acid was added along the sides of the test tube. The upper layer turns red, and the lower layer turns yellow with green fluorescence, indicating the presence of the steroids 24.
J) Test for Flavonoids:
NaOH Tests: To 2-3 ml extract, few drops of sodium hydroxide were added in a test tube. Formation of intense yellow color that becomes colorless on the addition of a few drops dilutes HCl indicates the presence of flavonoids 25.
Pew’s Tests: To 2-3 ml extract, zinc powder was added in a test tube, followed by dropwise addition of concentrated HCl. Formation of purple-red or cherry color indicates the presence of flavonoids 26.
K) Test for Coumarins: 10% NaOH (1 ml) was added to 1 ml of the plant extracts, the formation of yellow color indicated the presence of coumarins 27.
L) Test for Carbohydrates:
Molisch Test: To 2–3 ml of the aqueous extract, add two drops of alpha-naphthol solution in alcohol shake and add concentrated sulphuric acid from the sides of the test tube. Violet ring is formed which indicate the presence of carbohydrates 16.
Antimicrobial Activity of Medicinal Plants: The chloroform and methanol extracts of aerial and root parts of Aerva lanata were chosen for the antimicrobial studies. Antibiotic susceptibility tests were determined by the agar disc diffusion (Kirby–Bauer) method. Bacterial strains were swabbed using sterile cotton swabs in the nutrient agar plate. A disc of 6 mm was punched from Whatman no.1 filter paper. Up to 10 μl of each concentration of the extract were respectively introduced in the discs using sterile pipettes.
The disc was then placed on the surface of the medium, and the compound was allowed to diffuse for 5 min, and the plates were kept in incubation at 37 °C for 24 hours. At the end of incubation, inhibition zones were examined around the disc and measured with a transparent ruler in millimeters 28.
RESULTS AND DISCUSSION:
Physico-Chemical Evaluation: Physico-chemical parameters of a crude powder of Aerva lanata were shown in Table 1.
TABLE 1: PHYSICO - CHEMICAL PARAMETERS OF CRUDE POWDER OF AERVA LANATA
| S.
no |
Name of the physico-
chemical constants |
A. lanata
(% of the composition) aerial |
A. lanata
(% of composition) root |
| 1 | Moisture content | 37.5 | 35.33 |
| 2 | Total ash | 9.5 | 9.65 |
| 3 | Acid-insoluble ash | 4.45 | 5.55 |
| 4 | Water soluble ash | 3.8 | 4.95 |
The physicochemical parameter values of aerial and root parts of Aerva lanata reveals that the moisture content of Aerva lanata aerial and root parts was found to be 37.5% and 35.33% respectively. Thus, there is a less possibility of microbial growth, fungi, and contamination. The total ash value of aerial and root parts of Aerva lanata is found to be 9.5% and 9.65%. The total ash of the crude powders is not too high. So the plant may contain the appropriate amount of physiological and non-physiological ash. The acid insoluble ash value of aerial and root parts of Aerva lanata is found to be 4.45% and 5.55% respectively. The values of acid insoluble ash show that there is less possibility for each plant to be contaminated with silica materials. The water-soluble ash value of aerial and root parts of Aerva lanata was found to be 3.8% and 4.95%, which indicates that the constituents present in root parts of Aerva lanata were more extracted by water than root parts.
TABLE 2: FLUORESCENCE ANALYSIS OF AERIAL PARTS OF AERVA LANATA
| Treatment | U.V light | Visible light |
| Powder as such | Green | Pale yellow |
| Powder + methanol | Green | Light green |
| Powder + hexane | Light green | Light green |
| Powder + chloroform | Black | Violet |
| Powder + ethanol | Green | Light green |
| Powder + acetone | Green | Pale green |
| Powder + acetic acid | Pale green | Pale green |
| Powder + propanol | Dark green | Violet |
| Powder + acetonitrile | Green | Pale violet |
| Powder + ethyl acetate | Green | Green |
| Powder + water | Green | Yellow-green |
Fluorescence Analysis: The powdered aerial and root parts of Aerva lanata were subjected to fluorescence analysis. The results of fluorescence analysis of Aerva lanata were expressed in Table 2 and Table 3.
TABLE 3: FLUORESCENCE ANALYSIS OF ROOT PARTS OF AERVA LANATA
| Treatment | U.V light | Visible light |
| Powder as such
Powder + methanol Powder + hexane Powder + chloroform Powder + ethanol Powder + acetone Powder + acetic acid Powder + propanol Powder + acetonitrile Powder + ethyl acetate Powder + water |
Grey
Green Green Blue Green Dark green Dark green Blue Dark brown Brown green Blue |
Grey
Grey Grey Grey Violet Grey Violet Violet Grey Brown Brown |
The powdered aerial parts of Aerva lanata as such, and on treatment with methanol, hexane, ethanol, acetone, acetic acid, propanol, acetonitrile, ethyl acetate and water exhibit green colour in U.V light which may be due to the presence of compounds such as furanocoumarins, phytosterols, steroids, alkaloids, polar compounds in the crude powder 29-33. The aerial parts treated with methanol, hexane, ethanol, acetone, acetic acid and ethyl acetate in visible light shows green color this may be due to saponins, terpenoids in the crude powder 31, 34.
The violet color obtained on treatment with chloroform, propanol, and acetonitrile in visible light is due to saponins, sterols 34, 35 present in the sample. The crude powder with chloroform in the presence of U.V light fluoresces black which may be due to the flavonoids present in the powder 36. The crude powder as such shows pales yellow color in visible light which indicates the presence of flavonoid compounds in the samples. The powder with water shows yellow-green color under visible light may be due to the presence of ‘chlorophyllide b’ compound present in sample 36.
The powdered root parts of Aerva lanata as such grey in color in U.V light, this may be due the presence of flavonoids in sample 37 and grey color obtained in visible light on treatment with methanol, hexane, chloroform, acetone, acetonitrile is due to the presence of ‘pheophytin a’ present in the sample 36. The powder when treated with methanol, hexane, ethanol, acetone, acetic acid under U.V light shows green color which may be due to the presence of furanocoumarins, phytosterols, steroids, alkaloids, polar compounds in the crude powder 29-33. The powder treated with acetonitrile exhibit brown color under U.V light may be due to polyphenols, flavonoids in sample 38, 33. The powder with ethyl acetate in U.V light shows brown-green color which may be due to the presence of most flavones, glycosides, bioflavonoids and unusually substituted flavones 36.
The powder shows blue color in U.V light when treated with chloroform, propanol, water which may be due to the presence of flavonoids in sample 34. The crude powder as such, on treatment with methanol, hexane, chloroform, acetone, acetonitrile in visible light shows grey color this may be due to ‘pheophytin a’ present in sample 36. Violet color was obtained when the powdered samples were treated with ethanol, acetic acid, propanol under visible light may be due to saponins, steroids present in sample 34, 35. The brown color obtained on treatment with ethyl acetate and water in visible light indicates the presence of glycosides 39.
Qualitative phytochemical screening was done in various aerial extracts of Aerva lanata are presented in Table 4 showed the presence of alkaloids, saponins, phytosterols, triterpenes, proteins, glycosides, coumarins, flavonoids. Saponin is found in only in chloroform extract. Coumarin, Protein, Glycosides were found almost in all extracts. The maximum numbers of phytochemical constituents are found in chloroform, ethyl acetate, and water extract. The minimum number of phytochemical constituents is found in acetone extract.
Qualitative phytochemical screening was done in various root extracts of Aerva lanata are presented in the Table 5 showed the presence of alkaloids, saponins, phenols, coumarins, steroids, proteins, triterpenes, glycosides, flavonoids, carbohydrates, tannins and phytosterols. The phytochemical coumarin is observed only in the acetone extract. The carbohydrates and tannins are found in methanol and water extracts. Glycosides were found in all the extracts and proteins were found in all extracts except hexane. The maximum amount of phytochemical constituents is found in methanol extract, and the minimum number of phyto-chemical constituents is found in hexane and ethyl acetate extracts.
TABLE 4: PHYTOCHEMICAL SCREENING OF AERIAL EXTRACTS OF AERVA LANATA
| Chemical category | Name of the test | H | C | E | A | M | W |
| Alkaloids
|
Mayer’s test | +
|
-
|
-
|
-
|
-
|
+
|
| Wagner’s test | |||||||
| Saponins | Foam test | - | + | - | - | - | - |
| Froth test | |||||||
| Phytosterols | Liberman’s Burchard’s test | - | + | + | - | - | - |
| Triterpenes | Salkowski test | - | - | + | + | - | + |
| Proteins | Xanthoprotein test | + | - | + | - | + | + |
| Glycosides | Glycoside test | - | + | - | + | + | + |
| The concentrated sulphuric acid test | |||||||
| Coumarins | 10%NaOH+1ml plant extract | + | + | + | - | - | + |
| Flavonoids | Pew’s test | - | + | + | - | + | - |
| NaOH test |
H = Hexane extract, C = Chloroform extract, E = Ethyl acetate extract, A = Acetone extract, M = Methanol extract, W = Water extract, + = present, - = absence
TABLE 5: PHYTOCHEMICAL SCREENING OF ROOT EXTRACTS OF AERVA LANATA
| Chemical category | Name of the test | H | C | E | A | M | W |
| Alkaloids | Mayer’s test | + | + | - | - | + | - |
| Wagner’s test | |||||||
| Saponins | Foam test | + | + | + | - | + | - |
| Froth test | |||||||
| Phenols | Lead acetate test | - | - | - | - | + | + |
| Coumarins | 10%NaOH +1 ml plant extract | - | - | - | + | - | - |
| Steroids | Salkowski test | - | + | - | + | + | + |
| Proteins | Xanthoprotein test | - | + | + | + | + | + |
| Triterpenes | Xanthoprotein test | + | - | - | - | - | + |
| Glycosides | Glycoside test | + | + | + | + | + | + |
| A concentrated sulphuric acid test | |||||||
| Flavonoids | Pew’s test | - | + | - | + | + | - |
| NaOH test | |||||||
| Carbohydrates | Molisch’s test | - | - | - | - | + | + |
| Tannins | Ferric chloride test | - | - | - | - | + | + |
| Phytosterols | Liberman’s Burchard’s test | - | + | - | + | + | + |
H = Hexane Extract, C = Chloroform Extract, E= Ethyl acetate Extract, A = Acetone Extract, M = Methanol Extract, W = Water Extract, + = present, - = absent.
TABLE 6: ANTIMICROBIAL ACTIVITY OF CHLOROFORM EXTRACTS OF AERVA
| Organism | C2 | C3 | Positive Amikacin | Negative |
| Escherichia coli | NZ | NZ | 20 mm | - |
| Staphylococcus aureus | 9 mm | 9 mm | 20 mm | - |
| Bacillus substilis | 10 mm | 0 mm | 20 mm | - |
| Pseudomonas aeruginosa | 11 mm | 0 mm | 22 mm | - |
| Klebsiella pneumoniae | 11 mm | 0 mm | 22 mm | - |
| Streptococcus mutants | 10 mm | 10 mm | 20 mm | - |
NZ = No Zone of inhibition, C1= Chloroform extract of Dichrostachys cinerea leaves, C2, C3= Chloroform aerial and root extracts of Aerva lanata.
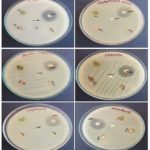
Antimicrobial Activity of Chloroform Extracts of Aerva lanata: The chloroform extract parts of Aerva lanata showed no inhibitory effect against Escherichia coli. In chloroform aerial extracts of Aerva lanata the maximum inhibitory effect was observed against Pseudomonas aeruginosa (11 mm), Klebsiella pneumonia (11 mm). The moderate activity was observed against Bacillus substilis and Streptococcus mutants (10 mm) and Escherichia coli (9 mm).
In chloroform root extracts of Aerva lanata, the maximum zone of inhibitory activity was found against Bacillus substilis, Pseudomonas aeruginosa, Klebsiella pneumonia and Streptococcus mutants (10 mm) and moderate inhibitory activity were found in Staphylococcus aureus (9mm). From the above observation, the maximum inhibitory activity was found in chloroform aerial extracts of Aerva lanata against Pseudomonas aeruginosa, Klebsiella pneumonia than other extracts. Thus, comparing the antimicrobial activity of gram positive and gram negative bacteria, the maximum inhibitory activity was found against gram-negative bacteria than gram-positive bacteria.
FIG. 1: SHOWING THE ANTIMICROBIAL ACTIVITY OF CHLOROFORM EXTRACTS OF AERVA LANATA. C1, C2, C3 - represents the antimicrobial activity of chloroform extracts of Dichrostyachys cinerea leaves, aerial and root parts of Aerva lanata
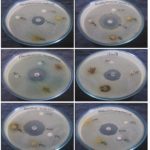
Antimicrobial Activity of Methanol Extracts of Aerva lanata: The methanol aerial extracts of Aerva lanata showed maximum inhibitory activity (15mm) against Escherichia coli whereas moderate antimicrobial activity was observed against Staphylococcus aureus and Klebsiella pneumonia (10mm), but the antimicrobial activity was not observed against Bacillus substilis, Pseudomonas aeruginosa, and Streptococcus mutants. In methanol root extracts of Aerva lanata the maximum zone of inhibition was obtained against Bacillus substilis (10 mm) whereas moderate zone of inhibition is observed against Escheria coli, Pseudomonas aeruginosa and Streptococcus mutants (8 mm) but the antimicrobial activity was not observed against S. aureus and K. pneumonia.
TABLE 7: ANTIMICROBIAL ACTIVITY OF METHANOL EXTRACTS OF AERVA LANATA
| Organism | M2 | M3 | Positive Chloramphenicol | Negative |
| Escherichia coli | 15 mm | 8 mm | 20 mm | _ |
| Staphylococcus aureus | 10 mm | NZ | 30 mm | _ |
| Bacillus substilis | NZ | 10 mm | 30 mm | _ |
| Pseudomonas aeruginosa | NZ | 8 mm | 25 mm | _ |
| Klebsiella pneumoniae | 10 mm | NZ | 25 mm | _ |
| Streptococcus mutants | NZ | 8 mm | 18 mm | _ |
NZ= No Zone of inhibition, the M1= Methanol extract of Dichrostachys cinerea leaves, M2, M3= Methanol extracts of aerial and root parts of Aerva lanata
FIG. 2: SHOWING THE ANTIMICROBIAL ACTIVITY OF METHANOL EXTRACTS OF AERVA LANATA. M1, M2, M3 - represents the antimicrobial activity of methanol extracts of Dichrostyachys cinerea leaves aerial and root parts of Aerva lanata.
When we compare the results of antimicrobial activity of chloroform and methanol extracts the maximum amount of antimicrobial activity was obtained from the aerial extracts of Aerva lanata against gram-negative bacteria.
CONCLUSION: Standardization is an essential measure for quality, purity and sample identification. Evaluation of extractive values is helpful in the estimation of specific constituents soluble in a particular solvent.
Physicochemical and chemical analysis confirms the quality and purity of the plant and its identification. Physicochemical parameters like moisture content of the selected plants are found to not high, and there be a less possibility of microbial attack. Total ash of the plants was found to be less, so the plants have less amount of carbonates, silicates, etc. Acid-insoluble ash, water-soluble ash values are less; this reveals that the plants are very less affected by contamination and inorganic compounds. Phytochemical analysis showed the presence of active phytoconstituents present in the extracts of each plant taken for the study. The antimicrobial activity is found mostly in gram-negative bacteria.
From the above results, the physicochemical parameters will help an individual, whether the plant material is undergone adulteration or not. Phytochemical analysis of the plant is very important commercially and has greatly involved in pharmaceutical companies for the production of new drugs for curing various diseases. The preliminary phytochemical screening reveals the useful findings of the chemical constituents present in the plants. The Thin Layer Chromatography performed on various extracts showed the presence phytochemicals present in the plants. From the antimicrobial study, medicinal plants can be used as antimicrobial agents in new drugs for therapy of infectious diseases in human. Here, the information collected was useful for further pharmacological and therapeutical evaluation along with the standardization of plant material.
ACKNOWLEDGEMENT: The authors are thankful to the authorities of Scott Christian College (autonomous), Nagercoil, for granting permission to do the work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Pal S and Shukla Y: Herbal medicine: current status and the future. Asian journal of Cancer Prevention 2003; 4: 281-288.
- Dinesh Kumar C: Pharmacognosy can help minimize accidental misuse of herbal medicine. Current Science 2007; 3: 1356- 1358.
- Chanda S: Importance of pharmacognostic studies of medicinal plants an overview. Journal of Pharmacognosy and phytochemistry 2014; 2: 69-73.
- Khanvilkar V, Patil L and Kadar V: Standardization of chitrakadi vati: An Ayurvedic polyherbal formulation. International Journal of Pharmaceutical Sciences and Drug Research 2014; 6: 303-309.
- Fazal H, Ahmad N and Khan MA: Physico-chemical, phytochemical evaluation and DPPH- scavenging antioxidant potential in medicinal plants used for herbal formulation in Pakistan. Pakistan Journal of Botany 2011; 43: 63-67.
- Handa SS, Khanuja SPS, Longo G and Rakesh DD: Extraction Technologies for medicinal and aromatic plants. International Centre for Science and High Technology Trieste 2008; 21-25.
- Bhattacharjee B and Islam SMS: Assessment of antibacterial and antifungal activities of the extracts of R. retusa blume- a medicinal orchid. World Journal of Pharmacy and Pharmaceutical Science 2015; 4: 74-87.
- Kokate CK: Practical Pharmacognosy. Vallabh Prakashan, Delhi, Edition 4th, 2001: 108-111,125.
- Harborne JB: Phytochemical methods. Chapman and Hall, NewYork, 1984: 6.
- Singh H, Mishra SK and Pande M: Standardization of Arjunarishta formulation by TLC method. International Journal of Pharmaceutical Sciences 2010; 2: 25-28.
- Kokate CK, Purohit AP and Gokhale SBP: Pharmacognosy. Nirali Prakashan, Pune, Edition 13th, 1994: 593-597.
- Bele AA and Khale A: Standardization of herbal drug. A review. International Journal of Pharmacy 2011; 2: 56-60.
- Vaghasiya Y, Nair R, Baluja S and Chanda S: Antibacterial and preliminary phytochemical analysis of Eucalyptus citriodora HK. Leaf. Natural Product Research 2008; 22: 754-762.
- Chase CR and Pratt R: Fluorescence of powdered vegetable drugs with particular reference to the development of a system of identification. Journal of the American Pharmaceutical Association (Scientific ed.) 1949; 38: 324-331.
- Kumar M, Monal P, Borah S and Mahato K: Physicochemical evaluation, preliminary phytochemical investigation, fluorescence and TLC analysis of leaves of the plant Lasia spinosa (Lour) Thwaites. Journal of Pharmacy and Pharmaceutical Sciences 2013; 5: 306-310.
- Neelima N, Devidas NG, Sudhakar M and Kiran VJ: A preliminary phytochemical investigation on the leaves of Solanum xanthocarpum. International Journal of Research in Ayurveda and Pharmacy 2011; 2: 845-850.
- Ashvin G, Swant R and Sutar A: Phytochemical analysis of an ethanolic extract of roots of Carrisa carandus Linn. Rasayan Journal of Chemistry 2012; 5: 456-459.
- Sanmugarajan V, Thabrew I and Sivapalan SR: Phyto, physicochemical standardization of medicinal plant Enicostemma littorale, Blume. ISOR Journal of Pharmacy 2013; 3: 52-58.
- Adegoke AA, Iberi PA and Mboto DAC: Studies on phytochemical screening and antimicrobial potentials of Phyllanthus amarus against multiple antibiotic-resistant bacteria. International Journal of Applied Research in Natural Products 2010; 3: 6-12.
- Boxi M, Rajesh Y, Raja Kumar V, Praveen B and Mangamma K: Extraction, phytochemical screening and in-vitro evaluation of antioxidant properties of Commicarpus chinesis (aqueous leaf extract). International Journal of Pharma and Biosciences 2010; 1: 537-547.
- Tiwari P, Kumar B and Kaur M, Kaur G and Kaur H: Phytochemical screening and Extraction: A review. Internationale Pharmaceutical Science 2011; 1: 98-106.
- Geetha TS and Geetha N: Phytochemical screening, quantitative analysis of primary and secondary metabolites of Cymbopogan citratus Stapf. Leaves from Kodaikanal Hills, Tamil Nadu. International Journal of Pharma Tech Research 2014; 6: 521-529.
- Kandewal KR: Practical Pharmacognosy. Nirali Prakashan Pune, Edition 19th, 2009: 149- 156.
- Shibu A and Dhanam S: Phytochemical screening of Caulerpa recemosa collected from Gulf of Mannar, Tamil Nadu. Asian Journal of Biochemical and Pharmaceutical Research 2015; 5: 40-45.
- Halilu ME, Abubakar A, Garba MK and Isah AA: Antimicrobial and preliminary phytochemical studies of methanol extract of root bark of Crossopteryx febrifuga (Rubiaceae). Journal of applied Pharmaceutical science 2012; 2: 60-70.
- Peach K and Tracey MV: Modern methods of plant analysis. springer Verlage, Berlin, 1956: 3: 33.
- Vijayalakshmi R and Ravindhran R: Preliminary comparative phytochemical screening of root extracts of Diospyrus ferrea (wild). Bakh and Aerva lanata (L.) Juss- Ex Schultes. Asian Journal of Plant Science and Research 2012; 2: 581-587.
- Bauer AW, Kirby WM, Sherris JC and Turck M: Antibiotic susceptibility testing by a standardized single disk method. American Journal of Clinical Pathology 1966; 45: 493-6.
- Borde V, Sonwane B, Sonakke V and Somwanshi B: Isolation and purification of alkaloids from medicinal plants by HPLC. International Journal of current Microbiology and Applied Sciences 2014; 3: 414-423.
- Kavitha.R: Fluorescence and FT-IR analysis of leaf and fruit of Trichosanthes dioica Roxb. World Journal of Pharmacy and Pharmaceutical Sciences 2014; 3: 563-572.
- Seasotiya L, Siwach P, Malik A, Bai S, Bharti P and Dalal S: Phytochemical evaluation and HPTLC finger print profile of Cassia fistula. International Journal of Advances in Pharmacy, Biology and Chemistry 2014; 3: 604-611.
- Mohandass S: Identification of bioactive compounds in Solanum incanum fruit by Thin Layer Chromatography and HPTLC. International Journal of Scientific Research 2013; 2: 22-25.
- Maruthamuthu V and Kandasamy R: Phytochemical Screening of secondary metabolites from Myxopyrum seratulum A.W. Hills by GC-MS, HPTLC Analysis. International Research Journal of Pharmacy 2016; 7: 51-57.
- Senguttuvan J and Subramaniam P: HPTLC fingerprints of various secondary metabolites in the traditional medicinal herb Hypochaeris radicata L. Journal of Botany 2016; 1-11.
- Dalal S, Seasotiya L, Bai S, Bharti P, Malik A and Taneja R: Preliminary phytochemical evaluation and HPTLC fingerprinting of leaves of Azadirachta indica. American Research Journal of Biosciences 2015; 1: 42-47.
- Harborne JB: Phytochemical methods, Chapman and Hall, Springer, Edition 3rd, 1998: 187- 278.
- Karthika K, Jamuna S and Paulsamy S: TLC and HPTLC fingerprint profiles of different bioactive components from the tuber of Solena amplxicanlis. Journal of Pharmacognosy and Phytochemistry 2014; 3: 198-206.
- Varghese S, Narmadha R, Gomathi D, Kalaiselvi M and Devaki K: Phytochemical screening and HPTLC fingerprinting analysis of Citrullus lanatus (Thunb) seed. Journal of Acute Diseases 2013; 2: 122-128.
- Hemmalakshmi S, Priyana S and Devaki K: Phytochemical screening and HPTLC fingerprinting analysis of the ethanolic extract of Erythrina variegata L. flowers. International Journal of Pharmacy and Pharmaceutical Sciences 2016; 8: 210-217.
How to cite this article:
Anita and Malar AR: Screening of Aerva lanata: Linn. for pharmacognostic and antimicrobial activity. Int J Pharmacognosy 2017; 4(4): 99-108. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(4).99-108.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
99-108
729
843
English
IJP
Anita and A. R. Malar *
Department of Chemistry and Research Centre, Scott Christian College (Autonomous), Nagercoil, Tamil Nadu, India.
anitavasantham@gmail.com
04 January 2017
01 March 2017
14 March 2017
10.13040/IJPSR.0975-8232.IJP.4(4).99-108
01 April 2017