REGULATION OF WNT GENES IN STEM CELLS DEVELOPMENT AND ORGANOGENESIS
HTML Full TextREGULATION OF WNT GENES IN STEM CELLS DEVELOPMENT AND ORGANOGENESIS
Rajendran Prakash
Department of Biotechnology, VMKV Engineering College, Vinayaka Missions University, Salem - 636308, Tamil Nadu, India.
ABSTRACT: Stem cell biology, many signals regulated stem cells and organ development. Wnt factors play a crucial role in stem cells development and induced signaling pathway in stem cells. Wnt signals classified as a canonical and non-canonical pathway. Wnt factors regulated both pathways for stem cells self-renewal, proliferation, and differentiation. Wnt canonical pathway induced by Wnt1, Wnt2, Wnt2b, Wnt3, Wnt3a, Wnt4, Wnt5a, Wnt6, Wnt7a, Wnt7b, Wnt8a, Wnt8b, Wnt10a and Wnt10b. Wnt non-canonical pathway regulated by Wnt factors such as Wnt1, Wnt4, Wnt5a, Wnt5b, Wnt6, Wnt7a, Wnt7b, Wnt9a, Wnt9b, Wnt10a, Wnt10b, Wnt11, and Wnt16. These all factors have a specialized role in stem cells and development. These factors also regulated wound healing mechanism and variety of organ regeneration. Finally, abnormality or aberrant expression of Wnt signals regulated a variety of cancers.
| Keywords: |
Stem cells, Wnt factors, Wound healing and regeneration, Abnormality, Cancer
INTRODUCTION: More than 31 years ago, int-1 was identified as a proto-ongogene activated by mouse mammary tumor virus, and later it was renamed as Wnt1 gene 1. Further development, 19 Wnt genes were discovered in our animal kingdom from hydra to human. These Wnt proteins are induced by three different pathways such as Wnt/β-catenin, Planer cell polarity (PCP) and Wnt/Ca2+ pathway. Wnt signaling pathways were mainly involved in stem cells development and carcinogenesis. In stem cells development, Wnt proteins have a crucial impact in the embryogenesis, adult tissue formation and regeneration.
During development, Wnt molecules involved in many specifications, such as germ layer formation, body axis formation and patterning, cell fate specification, limb and facial development, adult organ development and finally regeneration 2.
Wnt Genes and Pathways: Wnt genes were regulated that specific Wnt signaling pathways and 19 Wnt genes were involved in the canonical and non-canonical pathway. In canonical pathway, Wnt/β-catenin signal was induced by Wnt1, Wnt2, Wnt2b, Wnt3, Wnt3a, Wnt4, Wnt5a, Wnt6, Wnt7a, Wnt7b, Wnt8a, Wnt8b, Wnt10a and Wnt10b 3.
Non-canonical pathway, Wnt/Planar cell polarity pathway was regulated by Wnt3a, Wnt4, Wnt5b, Wnt7a, Wnt7b, Wnt9a, Wnt9b, and Wnt11. Wnt/Ca2+ signaling pathways was also stimulated by Wnt1, Wnt4, Wnt5a, Wnt5b, Wnt6, Wnt7a, Wnt9a, Wnt9b, Wnt10a, Wnt10b and Wnt16. Here, Wnt4, Wnt7a genes regulated both of canonical and non-canonical pathways. Finally, Wnt5b, Wnt9a, Wnt9b, Wnt11, and Wnt16 genes were stimulated only Wnt non-canonical signaling pathways 4, 5.
Wnt Genes in Stem Cells and Development: In stem cells signaling, Wnt signals play a major crucial role in stem cells development and regeneration. Wnt signals expression was disco-vered from zygote to mature adult cells and also Wnt signals involved in the wound healing process and repair mechanism Table 1.
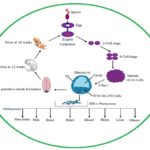
Embryonic Development: Wnt genes were involved embryonic development in a variety of organism, both of vertebrates and invertebrates Fig. 1. Wnt gene was first identified in Drosophila, and it was involved in segment polarity. Many Wnt genes were involved in the development process such as germ layer formation, cell fate determination, ectoderm, and mesoderm differentiation, limb and facial development, fetus formation, nervous system and spinal cord development 2, 6.
TABLE 1: WNT GENES IN STEM CELLS AND DEVELOPMENT
| Wnt genes | Pathway | Function in organ developments | References |
| Wnt1 | Wnt/β-catenin and Wnt/Ca2+ | Central nervous system (CNS) development, cell fate, and embryonic patterning, endothelial progenitor cells, neural crest progenitors, liver regeneration, epidermal stem cells differentiation, human embryonic stem cells self-renewal and proliferation. | 12, 15, 19, 21-25 |
| Wnt2 | Wnt/β-catenin | Limb and facial development, otic vesicle, heart, lung and body wall development, foregut endoderm development, hematopoietic, endothelial and cardiac lineages. | 12, 13, 26 |
| Wnt2B | Wnt/β-catenin | Branchial arch, CNS development, otic vesicle, eye, lung and genitourinary, liver formation, retinal cell differentiation, lung progenitor in foregut and hepatopancreatic specification. | 12, 27-30 |
| Wnt3 | Wnt/β-catenin | Ectoderm, facial and CNS development, heart and lung, head regeneration, hematopoietic differentiation. | 12, 31, 32 |
| Wnt3A | Wnt/β-catenin and planar cell polarity (PCP) | CNS development, digestive tract and tail formation, osteoblasts and fate differentiation, mesoderm formation and cardiomyogenesis, mesenchymal stem cells and hematopoietic, epidermal self-renewal and progenitors differentiation. | 12, 33-36 |
| Wnt4 | Wnt/β-catenin, PCP and Wnt/Ca2+ | Limb, facial and CNS development, otic vesicle, digestive tract, genitourinary and ectoderm, muscle regeneration and osteogenesis, hematolymphopoiesis, skeletogenesis, ovarian follicle development. | 12, 16, 20, 37, 38 |
| Wnt5A | Wnt/β-catenin and Wnt/Ca2+ | Limb, facial and CNS development, lung, digestive tract, genitourinary, real line and head mesenchymal, osteogenic differentiation and bone formation, cardiogenesis and spermatogenesis, endothelial differentiation and regeneration. | 12, 39-42 |
| Wnt5B | PCP and Wnt/Ca2+ | Limb, facial and CNS development, otic vesicle, eye, digestive tract, genitourinary and tail, mesenchymal cell aggregation and chondrocyte differentiation. | 12, 43 |
| Wnt6 | Wnt/β-catenin and Wnt/Ca2+ | Ectoderm, facial and CNS development, otic vesicle, genitourinary and real line, epidermis and epithelial development, extraembryonic endoderm formation, osteoblastogenesis, stromal cell proliferation | 12, 44-47 |
| Wnt7A | Wnt/β-catenin, PCP and Wnt/Ca2+ | Ectoderm, facial and CNS development, otic vesicle, eye and digestive tract, satellite stem cells expansion | 12, 17 |
| Wnt7B | Wnt/β-catenin, PCP | Ectoderm, facial and CNS development, otic vesicle, eye, lung, and digestive tract, hair follicle stem cells regulation and homeostasis, replication of epithelium and mesenchyme | 12, 14, 48 |
| Wnt8A | Wnt/β-catenin | Otic vesicle and eye development, axis and mesoderm development, | 12, 49, 50 |
| Wnt8B | Wnt/β-catenin | CNS development, retinal progenitor formation | 12, 51 |
| Wnt9A | PCP and Wnt/Ca2+ | CNS and otic vesicle development, palate morphogenesis, early patterning of oral-pharyngeal ectoderm and mesendoderm, hepatic epithelial morphogenesis | 12, 52-54 |
| Wnt9B | PCP and Wnt/Ca2+ | Facial, genitourinary and surface ectoderm, early patterning of oral-pharyngeal ectoderm and mesendoderm, heart, and pectoral fin bud morphogenesis | 12, 53, 55 |
| Wnt10A | Wnt/β-catenin and Wnt/Ca2+ | Ectoderm, branchial arch and heart development, osteoblastogenesis, odontoblast differentiation, and tooth morphogenesis, apical ectodermal ridge and limb development, tooth, face, and skin development | 12, 56-59 |
| Wnt10B | Wnt/β-catenin, Wnt/Ca2+ | Ectoderm, facial and oral line, stimulates osteoblastogenesis, development of limbs, tooth, face and skin | 12, 56, 59 |
| Wnt11 | PCP | Limb, facial, CNS and PNS development, otic vesicle, heart, oral line, tail and body wall | 12 |
| Wnt16 | Wnt/Ca2+ | Head mesenchymal and CNS development, otic vesicle, heart, digestive tract and tail | 12 |
FIG. 1: ORGAN DEVELOPMENT CYCLE FROM ZYGOTE TO ADULT HUMAN
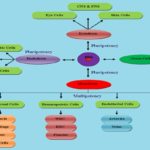
Wnt Genes in Axis Patterning and Cell Fate Determination: In embryonic development, body axis formation is critical steps and different axis including anteroposterior axis, dorsoventral axis, and right-left axis. Wnt genes were involved in the formation of the anteroposterior axis and dorsoventral axis 7. Cell fate determination or cell differentiation, undifferentiated cells were specialized to mature null potent cells. Wnt1, Wnt3a, Wnt6, Wnt8a, Wnt9a, and Wnt9b genes were regulated endoderm and mesoderm differentiation 8. Wnt1 gene was antagonized neural differentiation and also an important regulator in self-renewal of neural stem cells. Other Wnt genes were involved in sex determination, neural crest cell differentiation, gut tissue specification, germ cell determination, hair follicle development, lung, nephron and ovary development Fig. 2 9.
FIG. 2: SCHEMATIC REPRESENTATION OF STEM CELLS DIFFERENTIATION BASED ON THE INVOLVEMENT OF WNT SIGNALING PATHWAY
Cell Proliferation and Pluripotency: Undifferentiated cells were proliferated and migrated by Wnt signaling pathway. Wnt signals were induced differentiated and proliferated pluripotent stem cells to mesoderm and endoderm then proliferated to adult mature tissues type. Wnt3a was stimulated the proliferation of hematopoietic stem cells. Expression of several Wnt genes and Wnt pathway molecules in the blastocyst indicated the self-renewal and maintenance of pluripotency in the early embryo. Wnt3a was induced up-regulation of Wnt canonical pathway, and it was maintained self-renewal of human and mouse embryonic stem cells 10, 11.
Neuronal Development and Bone Formation: Wnt1, Wnt2b, Wnt3, Wnt3a, Wnt4, Wnt5a, Wnt5b, Wnt6, Wnt7a, Wnt7b, Wnt8b, Wnt9a, and Wnt16 proteins were involved in the formation of the central nervous system (CNS) by differentiation of mesoderm. Wnt11 was also stimulated both of CNS and peripheral nervous system (PNS) development. Wnt3a, Wnt4, Wnt5a, Wnt6, Wnt10a, and Wnt10b were induced in the development of bone formation 12.
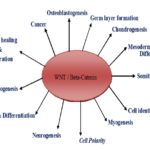
Wnt Molecules in Organ Development and Regeneration: In organ development, hemato-poiesis or blood formation was done by stimulation of Wnt2, Wnt3, Wnt3a, and Wnt4. Development of cardiogenesis was stimulated by Wnt2, Wnt3, Wnt3a, Wnt5a, Wnt10a, Wnt11 and Wnt16 12-14. Wnt2, Wnt3, and Wnt5a were involved in the development of lung. Wnt2b, Wnt5b, Wnt7a, Wnt7b, and Wnt8 were induced in the development of eye formation. Wnt3a, Wnt4, Wnt5a, Wnt5b, and Wnt7b were stimulated digestive tract formation Fig. 3. Formation of the genitourinary system was induced by Wnt2b, Wnt4, Wnt5a, Wnt5b and Wnt6 12.
Wnt1 was involved the most important function of human embryonic stem cells self-renewal and proliferation 15. Wnt2, Wnt3, Wnt4, Wnt5a, Wnt5b, and Wnt11 were induced limb and facial development from germ layer differentiation 12. Wnt 3a was also involved in epidermal and progenitor self-renewal and proliferation. Wnt4 especially stimulated ovarian follicle development, and over-expression was induced ovarian carcinogenesis 16.
FIG. 3: FUNCTION OF WNT SIGNALING PATHWAY IN STEM CELLS DEVELOPMENT AND CARCINOGENESIS
Wnt5a also has specialized role information of spermatogenesis and Wnt7a involved expansion of satellite stem cells 17. Wnt8b and Wnt9b were stimulated the development of retinal progenitor and bud morphogenesis 12, 18. In regeneration, Wnt signaling factors were involved in wound healing and regeneration. Different Wnt factors were stimulated different organ regeneration, and Wnt has a specialized role in wound healing. Wnt1 involved in liver regeneration and Wnt3 in head regeneration 19. Wnt4 and Wnt5a were induced muscle and endothelial regeneration and differentiation 12, 20.
CONCLUSION: Before 31 years ago, no one knows how Wnt signals were involved in stem cells and development. Nowadays, Wnt signaling pathways have a crucial role in stem cells development and carcinogenesis. Overexpression and mutations of Wnt factors were stimulated many varieties of cancer. In cancer treatment, few drugs only available against abnormality of Wnt signaling. In stem cell biology, Wnt signals play a crucial impact in stem cells maintenance, self-renewal proliferated and developed specialized organ. Wnt signals were also regulated wound healing and regeneration. Where and how Wnt signals are activated and which is the evolutionary origin. These all like unknown and well developed new specialized drug will identify when these are well known. Future work, identify and develop a new drug against cancer from an abnormality of Wnt signals.
ACKNOWLEDGEMENT: The authors are sincere thanks to Dr. A. Nagappan, Principal and Dr. C. K. Hindumathy, Dean- Biosciences and Dr. S. Anandakumar, Associate Professor, Vinayaka Mission’s Kirupananda Variyar Engineering College, Tamil Nadu for their support and carry out this work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Nusse R and Varmus HE: Many tumors induced by the mouse mammary tumor virus contain a provirus integrated into the same region of the host genome. Cell 1982; 31: 99-109.
- Nusse R and Varmus HE. Wnt genes. Cell 1992; 69: 1073-87.
- Katoh M andKatoh M: Comparative genomics on Wnt8a and Wnt8b genes. Int J Oncol 2005; 26: 1129-33.
- Quinlan R, Graf M, Mason I, Lumsden A andKiecker C: Complex and dynamic patterns of Wnt pathway gene expression in the developing chick forebrain. Neural Dev 2009; 4: 35.
- Pongracz JE and Stockley RA: Wnt signaling in lung development and diseases Respir Res2006; 7 : 15
- Nusse R:Wnt signaling and stem cell control. Cell Res 2008; 18 : 523-7.
- van Amerongen, R and Nusse R: Towards an integrated view of Wnt signaling in development.Development 2009; 136: 3205–14.
- Bakre MM,Hoi A, Mong JC, Koh YY, Wong KY and Stanton LW: Generation of multipotential mesendodermal progenitors from mouse embryonic stem cells via sustained Wnt pathway activation. J Biol Chem 2007; 282: 31703-12.
- Reya T andClevers H: Wnt signaling in stem cells and cancer. Nature 2005; 434: 843-50.
- Dravid G, Ye Z, Hammond H, Chen G, Pyle A and Donovan P: Defining the role of Wnt/β-Catenin signaling in the survival, proliferation, and self-renewal of human embryonic stem cells. Stem Cells 2005; 23: 1489-1501.
- Sokol SY: Maintaining embryonic stem cell pluripotency with Wnt signaling. Development2011; 138: 4341-50.
- Summerhurst K, Stark M, Sharpe J, Davidson D andMurphy P: 3D representation of Wnt & Frizzled gene expression patterns in the mouse embryo at embryonic day 11.5 (Ts19). Gene Expr Patterns 2008; 8: 331-48.
- Wang H,Gilner JB, Bautch VL, Wang DZ, Wainwright BJ and Kirby SL: Wnt2 coordinates the commitment of mesoderm to hematopoietic, endothelial, and cardiac line-ages in embryoid bodies. J Biol Chem 2007; 282: 782-91.
- Rajagopal J,Carroll TJ, Guseh JS, Bores SA, Blank LJ and Anderson WJ: Wnt7b stimulates embryonic lung growth by coordinately increasing the replication of epithelium and mesenchyme. Development 2008; 135: 1625-34.
- Cai L, Ye Z, Zhou BY, Mali P, Zhou C and Cheng L: Promoting human embryonic stem cell renewal or differentiation by modulating Wnt signal and culture conditions. Cell Res 2007; 17: 62-72.
- Boyer A,Lapointe E, Zheng X, Cowan RG, Li H and Quirk SM: WNT4 is required for normal ovarian follicle development and female fertility. FASEB J 2010; 24: 3010-25.
- Le Grand F,Jones AE, Seale V, Scimè A and Rudnicki MA: Wnt7a activates the planar cell polarity pathway to drive the symmetric expansion of satellite stem cells. Cell Stem Cell 2009; 4: 535-47.
- Jezewski PA,Fang PK, Payne-Ferreira TL and Yelick PC: Zebrafish Wnt9b synteny and expression during first and second arch, heart, and pectoral fin bud morphogenesis. Zebrafish 2008; 5: 169-77.
- Williams JM, Oh SH, Jorgensen M, Steiger N, Darwiche H andShupe T: The role of the Wnt family of secreted proteins in rat oval stem cell-based liver regeneration: Wnt1 drive Am J Pathol 2010; 176: 2732-42.
- Gozo MC,Aspuria PJ, Cheon DJ, Walts AE, Berel D and Miura N: Foxc2 induces Wnt4 and Bmp4 expression during muscle regeneration and osteogenesis. Cell Death Differ 2013; 20: 1031-42.
- Yang J, Brown A, Ellisor D, Paul E, Hagan N and Zervas M: Dynamic temporal requirement of Wnt1 in midbrain dopamine neuron development. Development 2013; 140: 1342-52.
- Gherghe CM, Duan J, Gong J, Rojas M, Klauber-Demore N and Majesky M: Wnt1 is a proangiogenic molecule, enhances human endothelial progenitor function, and increases blood flow to ischemic limbs in an HGF-dependent FASEB J2011; 25: 1836-43.
- Glejzer A, Laudet E, Leprince P, Hennuy B, Poulet C andShakhova O: Wnt1 and BMP2: two factors recruiting multipotent neural crest progenitors isolated from adult bone marrow. Cell Mol Life Sci 2011; 68: 2101-14.
- Li YC, Wu JJ, Bi JJ, Yang T, Yang GH and Wang BQ: Influence of Wnt-1 recombinant adenovirus on the differentiation of human epidermal stem cells. Zhonghua Shao Shang Za Zhi2008; 24: 187-90.
- Weisel KC, Kopp HG, Moore MA, Studer L and Barberi T: Wnt1 overexpression leads to enforced cardiomyogenesis and inhibition of hematopoiesis in murine embryonic stem cells. Stem Cells Dev 2010; 19: 745-51.
- Poulain M andOber EA: The interplay between Wnt2 and Wnt2bb controls multiple steps of early foregut-derived organ development. Development 2011; 138: 3557-68.
- Ober EA,Verkade H, Field HA and Stainier DY: Mesodermal Wnt2b signaling positively regulates liver specification. Nature 2006; 442: 688-91.
- Kubo F,Takeichi M and Nakagawa S: Wnt2b controls retinal cell differentiation at the ciliary marginal zone. Development 2003; 130: 587-98.
- Goss AM,Tian Y, Tsukiyama T, Cohen ED, Zhou D and Lu MM: Wnt2/2b and beta-catenin signaling are necessary and sufficient to specify lung progenitors in the foregut. Dev Cell 2009; 17: 290-8.
- Lancman JJ, Zvenigorodsky N, Gates KP, Zhang D, Solomon K and Humphrey RK: Specification of hepato-pancreas progenitors in zebrafish by hnf1ba and wnt2bb. Development 2013; 140: 2669-79.
- Chera S, Ghila L, Dobretz K, Wenger Y, Bauer C and Buzgariu W: Apoptotic cells provide an unexpected source of Wnt3 signaling to drive hydra head regeneration. Dev Cell 2009; 17: 279-89.
- Lako M,Lindsay S, Lincoln J and Cairns PM, Armstrong L and Hole N: Characterisation of Wnt gene expression during the differentiation of murine embryonic stem cells in-vitro: the role of Wnt3 in enhancing hematopoietic Mech Dev 2001; 103: 49-59.
- Qiu W,Chen L and Kassem M: Activation of non-canonical Wnt/JNK pathway by Wnt3a is associated with differentiation fate determination of human bone marrow stromal (mesenchymal) stem cells. Biochem Biophys Res Commun 2011; 413: 98-104.
- Tran TH, Wang X, Browne C, Zhang Y, Schinke M and Izumo S: Wnt3a-induced mesoderm formation and cardiomyogenesis in human embryonic stem cells. Stem Cells 2009; 27: 1869-78.
- Jia L,Zhou J, Peng S, Li J, Cao Y and Duan E: Effects of Wnt3a on the proliferation and differentiation of human epidermal stem cells. Biochem Biophys Res Commun 2008; 368: 483-8.
- Shang YC,Wang SH, Xiong F, Zhao CP, Peng FN and Feng SW: Wnt3a signaling promotes proliferation, myogenic differentiation, and migration of rat bone marrow mesenchymal stem cells. Acta Pharmacol Sin 2007; 28: 1761-74.
- Heinonen KM,Vanegas JR, Lew D, Krosl J and Perreault C: Wnt4 enhances murine hematopoietic progenitor cell expansion through a planar cell polarity-like pathway. PLoS One 2011; 6: e19279.
- Lee HH andBehringer RR: Conditional expression of Wnt4 during chondrogenesis leads to dwarfism in mice. PLoS One 2007; 2: e450.
- Brun J, Fromigue O, Dieudonne FX, Marty C, Chen J and Dahan J: The LIM-only protein FHL2 controls mesenchymal cell osteogenic differentiation and bone formation through Wnt5a and Wnt10b. Bone 2013; 53: 6-12.
- Cohen ED, Miller MF, Wang Z, Moon RT, Morrisey EE: Wnt5a and Wnt11 are essential for second heart field progenitor development. Development 2012; 139: 1931-40.
- Yeh JR,Zhang X and Nagano MC: Wnt5a is a cell-extrinsic factor that supports self-renewal of mouse spermatogonial stem cells. J Cell Sci 2011; 124: 2357-66.
- Yang DH, Yoon JY, Lee SH, Bryja V, Andersson ER, Arenas E: Wnt5a is required for endothelial differentiation of embryonic stem cells and vascularization via pathways involving both Wnt/beta-catenin and protein kinase C alpha. Circ Res 2009; 104: 372-9.
- Bradley EW,Drissi MH: Wnt5b regulates mesenchymal cell aggregation and chondrocyte differentiation through the planar cell polarity pathway. J Cell Physiol 2011; 226: 1683-93.
- Lavery DL, Davenport IR, Turnbull YD, Wheeler GN and Hoppler S: Wnt6 expression in epidermis and epithelial tissues during Xenopus organogenesis. Dev Dyn 2008; 237: 768-79.
- Hwang JT and Kelly GM: GATA6 and FOXA2 regulate Wnt6 expression during extraembryonic endoderm formation. Stem Cells Dev 2012; 21: 3220-32.
- Cawthorn WP, Bree AJ, Yao Y, Du B, Hemati N and Martinez-Santibañez G: Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a β-catenin-dependent mechanism. Bone2012; 50: 477-89.
- Wang Q,Lu J, Zhang S, Wang S, Wang W, Wang B, et al. Wnt6 Is Essential for Stromal Cell Proliferation During Decidualization in mice. Biol Reprod 2013; 88: 5.
- Kandyba E andKobielak K: Wnt7b is an important intrinsic regulator of hair follicle stem cell homeostasis and hair follicle cycling. Stem Cells 2013.
- Narayanan A, Thompson SA, Lee JJ and Lekven AC: A transgenic Wnt8a: PAC reporter reveals biphasic regulation of vertebrate mesoderm development. Dev Dyn 2011; 240: 898-907.
- Wylie AD,Fleming JA, Whitener AE and Lekven AC: Post-transcriptional regulation of wnt8a is essential to zebrafish axis development. Dev Biol 2014; 386: 53-63.
- Liu W: Focus on molecules: Wnt8b: a suppressor of the early eye and retinal progenitor formation. Exp Eye Res2012; 101: 113-4.
- Dougherty M, Kamel G, Grimaldi M, Gfrerer L, Shubinets V and Ethier R: Distinct requirements for wnt9a and irf6 in extension and integration mechanisms during zebrafish palate morphogenesis. Development 2013; 140: 76-81.
- Cox AA, Jezewski PA, Fang PK and Payne-Ferreira TL: Zebrafish Wnt 9a, 9b paralog comparisons suggest ancestral roles for Wnt9 in neural, oral-pharyngeal ectoderm and mesendoderm. Gene Expr Patterns 2010; 10: 251-8.
- Matsumoto K, Miki R, Nakayama M, Tatsumi N and Yokouchi Y: Wnt9a secreted from the walls of hepatic sinusoids is essential for morphogenesis, proliferation, and glycogen accumulation of chick hepatic epithelium. Dev Biol 2008; 319: 234-47.
- Jezewski PA, Fang PK, Payne-Ferreira TL and Yelick PC: Zebrafish Wnt9b synteny and expression during first and second arch, heart, and pectoral fin bud morphogenesis. Zebrafish 2008; 5: 169-77.
- Cawthorn WP, Bree AJ, Yao Y, Du B, Hemati N and Martinez-Santibañez G: Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a β-catenin-dependent mechanism. Bone 2012; 50: 477-89.
- Yamashiro T, Zheng L, Shitaku Y, Saito M, Tsubakimoto T and Takada K: Wnt10a regulates dentin sialophosphoprotein mRNA expression and possibly links odontoblast differen-tiation and tooth morphogenesis. Differentiation 2007; 75: 452-62.
- Narita T, Sasaoka S, Udagawa K, Ohyama T, Wada N and Nishimatsu S: Wnt10a is involved in AER formation during chick limb development. Dev Dyn 2005; 233: 282-7.
- Wang J andShackleford GM: Murine Wnt10a and Wnt10b: cloning and expression in developing limbs face and skin of embryos and in adults. Oncogene 1996; 13: 1537-44.
How to cite this article:
Prakash R: Regulation of Wnt genes in stem cells development and organogenesis. Int J Pharmacognosy 2014; 1(6): 366-72. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(6).366-72.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
366-372
688
1388
English
IJP
R. Prakash
Department of Biotechnology, VMKV Engineering College, Vinayaka Missions University, Salem, Tamil Nadu, India
yokaprakash0007@gmail.com
20 April 2014
23 May 2014
28 May 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.1(6).366-372
01 June 2014