PSORALEN: A PROMISING BOON IN TOPICAL MANIFESTATIONS
HTML Full TextPSORALEN: A PROMISING BOON IN TOPICAL MANIFESTATIONS
S. Kumar and R. Rao *
Department of Pharmaceutical Sciences, Guru Jambeshwar University Science and Technology, Hisar - 125001, Haryana, India.
ABSTRACT: Extensive studies have been carried out in the last decade to assess the pharmaceutical potential and screening of the phytochemical constituent, psoralen. This component is found in many plants species and has gained attention nowadays because of its antibacterial, antifungal, antiviral, and anti-inflammatory properties. Psoralen is an active component of Babchi oil found in Psoralea corylifolia (P. crylifolia) belonging to family Leguminosae. This review presents an overview of the current status and knowledge on overview, chemistry, synthesis, analysis, irritation, and toxicity of psoralen. Also, formulations of psoralen are also presented here.
| Keywords: |
Psoralen, Babchi oil, Essential oil, Drug delivery systems
INTRODUCTION: Plants have been the basis of many traditional medicines throughout the world for thousands of years and continue to be a source of new remedies to mankind. Since, the beginning of human civilization, they have been one of the vital sources of medicines. The recent renascence, of plant remedies involving EOs from several factors, such as the effectiveness of plant medicines and lesser side effects in comparison to modern medicines 1. Essential oils and their components are composed of lipophilic and highly volatile secondary plant metabolites, possessing molecular weight less than 300. The large bioactivity of EOs has been ascertained in several studies, including antibacterial, antiviral, anti-inflammatory, anti-fungal, antimutagenic, anticarcinogenic, and antioxidant as well as other miscellaneous activities.
The extensive list of biological activities of EOs can be linked to the complexity and variability of their chemical composition 2. Plants produce furanocoumarins mainly in reverberation to abiotic or biotic insults such as irradiation, infections, wounding and exposure to ozone, pollutants, and other harsh environmental conditions. Furocoumarins (psoralens and isopsoralens) are natural bioactive and available especially in the Umbelliferae, Rutaceae and Leguminosae 3. Psoralen (also called psoralene) is the parent compound belonging to the family of natural products known as furocoumarins. It is structurally related to coumarin by the addition of a fused furan ring and may be considered as a derivative of umbelliferone.
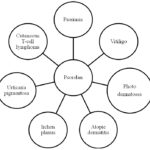
A furocoumarin compound, Psoralen is the pharmacologically active moiety of the several herbaceous plants belonging to the family, Leguminosae, like Psoralea corylifolia L. It is an important medicinal plant found in the tropical and subtropical regions of the world. It synthesizes diverse phenylpropanoids such as furanocoumarins, isoflavonoids, etc. 4 It is having numerous applications in the treatment of skin diseases such as psoriasis, vitiligo and chronic graft-versus-host, and is also known to possess anticancer, cytotoxic, antibacterial, cardiac, diaphoretic, diuretic, stimulant, aphrodisiac and tonic effects. A number of formulations rich in psoralen has been clinically used 5. Further, psoralen is commercially important for having a broad range of pharmacological activities such as photosensitizing, photobiological and phototherapeutic properties 6. Use of psoralen in various diseases is shown in Fig. 1.
FIG. 1: PSORALEN IN VARIOUS MANIFESTATIONS 7
Despite, its relevance for consumers, there is limited information available addressing psoralen. So, this review is aimed at assembling various scattered data on chemical properties, analytical techniques, various formulations, toxicity and irritation studies of this bioactive component.
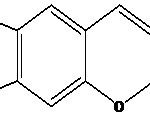
Chemistry of Psoralen: Bioactive molecule, psoralen belongs to naturally anticipating tricyclic furocoumarins. Its derivative, chiefly of plant origin, is 8-methoxypsoralen (xanthotoxin, methoxsalen,) is wildly exploited in photo-chemotherapy. Also, it can be assessable as a synthetic compound. 4, 5, 8-trimethylpsoralen (trioxsalen), a synthetic active moiety is used in vitiligo disease, was found less phototoxic upon oral administration and is commonly used for the treatment of vitiligo. Upcoming psoralens include 5-methoxy psoralen (bergapten), 3-carbethoxy-psoralen and angelicin 8, 9. The photobiological activity of angelicin is found low 10. The introduction of one or more methyl groups results in augmented photobinding capacity of angelicin towards DNA. 4,4,6-methyl angelicin is the most potential of the new angelicins.
FIG. 2: CHEMICAL STRUCTURE OF PSORALEN
Synthesis of Psoralen: It is very difficult to synthesize psoralen since umbelliferone leads to substitution at position 8 rather than position 6. Benzofuran reacts mainly with furan ring instead of a benzene ring.
However, the 7-hydroxy derivative of 2, 3-dihydrobenzofuran (also called coumaran) does undergo substitution at the desired 6-position allowing the resulting synthesis of the coumarin system via a Gattermann-Koch reaction followed by a Perkin condensation using acetic anhydride. The synthesis gets completed by dehydrogenation of the five-membered ring producing the furan ring 6.
Biosynthesis: Psoralen originates from coumarins by the shikimate pathway. The aromatic ring in 6 is activated at positions ortho to the hydroxyl group and is alkylated by 5, an alkylating agent. The dimethylallyl group in 7 then undergoes cyclization with the phenol group resulting in 8. A cytochrome P450-dependent monooxygenase17 (psoralen 5-monooxygenase), and cofactors (NADPH) and molecular oxygen catalyse this transformation 6. A second P-450-dependent monooxygenase enzyme (psoralen synthase) then cleaves off 10 (in the form of 11) from 8 to give 1. This pathway does not involve any hydroxylated intermediate, and cleavage is postulated to be initiated by a radical reaction 6.
Mechanism of Action: The exact mechanism for cutaneous photosensitivity reactions by psoralen is not precisely known. Mode of action of psoralens occurs but at several levels simultaneously including cellular RNA proteins, cell membrane lipids, DNA, mitochondria, etc. Psoralen induced skin photosensitization involves two distinct types of reactions when psoralen treated skin is exposed to 320 – 400 nm radiation in the skin of psoriatic patients and normal skin 7.
- Type I is an anoxic reaction which does not require oxygen and the site of cellular damage, is primarily in the DNA of cell nuclei.
- Type II is a sensitized reaction which depends on oxygen and involves the formation of reactive oxygen species such as O2, O-2, and free radicals.
In both of the above mentioned reactions, the reactive form of psoralen is its triplet state. The sites of reaction are:
- The cell membrane of the epidermal, dermal and endothelial cells.
- Cell nuclei (DNA and chromatin).
- Membrane lipids involving a photodynamic reaction and the production of reactive oxygen species.
- Cytoplasmic constituents (enzymes, RNA, lysosomes, etc.).
The major photochemical reaction is Type I reaction contributing to the cellular damage is the formation of monofunctional and bifunctional psoralen adducts with pyrimidine bases in DNA. The formation of these adducts in DNA results in psoriasis inhibition of DNA synthesis. As a result, cell proliferation is also inhibited in patients who receive repeated Psoralen Ultraviolet A (PUVA) treatment. PUVA leads to photoconjugation of psoralens to DNA and subsequent mitosis suppression, DNA synthesis, and cell proliferation. Possibly, PUVA may affect specific cells like lymphocytes or polymorphonuclear leucocytes which are included in the pathophysiology of psoriasis 11. Immunological alteration acts as an essential component of the therapy. A decrease in the circulating T-lymphocytes following PUVA treatment has been observed 12.
The mechanism by which PUVA induces repigmentation in the skin has been speculated as follows 11.
- By inducing melanocytes hypertrophy and an enhanced arborization of their dendrites.
- By enhancing the number of functional melanocytes as a result of mitosis or by activation of dormant melanocytes in the appendages and epidermis.
- By stimulating the activity of tyrosinase.
- By increasing the development and melanization of melanosomes and augmenting the transfer of melanosomes to keratinocytes.
- By generating a suppressor cell population which suppresses the melanocyte destruction stimulus.
- By increasing the migration of activated melanocytes from skin appendages 13.
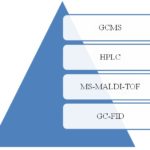
Analysis: Regarding analysis, numerous analytical methods have been devised to determine psoralen.
High-Performance Liquid Chromatography (HPLC): A sample preparation and analysis method using High - Performance Liquid Chromatography with UV detection (HPLC-UV) were developed for analysis of bergapten, 5-[3-(4, 5-dihydro-5, 5-dimethyl-4-oxo-2- furanyl)-butoxy]-7H-furo[3-2-g][1]benzopyran-7-one and psoralen in tablets and capsules employed in Brazil. The accuracy, linearity, the inter- and intra-day precision of the procedure was checked. Analytical curves for furanocoumarins were found linear in the range of 1-50 mg/ml. The percent coefficient of variation for the analysis of the furanocoumarins was observed under 5%, and the recoveries of the furanocoumarins were 97.3-99.5%. Gas Chromatography (GC) was used for inter-equipment study 14.
FIG. 3: ANALYTICAL TECHNIQUES USED FOR PSORALEN DETECTION
For the analysis of psoralen in solid lipid nanoparticles (SLNs) and marketed formulations, a quantitative High - Performance Thin - Layer Chromatography (HPTLC) method using precoated silica gel-60 has been developed and validated. Densitometric analysis was carried out at 250 nm using absorbance mode. At Rf 0.32 ± 0.02 compact bands of psoralen were obtained. The method was validated for precision, linearity, robustness, specificity, sensitivity, and recovery. Linearity (r2 = 0.995), limit of quantification (18.1 ng band−1), limit of detection (8.0 ng band−1), precision (≤0.74) and recovery (98.06–99.64%) were found satisfactory. The developed method for quantification of psoralen in solid lipid nano-particles and marketed formulations is established by statistical analysis selective and reproducible 15.
Gas Chromatography-FID-Quantification: P. corylifolia oil samples, consisting of psoralen, bakuchiol, and β-caryophyllene, were investigated using GC–MS (Shimadzu QP-2010) fitted with FID and a QP-5000 (Quadrapole) mass spectro-photometer. A fused silica capillary column DB5 (30 m × 0.25 mm, with a film thickness of 0.25 m) was used. Analytical grade Psoralen (Sigma–Aldrich), -caryophyllene (Sigma–Aldrich) and bakuchiol (Sigma–Aldrich) were used for quantifying P. corylifolia oil constituents. Every standard was used to prepare the calibration curve using FID response data. Response factors (RF) and regression coefficients were imposed independently for linearity. Equation = DR/C was used for calculation of Response factor (RF) where C is the concentration of the analyte and DR was the detector response in peak area (PA). The chromatograms of each of the essential oil samples including replicates and control were compared to the standard chromatograms. Target peaks were confirmed from retention time. Each chemical constituent percentage in the essential oil was confirmed using integrated peaks.
The RF of the target chemical constituent was used to determine the percentage of the constituent in each essential oil sample by equation (PA/RF/C) × 100 = % (peak area/response factor/concentration). Bhat et al., investigated the antiliorative response of some essential oil furanocoumarins and proteins from P. corylifolia against oxidative stress 16.
Formulations of Psoralen: Cassidy and its research group fabricated various triplex formation oligonucleotides (TFOs) conjugated with psoralen. The pyrimidine TFOs that have chimeric methylphosphonate bond form stable triplexes with envelope gene of HIV proviral DNA (enV-DNA). This chimeric skeleton was found well matched with the insertion of other modifications, like 5-propynyl-U or O-methyl ribose sugars, enhancing triplex stability. The efficacy of chimeric methylphosphonate TFOs for adhering to DNA, with their resistance to abasement by serum 3-exonucleases, indicated significant use in biological experiments 17.
Kim and its research group employ triplex-forming peptide nucleic acids (PNAs) and oligonucleotides (TFOs) as DNA-binding molecules to boost recombination and to carry out site-specific mutations at selected sites genome. To further evaluate the utility of PNAs, psoralen-dimeric bis-PNAs conjugates were examined to measure PNAs utility for site-specific gene modification. Fabricated PNAs were used in the formation of triplex-invasion complex in an episomal shuttle vector as well as to address site-specific photo adduct formation through the conjugated psoralen. This conjugate was observed to help photo adduct formation. The SV40-based shuttle vector assay was used to study the PNA-targeted photo adducts effects in a mammalian system. Upon conclusion of the results, the psoralen conjugates demonstrated the capability to mediate site-specific gene modification, leading as promising tools in gene-targeting 18.
To enhance the pharmacological potency of photopheresis and PUVA therapy, poly- (ethylene glycol) (PEG)-psoralen conjugates were fabricated to boost tumor targeting through increased retention and permeability effect. Peptide linkers were employed to achieve specific enzymatic cleavage through lysosomal proteases. The hydroxy group promotes to explore different schemes for PEG conjugation, and linkages with different stability such as urethane or ester prevailed. The new psoralen derivative was conjugated through covalent bond by PEG through four different linkages viz. ester linkage with a peptide spacer, direct ester bond, a carbamic linker with a peptide spacer and a carbamic linker. The stability profile of these conjugates was measured at different conditions. In-vitro investigation of the cytotoxic activity in the presence and absence of light was performed in cell lines (A375 and MCF-7 cells). Whereas, antiproliferative activity became evident upon UV irradiation. mPEG-NHCO-Gly-Phe-Leu-Gly-NHCOOMeP conjugate found to be great potential in tumor targeting if in-vivo studies to be carried out 19.
Fang and co-workers developed psoralen loaded solid lipid nanoparticles (SLN) consisting of Precirol ATO 5 as the solid core, whereas, psoralen loaded nanostructured lipid carriers (NLC) prepared by using squalene and Precirol, a liquid lipid along with above mentioned solid lipid. The comparative evaluation was carried out concerning mean particle size, polarity, viscosity, and differential scanning calorimetry (DSC) study to characterize the physicochemical properties of both SLN and NLC.
From DSC thermograms, incorporation of squalene into the solid lipid cores (of NLC), alter the crystalline lattice of the lipid cores leading to smaller particle sizes. NLC promotes permeation and psoralen delivery in a controlled manner in comparison to the conventional emulsion. Psoriasis-like skin obtained through repeated strippings in the dorsal skin of a nude mouse was taken as a permeation barrier. The results indicated that the encapsulation of psoralen in nanoparticulate systems could significantly overcome the permeation differentiation between hyperproliferative skin and normal concerning the free active moiety in control (aqueous) 20.
In the drug delivery systems, the use and biocompatible discovery polymers have become the focal point of research. In 2008, Gomes and co-workers fabricated the poly (DL-lactide-co-glycolide) (PLGA) microspheres encapsulating psoralen through solvent evaporation method. The prepared formulation was used in PUVA therapy (psoralen and UVA light (ultraviolet A, 320-400 nm) to treat psoriasis. The light microscopy was used in the evaluation of the formulation. The identification of microspheres having a non-fluorescent material identified using alteration in fixative agents or stains. These modifications improved the identification and preservation of microspheres in tissues in histological examinations indicated the promising protocol in pathological laboratories 21.
Patel and his research group formulated topical gel and cream containing psoralen to enhance drug transport through the skin. The oil in water creams of psoralen (0.05%) was designed using emulsifying agents such as Apifil and plurol strearique WL 1009 as well as aqueous cream (cream base materials). On the other hand, psoralen (0.05%) containing hydroalcoholic transparent gel was formulated using a gelling agent (hydroxypropyl cellulose). FTIR and DSC analytical techniques used for physicochemical compatibility estimation. Various parameters viz. viscosity, drug content uniformity, pH, limpidity and stability were measured. In addition to this, penetration enhancement of menthol (12.5%) was observed on the percutaneous flux of psoralen using excised epidermis of the rat as compared to gel and cream in the absence of menthol. The authors indicated the potential of penetration enhancer to minimize the lag time between drug application and UVA light 22. Rani and co-workers formulated psoralen loaded ethyl cellulose microsponges through quasi-emulsion solvent diffusion technique. The drug-loaded microsponges were incorporated into the gel base. The optimum stable formulation was selected by entrapment efficiency, physicochemical characteristics and drug permeation studies. In addition to reducing the skin irritation, psoralen microsponges delivered the fast recovery of normal complexation in comparison to conventional psoralen gel in anisole induced leucoderma mice model 23.
Luxmi and research group designed the psoralen gel using natural gums and polymers. The physicochemical compatibility of the formulation was elucidated by FTIR. The release pattern was measured and found to be best fitted to the Higuchi model. The potential drug incorporation was observed in a gel having xanthan gum and egg albumin. The antipsoriatic activity carried out through a mouse tail model indicating significant effectiveness of the formulation for topical delivery 24. In 2013, Kaur and co-workers designed psoralen transdermal spray using film forming agent (ethyl cellulose). The various parameters such as clarity of solution, pH, viscosity, spray pattern, dermal adhesion of film, area covered by each spray, flexibility of film, leakage from container, average weight of prepared spray formulation per spray, water washability of film, film formation time, drug content per spray, drug content uniformity, in-vitro drug transport, were calculated after a 10th, 15th, 30th and 40th spray.
The stability studies carried out as per ICH guidelines indicating the good stability profile. In addition to this, performed diffusion studies indicated the release profile of the drug and skin irritation study correlated the photosensitization property of the formulation. From the obtained results, the authors give an innovative approach for delivery of psoralen transdermally 25. Kumari and Pathak formulated composite ethosomes using the injection method. Psoralen was complexed with cavamax W7 through kneading technique. The evaluation was done by permeation characteristics. The permeation efficacy of the designed formulation was significantly more than standard ethosomal formulation even in the absence of permeation enhancers. This leads to clinical importance especially in psoriatic condition for topical delivery of drugs. Conclusively, cavamax W7 psoralen composite ethosomes found to be a potential carrier for topical delivery of psoralen by minimizing side effects of psoralen 26.
Kumar et al., designed a novel water-soluble cationic porphyrin–psoralen conjugate through Cu (I)-catalyzed azide-alkyne cycloaddition reaction. The designed conjugate of psoralen showed profound photocytotoxicity in the direction of A549 cancer cells in comparison to the potent photosensitizing and tumor-localizing agent, TMPyP. The authors suggested that porphyrin–psoralen conjugate must be a significant candidate for photo dynamic therapy 27. Zhang and co-workers formulated psoralen loaded ethosomes and liposomes through Touitou’s method and the conventional mechanical dispersion method. The designed formulations were compared by performing permeability and deposition study of the loaded psoralen. The quantity of psoralen loaded liposome that penetrated via skin enhanced upon a decrease in skin viability.
On the other hand, from both formulations, there was a reduction in psoralen deposition in the skin. Liposomal psoralen showed more effective delivery in human-immortalized epidermal cells; on the other hand, ethosomal psoralen delivery found more significant in human embryonic skin fibroblast cells. From in-vivo studies relating to drug deposition in the skin, ethosomes as a nanosystem, indicating a promising idea for enhancing targeting and permeability of psoralen in the skin through intracellular and intercellular transport pathways. Hence, the skin viability effects have a significant role in the evaluation of nanosystem-mediated drug permeation through skin 28. In the same year, by a similar group of Zhang designed ethosomal transdermal delivery system of psoralen to target deeper layers of skin by passing through stratum corneum.
The in-vitro skin permeation studies, the measured psoralen skin deposition, and transdermal flux variables pointed out the efficiency of ethosomes as compared the liposomes. The in-vivo studies carried out on the human embryonic skin fibroblasts suggested the significant biocompatibility due to the presence of phosphatidylcholine in ethosomes. Finally, these finding regarding ethosomes helps to signify the psoalen delivery in deep skin layers in psoriasis treatment 28. In the same year, to investigate the drug release of psoralen in the deep skin layers in rats, Zhang et al., modified the injection method to formulate ethosomes.
The prepared formulation was evaluated in-vitro and in-vivo in comparison with alcohol (ethanol) tincture. The in-vivo skin microdyliasis was performed and showed the area under the curve and peak concentration of psoralen about 2.34 and 3.37 fold higher, respectively, as compared to that of ethanol tincture. In addition to this, ethosomal percutaneous permeability was measured higher when applied to the abdomen in comparison when applied to scapulas or chest. The promising results relating to permeation and skin deposition of active molecule presented through ethosomes should be taken in to account in reduction of toxicity and enhancing the potency of the drug in long term treatment 29. Other carrier systems reported for psoralen encapsulation are mentioned in Table 1.
TABLE 1: LIST OF CARRIER SYSTEMS ENCAPSULATING PSORALEN
| S. no. | Carrier system | Method of preparation | Remarks | References |
| 1 | SLN | - | Method development by HPTLC | 15 |
| 2 | PEG-Psoralen Conjugates | Polymer conjugation | Assessed biological profile of conjugates | 19 |
| 3 | Conjugated Chimeric Triples forming Oligonucleotide | - | Enhancement in nuclease
A resistance of chimeric backbone |
17 |
| 4 | SLN and NLC | Hot homogenization and ultrasonication | Improved drug permeation for
psoriasis |
20 |
| 5 | PLGA microspheres | Solvent evaporation method | Identified drug-loaded-PLGA microspheres in rat skin. | 21 |
| 6 | Modified transport psoralen transdermal spray | - | Enhanced physical and chemical compatibility | 25 |
| 7 | Peptide nucleic
acids conjugates |
- | Site specific
Gene modification technique |
18 |
| 8 | Porphyrin–psoralen conjugates | A Cu(I)-catalyzed azide-alkyne cycloaddition reaction | Efficient DNA photocleavage activity | 27 |
| 9 | Ethosomal gel and solid complex gel | Injection method | More efficient ethosomal
gel |
26 |
| 10 | Antipsoriatic gel | - | Effective in psoriasis treatment | 24 |
| 11 | Topical gel and cream | - | Higher release profile in gel formulation | 22 |
| 12 | Microsponges | Quasi-emulsion solvent diffusion method | Effective delivery in induced leucoderma black mice | 23 |
| 13 | Ethosomes and
liposomes |
Touitou’s method and
Conventional mechanical dispersion method. |
Enhanced drug permeability and targeting in deep skin | 28 |
| 14 | Ethosomes | modified injection method | Enhanced permeation and skin deposition of the drug by microdialysis | 29 |
| 15 | Ethosomes and
liposomes |
Touitou’s method and ethanol injection method | Improved transdermal
delivery of drug |
30 |
Pharmacological Applications:
Insecticidal & Genotoxic Activity: Indiscriminate use of synthetic insecticides for mosquito’s eradication has resulted in physiological resistance. Plants are a source of biochemical compounds which possess an inhibitory effect on mosquitoes. In a study, the adulticidal, larvicidal, and genotoxic activity of P. corylifolia Linn. Essential oil against Culex quinquefasciatus Say was investigated. From the seeds of P. corylifolia Linn. essential oil was isolated Larvicidal and adulticidal bioassay of Cx. quinquefasciatus was done using the WHO method. Genotoxic activity of samples was carried out by comet assay. Gas chromatography-mass spectrometry was used for Identification of different compounds.
LC50 and LC90 values of essential oil were reported as 63.38 ± 6.30 and 99.02 ± 16.63 ppm, respectively against C. quinquefasciatus larvae. The obtained LD50 and LD90 values were 0.057±0.007 and 0.109 ± 0.014 mg/cm2. Genotoxicity of adults was determined at 0.034 and 0.069 mg/cm2. The mean comet tail length was 6.2548 ± 0.754 μm and 8.47 ± 0.931 μm and in comparison to controls the respective DNA damage was significant i.e. 6.713% and 8.864%. GCMS analysis of essential oil revealed 20 compounds. The major eight compounds include caryophyllene oxide (40.79%), caryophyllene (17.84%) phenol,4-(3,7-dimethyl-3-ethenylocta-1,6-dienyl) (20.78%), , α-humulene (2.15%), (+)- aromadendrene (1.57%), 1, 2, 3, 4-tetrahydro-1, 6- dimethyl-4-(1-methyl)-, naphthalene (1S-cis) (1.53%), methyl hexadecanoate (0.67%) and trans- caryophyllene (0.75%).
Results showed essential oil obtained from the P. corylifolia seeds showed potent toxicity against larvae and adult C. quinquefasciatus. The present work revealed that the P. corylifolia essential oil could be used as potential larvicidal and adulticidal agent for control of mosquitoes 31.
Toxicity Studies: Residual edema of the legs and cutaneous damage may occur when psoralen and its derivatives are used for sun tanning. In some cases, acute dermatitis with edema, blistering, and possibly renal complications have been observed. Other side effects include nausea, insomnia, vomiting, malaise, headache, loose motions, hepatotoxicity, and mental depression. Extensive chromosome damage took place in mammalian cells by psoralen treatment and high-intensity long wavelength irradiations. Therefore, caution should be needed while the use of psoralen and light therapy because this in may result from later malignancy. Long term therapy has been found to affect the liver, eyes, and immune system 32. A mixture of psoralen, isopsoralen, and imperatorin resulted in hypertrophy of kidney, liver, and spleen in rats at a daily dose of 2.5 mg/75 g for 60 days 33.
Acute toxicity 34 for psoralen LD50 oral in mice is 625 mg/kg; LD50 S.C. in mice is 480 mg/kg whereas, LD50 oral in rat is 1330 mg/kg and LD50 S.C. in rat is 830 mg/kg1.
CONCLUSION: The current trend is to promote alternative therapies as they are viewed favourably by many patients because they are often not being helped by conventional therapy and they believe that there are fewer detrimental side effects. Psoralen has a good potential to be developed as an alternative therapy. A wealth of in-vitro data now supports the long-held beliefs that psoralen has antimicrobial, antiviral, anti-biofilm and anti-inflammatory properties. Still, there is a great need for phytochemical investigations on active constituent psoralen for development of an effective natural remedy responsible for the wide range of activities.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Khushboo PS, Jadhav VM, Kadam VJ and Sathe NS: corylifolia Linn. - “Kushtanashini.” Pharmacognosy Reviews 2010; 4(7): 69-76.
- Pedro AS, Santo IE, Silva CV, Detoni C and E: Albuquerque E: The use of nanotechnology as an approach for essential oil-based formulations with antimicrobial activity. Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education 2013; 1364-1374.
- Acqua FD: Psoralens: A Review, Photosensitisation 15: 269-278.
- Boardley M, Stirton CH and Harborne JB: A chemotaxonomic survey of the tribe Psoralea in Africa. Biochemical Systematics and Ecology 1986; 14(6): 603-613.
- Gu Y, Si D, Gao J, Zeng Y and Liu C: Simultaneous quantification of psoralen and isopsoralen in rat plasma by Ultra-Performance Liquid Chromatography/Tandem Mass Spectrometry and its application to a pharmacokinetic study after oral administration of Haigou Pill. Journal of Chromatography B 2009; 877: 3137-3143.
- Dewick, PM: Medicinal Natural Products: A Biosynthetic Approach. Wiley 2009; 3:164-165.
- Srinivas CR and Pai S: Psoralens. Indian Journal of Dermatology, Venereology and Leprology 1997; 63: 276287.
- Honigsman H, Jaschke E and Gschnait F: 5 Methoxypsoralen in photochemotherapy of psoriasis. British Journal of Dermatology 1975;105: 369378.
- Dubertret L, Averbeck D and Zogdela F: Photo-chemotherapy (PUVA) of psoriasis using 3carbethoxy psoralen, a noncarcinogenic compound in mice. British Journal of Dermatology 1979; 101: 379389.
- Baccichetti F, Carlssare F and Bordin F: 4,4,6 Trimethyl angelicin a new very photoreactive and non skin phototoxic monofunctional furocoumarin. Photochemistry and Photobiology 1984; 39: 525-529.
- Honigsmann H, Wolff K and Fitzpatrick TB: Oral photochemotherapy with psoralens and UVA (PUVA): Principles and Practice. In: Dermatology in General Medicine, Mc Graw Hill Book Company, New York, Edition 3rd, 1987: 1533-1558.
- Volden G: PUVA, Textbook of Psoriasis, Edition 1st, 1986: 211232.
- Srinivasan S and Sarada DV: Antifungal activity of phenyl derivative of Pyranocoumarin from Psoralea corylifolia Seeds by inhibition of acetylation activity of trichothecene 3-O-acetyltransferase (Tri101). Journal of Biomedicine and Biotechnology 2012; 310850.
- Cardoso CA, Pires AE and Honda NK: A method for quantitative determination of furanocoumarins in capsules and tablets of phytochemical preparations. Chemical and Pharmaceutical Bulletin 2006; 54(4): 442-447.
- Akhtar N, Faiyazuddin MD, Mustafa G, Sultana Y, Baboota S and Ali J: High-Performance Thin-Layer Chromatographic analysis of psoralen in marketed formulations and manufactured solid lipid nanoparticles (slns): validation of the method. Acta Chromatographica 2012; 24(4): 603-613.
- Bhat TM, Choudhary S, Ansari MYK, Aslam R and Dar SA: Ameliorative response of some essential oil furanocoumarins and proteins from Psoralea corylifolia against gamma-irradiation induced oxidative stress. Industrial Crops and Products 2015; 76: 422-431.
- Cassidy RA, Kondo NS and Miller PS: Triplex formation by psoralen - conjugated chimeric oligonucleoside methylphosphonates. Biochemistry 2000; 39: 8683-8691.
- Kim KH, Nielsen PE and Glazer PM: Site-specific gene modification by PNAs conjugated to psoralen. Biochemistry 2006; 45: 314-323.
- Bettio F, Canevari M, Marzano C, Bordin F, Guiotto A, Greco F, Duncan R and Veronese FM: Synthesis and biological in-vitro evaluation of novel PEG-psoralen conjugates. Biomacromolecules 2006; 7: 3534-3541.
- Fang JY, Fang CL, Liu CH and Su YH: Lipid nanoparticles as vehicles for topical psoralen delivery: Solid lipid nanoparticles (SLN) versus nanostructured lipid carriers (NLC). European Journal of Pharmaceutics and Biopharmaceutics 2008; 70: 633-640.
- Gomes AJ, Lunardi CN, Lunardi LO, Pitol DL and Machado AEH: Identification of psoralen loaded PLGA microspheres in rat skin by light microscopy. Micron 2008; 39: 40-44.
- Patel NA, Patel NJ and Patel RP: Comparative development and evaluation of topical gel and cream formulations of psoralen. Drug Discoveries & Therapeutics 2009; 3(5): 234-242.
- Rani KRV, Prakash SEL and Manavalan R: Formulation of microsponge drug delivery of psoralen-isolated from Psoralea corylifolia. International Journal of Pharmacology and Biological Sciences 2009; 3(2): 119-132,
- Laxmi RJ, Karthikeyan R, Babu PS and Babu RVVN: Formulation and evaluation of antipsoriatic gel using natural excipients. J of Acute Disease 2013; 115-121.
- Kaur S, Kaur N, Sharma AK and Kanwar K: Development of modified transdermal spray formulation of psoralen extract. Der Pharmacia Lettre 2013; 5(2): 85-94.
- Kumari S and Pathak K: Cavamax W7 composite psoralen ethosomal gel versus cavamax W7 psoralen solid complex gel for topical delivery: A comparative evaluation. International Journal of Pharmaceutical Investigation 2013; 3(4): 171-182.
- Kumar D, Mishra BA, Shekar KPC, Kumar A, Akamatsu K, Kuriharab R and Ito T: Novel porphyrin-psoralen conjugates: synthesis, DNA interaction and cytotoxicity studies. Organic & Biomolecular Chemistry 2013; 11: 6675-6679.
- Zhang YT, Shen LN, Wu ZH, Zhao JH and Feng NP: Comparison of ethosomes and liposomes for skin delivery of psoralen for psoriasis therapy. International Journal of Pharmaceutics 2014; 471(1-2): 449-52.
- Zhang YT, Shen LN, Zhao JH and Feng NP: Evaluation of psoralen ethosomes for topical delivery in rats by using in vivo microdialysis. International Journal of Nanomedicine 2014; 9: 669-678.
- Zhang YT, Shen LN, Wu ZH, Zhao JH and Feng NP: Evaluation of skin viability effect on ethosome and liposome-mediated psoralen delivery via cell uptake. Journal of Pharmaceutical Sciences 2014; 103: 3120-3126.
- Dua VK, Kumar A, Pandey AC and Kumar S: Insecticidal and genotoxic activity of Psoralea corylifolia (Fabaceae) against Culex quinquefasciatus Say, 1823. Parasite Vectors 2013; 6: 30.
- Rajpal V: Standardization of Botanicals. Eastern Publishers, New Delhi, Vol. 2, 2005: 284-295.
- Sharma PC, Yelne MB and Dennis TJ: Database on Medicinal Plants used in Ayurveda. Central Council for Research in Ayurveda and Siddha, New Delhi, Vol. 2. 2001: 8993.
- http://www.mdidea.com. Accessed on 22- 06-2016
How to cite this article:
Kumar S and Rao R: Psoralen: A promising boon in topical manifestations. Int J Pharmacognosy 2016; 3(9): 375-83. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(9).375-83.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
375-383
616
964
English
IJP
S. Kumar and R. Rao*
Department of Pharmaceutical Sciences, Guru Jambeshwar University Science and Technology, Hisar, Haryana, India.
rekhaline@gmail.com
22 July 2016
11 September 2016
27 September 2016
10.13040/IJPSR.0975-8232.IJP.3(9).375-83
30 September 2016